Abstract
Background
Doublecortin-like kinase 1 (DCLK1) isoforms play distinct roles in the progression of gastrointestinal cancers. For the first time ever, the current study aimed to generate DCLK1-S-specific monoclonal antibodies (mAbs) to evaluate the clinical value of DCLK1-S (short isoform) in gastric cancer (GC).
Materials and methods
Mice were immunized with a unique 7-mer synthetic peptide of DCLK1-S conjugated with keyhole limpet hemocyanin (KLH). Immunoreactivity of hybridomas and mAbs was determined by ELISA assays and immunohistochemistry (IHC). DCLK1-S expression in two GC cell lines was assessed by flow cytometry. After characterization, the expression pattern of DCLK1-S was investigated in different histological subtypes of GC (n=217) and adjacent normal tissues (n=28) using IHC on tissue microarrays. The association of clinical prognostic values with DCLK1-S expression was also investigated.
Results
ELISA findings demonstrated that the generated monoclonal antibody (mAb) exhibited strong immunoreactivity towards the immunizing peptide. Positive control tissues, including GC and colorectal cancer, showed strong positive immunoreactivity with anti-DCLK1-S mAb whereas negative reagent control sections represented no staining, demonstrating the specificity of produced mAb. Flow cytometry analysis confirmed that the newly developed mAbs effectively recognized DCLK1-S on the cell surface. A mixture pattern of membranous, cytoplasmic, and nuclear DCLK1-S expression in the GC cells was observed. A significant and inverse association was identified between the expression DCLK1-S in the cell membrane and cytoplasm and PT stage, muscolarispropia, subserosa, and perineural invasion in intestinal subtype, respectively. In signet ring cell type, however, nuclear DCLK1-S expression was adversely associated with tumor size and PT stage. Furthermore, patients with low DCLK1-S expression had a shorter survival than patients with high expression, however, without a statistically significant association.
Conclusion
An efficient and precise tool for detecting DCLK1-S in cancer tissues has been developed. Moreover, DCLK1-S overexpression might be considered a favorable clinical factor in GC patients.
Keywords
Introduction
Gastric cancer (GC) is the fifth most commonly diagnosed malignancy and third leading cause of cancer-related mortality worldwide,1,2 with over 770,000 GC-related deaths, a five-year survival rate of less than 30%, and high prevalence in Asian, Eastern European, and South American countries. 3 Despite important advances in the multimodal therapeutic approaches, the global burden of GC still remains considerable with a particularly dismal prognosis. In addition, late-stage diagnosis, lack of adequate predictive biomarkers, progression to aggressive and metastatic forms, and treatment failure are the main challenges that hinder prognosis. 4 Of note, biomarkers represent valuable promising tools that could optimize early-stage diagnosis and prognosis, provide information regarding gastric malignancy pathophysiology, and predict clinical outcomes. 5
A growing body of evidence has highlighted the pivotal function of cancer stem cells (CSCs) in cancer initiation, progression, metastasis, and chemoresistance; in light of this, multiple strategies have been developed to recognize and target specific CSC markers in different types of cancers, including GC. 6 More recently, doublecortin-like kinase 1 (DCLK1), a microtubule-associated protein, has been suggested as a novel, tumor-specific stem cell marker in gastrointestinal cancers. Nakanishi et al. first discovered that DCLK1 can be used as a biomarker for distinguishing between cancer and normal stem cells in the intestine. 7 Thereafter, various investigations indicated that downregulating DCLK1 expression results in growth arrest in colorectal, 8 renal, 9 and pancreatic carcinomas. 10 Mechanistically, DCLK1 modulates NF-kB, NOTCH, AKT, and KRAS molecular signaling cascades that support tumor progression and promote stemness and EMT of cancer cells.11–13 Additionally, many of the investigations performed by our and other research teams have identified DCLK1 as being highly expressed in several malignancies, including colorectal, 14 pancreatic, 15 hepatocellular, 16 renal, 9 and bladder cancers. 17
The human DCLK1 gene consists of two promoters, a 5′ (α) promoter which drives the transcription of the long (L)-isoform (DCLK1-L, ∼ 80–82 KDa) and a 3′ (β) promoter which is responsible for the short (S)-isoform expression (DCLK1-S, ∼ 45–50 KDa). 18 Greater than 98% homology exists at the C-terminal end of both short and long isoforms. In contrast, the L-isoform and S-isoform differ mostly at the N-terminal end, including the absence of the doublecortin domain as well as a different sequence of six amino acids at the N-terminal of the DCLK1-S isoform. 19 Recent findings have shown the isoform particular role of DCLK1 in cancer progression. Evidence indicates that the α-promoter, which drives the expression of the DCLK1-L isoform, is epigenetically hypermethylated and silenced during the adenoma–carcinoma sequence of gastrointestinal cancers. 18 Thus, it has been suggested that DCLK1-L marks only normal human cells and early stage cancer cells. In contrast, the alternate β-promoter is used for transcription of the short DCLK1 isoform at the advanced stages of malignancies, such as CRC. 18 Importantly, several recent studies have shown the correlation of DCLK1-S overexpression with shorter survival outcomes in CRC 20 and esophageal squamous cell carcinoma (ESCC) patients. 21 Our recent findings also strengthen the crucial role of DCLK1-S, as an independent prognostic factor, in cancer aggressiveness in colorectal patients. 22
Regarding the importance of DCLK1 in GC, DCLK1 was first recognized as a potential driver based on a comprehensive genomic and molecular analysis of 100 primary GCs. 23 Limited research by our and other groups has identified the significance of DCLK1 in GC patients.14,24,25 However, all these studies used commercially available antibodies that target the C-terminal end of DCLK1 proteins, which is common to both long and short isoforms, or only the N-terminal doublecortin domains of L-isoform, which the short isoform lacks these domains. To the best of our knowledge, monoclonal antibodies (mAbs) specific for short isoforms with immunohistochemical application are not commercially available. The detection of DCLK1-S using mAb in cancerous and normal tissues can be of great value for cancer investigations. Additionally, the expression pattern and prognostic importance of DCLK1-S in most carcinomas, including gastric cancer, remain unclear. Therefore, these gaps prompted us to develop anti-DCLK1-S mAbs against the unique N-terminal six amino acids sequence present only in DCLK1-S. Afterwards, the expression and clinicopathological prognostic value of DCLK1-S were evaluated, using our laboratory produced anti-DCLK1-S mAbs, in a large series of GC tissue microarrays (TMAs) by immunohistochemistry (IHC) technique.
Material and methods
Generation and characterization of anti-DCLK1-S-specific monoclonal antibodies
Preparation of immunogen
High-purity synthetic peptide selected from the unique sequence of DCLK1-S isoform (NH2-Met-Leu-Glu-Leu-Ile-Glu-Cys-COOH) was purchased from BIO BASIC INC (Canada). Imject Maleimide-activated bovine serum albumin (BSA) (Thermo Scientific, Rockford, IL, USA) and Maleimide-activated keyhole limpet hemocyanin (mcKLH) (Thermo Scientific, Rockford, IL, USA) were utilized to be conjugated separately with peptide. To investigate the conjugation efficacy, the sodium salt-polyacrylamide gel electrophoresis (SDS-PAGE) pattern of the BSA conjugate was investigated.
Immunization protocol
Female BALB/c mice (6–8 weeks old) were purchased from Pasteur Institute of Iran (Tehran, Iran) and maintained in standard conditions at the animal facilities of Iran University of Medical Sciences (IUMS). All protocols were approved by the animal research committee of IUMS. Each mouse was immunized intraperitoneally five times at three-week interval between the first and second injection and two-week intervals between next injections. For the first immunization, 50 μg KLH-peptide emulsified in complete Freund's adjuvant was utilized for the intraperitoneal immunization of each BALB/c mouse. For the subsequent four booster immunizations, each mouse was immunized with 50 μg the KLH-peptide in incomplete Freund's adjuvant. Subsequently, blood from the tail vein was obtained, ten days after the last injection, and the antibody titer was determined in each mouse sera using enzyme-linked immunosorbent assay (ELISA) against DCLK1-S peptide, as described below. 26 According to ELISA results, three days prior to cell fusion, 20 μg of the antigen was injected intravenously into the mouse with the highest antibody titer.
Indirect ELISA
Indirect ELISA was applied to assess and confirm the reactivity of mice sera, secreting hybrids, and the purified mAbs against DCLK1-S. Briefly, 96-well ELISA plates were coated with a total volume of 100 μl DCLK1-S peptide (10 µg/ml) diluted in phosphate-buffered saline (PBS, 0.15 M, pH 7.2). Following incubation at 37 °C for 1 h, the plates were incubated at 4 °C overnight. The following day, after three times washing with PBS containing 0.05% Tween 20, the wells were blocked for one hour with 2.5% skimmed milk at 37 °C. Following that, the wells were incubated with serial dilutions of mice sera, secreting hybridomas, or serial dilutions of purified anti-DCLK1-S mAbs for 1.5 h at 37 °C. Following washing, 1:1000 dilution of horseradish peroxidase (HRP)-conjugated sheep anti-mouse immunoglobulin (Ig) (Avicenna Research Institute, Tehran, Iran) was added to all wells at 37 °C for 1.5 h. After washing, tetramethylbenzidine substrate (TMB) substrate (Sigma-Aldrich Chemie GmbH, Germany) was applied for each well and H2SO4 (20%) was added to stop the reaction. Finally, signals were measured on a microplate reader (Synergy HTXMulti-Mode Reader, BioTek, USA) at 450 nm.
Cell culture
Murine myeloma cell line (SP2/0) and gastric cancer cell lines (MKN-45 and AGS) (all from National Cell Bank of Iran, Pasteur Inst. of Iran, Tehran) were cultured in either RPMI 1640 (Gibco) (for SP2/0, MKN-45) containing 10% fetal bovine serum (FBS) (Gibco) or Dulbecco's modified Eagle medium (Gibco, Invitrogen, CA, USA) (for AGS) containing 10% FBS. Cultured cells were maintained within an incubator containing humidified 5% CO2 atmosphere at 37 °C.
Generation of anti-DCLK1-S monoclonal antibodies
mAbs were generated by standard hybridoma technology, as described elsewhere.26,27 Briefly, spleen lymphocytes were isolated from immunized mice and fused with SP2/0 cell line through polyethylene glycol (PEG) 1500 (Sigma) at a 5:1 ratio. Then, RPMI medium containing hypoxanthine-aminopterin-thymidine (HAT) and 20% FBS was used for the selection of hybridisms. After 10–12 days of fusion, indirect ELISA was applied to screen the supernatants of hybridomas. Using limiting dilution method, hybridomas capable of producing specific antibody were cloned four times. According to the results obtained from screening steps, finally, two clones, including 2D5C2 and 2H3D5, were opted for the subsequent experiments. Protein G affinity chromatography (Amersham Biosciences, the Netherlands) was applied to purify the monoclonal antibodies. The concentration of purified mAbs was spectroscopically determined at 280 nm. The purity of the mAbs was assayed by electrophoresis and the reactivity of the purified antibodies was evaluated using indirect ELISA technique.
Monoclonal antibody characterization by immunohistochemistry
The specificity of anti-DCLK1-S mAb was determined by IHC. In this study, humans colorectal and gastric cancer tissues were utilized as positive control for our generated mAb against DCLK1-S. In isotype control slides, mouse IgG (Avicenna Research Institute, Tehran, Iran), instead of the primary antibody, was utilized to validate anti-DCLK1-S mAbs specific bindings. Additionally, prior to the application of the primary antibody solution to the TMA sections, primary antibody was preincubated with a saturating concentration of immunizing peptide (1:100 molar ratio) in negative reagent control slides. For further verification of the specificity of our mAb, according to protein atlas databases, normal tissue of testis as well as skin and ovary cancerous tissues as negative control tissues were also utilized.
Analysis of DCLK1-S expression in gastric cancer cell lines by flow cytometry
Flow cytometry was applied to investigate the DCLK1-S expression in GC cell lines. MKN-45 and AGS cells at 70%–80% confluency were harvested. Following three times wash with cold PBS (pH 7.2, 0.15 M), 5% sheep serum was added to cells for 20 min on ice. Sequentially, cells were treated with either anti-DCLK1-S mAb diluted in PBS-2%FBS (10 μg/mL) or equal concentration of non-immunized mouse Ig (as negative control) on ice for 45 min. After three times washing, cells were incubated with FITC-conjugated sheep anti-mouse Ig (1:75, Avicenna Research Institute, Iran) for 30 min. Finally, cells were washed three times. Data was obtained via an Attune NxT flow cytometer (Thermo Fisher Scientific, USA) and analyzed by FlowJo software VX.
Population study
Sample collection and patients characteristics
In the current cross-sectional study, a total of 217 formalin-fixed, paraffin-embedded (FFPE) specimens from GC cases were enrolled from university-based referral hospitals in Tehran, Iran. Cases did not receive any preoperative chemotherapy or radiotherapy prior to surgery. Patient's hematoxylin and eosin (H&E) stained pathology sections and medical archived data were evaluated to obtain the clinical and pathological features: gender, age, tumor size (maximum tumor diameter), histological grade, histological subtype, tumor site, PT stage, tumor extension, lamina propria involvement, muscularis mucosa involvement, muscularis propria involvement, subserosa involvement, perineural invasion (PI), lymphovascular invasion, distant metastasis, and tumor recurrence. In addition, 28 adjacent normal tissues were utilized for assessment of DCLK1-S expression in comparison to cancerous specimens. Additionally, the patient's survival outcomes, including disease-specific survival (DSS) and progression-free survival (PFS), were reviewed. DSS was considered as the interval from surgery until death from patient's cancer. The PFS was calculated as the length of time between the primary surgery and the last follow-up with no evidence of disease, distant metastasis, or recurrence. This research was performed with Ethical approval by the Ethics Committee of the Iran University of Medical Sciences (Ref No: IR.IUMS.REC-98–4-28-16306) and informed written consents were obtained from all patients before inclusion.
Tissue microarray (TMA) construction
The GC TMA blocks construction was conducted, as reported previously.28,29 Briefly, to prepare TMA blocks, three most representative regions from different areas of the tumor and normal tissues were opted and marked after matching with H&E slides. Afterward, the marked tumor regions of each donor block with 0.6-mm diameter were punched out by tissue arrayer instrument (ALPHELYS, Plaisir, France) and transferred into the TMA recipient paraffin blocks. Finally, the sectioning of GC TMA blocks was performed at 4 μm thickness and prepared sections were transferred to microscope slides (SuperFrost plus, Thermo Scientifc™, Germany). In this research, each tumor sample was constructed in three cores to cover tumor heterogeneity challenge during the TMA process, and, then, scored individually for precise evaluation of the specimens. To increase the validity and accuracy of the data analysis, the final scores for tissue specimens were calculated from the mean of the three cores.
Immunohistochemistry
Immunostaining of GC TMA slides for DCLK1-S was conducted through our previously described laboratory protocol.22,30 Briefly, after deparaffnization of all TMA slides (at 60 °C and then by xylene), the sections were rehydrated under serial dilutions of ethanol. Afterward, to block endogenous peroxidase activity, tissue slides were incubated in methanol containing 3% hydrogen peroxide (H2O2) at room temperature for 20 min. After three times wash of the slides in Tris Bufered Saline (TBS), antigen retrieval was conducted through autoclaving the slides in sodium citrate buffer (pH 6.0) for 10 min. Afterwards, following three times wash, the slides were treated with our generated primary anti-DCLK1-S mAb (5 µg/ml) at 4 °C overnight. Additionally, mouse IgG (Avicenna Research Institute, Iran) was used at 5 µg/ml for isotype control. The following day, TMA sections were washed three times with TBS, followed by incubation for 1 h with the EnVision™ + /HRP, DualLink Rabbit/Mouse (Dako, Denmark), as secondary antibody, at room temperature. Then, all sections were visualized through chromogen, named 3, 3′-diaminobenzidine substrate (DAB, Dako, Glostrup, Denmark) during 10 min, followed by counterstaining through Mayer's hematoxylin dye (Dako, Glostrup, Denmark) for 2 min. Finally, the slides were dehydrated in graded ethanol, cleared in xylenes, and mounted for assessment by pathologists. Images were photographed by an H550S microscope equipped with a Digital sight DS-LS camera (Nikon, Japan).
Scoring system of IHC slides
Our pathologist (NM) applied a semi-quantitative scoring system to assess the immunostaining of tissue arrays in a blinded manner. The expression level of DCLK1-S was evaluated by three scoring methods, namely the staining intensity, positively stained cells percentage, and the Histochemical score (H-score). The immunostaining intensity of DCLK1-S was scored in a 4-point scale (absent/negative: 0, weak: 1, moderate: 2, or strong: 3) and the percentage was valued based on 1–100% of positively stained cells. The H-score was acquired by multiplying the staining intensity by the percentage, given an overall score of 0–300 for each core.31–33 The final H-score of each sample was achieved from the average of three replicates. In the present study, for analysis, the H-scores were categorized into two groups (low or high expression) based on the median DCLK1-S expression. The median H-score value was 180 in total tumor samples and intestinal type for membranous, cytoplasmic, and nucleolus localizations. However, in signet ring cell type, the median H-score values (cutoffs) were 140 for membranous staining, 170 for cytoplasmic staining, and 160 for nucleolus staining.
Statistical analysis
Pearson's chi-square (χ2) and Spearman's analyses have been applied to assess the association and correlation between DCLK1-S expression and clinicopathological characteristics, respectively. Kruskal–Wallis and Mann–Whitney U analyses were utilized for pairwise comparison of DCLK1-S expression across the groups. The Kaplan–Meier analysis was applied to generate the DSS and PFS curves. Furthermore, to compare survival outcomes across the groups (low and high expression of DCLK1-S), log-rank analysis was conducted. The classified data were reported by N (%), and quantitative data by mean with standard deviation (SD) and median with quartile (Q1, Q3). The Cox proportional hazards regression model was utilized to conduct both univariate and multivariate analyses. The SPSS statistical software package version 25 (SPSS, Chicago, IL, USA) was applied to statistically analyze all clinical data. P value less than 0.05 (P ≤0.05) was considered as statistically significant.
Results
Generation of anti-DCLK1-S monoclonal antibody and characterization
Preparation of immunogen
Electrophoresis of the BSA-conjugate was applied to assess the efficacy of immunogen-carrier conjugation. The smear pattern of the DCLK1-S-conjugated BSA and the lack of free peptide confirmed peptide-carrier conjugation (Figure 1A).
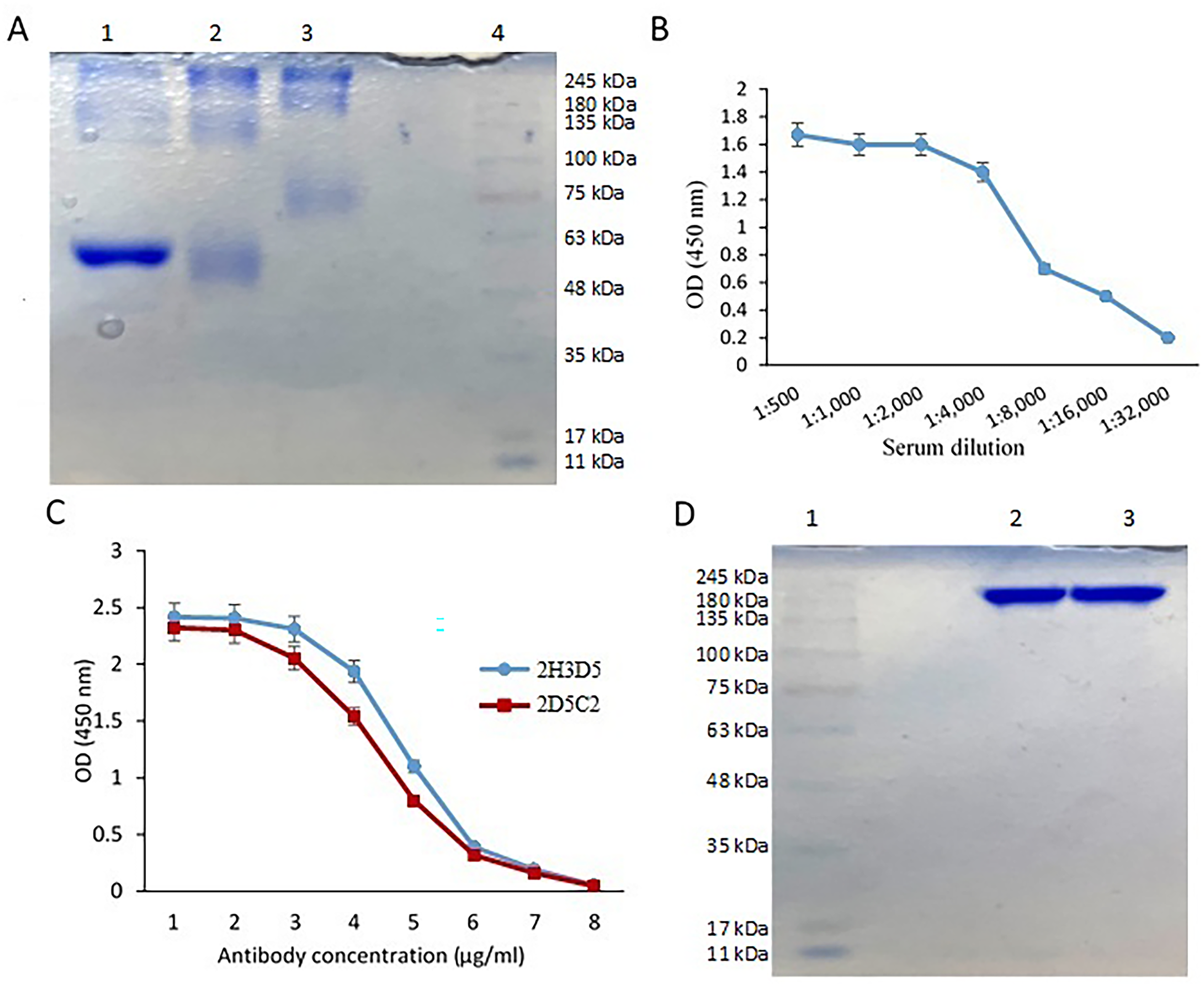
Generation of anti-DCLK1-S monoclonal antibody. (A) DCLK1-S peptide was conjugated to a carrier protein and conjugation efficacy was confirmed by SDS-PAGE analysis. BSA (lane 1), BSA-linker (lane 2), and BSA-conjugated peptide (lane 3), Molecular weight markers (lane 4). (B) Titration of anti-DCLK1-S antibody in immunized mice serum by indirect ELISA. Hyperimmunized serum was serially titrated on a peptide-coated plate. (C) Reactivity assessment of purified anti-DCLK1-S monoclonal antibodys (mAbs) by indirect ELISA. Purified antibodies indicated excellent reactivity with immunizing peptide in an indirect ELISA. (D) The purity of the mAbs was confirmed by SDS-PAGE. Molecular weight markers (lane 1), Clone 2H3D5 (lane 2), and Clone 2D5C2 (lane 3).
Immunization of mice and selection of hybridomas
After last immunization of mice, the production of anti-DCLK1-S antibody was confirmed by an optimized indirect ELISA. Both sera of the immunized mice showed a relatively high titer of anti-DCLK1-S antibodies. The mouse showing higher level of antibody titer was selected for fusion (Figure 1B). Following cell fusion, the hybridoma culture supernatants were screened for DCLK1-S peptide-specific antibodies, as described for the serum ELISA above. After four successive cloning, finally, two stable clones, named 2H3D5 and 2D5C2, were obtained and further characterized. The protein G affinity columns were applied to purify both clones.
Reactivity assessment of purified mAbs
Indirect ELISA was used to verify the immunoreactivity of purified mAbs. The findings indicated high immunoreactivity of both mAbs against immunizing peptide (Figure 1C). Both 2H3D5 and 2D5C2 mAbs showed partly similar reactivity reaching to plateau as low an antibody concentration of 5 μg/mL. Additionally, the mAbs’ purity was confirmed using SDS-PAGE (Figure 1D).
Validation of anti-DCLK1-S monoclonal antibody
To evaluate the specificity and reactivity of generated anti-DCLK1-S mAbs, immunohistochemical experiments were performed on GC and CRC tissues as positive control samples and human normal testis, as well as skin and ovary cancer tissues as negative control samples (Figure 2). Additionally, as negative reagent controls, primary antibody pre-adsorbed with immunizing peptide and isotype control were applied (Figure 2). Immunostaining of GC tissues showed that 2H3D5 clone against DCLK1-S have more specificity of reactivity compared to 2D5C2 clone (supplementary Figure 1). Therefore, 2H3D5 mAb was used for the following investigations. Positive control tissues, including GC and CRC, showed strong positive immunoreactivity with anti-DCLK1-S mAb whereas negative reagent control sections represented no staining, demonstrating the specificity of the produced mAb (Figure 2). Additionally, negative control tissues did not show any staining, confirming the particular immunoreactivity of the generated mAb against DCLK1-S (Figure 2). GC cells showed a variable immunostaining pattern of expression of DCLK1-S in their membrane, cytoplasm, and nucleus (Figure 3).
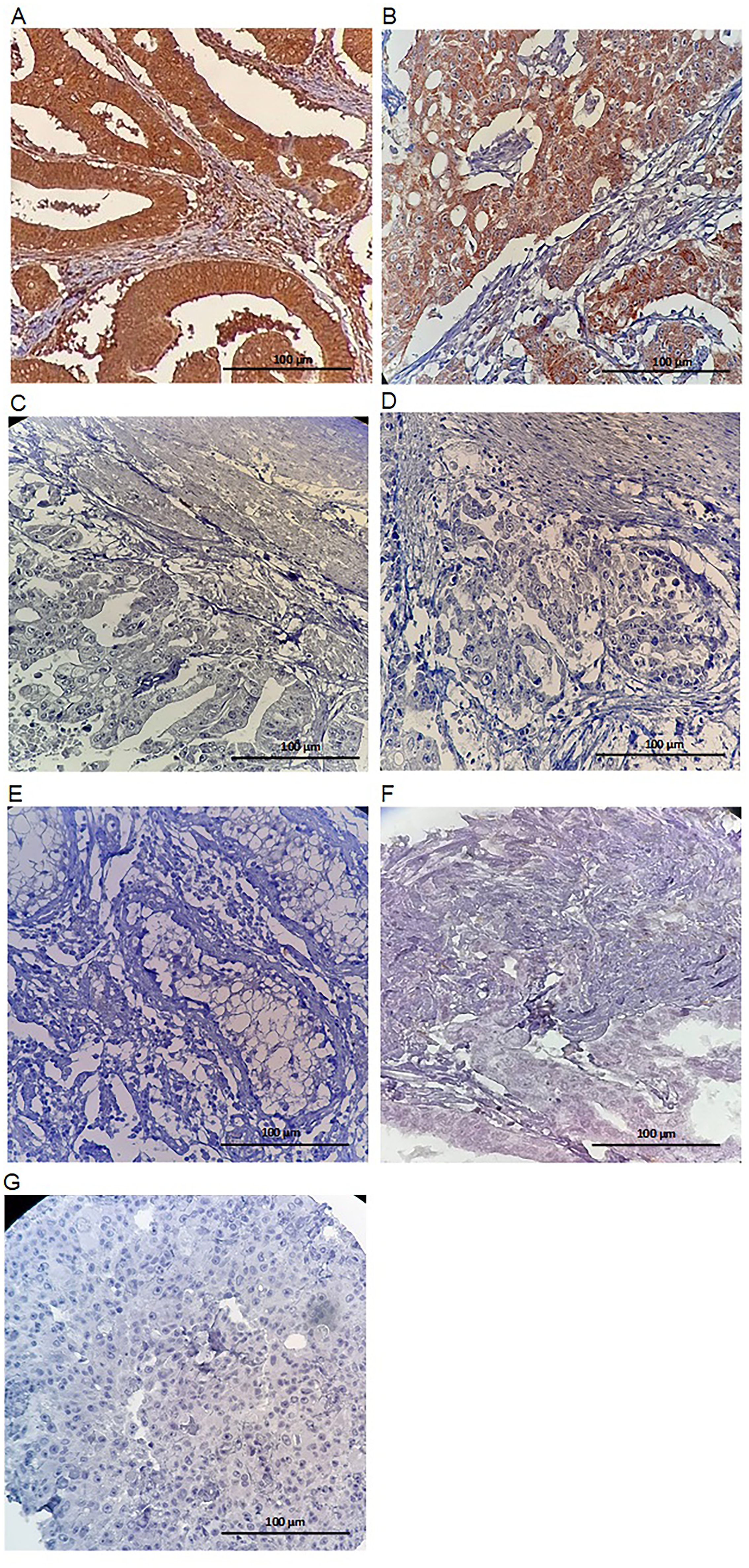
Characterization of anti-DCLK1-S monoclonal antibody. (A, B) The reactivity of the generated 2H3D5 mAb against DCLK1-S was examined by immunohistochemistry on colorectal (A) and gastric (B) cancer tissues as positive control samples. (C, D) Negative reagent controls, including sections incubated with non-immunized mouse IgG instead of primary antibody (C) and primary antibody was pre incubated with 100 fold molar concentration of immunizing peptide (D). (E, F, G) Immunohistochemical staining of DCLK1-S in negative control tissues, including Normal testis (E), ovarian cancer (F), and squamous cell carcinoma (SCC) tissues (G). (Images were taken at 200x magnification).
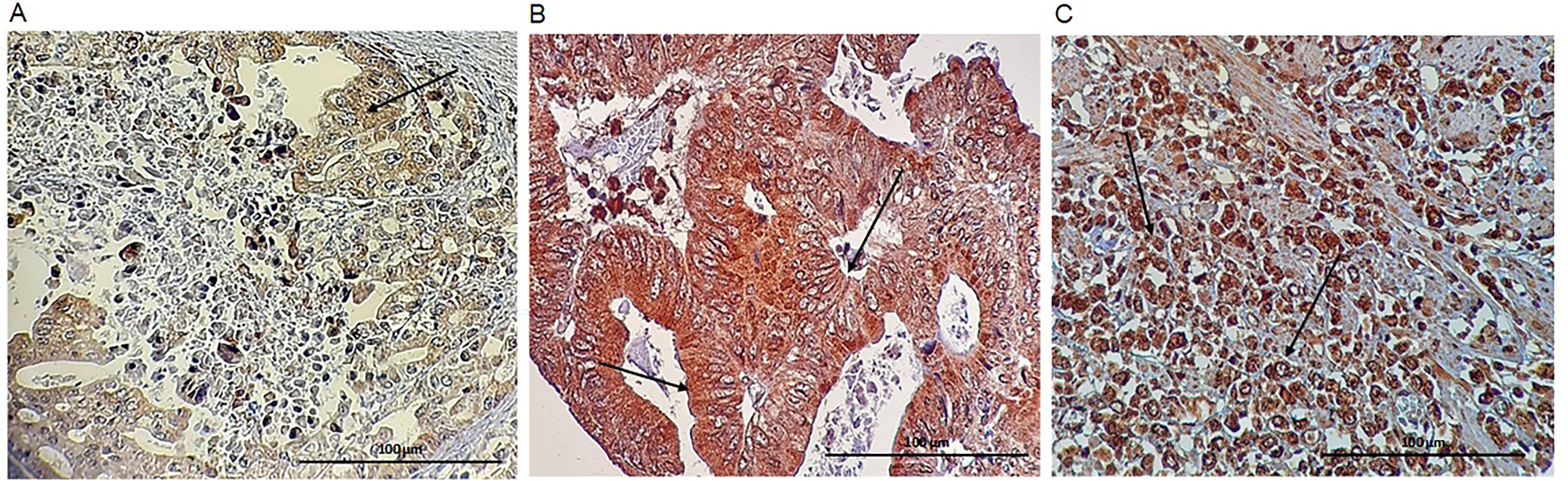
Different DCLK1-S expression patterns in gastric cancer tissues using immunohistochemical staining: (A) cytoplasmic expression (black arrow), (B) membranous and cytoplasmic expression (black arrow), and (C) nuclear expression of DCLK1-S (black arrow) (all images were taken at 400x magnification).
Expression of DCLK1-S in gastric cancer cell lines by flow cytometry
Flow cytometry was applied to assess the surface expression of DCLK1-S in two gastric cancer cell lines, including MKN-45 and AGS. Our results showed the surface DCLK1-S expression in both cell lines with varying levels. AGS cells (58.2%) expressed higher surface DCLK1-S levels compared to MKN-45 (23.6%) (Figure 4).
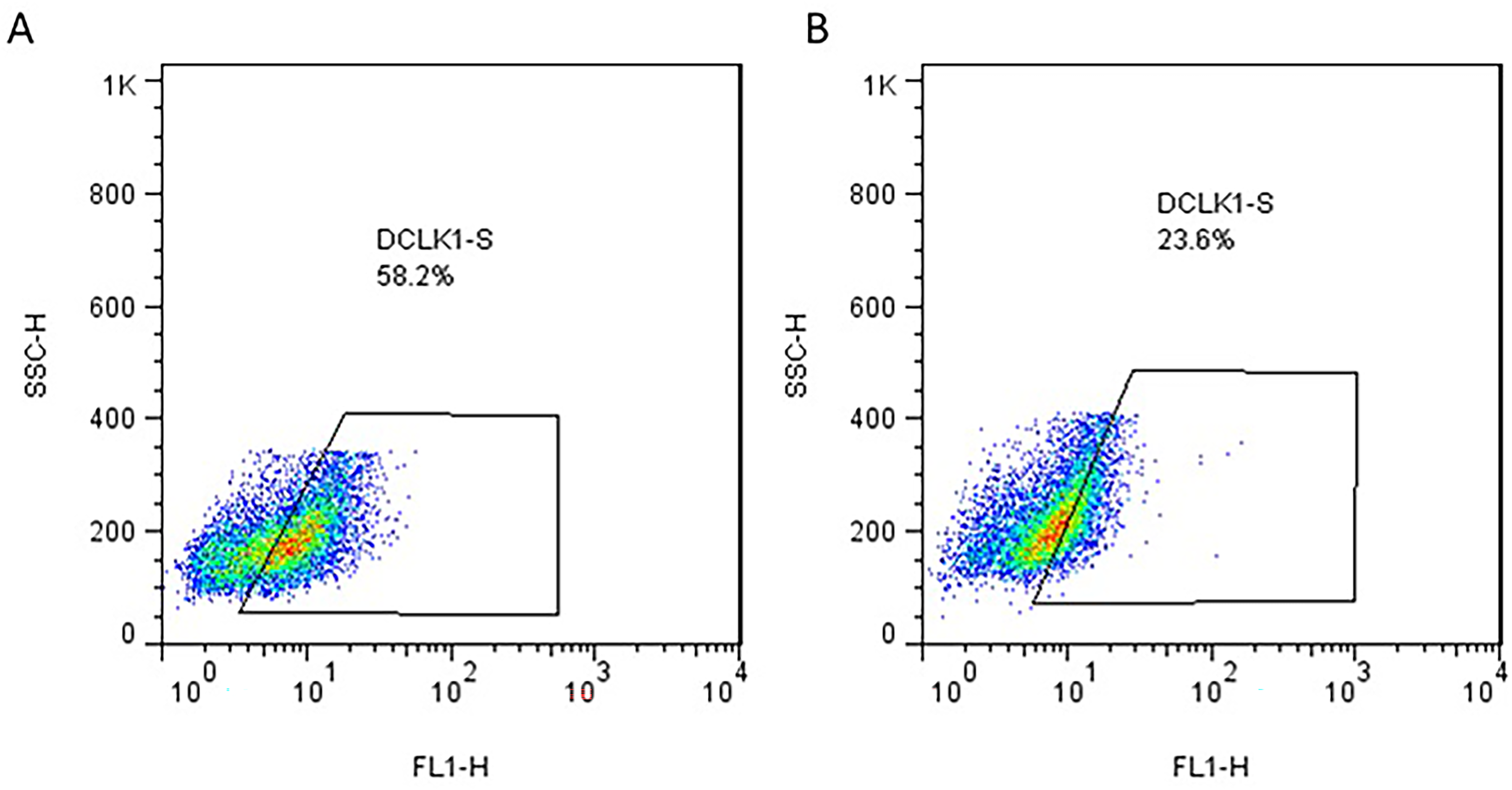
Expression profiling of DCLK1-S in human gastric cancer cell lines. Surface DCLK1-S expression in (A) AGS and (B) MKN-45 gastric cancer cell lines was carried out using flow cytometry.
Immunohistochemical evaluation by anti-DCLK1-S monoclonal antibody in gastric cancer tissues
Patient characteristics
To evaluate the clinicopathological importance of DCLK1-S, its expression was investigated in 217 GC tissues. Overall, the population median age was 64 years (range 24–85). The study contained 161 (74.2%) male and 56 (25.8%) female patients that male-to-female ratio was 2.8. In this study, first, total GC samples (N = 217) were analyzed and then, investigation was performed based on categorization to each gastric cancer histological subtype, including intestinal subtype (N = 155) and signet ring cell carcinoma subtype (N = 61). Clinical and pathological data of total samples and subtypes of GC are demonstrated in Table 1.
Patients and tumor clinicopathological characteristics of total gastric cancer samples and different histological subtypes.
DCLK1-S protein expression in gastric cancer patients, histological subtypes and adjacent normal tissues samples
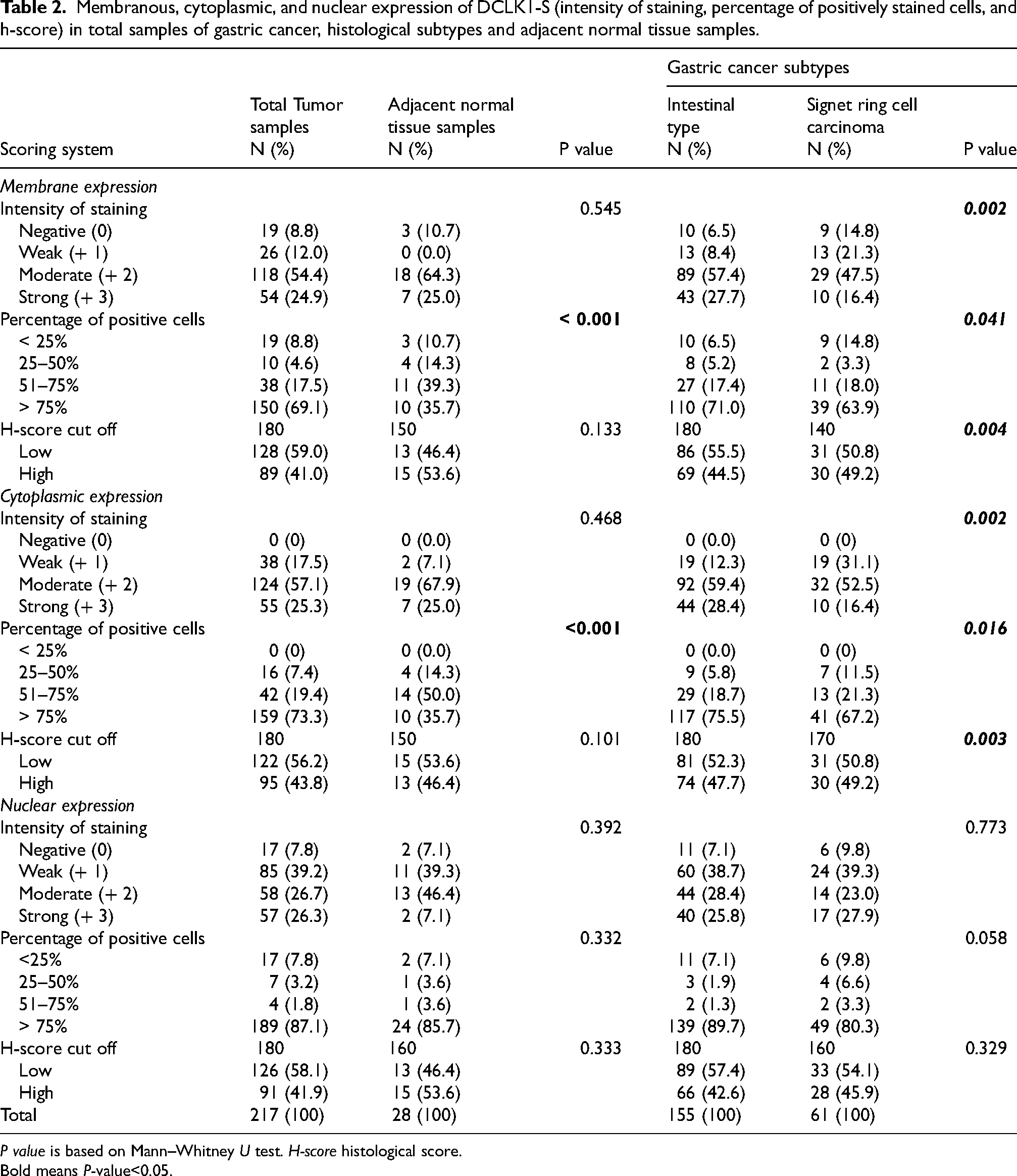
The level of DCLK1-S expression in each GC was evaluated based on TMA–immunohistochemistry staining via three scoring methods: staining intensity, percentage of positively stained tumor cells and H-score. The DCLK1-S expression was observed at various intensities in the membranous, cytoplasmic and nuclear compartments in total tumor samples, both histological subtypes of GCs, and adjacent normal tissues (Figure 5 and Table 2). First, total GC samples (N = 217) were analyzed and then, investigation was performed based on categorization to each gastric cancer histological subtype. For all three localizations, GC were classified into either low (≤median of H-scores) or high (> median of H-scores) expression using the median H-score value as the cutoff. The median H-score value was 180 in total tumor samples and intestinal type for membranous, cytoplasmic, and nucleolus localizations. However, in signet ring cell type, the median H-score values (cutoffs) were 140 for membrane, 170 for cytoplasm, and 160 for nucleolus to categorize the samples into either low (≤median of H-scores) or high (> median of H-scores) expression. A low expression of DCLK1-S was detected in 128 (59.0%), 122 (56.2%) and 126 (58.1%) of GC samples in the membrane, cytoplasm and nuclear, whereas higher DCLK1-S expression was found in 89 (41.0%), 95 (43.8%) and 91 (41.9%) samples, respectively (Table 2). The DCLK1-S expression in adjacent normal tissues was almost similar to that seen in GC samples based on staining intensity and H-score, as Kruskal-Wallis and Mann-Whitney U tests demonstrated non-significance difference between the median membranous, cytoplasmic and nuclear DCLK1-S expression of tumural and normal tissues (table 2). However, a statistically significant difference was distinguished in the percentage of positively stained cells for membranous and cytoplasmic expressions of DCLK1-S between gastric cancer and normal samples (for both; P<0.001) (Figure 6 and table 2).
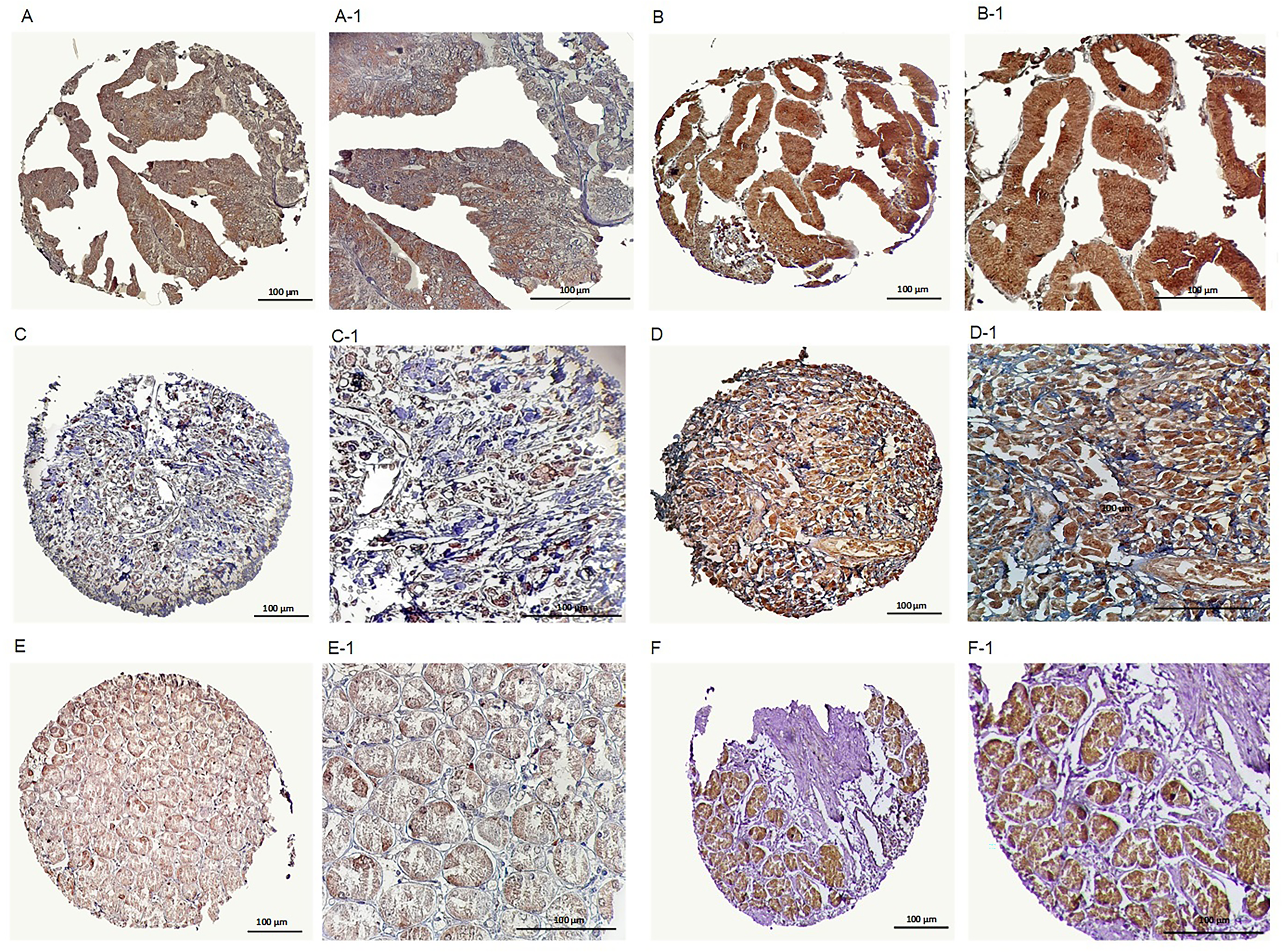
Immunohistochemical evaluation of DCLK1-S protein expression in gastric cancer samples. DCLK1-S expression in intestinal type: (A, A-1) low expression, (B, B-1) high expression. DCLK1-S expression in signet ring cell carcinoma: (C, C-1) low expression, (D, D-1) high expression. DCLK1-S expression in in adjacent normal tissue: (E, E-1) low expression, (F, F-1) high expression (Images were taken at 100x and 200x magnification).
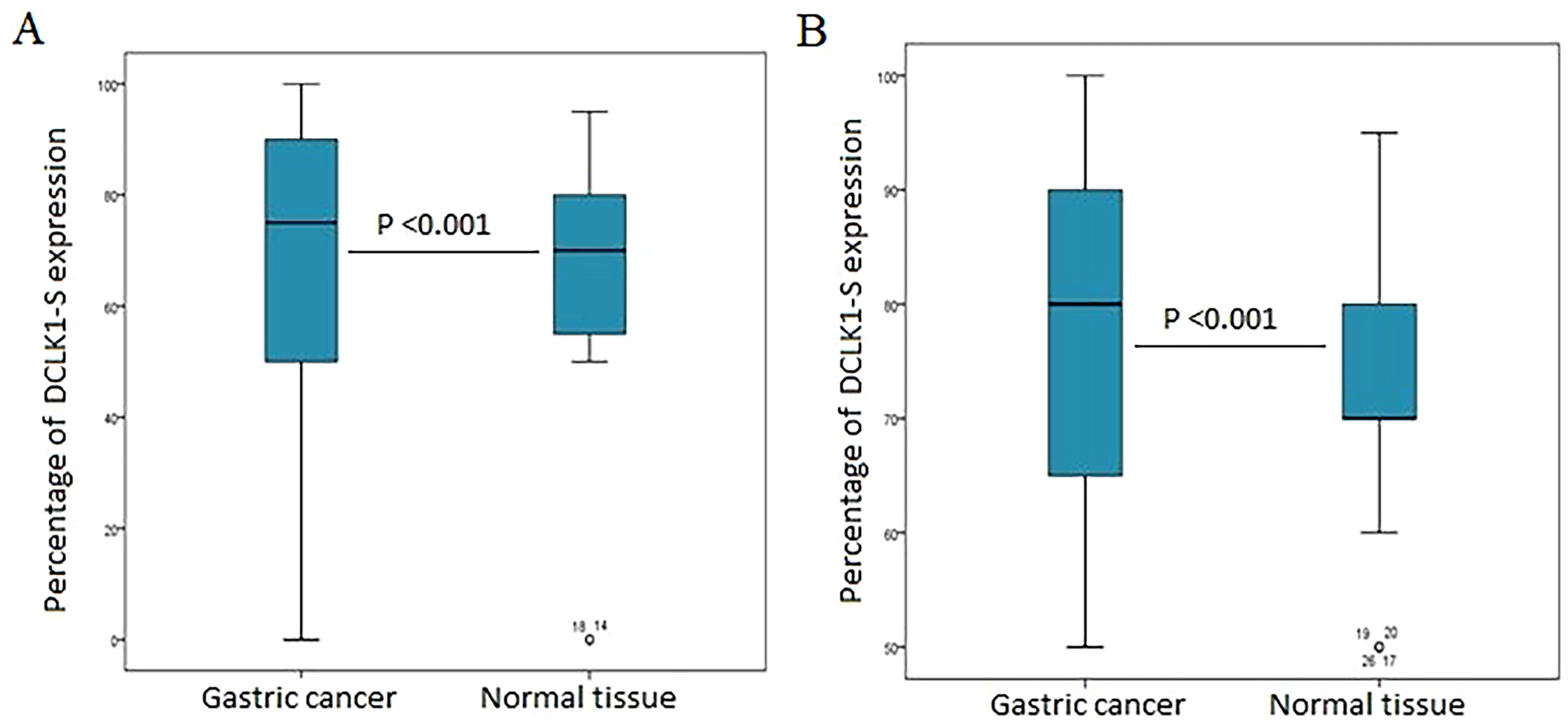
Box plot analysis of the DCLK1-S expression in gastric cancer (gc) patients versus adjacent normal tissues based on percentage of positively stained cells. The results of Mann-Whitney U test indicated that there is a statistically significant differences in the median expression DCLK1-S levels among GC and adjacent normal tissues in (A) membrane (P < 0.001) and (B) cytoplasm (P < 0.001).
Membranous, cytoplasmic, and nuclear expression of DCLK1-S (intensity of staining, percentage of positively stained cells, and h-score) in total samples of gastric cancer, histological subtypes and adjacent normal tissue samples.
P value is based on Mann–Whitney U test. H-score histological score.
Bold means P-value<0.05.
Mann-Whitney U analyses were also applied to compare the DCLK1-S protein expression between the histological subtypes of GCs. A statistically significant difference was identified for the cytoplasmic and membranous expressions of DCLK1-S between GC histological subtypes in all scoring methods (Table 2). The DCLK1-S expression data is summarized in GC patients, histological subtypes, and adjacent normal tissues in Table 2.
Association of DCLK1-S expression with clinicopathological characteristics in the total gastric cancer patients and histological subtypes
Total gastric cancer samples
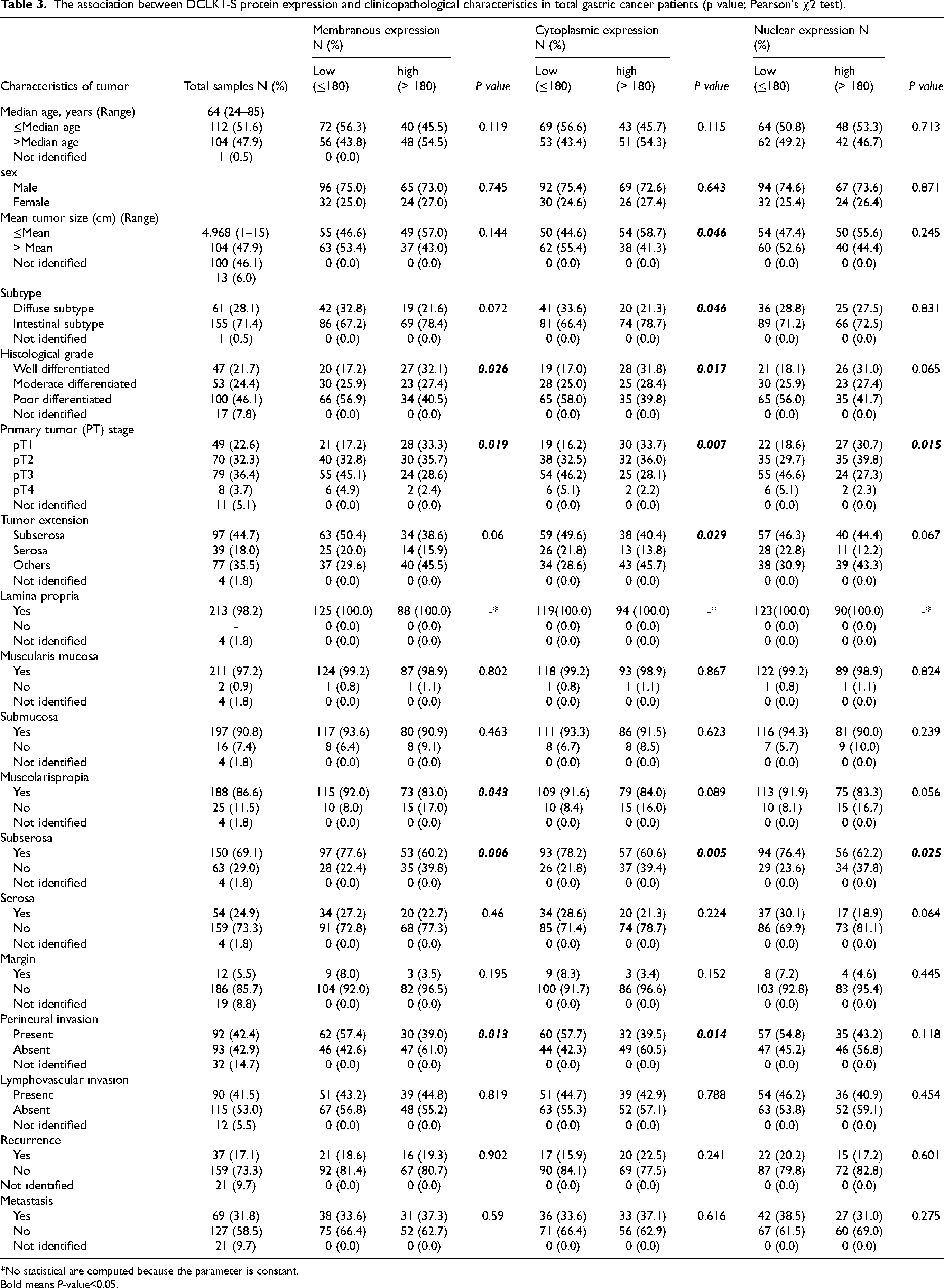
To assess the association of expression of DCLK1-S with clinicopathological features, Pearson's χ2 analysis was performed. As demonstrated in Table 3, a statistically significant association was identified between the membranous expression of DCLK1-S and histological grade (P = 0.026), PT stage (P = 0.019), muscolaris propia (P = 0.043), subserosa (P = 0.006), and perineural invasion (P = 0.013) in GC patients (in terms of H-score). Furthermore, there were significant association between cytoplasmic DCLK1-S expression and important clinicopathological parameters, including mean tumor size (P = 0.046), subtype (P = 0.046), histological grade (P = 0.017), PT stage (P = 0.007), tumor extension (P = 0.029), subserosa (P = 0.005), and perineural invasion (P = 0.014). Nuclear DCLK1-S expression was only associated with PT stage (P = 0.015), and subserosa (P = 0.025).
The association between DCLK1-S protein expression and clinicopathological characteristics in total gastric cancer patients (p value; Pearson's χ2 test).
No statistical are computed because the parameter is constant.
Bold means P-value<0.05.
The correlation between DCLK1-S expression and clinical features was evaluated using Spearman's correlation test. Spearman's correlation test results showed a significant inverse correlation between membranous, cytoplasmic, and nuclear expression of DCLK1-S and the above mentioned clinicopathological features, indicating that the higher DCLK1-S expression is more often found in GC tissues with well differentiation, lower PT stages and lack of perineural invasion of GC tissues.
Intestinal type
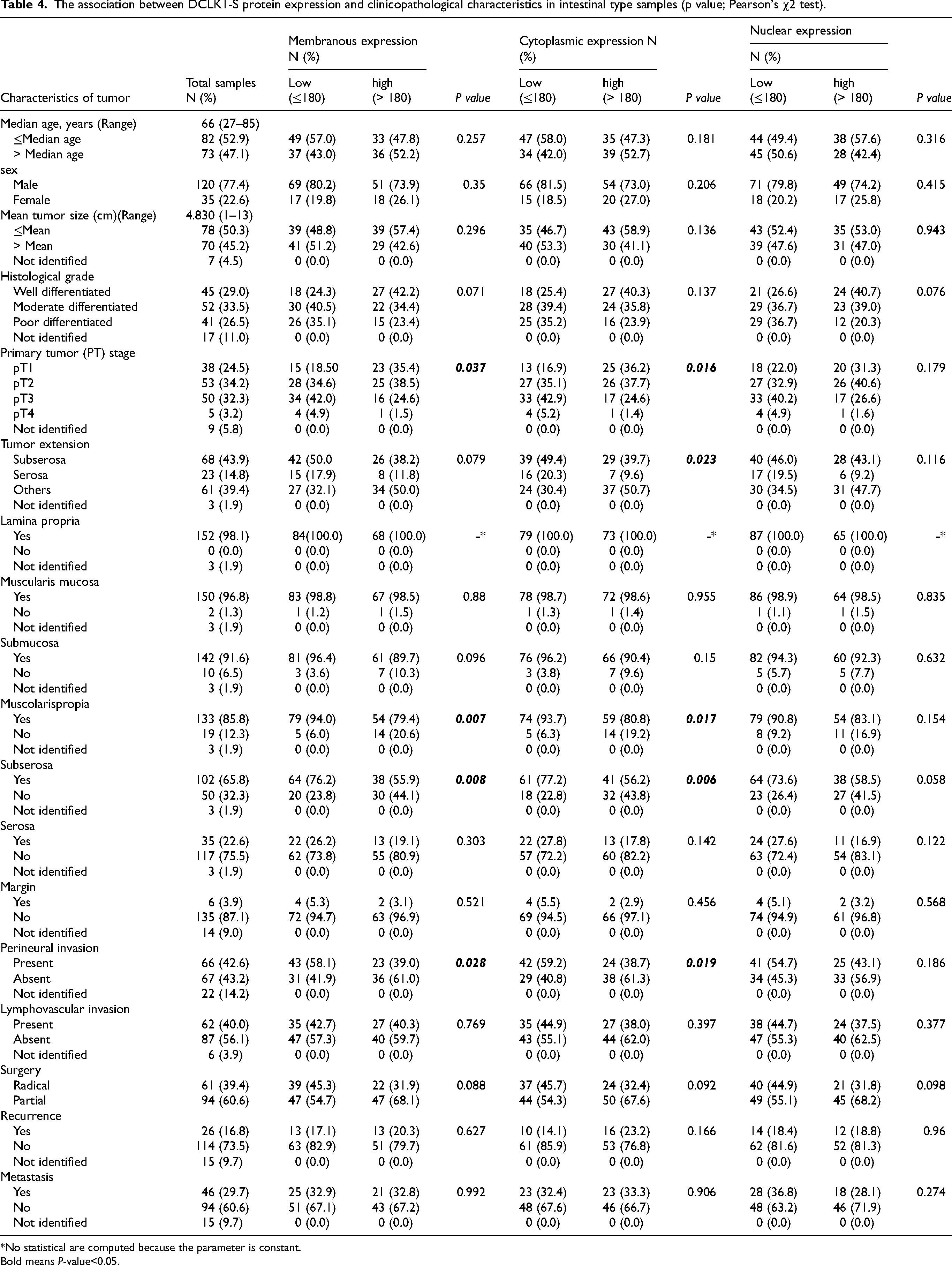
Pearson's χ2 test showed that membranous DCLK1-S expression is statistically associated with PT stage (P = 0.037), muscolarispropia (P = 0.007), subserosa (P = 0.008), and perineural invasion (P = 0.028) (Table 4). In addition, negative correlation was found between these clinicopathological parameters and membranous DCLK1-S expression.
The association between DCLK1-S protein expression and clinicopathological characteristics in intestinal type samples (p value; Pearson's χ2 test).
No statistical are computed because the parameter is constant.
Bold means P-value<0.05.
Based on Pearson's χ2, we also observed a statistically significant association between cytoplasmic expression of DCLK1-S and PT stage (P = 0.016), tumor extension (P = 0.023), muscolarispropia (P = 0.017), subserosa (P = 0.006), and perineural invasion (P = 0.019) (Table 4) with an adverse correlation via Spearman's correlation analysis results.
As demonstrated in Table 4, there was no significant association between nuclear DCLK1-S expression and clinical parameters in the intestinal type cases.
Signet ring cell type
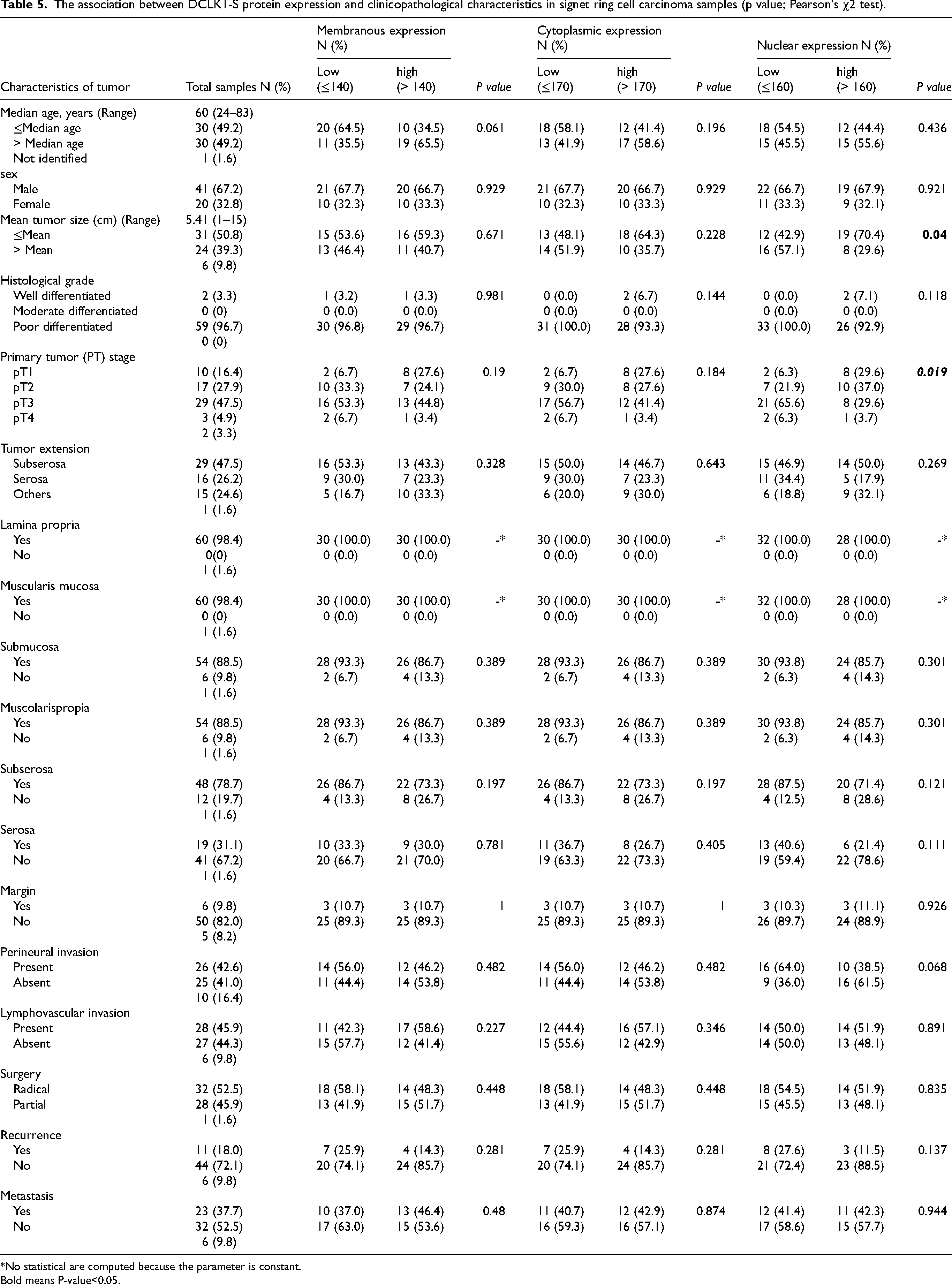
In Signet Ring Cell Type, a significant association was only identified between nuclear DCLK1-S expression and tumor size (P = 0.040) as well as PT stage of patients (P = 0.019) (Table 5). Spearman's correlation analysis results indicated an inverse correlation between these clinicopathological parameters and nuclear DCLK1-S expression.
The association between DCLK1-S protein expression and clinicopathological characteristics in signet ring cell carcinoma samples (p value; Pearson's χ2 test).
No statistical are computed because the parameter is constant.
Bold means P-value<0.05.
However, membranous and cytoplasmic DCLK1-S expressions were not associated with clinicopathological features in signet ring cell type samples.
Prognostic value & survival outcomes of DCLK1-S expression in gastric cancer patients and histological subtypes
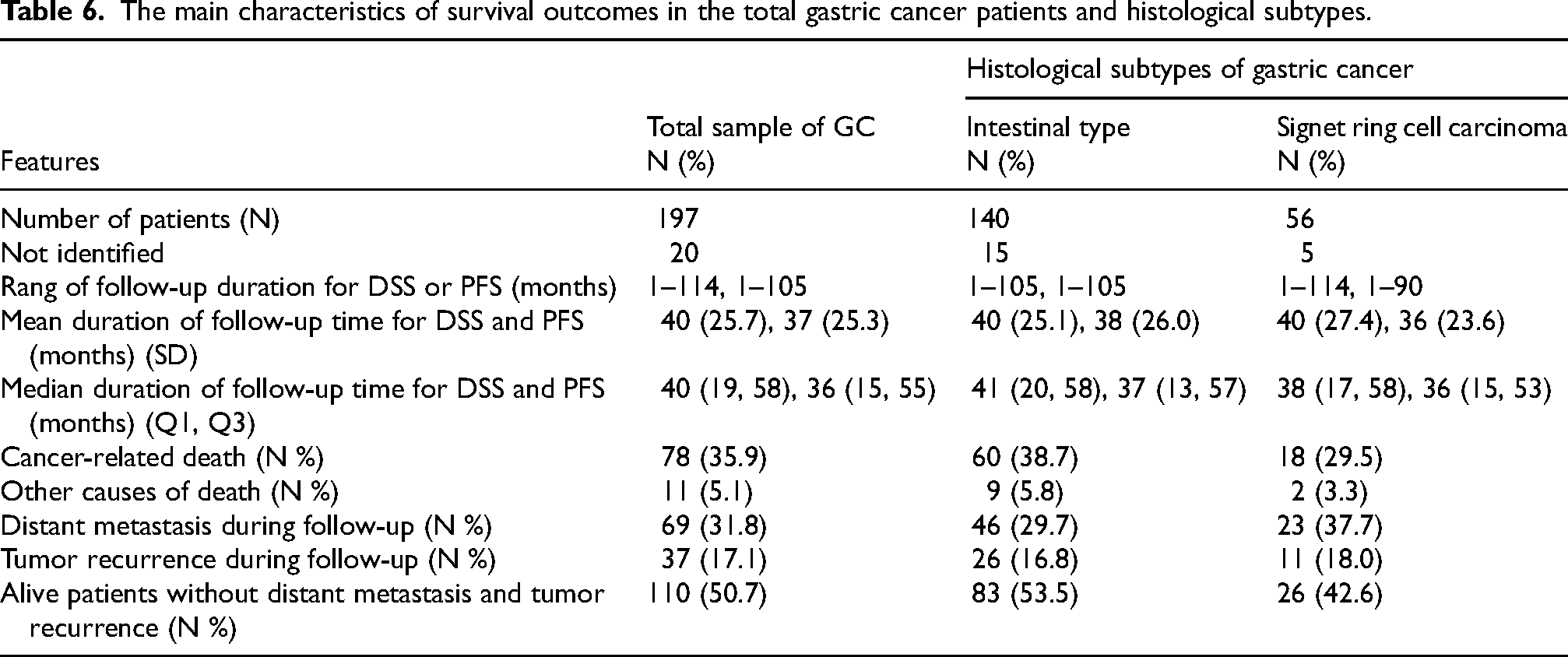
Kaplan–Meier curves (with log-rank test) were applied to analyze the association between DCLK1-S expression and DSS as well as PFS on 197 of 217 GC samples for whom follow-up data was accessible. For DSS, the median and mean follow-up duration was 40 (Q1, Q3; 19, 58) and 40 (SD = 25.7) months, respectively, ranging 1 to 114 months in total GC samples. For PFS, the median and mean duration of the follow-up was 36 (Q1, Q3; 15, 55) and 37 (SD = 25.3) months, ranging 1 to 105 months, respectively. During the follow-up period, 78 (35.9%) patients suffered disease-related death. For tissues stained with DCLK1-S, distant metastasis and tumor recurrence occurred in 69 (31.8%) and 37 (17.1%) patients, respectively. The main characteristics of survival outcomes were described in table 6 in the total GC samples and histological subtypes.
The main characteristics of survival outcomes in the total gastric cancer patients and histological subtypes.
Total gastric cancer samples
Based on the Kaplan–Meier analyses, there were no significant differences in DSS and PFS between cases with high and low membranous and cytoplasmic expression of DCLK1-S (DSS: P = 0.315, P = 0.433; PFS: P = 0.216, P = 0.377, respectively) (Supplementary Figure 2). Further, the Kaplan–Meier curve of nuclear DCLK1-S expression showed that cases with DCLK1-S overexpression have a longer DSS or PFS in comparison with those with low expression of DCLK1-S, however, without a statistically significant association (P = 0.196, P = 0.104, respectively) (Supplementary Figure 2). Univariate and multivariate analyses were also applied to assess the clinical significance of various parameters that might affect DSS in patients with SOC. In total gastric cancer patients, tumor size (P = 0.002) and tumor extension (P = 0.039) affected the DSS of patients in the univariate analysis. However, only tumor size was independent prognostic factor affecting the DSS based on multivariate analysis. Other variables did not show any significant results using the univariate analysis (Supplementary Table 1).
Intestinal type
In the intestinal type specimens, the Kaplan–Meier survival analyses showed no significant association between membranous, cytoplasmic and nuclear expression of DCLK1-S and DSS or PFS (DSS: P = 0.732, P = 0.978, P = 0.552; PFS: P = 0.545, P = 0.921, P = 0.334, respectively) (Figure 7). Additionally, tumor extension (P = 0.030) in intestinal type samples affected the DSS of patients in the univariate analysis. However, multivariate analysis was not performed because only one variable showed meaningful results (Supplementary Table 2).
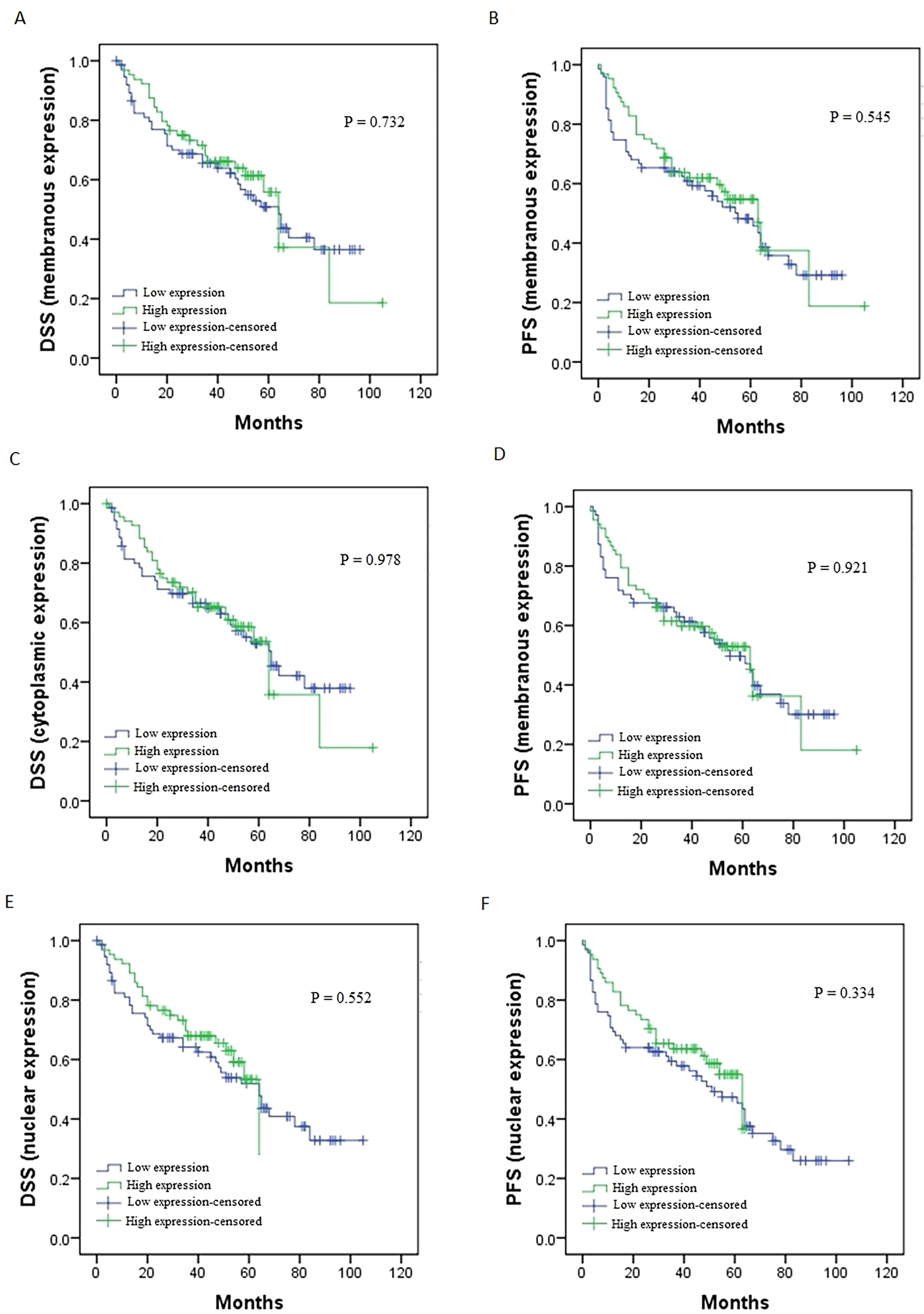
Survival analysis for membranous, cytoplasmic and nuclear DCLK1-S expression in intestinal type of gastric cancer patients. The Kaplan–Meier survival methods indicated no significant differences in disease-specific survival (DSS) and progression-free survival (PFS) between cases with high and low membranous (A, B), cytoplasmic (C, D) and nuclear (E, F) expression of DCLK1-S protein.
Signet ring cell type
In the cases of signet ring cell type, the Kaplan–Meier curves exhibited that cases with high membranous, cytoplasmic and nuclear DCLK1-S expression had a longer DSS or PFS than those with high DCLK1-S expression, however, without a statistically significant association (DSS: P = 0.233, P = 0.179, P = 0.129; PFS: P = 0.188, P = 0.152, P = 0.119, respectively) (Figure 8). Furthermore, the tumor size (P = 0.008) in samples of the signet ring cell carcinoma had an impact on the DSS of patients, as observed in the univariate analysis. However, the multivariate analysis could not be conducted due to the presence of only one variable that yielded significant findings (Supplementary Table 3).
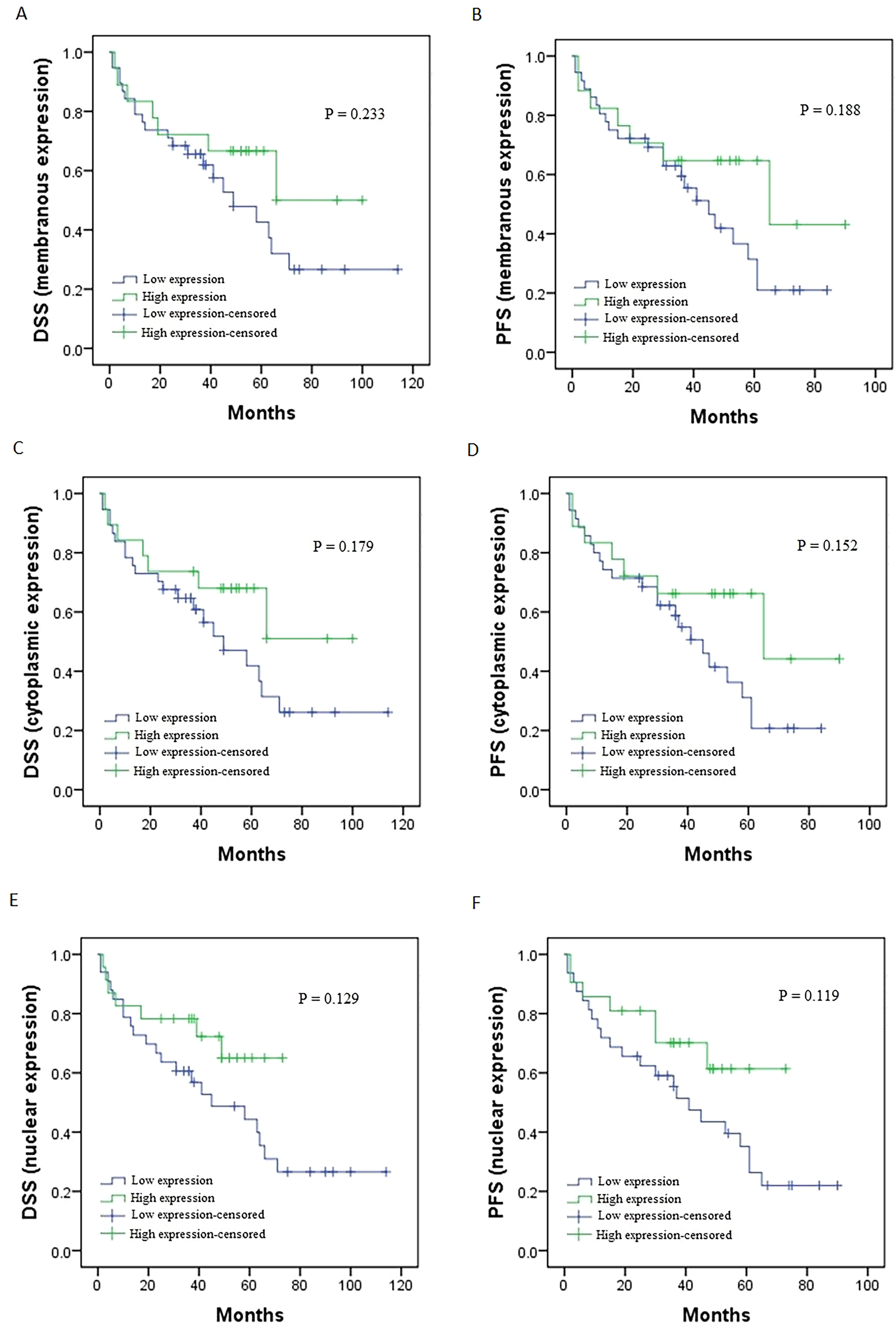
Survival analysis for membranous, cytoplasmic and nuclear DCLK1-S expression in signet ring cell type of gastric cancer patients. The Kaplan–Meier survival methods indicated differences in disease-specific survival (DSS) and progression-free survival (PFS) between cases with high and low membranous (A, B), cytoplasmic (C, D) and nuclear (E, F) expression of DCLK1-S protein, however, without a statistically significant association.
Discussion
Cancer biomarker discovery could be an essential step to understanding gastric cancer biology. In this regard, mAbs, with their specific pattern of reactivity, may be helpful tools for diagnostic methods and research. Evidence accumulated by our research group and others highlight the significance of DCLK1 expression as an independent predictor of prognosis, therapeutic resistance, and recurrence in diverse tumors.8,14,17,34–38 Human DCLK1 has two major isoforms, the long isoform (DCLK1-L) and the short isoform of DCLK1 (DCLK1-S). We have previously studied DCLK1 as a novel CSC marker with different commercial antibodies against DCLK1-L or against both DCLK1-S and -L isoforms.17,24 Since there were no commercially available antibodies specifically targeting DCLK1-S at that time, we initially developed a polyclonal antibody against DCLK1-S. 22 As no particular anti-DCLK1-S mAbs was generated, the current research focused on producing and characterizing mAb against the short isoform of DCLK1. This will provide more opportunities to researchers to acquire further reliable information regarding the DCLK1-S expression pattern.
The present study aimed to investigate expression, localization and clinical significance of DCLK1-S using our produced mAb on a large series of GC tissue samples as a preliminary study to find a potential suitable marker for immunotherapy.32,39,40 Generated clones, even at low antibody concentrations, recognized mouse DCLK1-S peptides well in ELISA, displaying high binding affinity. However, the immunostaining pattern of the 2H3D5 clone was more specific with GC tissue specimens with no non-specific reactivity. In particular, the specificity of 2H3D5 mAb against DCLK1-S was proven through IHC staining of normal testis tissue, cancerous ovarian and skin tissues, and GC sections in which primary antibody was pre-adsorbed with immunizing peptide or replaced with mouse IgG. No positive staining was identified in these specimens at the protein level. These results indicated that 2H3D5 mAb specifically recognizes DCLK1-S in gastric cancer. Therefore, 2H3D5 mAb was selected to investigate its potential for monitoring cancer cells with DCLK1-S expression.
Regarding the specific functions of DCLK1 isoforms, two recent studies have shown that the DCLK1-S isoform plays a function in the progression of colon cancer and ESCC, however, similar findings for DCLK1-L were not acquired in these studies.19,21 In addition, our previous study also showed the critical relationship of DCLK1-S with cancer aggressiveness and poor DSS in CRC. 22 These results emphasize the significance of evaluating DCLK1 isoforms separately in various cancers where their expression and function may differ. Nonetheless, no studies have investigated DCLK1-S importance in gastric carcinoma. For the first time, the current study investigated the expression pattern, clinicopathological importance, and prognostic implications of DCLK1-S through IHC analysis of TMA cores in a large series of GC tissues. The assessment of stained GC tissue sections demonstrated the heterogeneous membranous, cytoplasmic, and nuclear patterns of DCLK1-S expression in cancer cells with a variety of intensities. This result is in line with the findings of Sarkar et al., who reported that in addition to membranous and cytoplasmic DCLK1-S expression, its expression was observed in the nuclei and mitochondria of neoplastic cells by immune-electron-microscopy (IEM) test in colorectal cell lines, unlike the L-isoform with primary accumulation in the plasma membranes and cytosol of the cells.19,41,42 Differential localization of the two isoforms may be due to the lack of microtubule-binding domain in the short isoform. Variations in the biological activities of the DCLK1-L/S isoforms may be rooted in differences in subcellular locations of the two isoforms. 19 In addition, according to the positively stained cells percentage, DCLK1-S expression was considerably higher in the membrane and cytoplasm of cancer cells in GC tissues in comparison with adjacent normal tissues, in accordance with other investigations.21,22
According to the authors’ findings, the lower DCLK1-S expression in the membrane and cytosol of intestinal type samples was associated with increased tumor progression. However, for nuclear expression, such association was not observed. On the other hand, in the signet ring cell type, a significant association was identified between high nuclear expression of DCLK1-S and smaller tumor size as well as lower PT stage of patients. These findings indicate possible different functions of DCLK1-S in different subtypes of gastric carcinoma. Intestinal type and signet ring cell type have different macroscopic appearances and microscopic growth patterns, revealing notable differences in their molecular signaling pathways. Coordinating with this data, our results indicated that tumors with higher DCLK1-S expression, especially in nuclear, had longer DSS or PFS than tumors with low expression of DCLK1-S, although without a statistically significant association in either GC subtype. It should be stressed that this data could be reinforced with an extended follow-up time and more information from a larger cohort.
There is an absence of robust studies on DCLK1-S expression in the majority of cancers, especially gastric carcinoma. Some studies have analyzed the potential of DCLK1-S in colorectal and renal cancers, and their results can be used for the purpose of our comparison. However, there seems to be some controversy in the literature regarding the clinical value of DCLK1-S. In agreement with the findings from the current study, Ge et al. demonstrated a significant inverse relationship between high S-isoform expression and stage, tumor size, and lymph node status in clear cell renal cancer. Additionally, its expression predicted increased survival. 42 By contrast, a study by our research group indicated the association of cytoplasmic DCLK1-S expression with cancer aggressiveness and worse DSS in CRC cancer. 22 In addition, the positive clinical impact of high DCLK1-S expression in the authors’ GC tissue samples is in contrast to the report of a study on ESCC. 21 There are other studies in accordance with our findings; however, these investigations used commercial antibodies against DCLK1 that identify sequence homology epitopes of both isoforms, DCLK1-S/L, or only the DCLK1-L isoform. For example, a study by our group revealed a significant correlation between high DCLK1 expression and early-stage GC tissues. 24 Tao et al. reported that DCLK1 expression has a high tendency to diminish with the progression of pancreatic cancer from early stages to advanced stages.41,43
As the identification of surface markers is crucial for immunotherapy, our objective in this study, therefor, was to examine the cell membranous expression of DCLK1-S on selected GC cell lines (MKN-45 and AGS) using flow cytometry to determine whether cell lines with different invasive potentials display distinct expression patterns of DCLK1-S, comparable to what we observed in gastric cancer tissue using IHC. MKN-45 and AGS cell lines differ in terms of their invasive abilities.44,45 Interestingly, we found that MKN-45 cells with higher invasive potential expressed a lower surface DCLK1-S level than AGS cells with less invasiveness. These findings in accordance with our clinicopathological results, indicating that GC specimens with a higher histological grade and PT stage expressed lower DCLK1-S levels. These findings may strengthen the concept that there is a negative correlation between DCLK1-S level and GC invasiveness.
This protective role may be ascribed in part to the fact that DCLK1 interaction is pivotal for ataxia-telangiectasia mutated (ATM) phosphorylation and activation in the epithelial cells to modulate ATM-mediated DNA damage repair (DDR). 46 Another study provided clear evidence that cells lacking DCLK1 are unable to restore the impaired quality of the tight junctions. 47 Because of the indispensable role of DCLK1-S in the generation of functional and productive DDR and stable tight junctions, the high expression of DCLK1-S can be used as a potential favorable clinical marker in GC patients. In addition, it can be assumed that the contradictory findings with regards to the clinical impact of DCLK1-S may be the result of differences in the organ specificity, cancer type, DCLK1-S-expressing cell type, antibodies used, criteria employed to determine the cutoff for classification of H-score, follow-up time, and sample sizes used in other studies.18,21
Conclusion
This research is the first of its kind to examine the generation and characterization of a mAb against the short isoform of DCLK1. We demonstrated that our generated mAb could properly detect DCLK1-S in IHC in both subtypes of gastric cancer. For both histological subtypes of GC, DCLK1-S overexpression was associated with decreased tumor progression and early-stage disease. Moreover, our findings suggest that DCLK1-S expression, specially nuclear expression, might prove to be a favorable prognostic marker in GC cases if the follow-up time is prolonged and the number of cases is increased. Further studies are needed to decipher the contradictory results obtained from our study and other studies on DCLK1-S expression in GC and other cancer types.
Supplemental Material
sj-docx-1-cbm-10.1177_18758592241301691 - Supplemental material for Clinical significance of “S” isoform of DCLK1 in different gastric cancer subtypes using newly produced monoclonal antibody
Supplemental material, sj-docx-1-cbm-10.1177_18758592241301691 for Clinical significance of “S” isoform of DCLK1 in different gastric cancer subtypes using newly produced monoclonal antibody by Mahdieh Razmi, Ali-Ahmad Bayat, Nafiseh Mortazavi, Elham Kalantari, Leili Saeednejad Zanjani, Sima Saki, Roya Ghods and Zahra Madjd in Cancer Biomarkers
Footnotes
Ethics approval and consent to participate
All procedures performed in this study were approved by the Human Research Ethics Committee and the animal research committee of Iran University of Medical Sciences. Informed consent was obtained from all individual participants included in the study.
Author contributions
INTERPRETATION OR ANALYSIS OF DATA: MR, AB, NM, LS, EK, and SS.
PREPARATION OF THE MANUSCRIPT: MR.
REVISION FOR IMPORTANT INTELLECTUAL CONTENT: ZM and RGh.
SUPERVISION: ZM and RGh.
All authors read and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has received research support from Oncopathology Research Center, Iran University of Medical Sciences (No: 98-4-28-16306).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
