Abstract
Keywords
Introduction
Malignant pleural effusion (MPE) is a common clinical problem worldwide and most cases are secondary to pleural metastasis of lung and breast carcinoma. 1 With the rising incidence of cancer and improved survival rates due to advances in systemic therapy, MPE has become a growing burden on healthcare systems. 2 Despite this, MPE is a neglected area of research with insufficient therapeutic developments.2,3 The current guidelines recommend talc pleurodesis, a technique first described in 1935, as the primary treatment for MPE. 4 However, indwelling pleural catheters, initially developed for patients who were not suitable for pleurodesis, are now increasingly used as first-line treatment due to their ability to provide ambulatory drainage and offer similar symptom relief with a relatively low risk of complications. 5
At present, the basics of current treatment modalities for managing MPE involve either promoting the adherence of pleural layers to prevent the accumulation of fluid or draining the fluid from the pleural cavity via catheters. Both of these modalities have limitations and drawbacks. Currently, there are no available etiologic therapies targeting the formation of MPE. 6 Consequently, there is a profound need for innovative treatments that target the underlying mechanisms of MPE formation, thereby minimizing the need for invasive procedures and hospitalizations. To achive this, it is essential to have a thorough understanding of the pathogenesis of MPE.
While it is well-established that cancer cells can spread to the pleura via lymphatic, hematogenous, or direct invasion routes, the exact mechanisms underlying the production of pleural effusions remain poorly understood. 2 The development of MPE is mainly attributed to impaired lymphatic drainage and excessive plasma leakage through a hyperpermeable pleural vasculature. The pleural microenvironment involves a complex interplay between metastatic cancer cells, resident mesothelial cells, and inflammatory cells. These cells produce a variety of mediators that, in combination with those derived from systemic circulation, induce vascular permeability, plasma extravasation, and subsequent fluid accumulation.7,8
Increasing evidence suggests that intrapleural expression of angiogenic and proinflammatory mediators plays an important etiologic role in the development of MPE. 9 Among these mediators, monocyte chemoattractant protein-1 (MCP-1) is a key chemokine that regulates the migration and infiltration of monocytes/macrophages. MCP-1 is produced by various cell types present in the pleural space, including mesothelial, endothelial and inflammatory cells. 10 Furthermore, there is evidence to suggest that cancer cells in the pleural cavity can also secrete MCP-1, potentially making it a potent inducer of MPE. 9 Experimental studies have shown that MCP-1 plays a role in the formation of benign pleural effusions, and inhibiting MCP-1 activity significantly reduces the formation of inflammatory pleural effusions.11,12 Moreover, the blockade of MCP-1 has been found to be effective against experimental MPE. 13
The aim of this study is to investigate the potential involvement of MCP-1 in the development of MPE in patients with metastatic pleural malignancies.
Materials and methods
Patients
This is a prospective case-control study which was approved by the local ethics commitee of Hacettepe University Medical Faculty (GO 19/638, 11.06.2019). All study subjects provided informed consent. The study included 100 adult patients who had undergone diagnostic or therapeutic thoracenthesis, insertion of pleural catheter, or video-assisted thoracic surgery for pleural pathology between June 30th, 2019 and January 30th, 2021. Patients with inconclusive diagnoses were excluded from the study.
The patients were divided into three groups: Group 1 (MPE, n = 56 patients), Group 2 (benign exudate, n = 27 patients), and Group 3 (transudate, n = 17 patients). Pleural fluid total protein, albumin, lactate dehydrogenase (LDH), glucose, adenosine deaminase (ADA) levels and concurrent peripheral blood protein, albumin, LDH, and glucose levels were recorded. Transudates and exudates were differentiated using Light's Criteria. MPE was diagnosed cytologically and/or histopathologically in the majority of the patients (n = 47, 84%). There were 9 patients (16%) who were diagnosed as MPE based on clinicoradiological findings such as pleural thickening, pleural nodules or masses on computed tomography (CT) of the thorax or high standardized uptake values on positron emission tomography with CT (PET/CT) related to pleural nodules/ masses. The accurate diagnosis of benign exudates was confirmed by clinical, laboratory, radiological, histopathological findings, and follow-up of treatment responses. Patients with inconclusive histopathological and microbiological data were followed up at least one year in accordance with the literature. 14
Estimation of pleural effusion amount
Radiological criteria were used to estimate the amount of pleural effusion. Either a postero-anterior (PA) chest radiograpy or a thorax CT was used depending on availability at the time of pleural effusion sampling. If both images were available, thorax CT was preferred. The amount of pleural effusion was estimated by quartiling the PA chest radiography or measuring the anteroposterior depth on thorax CT. On PA chest eadiography, a pleural effusion at the midclavicular line was considered “small” if it was in the first quartile (<25%); “moderate” if it exceeded the first quartile but less than 50%; and “large” if it exceeded the second quartile (>50%). On CT of thorax, a pleural effusion was quantifed as “small” if the anteroposterior depth was <3 cm; “moderate” if it was 3–10 cm; and “large” if it was >10 cm. 15
Pleural effusion analysis and measurement of MCP-1 levels
The levels of pleural fluid total protein, albumin, glucose, LDH, and ADA were measured using Beckman Coulter analyzers (AU5800, USA) at the time of patient inclusion in the study. Pleural fluid samples (8–10 ml) were collected in serum separator tubes, then centrifuged promptly (up to one hour) at 2350 g for 20 min and stored at −80°C until MCP-1 level measurement. The MCP-1 levels in pleural fluid samples were determined using an ELISA kit (USCN, Wuhan). Freshly prepared seven standards (1000 pg/mL, 500 pg/mL, 250 pg/mL, 125 pg/mL, 62.5 pg/mL, 31.2 pg/mL, 15.6 pg/mL) were used for standard curve. The MCP-1 concentration was measured by determining the absorbance at a wavelength of 450 nm. The assay had a detection range of 15.6–1000 pg/mL, with a minimum detectable dose of typically less than 6.4 pg/mL, intra-assay CV of <10%, and inter-assay CV of <12%.
Statistical analysis
The statistical analysis was performed using the SPSS 25.0 software for Windows. Numeric data was summarized using mean ± standard deviation or median (25–75th percentile), depending on the distribution of the data. Categorical data was presented as frequencies and percentages. To assess the differences in numerical variables between two independent groups, one-way ANOVA was used for variables with a normal distribution and homogeneous group variances. If group variances were heterogeneous, Welch ANOVA was used instead. The Kruskal-Wallis test was used when the distribution of the variables was not normal. Pairwise comparisons were conducted using Dunn's test. The relationship between categorical variables was assessed using the chi-square test. The Spearman correlation coefficient was used to evaluate the correlation between numerical variables. A p-value less than 0.05 was considered statistically significant.
Results
The study included 100 patients (61 male/ 39 female) with a mean age of 65.4 ± 14.2 years. The patients were divided into three groups: Group 1 (MPE), Group 2 (Benign exudates), and Group 3 (Transudates). The characteristics of the patients in the study groups are summarized in Table 1. Age and gender were similar among the groups. There were significant differences in pleural effusion side, appearance, and volume among the study groups. While large size effusions were more common in Group 1, serous and bilateral effusions were more common in Group 3. The most prevalent diagnoses were lung cancer (n = 25, 44.6%) in Group 1, parapneumonic effusion (PPE) (n = 14, 51.9%) in group 2, and congestive hearth failure (CHF) (n = 12, 70.6%) in Group 3.
The characteristics of study patients.
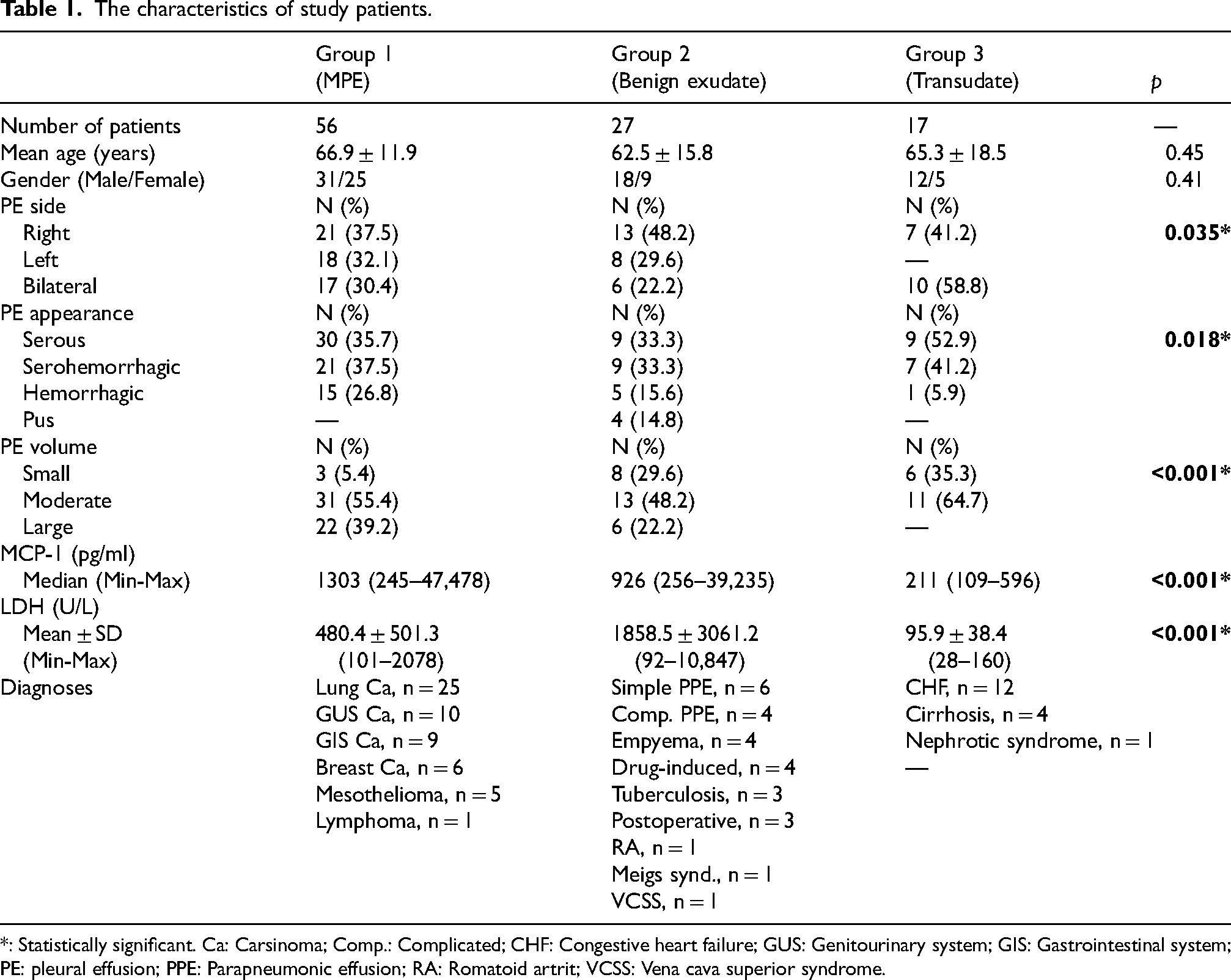
*: Statistically significant. Ca: Carsinoma; Comp.: Complicated; CHF: Congestive heart failure; GUS: Genitourinary system; GIS: Gastrointestinal system; PE: pleural effusion; PPE: Parapneumonic effusion; RA: Romatoid artrit; VCSS: Vena cava superior syndrome.
Median and minimum-maximum values of MCP-1 levels were measured as 1303 (245–47,478) pg/ml in Group 1; 926 (256–39,235) pg/ml in Group 2; and 211 (109–596) pg/ml in Group 3 patients. Median MCP-1 levels were significantly different among study groups (p < 0.001). MCP-1 levels were significantly higher in Group 1 (p < 0.001) and Group 2 (p = 0.001) compared to Group 3 but not significantly different between Group 1 and Group 2. Box-plot graphic of MCP-1 levels in the three study groups are shown in Figure 1.
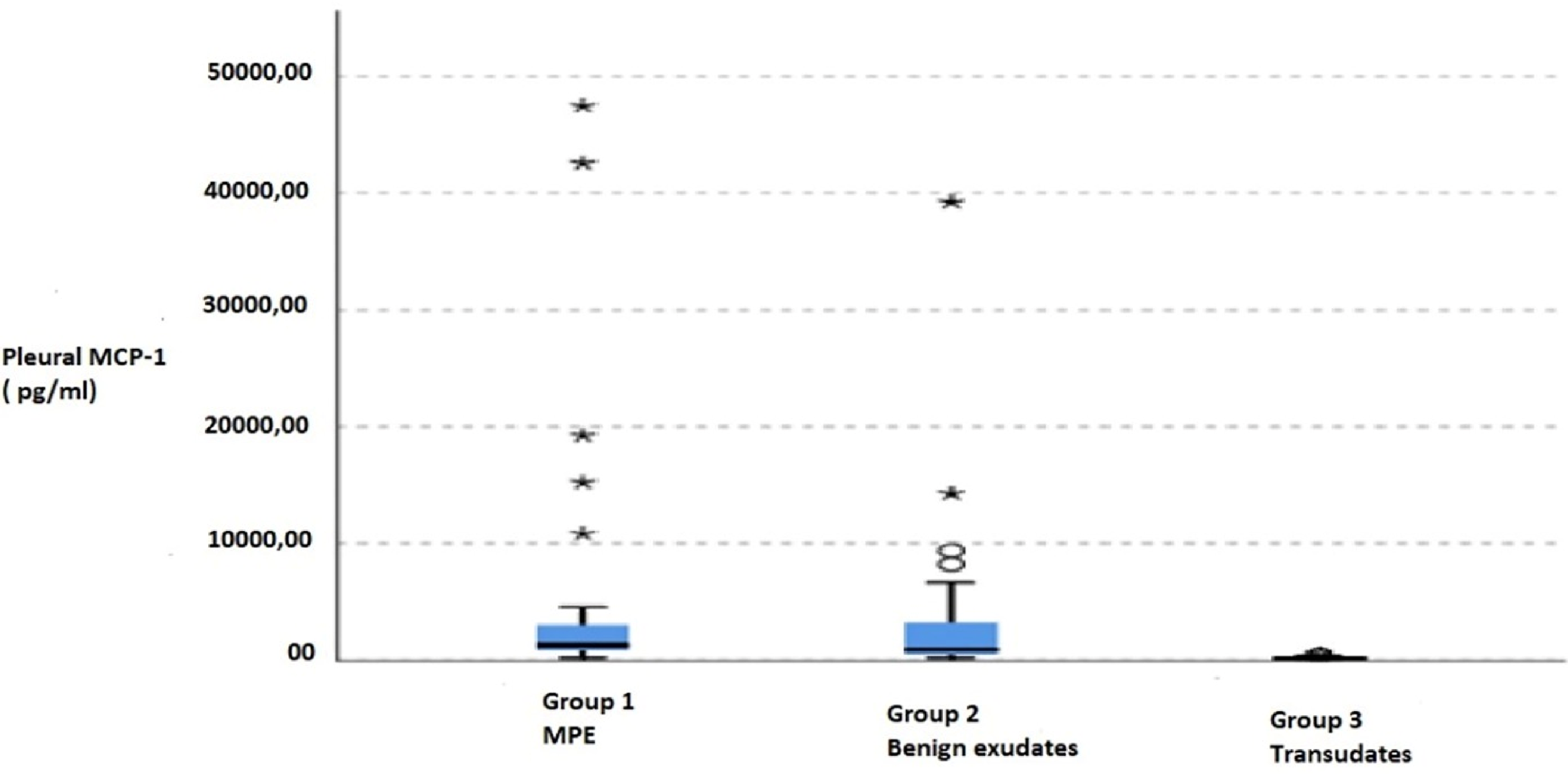
Box-plot graphic of MCP-1 levels. MCP-1 levels were significantly higher in Group 1 (p < 0.001) and Group 2 (p = 0.001) compared to Group 3. MCP-1 levels were not significantly different between Group 1 and Group 2.
There was a significant correlation between pleural MCP-1 levels and pleural LDH levels. This correlation was significant across all study patients (r = 0.61; p < 0.001), and also when patients from Group 1 and 2 were combined (r = 0.38; p = 0.001), as demonstrated in Figure 2(a) and (b), respectively.
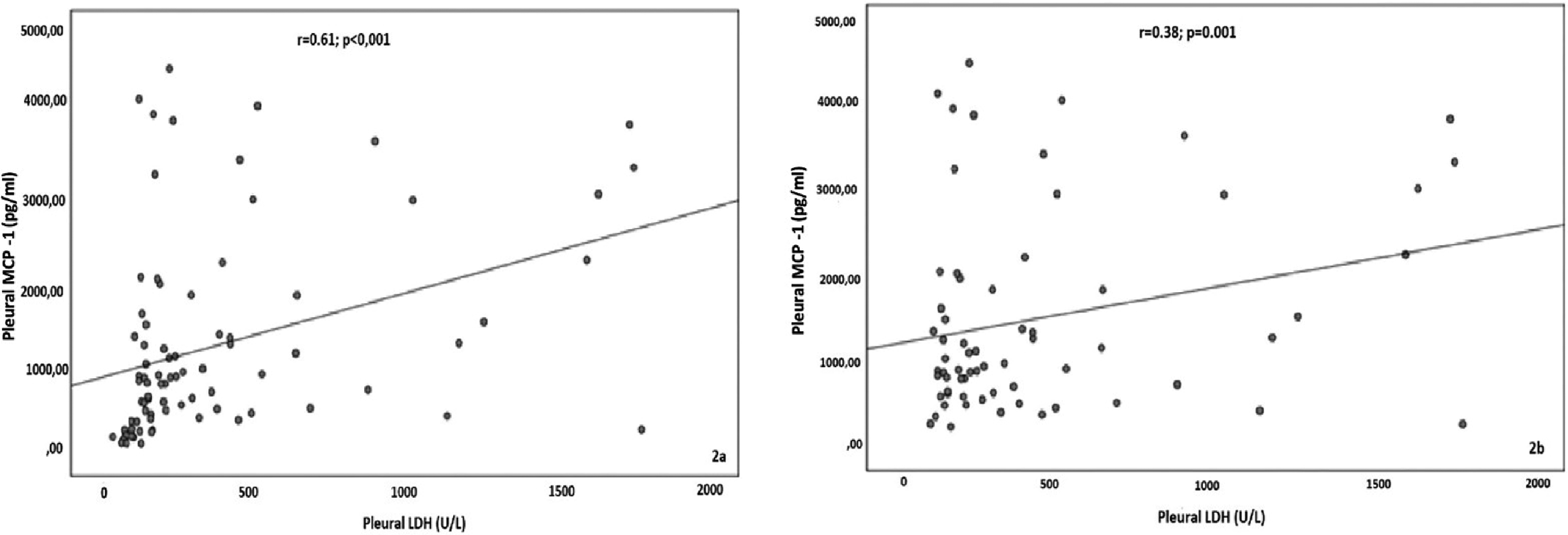
The correlation between MCP-1 and pleural LDH levels in all study patients (Figure 2(a)) and in patients of group 1 (MPE) and 2 (benign exudate) evaluated together (Figure 2(b)) (Spearman's rank correlation).
Table 2 displays the median MCP-1 levels based on pleural effusion size, while Figure 3 presents the box-plot graphics of the same data. Median MCP-1 levels were significantly different among patients who had small-moderate-large size pleural effusions when Group 1 and 2 patients (n = 83) were evaluated together (p = 0.007). As shown in Figure 3(a), MCP-1 levels increased as the pleural volume size increased. However, when only Group 1 (MPE) patients were considered, although MCP-1 levels also increased with the pleural effusion volume (Figure 3(b)), the median MCP-1 levels were not statisticaly different (p = 0.9) among patients who had small-moderate-large size pleural effusions.
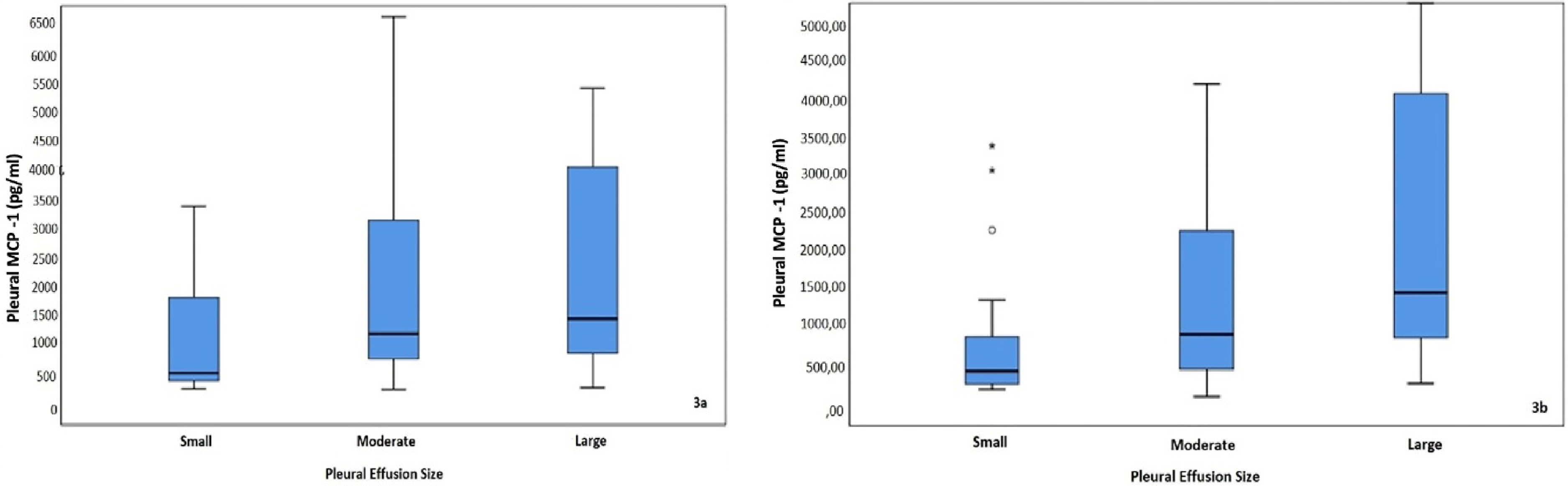
Distribution of MCP-1 levels according to pleural effusion size in Group 1 (MPE) and Group 2 (Benign exudates) patients evaluated together (Figure 3(a)) and in Group 1 (MPE) patients (Figure 3(b)).
Median pleural MCP-1 levels based on pleural effusion size.

*Statistically significant, Group 1: MPE patients, Group 2: Benign exudates.
Discussion
The present study aimed to investigate the potential role of MCP-1 in the development of MPE, and it yielded three key findings. First, it was found that MCP-1 levels were comparable between MPE and benign exudates, but significantly higher in comparison to transudates. Secondly, there was a positive correlation between MCP-1 levels and pleural LDH levels. Finally, MCP-1 levels increased in parallel with the amount of pleural fluid, suggesting a potential link between MCP-1 and the volume of pleural effusions.
Despite being a common clinical problem, the underlying mechanisms of MPE formation are still not fully understood. MPE is defined as the accumulation of pleural fluid, which is believed to result from direct infiltration of cancer cells into the pleural space. Although it is widely accepted that cancer cells can reach the pleura via lymphatics, hematogenous dissemination, or direct invasion, the precise mechanisms underlying the generation of pleural effusions by cancer cells remain poorly understood. 2
The pathogenesis of MPE is complex and involves various factors. Considerable effort had been put to understand the molecular mechanisms of tumor driven pleural fluid accumulation. Multiple mechanisms have been implicated, including the formation of new blood vessels, increased permeability of existing and newly formed blood vessels, and an inflammatory host response triggered by tumor cells in the pleural space.6–9 It is thought that there is a complex interplay between cancer cells, mesothelial cells and inflammatory cells in the pleural microenvironment. 7 These cells produce or release various angiogenic or proinflammatory mediators that induce vascular permeability and plasma extravasation. 9 Among these mediators, vascular endothelial growth factor (VEGF) has been shown to be a potent inducer of MPE, and anti-VEGF strategies have been effective in inhibiting fluid formation in preclinical studies.16–18 Zoledronic acid, a potent bisphosphonate with anti-angiogenic, anti-inflammatory, and anti-cancer activities, has also shown promise in experimental MPE. 19 However, clinical trials using these interventions have not been successful in humans. 20
MCP-1, a chemokine known for its ability to attract and activate mononuclear cells, has been linked to both inflammation and cancer development. 10 Studies suggest that MCP-1 plays a crucial role in the formation of exudative pleural effusios by inducing endothelial permeability, angiogenesis, and recruitment of inflammatory mediators.11,12 In addition, MCP-1 can stimulate the secretion of growth and survival factors by activated mononuclear cells, thereby enhancing tumor progression through the provision of angiogenic factors such as VEGF. 21 MCP-1 binds to a receptor called chemokine receptor 2 (CCR2) which is expressed on the surface of endothelial cells. This binding can promote angiogenesis and increase pleural vascular permeability, leading to the formation of pleural effusions. In the case of MPE, MCP-1 may be secreted by tumor cells themselves, or it may be produced by nearby stromal and/or endothelial cells that are stimulated by tumor cells.21,22 Elevated levels of MCP-1 have been found in several types of cancer that can lead to pleural and peritoneal effusions.22,23 In a study conducted by Stathopoulos et al., the pivotal role of MCP-1 in MPE development was demonstrated in a mouse Lewis lung cancer model. It was found that cancer cells in the pleural space secrete MCP-1, and inhibition of MCP-1 led to a reduction in effusion formation. 24 Additionally, MCP-1 has been found to be elevated in several pleural diseases such as MPE, PPE, empyema, and tuberculosis. In a study including 63 patients with MPE, empyema, PPE or tuberculosis, MCP-1 levels were found to be highest in patients with MPE. Moreover, patients with cytology positive MPE had higher levels of MCP-1 than those with cytology negative MPEs, and MCP-1 levels were correlated with the absolute number of monocytes present in the effusion. 25 In another study, 79 treatment-naive primary lung adenocarcinoma with MPE were studied alongside 23 patients with tuberculous pleurisy. MCP-1 levels were significantly higher in the MPE group. A MCP-1 level greater than 3187 pg/mL indicated failure to control MPE and MCP-1 was confirmed to be an independent poor prognostic factor for progression free survival. 22
In the present study, MCP-1 levels were similarly high in MPE and benign exudates compared to transudates. The observed high levels of MCP-1 in both MPE and benign exudate groups can be explained by the inflammatory nature of these conditions. MPE and benign exudates are inflammatory effusions and the development of pleural effusion is often associated with an increased number of inflammatory cells in the pleural space. While neutrophils are the predominant cell type in PPEs, mononuclear cells are usually increased in MPEs and benign exudates. 26 It has been shown that inflammatory markers are highly expressed in MPEs, but they are not usefull in the differential diagnosis of exudative effusions. 27 The MCP-1 levels in our study showed a wide range of variability, which was attributed to various factors such as different tumor types, the heterogeneity of tumor cells within tumors, and varying disease timelines in the MPE group, and different etiologies in benign exudates. Pleural LDH, a well known indicator of pleural inflammation, was positively correlated with MCP-1 levels both in all study patients, as well as in patients from Group 1 (MPE) and Group 2 (Benign exudates) who were evaluated together. These study findings were consistent with the results of a recent study which evaluated the longitudinal changes in MPE by repeated sampling from indwelling pleural cathaters. The study showed that pleural fluid pH decreased over time, accompanied by a corresponding rise in LDH. The rising MCP-1 levels observed over time supported the pathobiological role of MCP-1 in MPE. 28
The ideal therapy for MPE should aim to improve dyspnea, increase quality of life, be well-tolerated, minimally invasive, cost-effective, and require minimal hospitalization. 29 In the last two decades, MPE management and research have made remarkable progress. However, current management strategies primarily focus on mechanical methods of drainage and pleural space sealing, In the future, significant efforts should be directed at more sophisticated biomarker analysis and validation and subsequent targeting of these with intrapleural immunological agents to “turn the tap off”. 30
Experimental models of MPE have shed light on the pathogenesis of pleural fluid formation and have also been used to test novel biological agents as potential treatment options. 9 It has been shown that blocking MCP-1 or its receptor CCR2 can lead to a significant decrease in pleural effusion volume in a mouse model of carrageenan induced pleurisy, which is a well-validated murine model of benign pleurisy. 11 In an experimental study which aimed to determine the mechanism of tissue plasminogen activator induced exudative pleural effusion, pleural fluid MCP-1 levels were significantly higher than the corresponding sera; and pleural fluid MCP-1 levels were significantly correlated with the effusion volume. Mice treated with anti-MCP-1 antibody or MCP-1 receptor antagonist demonstrated a significant decrease in pleural effusion volume. 12 A separate study comfirmed the effectiveness of two available CCL2 and CCL12 antagonists, either alone or in combination, in limiting MPE induced by murine and human adenocarcinoma. The treatment resulted in a significant reduction in inflammation, vascular formation, and permeability, which are essential processes for pleural fluid formation. 13 The present study is a clinical study that showed, for the first time, that MCP-1 levels increased as the amount of pleural fluid increased. This suggests that MCP-1 could be a promising target for the future treatment of MPE.
This study is a prospective clinical study with a relatively sufficient number of patients compared to current literature. However, the heterogeneity of tumors in the MPE group made it challenging to conduct subgroup analysis for certain tumor subtypes. Additionally, sampling of pleural effusions was not restricted to a specific time point during the disease course, they were colleceted at any time. Despite these limitations, demonstration of an increase in MCP-1 levels with the increase in pleural effusion volume is an important finding which supports the hypothesis that MCP-1 could be a promising target for preventing fluid formation. Blocking MCP-1 can turn the the tap off.
Conclusion
The current management options of MPE have remained unchanged for years. Hence, there is a need for novel therapies targeting molecules that play a role in fluid formation and can “turn the tap off”. One such molecule can be the proinflammatroty cytokine MCP-1. This study revealed that MCP-1 levels were similarly high in both MPE and benign exudates, suggesting that inflammation accompanying the tumor could contribute to the formation of pleural effusion. The correlation between pleural fluid MCP-1 and LDH levels supports this finding. Moreover, this clinical study, for the first time, demonstrated that MCP-1 levels significantly increased with the amount of pleural fluid. Therefore, it provides important clinical data supporting preclinical studies indicating that MCP-1 may be a promising target for future MPE therapy.
Highlights
Malignant pleural effusion (MPE) is a common clinical problem, and current avaliable management options have remained unchanged for years. Therefore, there is a need for novel therapies that can target the underlying mechanism of fluid formation in MPE. This study investigated the possible role of MCP-1 in the development of MPE. The results showed that MCP-1 levels were elevated in both MPE and benign exudates patients and were positively correlated with pleural LDH levels. The MCP-1 levels were observed to increase as the volume of pleural fluid increased. The findings of this study are significant in providing crucial clinical evidence to support preclinical studies indicating that MCP-1 could be a promising therapeutic target in the future treatment of MPE.
Footnotes
List of abbreviations
Acknowledgements
This study was supported by Hacettepe University Scientific Research Unit. ELISA kits used for the measurement of MCP-1 were purchased with the support of Hacettepe University Scientific Research Unit (Project ID: TTU-2020- 18474). The summary of this residency thesis was uploaded to the Hacettepe University residency thesis collection (openaccess.hacettepe.edu.tr:8080) The abstract of this study was presented in the annual congress of European Respiratory Society as an oral presentation in 2021.
Author contributions
Conception: DK, ZGD; Interpretation or analysis of data: SK, FT; Preparation of the manuscript: FT, RB, BA; Revision for important intellectual content: DA, ED, SO; Supervision: DK, ZGD; Writing content in the manuscript (FT, DK, ZGD, SK); Literature search (FT, RB, BA); Statistical analysis (SK); Collection of pleural fluid samples (FT, RB, BA, ED, DA); Evaluation of patologic materials of the study subjects (SO); Analysis of pleural effusions (ZGD); Critical revision and administration during manuscript writing (DA, ED, SO).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
