Abstract
Purpose:
The risk of implant-associated infection (IAI) is occasionally cited as an indication for routine removal of orthopaedic implants in children, but evidence is lacking. This study aimed at exploring the frequency of microbial colonization of paediatric orthopaedic implants by sonication, a gold standard for diagnosing IAI.
Methods:
Data of all patients aged <18 years at index implantation who underwent implant removal over a 34-month period at a single institution were retrospectively reviewed. Sonication culture results were classified according to microbial growth as negative (no/non-significant growth) or positive (significant growth/colonization). Descriptive statistics were performed, correlations were analysed via crosstabs and univariate ANOVA (
Results:
One hundred and twenty-nine sonicated devices from 63 patients were included. Mean patient age at implantation was 9.8 years (standard deviation (SD) 3.4; range 3.0–15.6), mean implant in situ time was 2.2 years (SD 1.5; range 0.6–8.8). In all, 63 tension band plates, 35 locking screw plates, 26 cannulated screws, 3 intramedullary nails and 2 K-wires were evaluated. In total, 128/129 (99%) of implant sites were asymptomatic. Sonication fluid cultures of 24/128 (19%) implants from 21 asymptomatic patients showed non-significant growth of pathogens mainly related to the human skin microbiome, suggesting contamination.
Conclusions:
Sonication of implants removed from asymptomatic implant sites showed no significant microbial growth. Thus, the risk of IAI should be irrelevant for scheduling implant removal in children without signs and symptoms of infection.
Significance of study:
This study investigates for the first time the role of sonication in non-spinal paediatric orthopaedics and provides important insights into the question of routine implant removal in children, providing a foundation for future research.
Keywords
Introduction
Implant removal ranks among the most frequently performed orthopaedic procedures worldwide, although its value to the patient is quite unclear to date.1–7 As any surgical procedure, implant removal bears risks.5–10 Available literature implicates complication rates ranging from 0% to >30% in adults, but largely lacks respective evidence for the paediatric population.1,5–16 However, in children, it is particularly common to remove orthopaedic implants upon attainment of their therapeutic goals, even if the hardware impairs neither growth nor the patient’s quality of life.7,8,14–21 Aims of implant removal include reduction of implant-associated discomfort and pain, regain of function and load-bearing capacity, prevention of implant migration, implant failure, deformity induction and stress-shielding effects causing bone loss, avoidance of long-term metal exposure, as well as prevention and/or therapy of implant-associated infection (IAI).18–20 Apart from deformity overcorrection following guided growth procedures, evidence supporting these indications in the paediatric population is scarce.17–19 In fact, data validating the approach of routine removal of asymptomatic paediatric orthopaedic implants to decrease IAI are non-existent.
IAI can occur up to decades after implantation of the hardware, originating from microorganisms colonizing the implant surfaces and producing biofilms.22,23 Sonication uses ultrasound to disintegrate biofilm from explanted implants.24,25 Cultures of sonication fluid have been proven to be more sensitive and more specific diagnostic tools to detect microbial colonization of implants and IAI than conventional peri-implant tissue cultures in adult orthopaedics.24–27 Thus, it seems logical to implement sonication in paediatric orthopaedics. 28
This retrospective evaluation was carried out to gain knowledge concerning microbial colonization of paediatric orthopaedic implants. It aimed at contributing to answering the question of whether routine removal of such implants should be based on an anticipated elevated risk of IAI.
Materials and methods
Study design and ethical approval
This retrospective cohort study was conducted in a tertiary healthcare centre. It was approved by the institutional ethical committee (No. EA2/255/21), and the need for informed consent was waived as the data utilized in this retrospective study have been de-identified. This study was performed in accordance with the most recent iteration of the Declaration of Helsinki.
Study population
Consecutive patients undergoing implant removal at our department between January 2019 and November 2021, who had been younger than 18 years of age at index implantation, were included in this cohort. Patients were identified via the institutional database. Data were extracted from the patient documentation system, including demographics, comorbidities, age at index implantation and implant removal, indication for index implantation, implant status, site and in-situ time of the index implant(s), complications during and following primary and removal surgery, clinical (e.g. local/systemic signs and symptoms of infection) and, if available, paraclinical (white blood cell count (WBC) and C-reactive protein (CRP)) features at presentation for implant removal, as well as microbiological and histopathological findings.
Definitions
Based on sonication fluid cultures, implants were classified according to microbial growth as negative (no growth or non-significant growth) or positive (significant growth). Sonication results were considered positive for significant growth if ≥50 colony-forming units (CFU)/ml of microbiota of the human skin (e.g. coagulase-negative staphylococci,
Similar to the approach used in several recent studies,24,29–33 IAI was diagnosed when at least one of the following criteria was fulfilled: (i) macroscopic purulence or presence of a sinus tract, (ii) isolation of a highly virulent organism from at least one sample OR isolation of the identical organism in two or more samples in case of low-virulence organisms (coagulase-negative staphylococci,
Standard operating procedures
Indication for implant removal
In our paediatric orthopaedic department, elective implant removal is performed routinely in growing individuals once the implant has served its purpose. Non-elective implant removal is performed as soon as deformity correction is accomplished by guided growth, and in cases of implant-associated complications, such as peri-implant infections, fractures, discomfort or implant loosening.
Perioperative routine
Implant removal and index implantation were performed under sterile conditions in the operating room with the patient under general anaesthesia. At index implantation and at implant removal, a single shot of cefuroxime (10 mg/kg bodyweight) was administered in all patients within 60 min prior to incision, and the skin had been disinfected with alcoholic povidone-iodine solution at least three times before incision. The implants were removed from the bone and immediately placed into a lidded sterile plastic container using sterile surgical instruments. The containers were closed airtight immediately. Implants from different sites of the same patient were put into separate containers and sonicated separately. If the implants consisted of different components (e.g. plate-screw constructions for osteosynthesis or epiphyseodesis), all removed components were considered a single implant and therefore placed in the same container and sonicated as a whole.
Sonication
Sonication of explanted orthopaedic devices has been a standard procedure in our tertiary care paediatric orthopaedic department since January 2019.
The retrieved implants are sent for sonication and processed within 6 h of removal, as previously described. 24 In brief, after adding normal saline covering most of the implant, the sonication box is vortexed for 30 s, sonicated for 1 min at 40 kHz (BactoSonic, Bandelin electronic, Berlin, Germany) and again vortexed for 30 s. The resulting sonication fluid is plated in aliquots of 0.1 ml onto aerobic and anaerobic sheep blood agar plates, and 1 ml is inoculated into thioglycolate broth. Cultures are incubated at 37°C for 14 days and inspected daily for microbial growth. Microorganisms on plates are enumerated as the number of CFU/ml of sonication fluid. The colonies of each microorganism morphology are identified by standard microbiological methods using the automated system VITEK 2 (bioMérieux, Marcy L’Etoile, France). Susceptibility testing is performed using the gradient-strip test (E-test) by the hospital microbiology laboratory.
Statistical analysis
Data were analysed via descriptive statistics (IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY, USA: IBM Corp.). Arithmetic mean, standard deviation (SD) and range were calculated for continuous values. Crosstabs were produced to look for correlations. The correlation between a metric and a nominal variable was performed via univariate ANOVA. A two-sided
Results
Patient characteristics
Patient demographics and comorbidities, indications for and types of primary surgery, that is, index implantation, are summarized in Table 1. This study includes 63 patients, 28 females and 35 males, who underwent implant removal at a mean age of 12.0 years (SD 3.6; range 4.8–19.1). Forty-five of these children had relevant comorbidities, including immunocompromising conditions in 23 cases (37% of the cohort).
Cohort characteristics: Patient demographics, comorbidities, relevant paediatric orthopaedic history and preoperative CRP.

CRP: C-reactive protein.
Neither intra- nor perioperative adverse events complicated primary surgery and/or implant removal in any patient. Particularly, no cases of wound infection, delayed or secondary wound and/or bone healing were recorded following primary surgery.
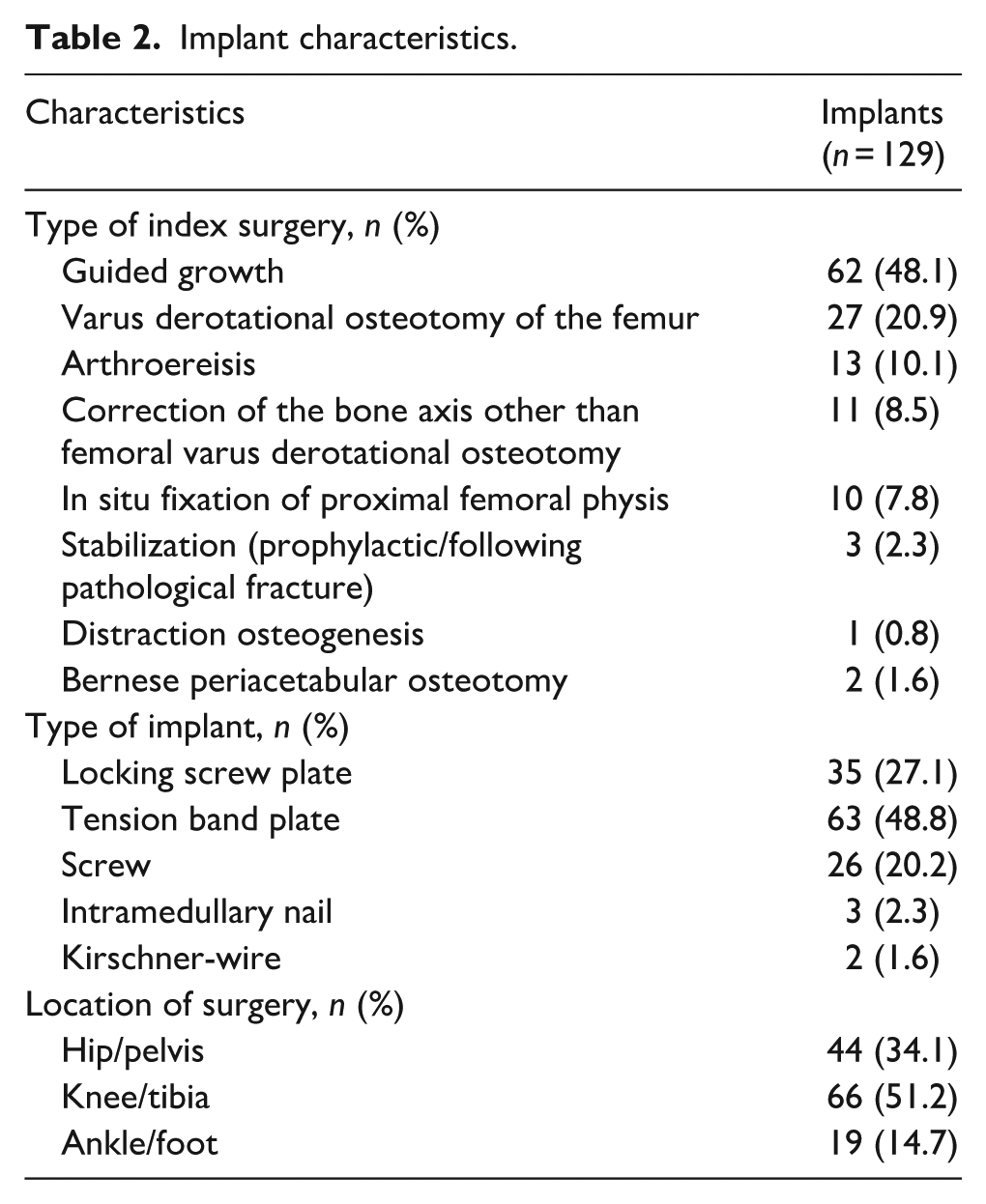
Implant characteristics
A total of 129 implants were analysed, including 63 tension band plates, 35 locking screw plates, 26 cannulated screws, 3 intramedullary nails and 2 Kirschner-wires retrieved from the knee/tibia region (
Due to the primary surgical indications, many children had been operated on more than one surgical site, generally in one session. One patient had five implant sites, 9 patients had four, 2 patients three and 31 patients two implant sites, and 20 patients had a single implant site. All but one patient had been asymptomatic at all implant sites before implant removal.
Infection diagnosis
According to the above-mentioned criteria, IAI was diagnosed in one patient at one implant site. The affected 13-year-old boy had a crouch gait associated with bilateral spastic cerebral palsy (Gross Motor Function Classification System level IV). Due to the acute onset of local redness, swelling and pain at one of his four implants and seven surgical sites, respectively, he presented 11 months after uneventful single-event multilevel surgery (bilateral proximal femoral varus derotational osteotomy, distal femur extension osteotomy and patellar tendon advancement, and left-sided tenotomy of the hamstrings). The locking screw plate from the only symptomatic site was removed in a timely manner in a non-elective setting. In this patient, the preoperative CRP level was 16.8 mg/l, white blood cell count was within physiological range. Blood cultures taken at presentation, cultures of intraoperatively sampled peri-implant fluid and of one swab taken from the subcutaneous fluid collection showed no microbial growth. Another swab taken at the bone-implant interface following implant removal revealed significant growth of
The following results refer to the 62 asymptomatic patients:
Sonication results
Sonication revealed bacterial growth on 24/128 devices (19%) from 21 asymptomatic patients (Table 2). In all of these cases, non-significant growth suggests contamination. The detected pathogens were mainly related to the human skin microbiome and are listed in Table 3.
Implant characteristics.
Sonication findings.
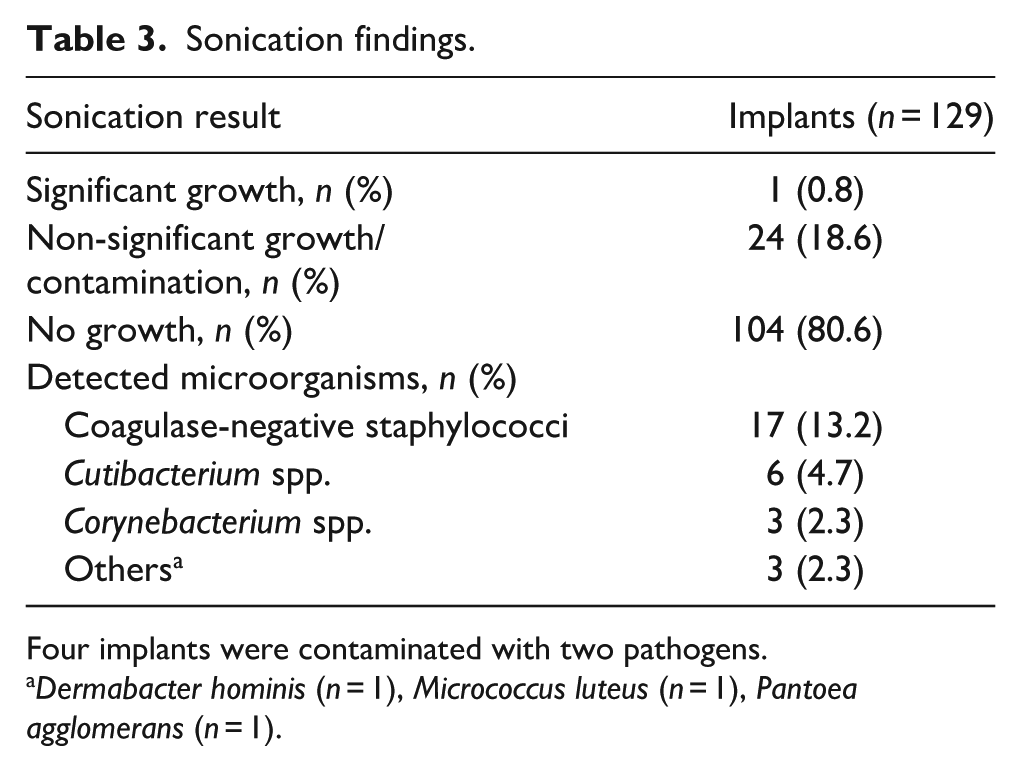
Four implants were contaminated with two pathogens.
Additional microbiological work-up
Cultures of peri-implant tissue/fluid or swab samples were analysed in 23 asymptomatic patients, and significant growth was detected in none. Non-significant growth of coagulase-negative staphylococci was detected in 6 implant sites of 5 patients, in all of which it was classified as contamination by infectious diseases specialists.
Non-microbiological work-up
Histopathological analyses of peri-implant tissue samples taken from 48 implantation sites of 26 asymptomatic patients revealed signs of acute low-grade inflammation at 3 implant sites of 2 patients, and signs of chronic low-grade inflammation at 11 implant sites of 9 patients (i.e. any inflammation in histopathology in 42% of patients). In none of these cases, histopathological findings were consistent with the applied definition of IAI. Microbiological analyses (sonication of 14/14 explanted implants; swabs taken at 13/14 implant sites) of the sites found to be inflamed in histopathology showed no significant microbial growth. No correlation was found between histopathological signs of inflammation and the implant’s in situ time (patient-wise
White blood cell count
WBCs were determined in 48 asymptomatic patients, and no elevation was detected.
C-reactive protein
CRP values were available from 51 asymptomatic patients (106 surgical sites) and were significantly elevated (i.e. >10 mg/l) in one of them (2%) with one implantation site.
Discussion
Despite paucity of valid respective data, a possible risk of IAI has occasionally been referred to as an indication for implant removal in asymptomatic paediatric patients. In this context, it has been repeatedly hypothesized but not yet demonstrated that IAI may arise from a previously clinically silent microbial colonization of the implant in certain scenarios, such as new onset or aggravation of immunodeficiency. 28 Therefore, the recognition of the incidence of asymptomatic significant microbial colonization of paediatric orthopaedic implants may be crucial to guide decision-making as to whether and when to schedule children and adolescents without signs and symptoms suggestive of infection for implant removal.
Sonication may be key in determining that incidence, as it has not only been shown to significantly increase sensitivity and specificity in detecting IAI but has also revealed unanticipated rates of clinically silent colonization of a multitude of implants used in various medical fields such as plastic surgery, interventional cardiology and neurosurgery.24-27,34–36 Still, to the best of our knowledge, only one study previously employed sonication in paediatric orthopaedics. Plaass et al.
28
used it to analyse bacterial colonization of vertical expandable titanium ribs in a 41-month longitudinal prospective cohort (
Considering its composition regarding patient characteristics (e.g. age range, sexes, comorbidities), orthopaedic histories and the multitude of standard non-spinal implants included, our cohort clearly differed from the cohort analysed by Plaass et al. but appears representative of a typical general paediatric orthopaedic clientele.
In our cohort, only one child underwent implant removal due to symptomatic IAI, accounting for an IAI prevalence of 1.6%. Sonication identified the pathogen in that single symptomatic patient, but did not reveal significant bacterial or fungal growth on any implant removed from an asymptomatic patient or asymptomatic implant site, respectively. Fuchs et al. 33 used sonication to evaluate bacterial colonization of osteosynthesis implants around the hip and knee in adult patients without clinical signs of infection, and applied the definitions of bacterial growth used in the present study. They found significant bacterial counts in 11% of sonication cultures. 33 Other studies focusing on adult orthopaedic or trauma surgery patients reported higher rates of ‘positive sonication cultures’ but did not discriminate colonization from contamination.33,37–39 However, our results suggest that in children, clinically silent, significantly colonized non-spinal orthopaedic implants are rarer than in adults, and their clinical relevance remains to be determined.
Considering the present study’s findings and current literature reporting orthopaedic implant removal being associated with rates of perioperative complication of up to >30%,5,8–15 routine implant removal in asymptomatic children as a preventive measure to reduce IAI rates does not seem reasonable.
There are several limitations to this study. Due to its retrospective design, details of medical histories might not always be complete. Furthermore, this study was not designed to evaluate the risk of infection related to the number or type of implant(s) in situ, or to comorbidities. It also remains unclear whether the routine perioperative single-shot antibiotic prophylaxis at implantation and explantation had an impact on bacterial colonization of the hardware. However, it has been shown that perioperative antimicrobial prophylaxis did not compromise the diagnostic sensitivity of tissue sample cultures in a prospective cohort of adults undergoing revision surgery for suspected periprosthetic joint infection. 40 Finally, a comprehensive diagnostic work-up, including inflammation parameters, histopathology and intraoperative tissue cultures, was rarely performed in this cohort, mainly including asymptomatic patients.
In summary, sonication demonstrated a positive microbiological yield in <1% of all explanted implants, 99% of which were removed from asymptomatic sites.
We conclude that an anticipated risk of IAI should not be relevant for scheduling implant removal in asymptomatic children. Further research elaborating the significance of sonication in paediatric orthopaedics is indispensable.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521251400162 – Supplemental material for Sonication findings do not support routine removal of paediatric orthopaedic implants
Supplemental material, sj-pdf-1-cho-10.1177_18632521251400162 for Sonication findings do not support routine removal of paediatric orthopaedic implants by Anastasia Rakow, Susanne Lebek, Nora Renz and Julia Funk in Journal of Children's Orthopaedics
Footnotes
Acknowledgements
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author contributions
We confirm that each of the listed authors meets each of the authorship requirements as stated in the Uniform Requirements for Manuscripts Submitted to Biomedical Journals. The authors contributed as follows: Anastasia Rakow: Conceptualization, Data curation, Formal analysis, Interpretation of results, Investigation, Manuscript preparation, Study design & methodology, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. Susanne Lebek: Conceptualization, Data curation, Formal analysis, Interpretation of results, Investigation, Study design & methodology, Validation, writing – review & editing. Nora Renz: Formal analysis, Interpretation of results, Investigation, Study design & methodology, Validation, Visualization, Writing – review & editing. Julia Funk: Data curation, Formal analysis, Interpretation of results, Investigation, Manuscript preparation, Project administration (incl. ethics proposal), Statistical analysis, Study design & methodology, Supervision, Validation, Visualization, Writing – review & editing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We gratefully acknowledge support for the Article Processing Charge from the DFG (German Research Foundation) and the Open Access Publication Fund of the University of Greifswald.
Ethical approval
This research study was conducted retrospectively from data obtained for clinical purposes at a university medical centre. All participants’ legal guardians or, if applicable, the participants themselves had given written informed consent to the use of their data for clinical research. Approval was granted by the Ethical Board Committee of the Charité – University Medicine (approval number EA2/255/21).
Availability of data
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
