Abstract
Background:
Metal implants are increasingly used in children for trauma and deformity correction. This review outlines the current knowledge on the types of metals used and explores reasons for removal and the potential for long-term health issues of metal implants.
Methods:
The literature pertaining to these aspects was studied and summarised in this review.
Results:
Types of metals used have evolved as well as the development of children-specific implants. Improvements in deformity correction are measurable with likely improved outcomes and reduced health costs. Indications for metal implant removal following successful treatment remain ill-defined; however, the risks of removal are known with a minimum 6% complication rate. Health costs could be reduced by around 6% by judicious decisions to leave metal in place. Implant removal should only be encouraged in the presence of infection, mechanical failure or symptoms that are truly attributable to the implant. In the domain of spinal implants, there is evidence of significant metal ion release, most notably titanium which remains elevated to many times baseline levels beyond 2 years. The detection of titanium at low levels requires special techniques. The long-term health effects on patients and/or their offspring are not well defined, although well described in animal models.
Conclusion:
The risks of metal removal are significant. Clinicians need to be aware of potential health risks in the use of metal implants and the potential for covert toxicity effects in children especially with their long life ahead. There is a need for greater awareness of metal alloy composition and implant design to minimise risks.
Level of evidence:
Level V.
Keywords
Introduction
Metal devices are mainly implanted in children for:
Trauma
Deformity correction.
Trauma
Closed reduction with cast immobilisation treatment is the most used, inexpensive and largely produces excellent results in young children (Figure 1). Plaster of Paris, synthetic cast or a removable splint are used and are effective.1,2 Two factors allow cast immobilisations to be so successful in children – firstly the child’s strong periosteum which helps tether the bones in place and assist in reduction manoeuvres and secondly, the ability of children’s fractures to remodel and realign so that perfect alignment is not initially required. Adults generally lack these advantages with thinner periosteum and limited remodelling potential.
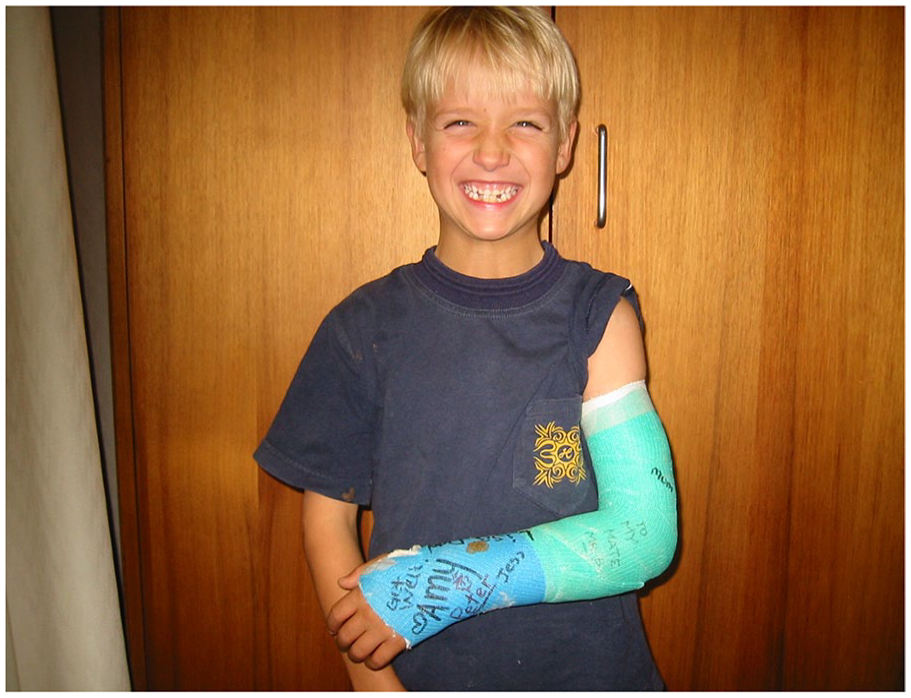
Most children’s fractures are treated by simple external fixation by a cast or splint. Here a child wears a waterproof synthetic cast which allows bathing and swimming.
History reveals other external means of immobilisation including animal skin, dried clay, bandages and bamboo splint. Bandages were soaked in egg whites, flour and other solidifying organic matter before application. In the 1800s, devices such as the Thomas splint (Crimean War 1852) were invented. Wars provide great stimulus for better management of fractures and injuries.
The earliest evidence of internal fixation of fractures was found on a slope of the Billingen Mountain in Sweden in 1928 and has been dated to between 1260 and 1527. 3 Folded around the mid shaft of a humerus was a copper plate which was fixed with three rivets. The bone underneath was healed (Figure 2). It is thought that the reason for the selection of copper was its ability to be easily moulded and its possible anti-bacterial effect.
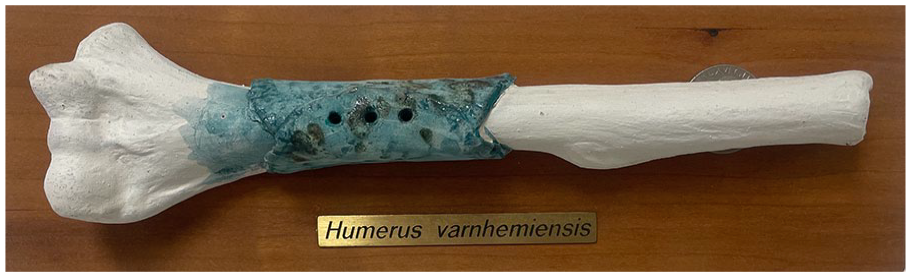
Humerus Varnhemiensis is believed to be the earliest evidence of internal fixation of fractures between 1260 and 1527 AD.
Metal implants have benefitted paediatric orthopaedics by way of fewer days of hospitalisation which permits diminished economic burden to the community and health systems, as well as less time off work for parents/carers.4 –6 Children are usually happier in their home environment.
Internal fixation appears to be increasingly used in paediatric trauma (Figure 3). Implants used for adults have been scaled to paediatric use and a plethora of internal fixation devices are being promoted by manufacturers. A paper from Finland in 2018 indicated that one in three shaft fractures of the tibia in children were being treated by open fixation. 7 The authors concluded that the surgeon’s personal preference, type of fracture and age of the patient were the main determinates of internal fixation or closed management. Polytrauma in children often demands selective methods of management with damage control surgery and delayed internal fixation of multiple fractures.
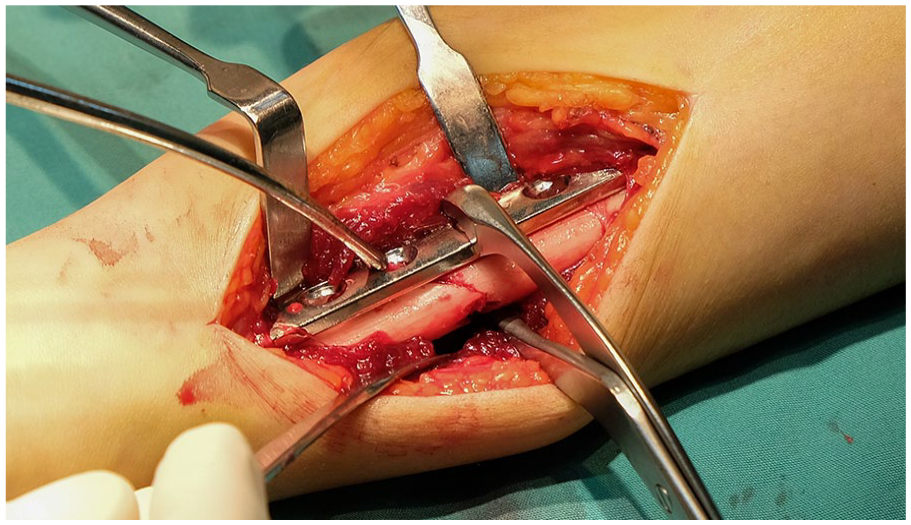
Plate and screw fixation of limbs in children is increasingly popular but may not always be necessary.
Growth plates at the end of each long bone have been a barrier to intra-medullary fixation; however, newer techniques with physeal-sparing insertion points have been facilitated by implant redesign. Femur fractures can be ‘rodded’ with care to avoid damage to the blood supply to the femoral head by avoiding the piriformis fossa region.
The current obesity epidemic in developed/developing countries may be changing our approaches to fracture management, especially with mobility issues for obese children who have sustained lower limb injuries. 8 Anaesthetic and wound healing risks are also increased in this cohort. However, internal fixation can help meet the challenges of nursing care and mobility for the obese child.
But are higher rates of internal fixation better for the child overall? An anaesthetic for the first operation for internal fixation, a second for removal of fixation later as well as the potential for complications such as infection, non or delayed union, disability associated with operative neurovascular injury and the potential for cognitive issues from anaesthesia. These questions require complex analysis at a public health level for assessment of personal and societal benefits.
Elective deformity correction
Deformity correction such as osteotomies for realignment of crooked long bones or spines usually requires some form of internal fixation to maintain the new position of the bones whilst the cut bone ends heal. Such devices include plates and screws, intramedullary rods or external frames with pins through the skin into the bones. It is not the scope of this review to contrast these techniques but suffice to say that each has specific attributes that are better in various conditions.
Advances in instrumentation have facilitated improvement in deformity correction, such that more reliable and robust constructs allow better correction, and many implants are now tailored to specific problems, such as osteotomy around the hip. Shorter hospital stays and reduced pain are promoted. Robust fixation often negates the need for external immobilisation such as a spica cast. As such, the use of metal implants is popular with a vast array available and matched by an ample supply of manufacturers who promote their use.
Should metal implants be removed and what are the risks?
The risks of removal are plentiful, and a non-exhaustive list is below. 9
Pathological fracture through the screw/device holes or stress-riser area
Infection
Neurovascular injury
A second anaesthetic for implant removal
More time off work for parents/carers
Difficult or abandoned removal if corrosion/cold welding of the screw to plate
Implant migration down the long bone or becoming encased in bone/incorporated
Challenges for the next surgeon with further surgery (such as hip replacement)
In January 2005, Hamlet Peterson published a sentinel paper titled ‘Metallic implant removal in children’. 10 This should be essential reading as it highlights the risks of routine implant removal. Notably, he provided historical context by stating ‘Because of the longer remaining life span of children, the possibility of corrosion, as well as the possibilities of allergic reaction, ion toxicity, malignant transformation, and stress shielding, was greater in children than in adults. Thus, through the first half of the 20th century, the accepted standard for children was to remove a metallic implant as soon as it had served its purpose’. Subsequent publications have taught us that the risk of malignant transformation effect is negligible or non-existent. Routine implant removal to minimise a long-term cancer risk does not appear justified in current proper scientific epidemiological studies.11,12
Amusingly, a reply to the Petersen article from Carl Stanitski 13 noted the frustrations that even experienced surgeons can face and quipped that ‘no one ever looked good on a hardware removal’. So, do surgeons have good reasons for hardware removal?
Paediatric trauma implants received an excellent review in a 2021 paper. 14 They noted that implant removal remains controversial and that surgeon surveys suggest up to 60% of surgeons routinely remove implants. This preference is unlikely to be evidence based and probably related to prior teaching and orthopaedic myths.
It seems that implant removal should remain discretionary except in situations as below:
Infection
Mechanical failure
Symptoms are truly attributable to implant impingement.
It can be argued that implants tend to become covered in bone in the long term in children, which increases the complexity of surgical removal. The surgeon needs to be certain that the symptoms described are reliably due to the internal fixation device or coming from other causes.
Do the patients receive benefits from implant removal? A unique retrospective patient-reported outcome study from Germany suggests some benefits. 15 In a subset of patients having implant removal because of pain or impaired function, 96% self-reported decreased pain and 72% patients an improvement in function. Patients reported a rate of 10% complications and 42% of patients felt no change in pain before and after the operation. Worryingly, 7% of patients without pain before implant removal complained about pain afterwards.
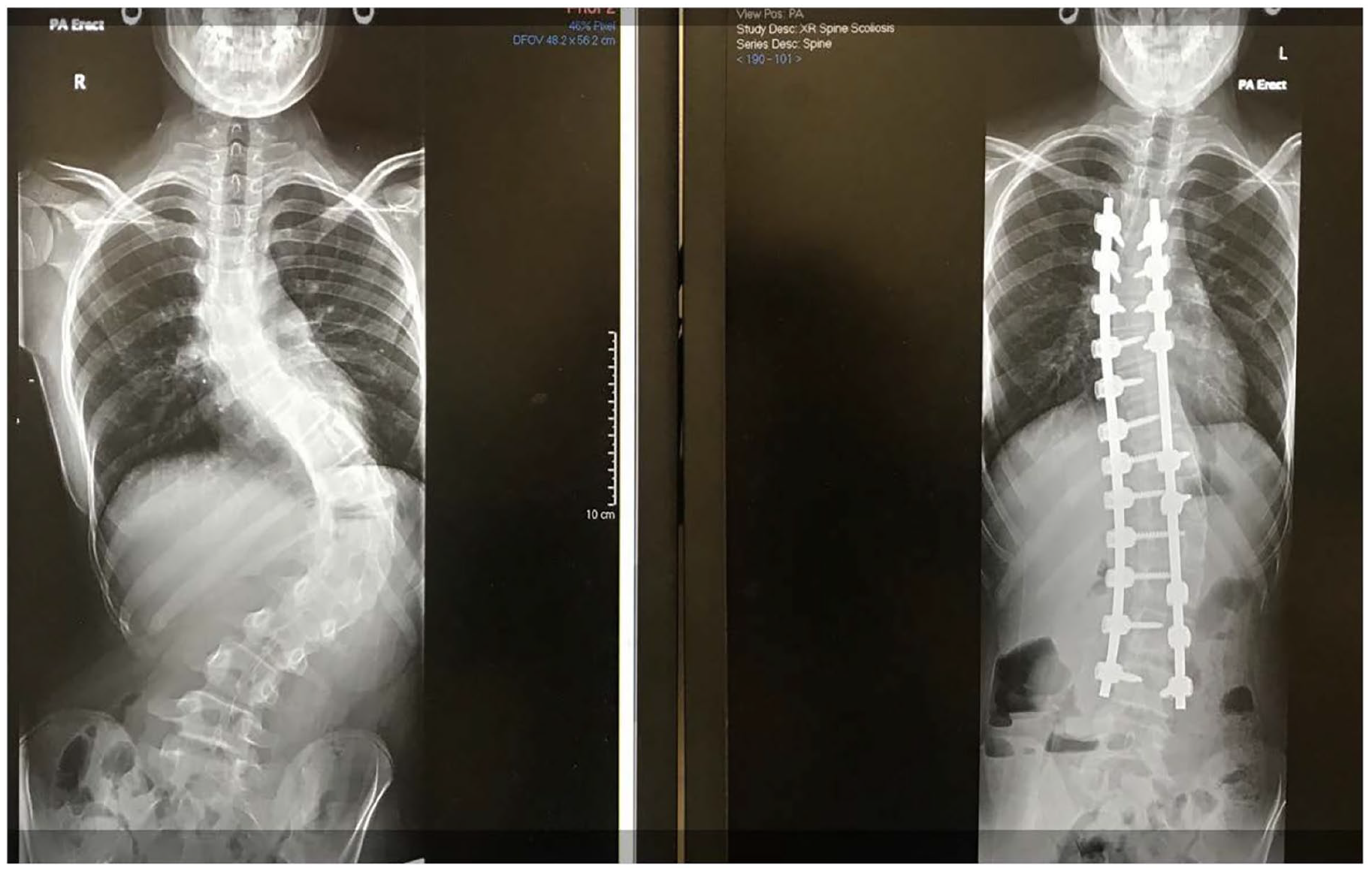
Some children do need their implants for life, such as the treatment of various dysplasias or osteogenesis imperfecta, where the metal implants add strength to weakened bones or are prone to recurrent deformity, fracture or non-union. Spinal rods and screws are usually left in permanently as they are associated with fusion and provide ongoing support to minimise the risk of deformity recurrence (Figure 4).
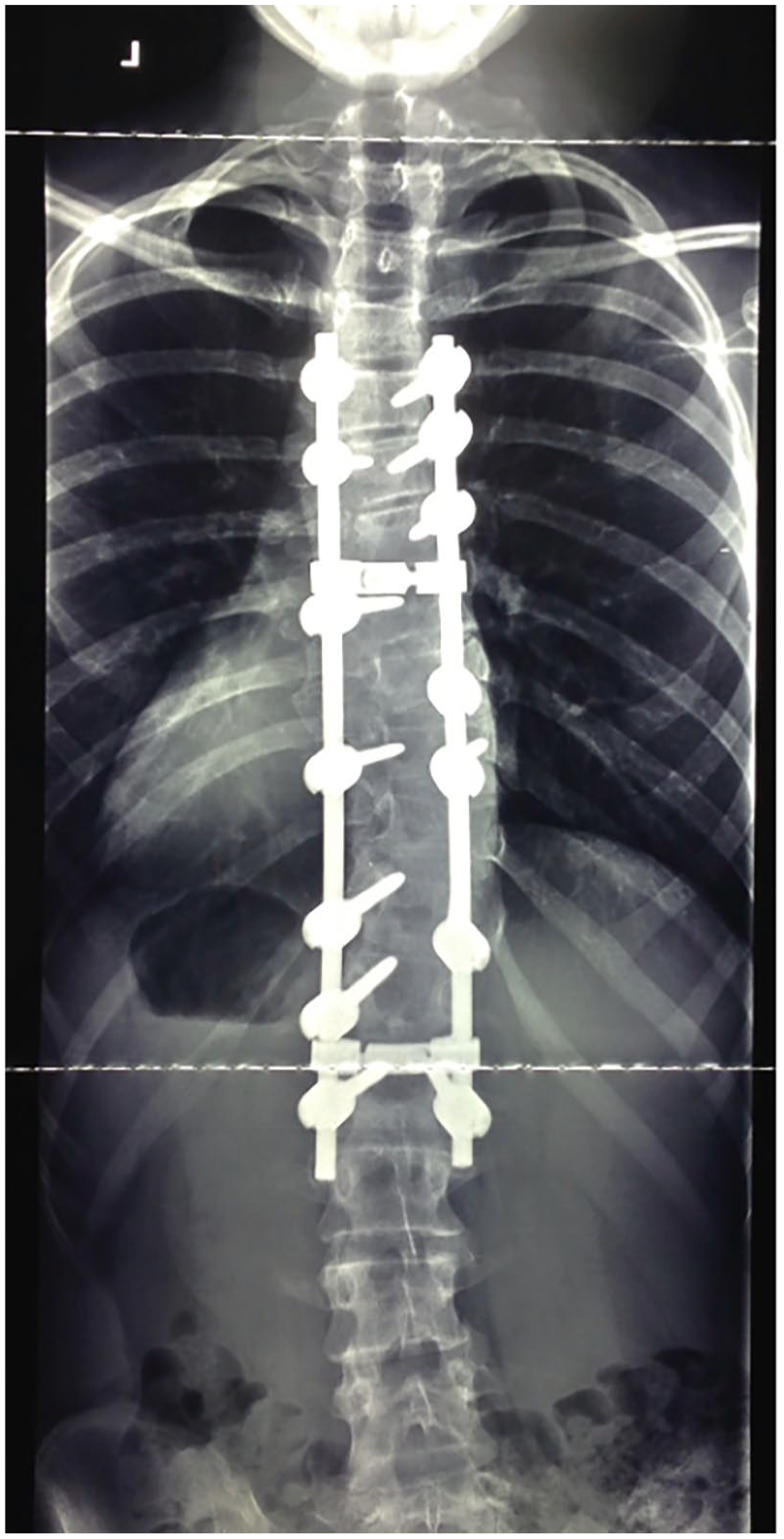
Spinal deformity surgery has been associated with systemic metal release with the persistence of raised titanium levels beyond 2 years.
Fracture following implant removal is not unknown. A systematic review in 2022 16 reviewed 15 studies (only 1 was prospective level 2, others were all levels 3 and 4) and indicated a fracture incidence of 3%–16% following implant removal in paediatric patients. The site and type of fixation influenced the fracture rate with the forearm and femur being the highest. Importantly, the rate of periprosthetic fracture appeared lower than fracture after implant removal. Compression plate and screw removal appear to have a higher rate of fracture and is probably related to the multiple screw holes as well as stress shielding along the length of the bone under the plate. Many recommend 6–12 weeks of modified activities but in children, this is often challenging to enforce. External fixation showed the highest rate of refracture at 14.5% in femoral shaft fractures although the reported series are small in numbers. Other studies report similar higher rates of re-fracture following implant removal.17 –20
An earlier report indicated a 28% complication rate for non-medically indicated implant removal surgery and recommended implant removal only when medically indicated by way of infection, mechanical problems or implant failure. 21 Patient demand without strict medical reasons seems best to be resisted, and perhaps retention of the implant should be recommended before initial implantation, thereby fostering realistic expectations for the future.
A recent review provided advice on the timing of implant removal and risks for refracture as well as the need for a shared decision with parents including costs and risks. 9 Notably, some healthcare insurers do not cover the costs of implant removal. Whilst clinicians focus on what is best for the child, health economics are increasingly relevant, and surgeons must be able to justify decisions and maximise efficiencies in the use of health monies. A study from the US Healthcare Cost and Utilization Project using the Kids’ Inpatient Database estimated the average costs of implant removal to be in the order of $21,040 when infection-related causes were excluded. 22
A 6% rule
By way of a broad-brush summary, it has been the authors’ overall observation of the literature that implant removal constitutes around 6% of the workload of hospital operating room activity and costs, and conveys around a 6% complication rate (with a wide range of 0%–43%). Clinicians should be aware of these statistics for evidence-based clinical management.
Slipped femoral capital epiphysis screw removal
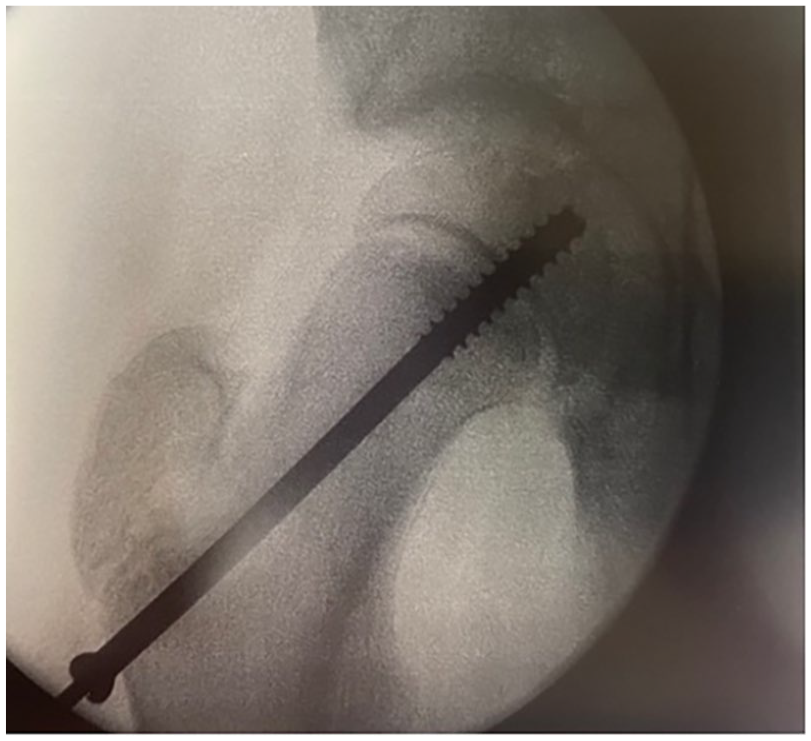
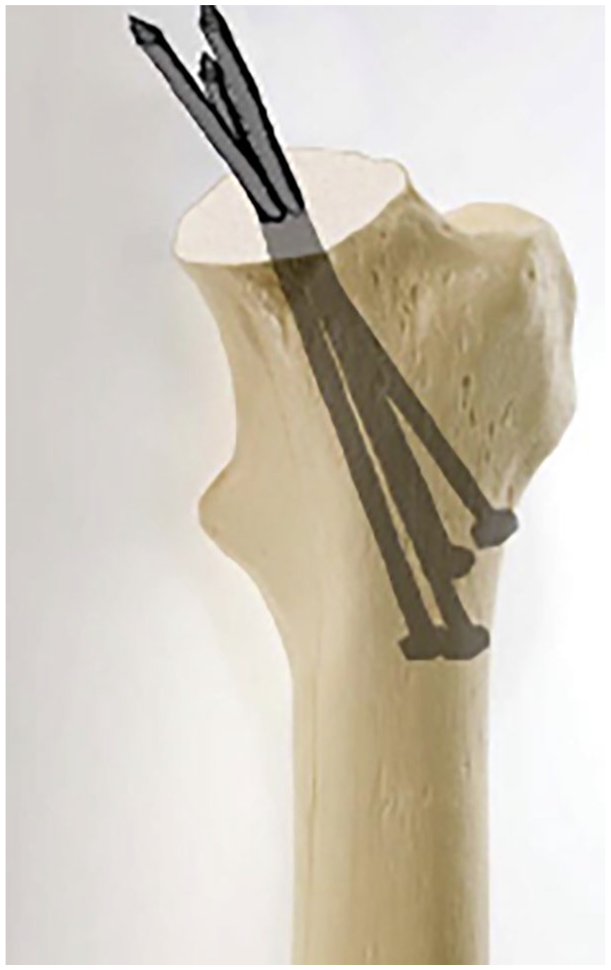
Removal of screws after treatment for slipped femoral capital epiphysis (SCFE) provides special challenges (Figures 5 and 6). It can be technically difficult and fraught with complications, not the least being abandonment due to stripping of the head, partial removal then becoming stuck and the often-needed larger scar and dissection to gain access. 23 Whilst some may argue that retention makes subsequent total hip arthroplasty more difficult, suitable techniques are described for removal at hip replacement surgery with regular neck osteotomy and sectioning of the head followed by retrograde tapping out of screws (Figure 7). 24 It is the authors’ preference that screws inserted for SCFE remain in place indefinitely and that this is informed at the time of initial surgery. Individual surgeons may have their preferences for screw removal particularly if magnetic resonance imaging (MRI) is thought to be needed in the future to assess vascularity or impingement issues. If the informed consent includes a surgeon-directed preference to not routinely remove the screw, this allows the family to have realistic expectations for the future. Similarly, the avoidance of fully threaded screws facilitates easier removal of screws at the time of total hip replacement (partially threaded screws are easier to tap out after head/neck osteotomy during hip replacement).
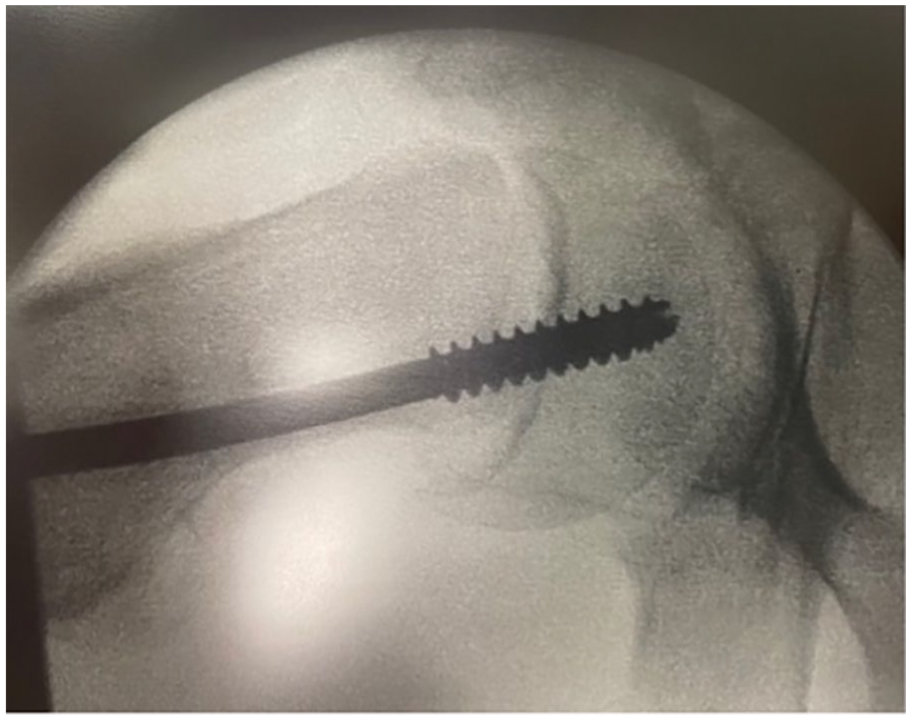
Screw fixation of slipped capital femoral epiphysis.
The risks of screw removal are likely to outweigh the benefits. Pathological fracture through the screw entry hole, infection, and difficult or abandoned screw removal account for many of the complications.
Screws may be removed at the time of hip replacement by femoral neck osteotomy and quartering osteotomies of the femoral head, then tapping the screws out in a lateral direction.
What types of metal are being used?
Vanadium steel was developed in the early 1900s with plates by Sherman using cross-headed screws. Issues of breakage and infection understandably arose in this era before antibiotics. In the 1920s, copper and nickel were tried with discoloration of the surrounding tissue as well as non-stainless steel and iron corroding quickly. Less reactive metals such as gold and silver did not have the strength and stiffness needed to stabilise bones. Soon after, the addition of chromium to create stainless steel was a major step forward. The principles of internal fixation, implants and instruments were promoted by an active group of Swiss surgeons in 1958 with the formation of Arbeitsgemeinschaft für Osteosynthesefragen/Association of the Study of Internal Fixation.
In the 1970s, titanium became popular with its excellent tissue compatibility, corrosion resistance and mechanical properties. Most orthopaedic surgeons probably look upon titanium as an ‘inert’ biomaterial that is well tolerated in the human body and may have an anti-bacterial effect. A silver coating has also been applied to surgical implants particularly when the patient is compromised by way of tumour resection, chemotherapy and an enhanced risk of infection. Silver has known antibacterial effects; however its systemic release and deposition in end organs with toxicity has limited its use in humans.
Titanium is commonly mixed with other metals to create titanium alloy and obtain superior mechanical properties. The titanium alloy Ti-6Al-4V includes aluminium and vanadium and was historically in common use but had low shear strength and is now not preferred for screws or plates. By contrast, the alloy Ti-6Al-7Nb (with aluminium and niobium) replaced the cytotoxic vanadium in the mid-1980s with niobium providing better ductility.25,26 It continues to be the most used titanium alloy with variations including the addition of nitrogen, chromium and cobalt for stiffness.
Titanium is not an inert substance and implants readily coat themselves in a passivating layer of titanium dioxide which protects the reactive surface of natural titanium. This is distinct from the so-called ‘noble metals’ such as gold, silver and platinum which are generally non-reactive but too soft for orthopaedic implants by themselves. Most notably, the commonly used implants are made of metals used in normal biological processes in the human body such as chromium, cobalt and iron – except titanium which has no natural biological role.
If the passivating layer of titanium dioxide is disturbed (such as by a connection or rubbing), then the bare metal underneath allows metal to be released whilst the area rapidly re-passivates with more titanium dioxide. This occurs in the salty environment of the human body and the development of this ‘corrosion’ is seen in many forms such as crevice corrosion, fretting or galvanic action. 27
Human cortical bone has Young’s modulus of elasticity in the order of 30–70 MPa, whereas the ‘softest’ titanium alloys are 110 MPa plus. As the implant increases in modulus of elasticity scale, there is greater potential for stress shielding and bone resorption. Titanium alloys are especially popular around spinal implants due to less MRI scatter and thus facilitating better visualisation of the spinal cord (Figure 8). The esoteric metallurgical variabilities are beyond the scope of this review but suffice it to say that alpha and beta titanium alloys exist and percentages of each of these types influence behaviour of the metal implant, alongside other characteristics such as strength, machinability, hardness and springback. 25

Metal-on-metal hip replacements have been associated with systemic metal release and local tissue reactions.
Physical connections between implant components (such as a screw to a plate, or pedicle screw to a spinal rod) or direct contact with adjacent bone promotes tribocorrosion with wear debris and subsequent inflammation and complex immunological reactions. Micro-motion occurs and mechanical models show how micro-motion under load induces wear such that the passivation layer (of titanium dioxide) is rubbed away and exposes the native titanium alloy – this is accelerated in salty environments. 28 Increased loads enhance this wear and the release of third-body particles. Such issues entered the public press and medical journals when some metal-on-metal hip implants failed at a higher rate as well as concern that cobalt and chromium blood levels produced severe health effects (Figure 8).
Spinal surgeons often bend the spinal rods to provide the expected correction and shape. Spinal rods that have a low Young’s modulus of elasticity tend to have a higher ‘springback’ value such that they will tend to straighten to their native shape. Physical bending of titanium alloy rods in the operating room has the effect of increasing the stiffness of that part of the rod. Adding chromium to titanium alloy also increases stiffness (Figures 9 and 10).26,29

A typical titanium alloy pedicle screw shows a connection to the spinal rod. The connection and micro-motion may facilitate metal release.
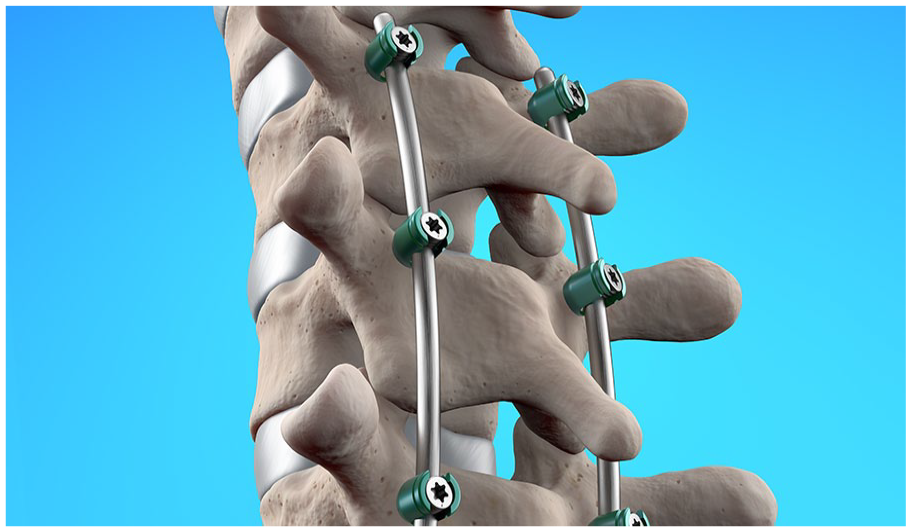
Spinal rods in place show fixation over many levels, with the potential for micro-motion when the child moves.
How do titanium implants react in the body?
Inevitably, with implant insertion, there is a host response that starts as an ‘injury’ due to the surgery involved in delivering the implant. This inflammatory response commences instantly with platelet activation, complement release and adherence of proteins to the surface of the implant. Interleukin factor 8 is activated, and macrophages gather. Good healing of tissues requires, in general, for this inflammatory reaction to resolve and then for local regeneration to commence and continue. In this regard, the persistence of the inflammatory response has the potential to impede healing – a good example being the presence of infection which promotes non-union of fractures via the persistence of inflammation and a toxic local environment for regrowth.
Macroscopic signs of metallosis are not infrequently seen by orthopaedic surgeons when removing implants with blackened fibrous tissue adjacent to screws in plates. Adult hip replacement surgeons sometimes witness severe adverse local tissue reactions in patients with metal-on-metal total hip replacements. This reflects local corrosion issues which can be of wear, fretting, galvanic and/or crevice corrosion type. In the salty environment of the human body, the reactions between different metals within alloys can be quite significant and variable.
Dendritic cells detect these foreign antigens (such as the blackened debris or titanium particles) and then have a significant role in activating T-cell function (Figure 11). Dendritic cells then migrate to lymphoid tissues and can also migrate to the brain. Interestingly, the sites of dendritic cell accumulation are similar to the target organs identified for titanium deposition in autopsy studies. The link below animates the process of dendritic cell function in response to foreign antigens or cancer cells (https://youtu.be/OtOLOSD-iXA).
Dendritic cells (pink) detect foreign antigens, activate T cells, and migrate to lymphoid tissues.
What happens to the released titanium ions or titanium dioxide particles?
Evidence of the release of metal ions in children with spinal constructs has been a focus for some researchers, including the authors of this review. The release of metal ions from static implants that are not ‘articulated’ (as distinct from a metal-on-metal hip replacement) was not expected from pilot studies that commenced in the 1990s. 30 Continued research has confirmed a release from most spinal constructs.31 –36
Extremity plates and screws release fewer metal ions than spinal implants (Figure 12). 36 This study showed a negligible increase in titanium levels in their extremity plates and screw cohort however spinal implants demonstrated a statistically significant increase in serum Ti levels from 0.57 ppb (parts per billion) pre-op to 3.3 ppb post-op.

Extremity plates and screws should be considered to remain in place if asymptomatic.
Growing rod constructs in children utilising articulations generally show much higher titanium levels equivalent to 8 ppb with a range extending to 18 ppb. 37
It is important for researchers in this field to be aware of the challenges associated with the measurement of titanium in blood, including issues of contamination, difficulties in measurement, inter-laboratory sensitivity and accuracy of measurement and units of measurement. 34
Multiple longitudinal studies of spinal implants in children in Australia, Sweden and the United States indicate that chromium and cobalt levels initially rise after surgery but approach or decline to baseline by 6 months (Figure 13).30 –36 By contrast, titanium continues to rise to 2 years when median values plateau at 5.4 times baseline levels. 34 To put this in context, baseline levels of titanium before surgery had a median value of 0.45 ppb, compared to the median level of 2.33 at 2 years (range 1.0–21 ppb). So, questions arise as to why titanium is still present in the blood beyond 2 years, and does it cause harm?
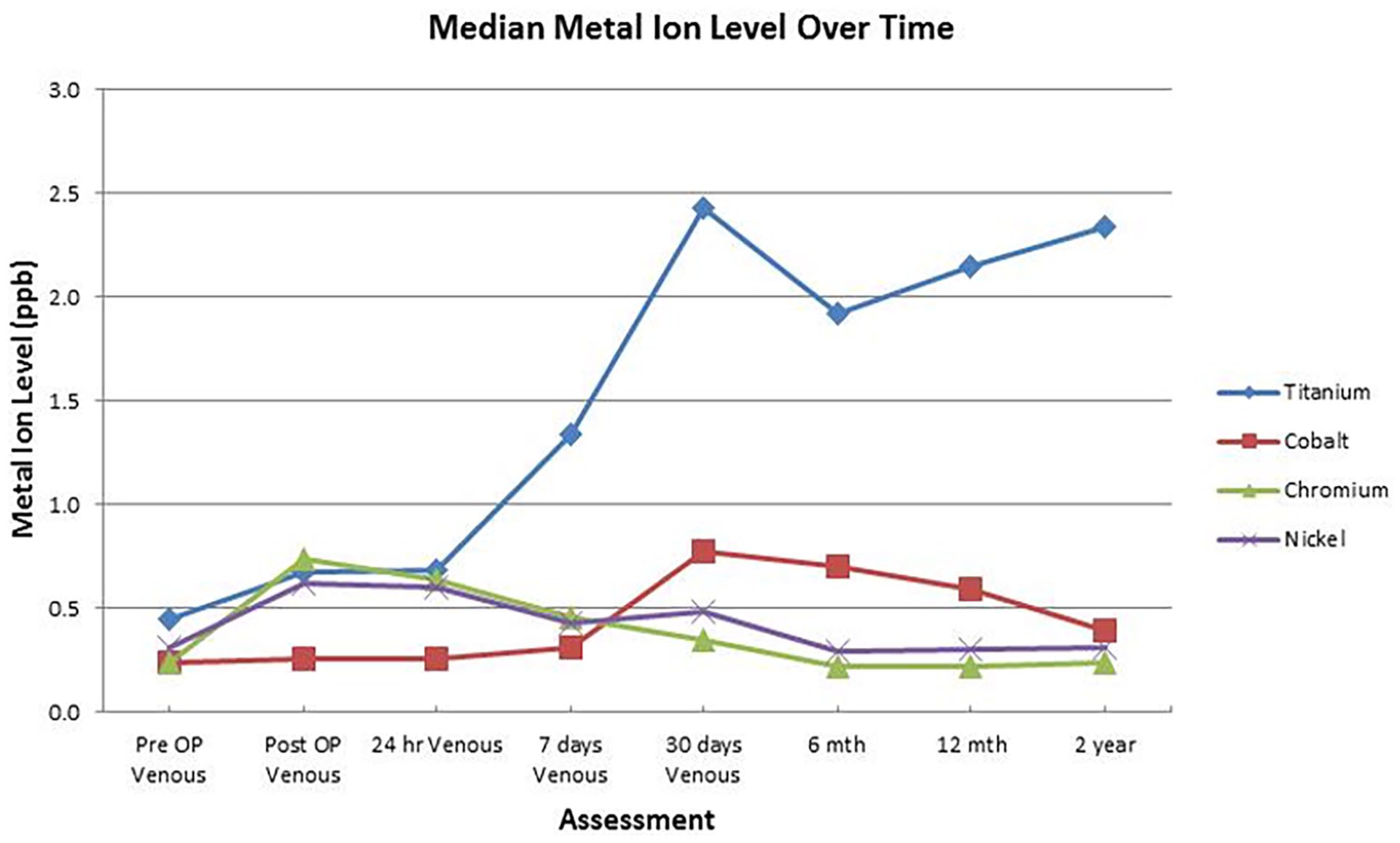
Graph showing persistence of median titanium levels in serum beyond 2 years from spinal deformity surgery, whereas chromium and cobalt approach pre-operative baseline levels. 34
Many metal implants develop fibrous encapsulation, whereas titanium mesh is believed to promote direct bone ingrowth so that the titanium alloy integrates with the body’s bone. Additionally, various coatings have been promoted to enhance body ingrowth such as glycosaminoglycans and hydroxyapatite (sometimes having a pro-inflammatory deleterious effect).
It is possible that the direct bone ingrowth associated with titanium implants does facilitate a direct release of titanium (via the bloodstream), as distinct from the scenario of fibrous encapsulation or physical barrier between metal and bone by way of bone cement (in cemented femoral stems in hip replacement) where a less vascular interface is present.
Titanium is not easily detectable. Current in vivo imaging techniques have not been developed and only biopsy can confirm solid organ depositions. As titanium is non-ferrous, it is not readily detectable by MRI scan. The spread of titanium ions has been demonstrated in animal models with solid organ deposition in the brain, heart, liver, spleen and lymph nodes. Adult human autopsy reports have documented deposition in these areas after metallic hip implants. A study of 15 human livers and spleens at autopsy using scanning electron microscopy with energy-dispersive X-ray detection confirmed titanium particles in all these solid organs. 38
Metal ions release into the blood and deposition in solid organs
Here are some questions that need answers.
Why is titanium consistently present in the circulation?
Despite titanium alloys having the ability to rapidly self-passivate with titanium dioxide, surface changes occur through various tribological effects. This is supported by the higher levels seen in growing rod constructs where articulations are used. This contrasts with posterior spinal fusions which are largely static. It is likely that articulations or micromovements result in abrasion of the titanium dioxide passivation layer and allow momentary exposure of the native metal alloy. The persistent level of titanium in fused spines was unexpected and currently seems best explained by the exposure of a large surface area of pedicle screws to the circulation or tribocorrosion at connections.
It is therefore possible that the continuous release of titanium then creates a steady state of titanium in the blood, and it is possible that a level of homeostasis develops with target solid organs continuing to seed the circulation without a natural path for sufficient excretion of titanium. The excretion of titanium in humans may be idiosyncratic and variable between individuals.
Some studies fail to detect raised levels of titanium. This likely reflects the testing apparatus used, especially as titanium can be difficult to assay at low levels reliably, particularly below 5 ppb. Titanium dioxide is not readily soluble and special techniques are required for assay. Some laboratories can only detect very high levels of titanium. Comparison of levels between studies is also challenging due to varied methods of analysis (high-resolution inductively-coupled mass spectrometry is currently preferred), as well as contamination during sampling, the use of whole blood versus serum and different laboratory analytical approaches. One study, analysing seven different laboratories, found variability in the order of 0.4–24.6 ppb of titanium and encouraged the use of a single laboratory using high-resolution machines. 39 Consistent techniques are mandatory for longitudinal studies.
Does it accumulate in solid organs or tissues of the human body?
The liver is the known main filtering organ for oral and inhaled titanium and autopsy studies reveal primary accumulation in the human liver. The main excretion of titanium is via bile and the alimentary tract. Of interest is the reported absence of urinary titanium in a prospective series of growing rod patients with early onset scoliosis despite mean serum levels of 4 ppb. 40 Excretion in rats after intravenous infusion appears to be faecal via bile from the liver. 41
Studies in mice have confirmed deposition of titanium in the midbrain substantia nigra. 42 A 2023 review indicated titanium dioxide nanoparticles readily crossing the blood–brain barrier with impairment of neurotransmitter metabolism and neurobehavioural effects in rodents.43 –45
The lack of a reliable method of detection of titanium in vivo, without biopsy, makes knowledge of its metabolism in humans challenging. There is a need for focused research in this area with new methods of detection and epidemiological large numbers to look for birth defects or later language/learning difficulties in offspring of affected high titanium mothers, as reported in rodent studies.
Epidemiological data on the effects of titanium exposure in humans are sparse. Most research has been on oral ingestion. The European Food Safety Authority concluded in 2020 that titanium dioxide cannot be considered a safe food additive and the European Commission instructed removal of its use. 46 More recently in May 2024 the Committee on Toxicity (UK) advised the Food Standards Agency (UK) that the oral absorption of titanium was low and allayed concerns regarding colonic crypts, immunotoxicity, inflammation, neurotoxicity and reproductive/developmental toxicity. 47 However, the release of titanium from implants is by a different mechanism (not oral) and is likely distributed via lymphatic drainage and the bloodstream.
Does titanium affect the offspring of persons with titanium spinal implants, particularly noting to predominance of teenage females having adolescent idiopathic scoliosis surgery?
Safe levels of titanium in the blood (serum) are not well defined; however, 1 ppb is commonly accepted as the upper range of normal. Titanium is known to readily cross the placenta from mother to foetus in animals and humans.
It is traditionally thought that the placenta acts as a barrier to environmental toxins and pathogens that could harm the growing foetus; however, nanoparticles of titanium have been shown to cross from the maternal to foetal circulation with titanium detected in the placenta and meconium (the neonate’s first stool) in humans. 48 Thus, there is evidence of the absorption of titanium by the foetus from the human mother.
Other studies using intravenous injections of titanium (albeit in very large doses) in rodents throughout gestation showed accumulation in the foetal liver, testis and brain with evidence of impairment of cognitive functions in the offspring, lower birthweight, lower sperm counts and smaller uteri.49,50 These studies employed short-term infusions of titanium, which is distinct from the potential human exposure in females who have titanium spinal implants. Here the foetus would be exposed to a low level of titanium throughout the entire (9 months) of pregnancy from time of conception to delivery. Whilst the effects in rodents are not always comparable to human responses, human investigations with proper epidemiological study appear warranted. There is a need for focused research in this area with large numbers to look for potential defects or later language/learning difficulties as reported in rodent studies. It is worth noting that for children having surgery for adolescent idiopathic scoliosis, the majority are female (75%–80%) (Figure 14).
Spinal deformity surgery usually requires the rods and screws to remain for life, with potential exposure of offspring to titanium whilst in-utero during pregnancy.
Are there specific (and modifiable) metallurgical properties that influence the release of metal ions?
The exact composition of the implants in current use requires further analysis. Manufacturers usually state the composition of the components supplied in their Information For Users pamphlets including indication of compliance with American Society for Testing and Materials (ASTM) International Standards with defined composition of the alloys and testing for fatigue. In a recent study, 34 there was a subset of patients with ‘all titanium’ implants (titanium pedicle screws and titanium rods) that demonstrated raised levels of chromium and cobalt compared to pre-operative baseline. This puzzling result could be ascribed to impurities being present in the metal stock being used in the manufacturing process, contamination in testing or undocumented metals being incorporated to adjust the strength of stiffness of the implants.
Medical device standards are controlled by most governments using internal assessments of safety and the ASTM recommendations. Devices are assessed differently by individual countries – in the United States by the Food & Drug Administration and in Europe by Notified Bodies. Different emphases are noted in the regulation of the materials used.
Whilst individual implant components have been assessed, the combining of dissimilar metals creates an environment for the phenomenon of galvanic corrosion. 51 This review cannot easily summarise the complex issues of the effects of combining the use of dissimilar metal alloys but suffice to say that such practice does enhance corrosion and the potential for metal release. This is frequently only visible at electron microscope level magnification with cracking of the surface, as well as crevice and pitting corrosion.
Some implants receive a coating of titanium to potentially aid in osseointegration. For example, spinal interbody fusion cages can be made of polyetheretherketone (PEEK) and have a nanoparticle coating of titanium or titanium alloy. A cross-sectional study of 32 adults having spinal deformity surgery demonstrated similar increases in titanium at pre-op and 1 year post-op with average serum titanium levels at almost 3.5 times baseline. 52 The authors made an interesting observation that the presence of an intervertebral body device (in association with a short one- or two-level lumbar posterior instrumentation and fusion) did not change titanium levels when it was made of PEEK, but levels of titanium did increase when the intervertebral device was ‘all titanium’. This supports the view that titanium release to the circulation is influenced by the surface area of titanium and the opportunity for surface changes. Mechanical fretting at connections was thought by them to not be the cause of raised titanium levels.
The future of metal implants in children
The long-term effects of metal implants in children have not been extensively studied. Whilst titanium alloy is known to be one of the more biocompatible materials and is ranked highly for corrosion resistance, some degree of release of titanium is likely to be universal. Many studies lack the ability to measure low levels of titanium, and this facilitates under-reporting of increased levels.
Will we see different or improved metals? Will implants be biodegradable? The latter shows some promising early results; however local tissue reactions appear an impediment to their wide acceptance and the potential systemic effects of the degraded product will need to be examined.
Will health economists ultimately decide that the financial burden of the occasional complication outweighs the immediate benefits of internal fixation and reduced hospital length of stay? Similarly, decisions should be made regarding the retention of implants to minimise harm and control costs.
The orthopaedic surgeon must have a role in this. Orthopaedic surgeons need an awareness of metal composition when choosing implants. We must remain vigilant and query what we place in patients, especially the young, to maximise long-term safety and minimise the risk of harm.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521241293954 – Supplemental material for Metal implants in children
Supplemental material, sj-pdf-1-cho-10.1177_18632521241293954 for Metal implants in children by Peter John Cundy and Nicole Williams in Journal of Children’s Orthopaedics
Footnotes
Acknowledgements
Georgia Antoniou, Research Officer, Department of Orthopaedic Surgery, Women’s & Children’s Hospital, Adelaide, Australia, for data collection and statistical advice. Dr. William Cundy, Orthopaedic Surgeon, Department of Orthopaedic Surgery, Women’s & Children’s Hospital, Adelaide, Australia, for specimen collection and processing. Associate Professor Abel Santos Alejandro, Associate Head of School (Research), School of Chemical Engineering, Faculty of Sciences, Engineering and Technology, University of Adelaide, for advice on metallurgy. Dr. Cheryl Suwen Law, PhD, Research Associate, School of Chemical Engineering, University of Adelaide. Faculty of Sciences, Engineering and Technology, University of Adelaide, for advice on metallurgy and testing of implant composition.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
