Abstract
Purpose:
Paediatric forearm fractures are among the most common childhood injuries, with diaphyseal fractures often requiring surgical management due to their instability and poorer remodelling potential. Traditional methods, such as elastic stable intramedullary nailing, are effective but require secondary procedures for implant removal, increasing healthcare burdens. Bioabsorbable intramedullary nails offer an alternative, eliminating the need for implant removal. This systematic review and meta-analysis evaluates the efficacy and safety of bioabsorbable intramedullary nails compared to conventional methods.
Methods:
Five studies, including 255 paediatric patients with 399 forearm fractures, were included. Of these, 159 were treated with bioabsorbable intramedullary nails. Meta-analyses assessed outcomes, including fracture healing time, complications, functional results and reoperation rates.
Results:
Bioabsorbable intramedullary nails demonstrated fracture healing times comparable to elastic stable intramedullary nailing (10.67 weeks; 95% CI: 8.92–11.42) with no significant differences. Complication rates were similar, with nine re-fractures in the bioabsorbable intramedullary nail group and 10 in the elastic stable intramedullary nailing group. Secondary displacement occurred in 3.1% of bioabsorbable intramedullary nail patients versus 4.7% for elastic stable intramedullary nailing. Functional outcomes showed improved pronation (78.5° versus 72.7°, p = 0.030) and elbow flexion (153.8° versus 144.8°, p = 0.001) for bioabsorbable intramedullary nails. Pain scores were comparable, with fewer bioabsorbable intramedullary nail patients reporting postoperative pain.
Conclusions:
Bioabsorbable intramedullary nails are a safe and effective alternative to elastic stable intramedullary nailing, offering equivalent clinical outcomes while eliminating the need for implant removal. Larger, long-term studies are needed to confirm these findings and evaluate the cost-effectiveness of bioabsorbable intramedullary nails in paediatric fracture management.
Introduction
Forearm fractures make up 41.1% of all paediatric fractures in the United Kingdom, with the majority being distal radius and/or ulna fractures, 1 although diaphyseal fractures account for almost 55% of fractures requiring surgery in children. 2 Diaphyseal fractures are often unstable and may remodel poorly compared with distal fractures, and have a higher rate of non-union, and poorer functional outcomes, particularly in older children with less remodelling potential; over the years, there has, therefore, increasingly been a shift towards surgical management for these fractures. 3
Indications for surgical management of forearm fractures commonly include open, irreducible or unstable fractures, with factors such as age, the location of the fracture, its angulation and/or rotation, and displacement being taken into account. 4
Surgical management of forearm fractures in children traditionally uses elastic stable intramedullary nailing (ESIN) or open reduction and internal fixation (ORIF) with plating. ESIN is effective and inserted using a minimally invasive technique, which can provide a better cosmetic result compared with ORIF, and allows for easier subsequent removal of metalwork. The major drawback of both techniques, however, is that both typically require further surgery to remove metalwork when the fracture has healed, thus further exposure to perioperative risks and potential complications. A retrospective review of the removal of 1223 implants from 801 children found an overall complication rate of 12.5% 5 ; another retrospective analysis following removal of ESIN from 384 paediatric patients, 233 of whom had been treated for diaphyseal forearm fractures, found an overall complication rate of 3.1%. 6
Cost is an important factor when considering treatment options. An analysis of cost-effectiveness of ESIN versus Kirschner wire (K-wire) and casting in Romania reported that ESIN cost significantly more than other methods; the mean cost of surgical management with ESIN was $632.76, and the mean cost of removal was $462.84, thus highlighting the additional financial burden associated with fixation methods requiring subsequent removal of metalwork. 7
Bioabsorbable intramedullary nails (BINs) have emerged as an alternative implant to the traditional elastic titanium nails used in ESIN; these implants, made from poly-L-lactide-co-glycolide (PLGA), are resorbed over time, thus eliminating the need for secondary removal surgery. Bioresorbable implants have previously been used in other areas of orthopaedic surgery, for example, in fixation of ankle fractures and chevron osteotomy for hallux valgus 8 ; a known potential complication of the use of bioresorbable implants is development of a non-specific foreign body reaction within the first 2–3 months, reported in around 2%–4% of patients,8,9 which may require aspiration, but has not been reported to affect bone healing. 9
This systematic review and meta-analysis aims to evaluate current literature regarding the use of BIN in the surgical management of paediatric forearm fractures, compared with current methods.
Methods
This systematic review and meta-analysis was conducted in accordance with the Cochrane Handbook for Systematic Reviews 10 and PRISMA-P statement. 11 The protocol was registered with PROSPERO on 03 July 2023 (CRD4202341673).
Search strategy
A comprehensive search of databases, including PubMed, Scopus, MEDLINE, Embase, CINAHL and Google Scholar, was conducted on 09 December 2024. The search was extended to include trial registers, grey literature sources and other relevant databases. All studies published before December 2024, with no restrictions, were included in the screening process.
Study selection
Two authors (FE and GA-D) separately screened all search results, blinded to each other, based on the inclusion and exclusion criteria. Any disagreements were resolved by a third author (MP) independently. The full screening process is outlined in the PRISMA flow diagram.
Criteria for inclusion
Participants
This review included studies with paediatric patients aged 18 or younger with radiologically confirmed forearm fractures (radius, ulna or both). Only human studies were included.
Interventions
The accepted intervention was fixation with any bioresorbable implant, including bioresorbable intramedullary nails, with or without open reduction and postoperative plaster immobilisation.
Types of study and controls
Observational and randomised controlled trials (RCTs) were included in this review; however, review articles, case reports, technical reports and letters to editors were excluded. Original articles were included if they were published in a peer-reviewed journal or available in a database, and the full text was available, whereas published abstracts were excluded. Any control intervention was accepted for RCTs.
Outcome measures
This review analysed postoperative and follow-up outcome measures to assess the effectiveness and safety of the treatment approach. The primary outcome was fracture healing time, as it directly reflects treatment success. Secondary outcomes included radiological outcomes, functional outcomes, complication rates, adverse effects, operative time, fracture displacement rate and length of hospital stay. These secondary measures provided further insight into the overall safety, efficiency and success of the treatment method. The average selling price of the BIN implant was obtained directly via personal communication with a company representative.
Data extraction and synthesis of results
Data for patient demographics and outcome measures were input into a spreadsheet. Where possible, meta-analysis was conducted using a random-effects model to assess primary outcomes and inverse variance meta-regression to identify predictors of poor outcomes. Meta-analyses and meta-regression were conducted by an author (FR) holding formal qualifications in biostatistics and surgical research, using Stata software (StataCorp LLC, Texas). 12
Heterogeneity was assessed using the I2 statistic, which describes the proportion of variation in effect sizes that is due to heterogeneity rather than chance; a low value indicates little variation between the results of the studies.
Risk of bias
Risk of bias was assessed using the ROB-2 tool for Risk Of Bias In Randomized Studies of Interventions 13 and ROBINS-I checklist for observational studies, 14 in accordance with the Cochrane Handbook for Systematic Reviews of Interventions. 10
Publication bias
Funnel plots were generated to assess the likelihood of publication bias. During the search and screening process, authors aimed to identify any study protocols for which no results were published, which could indicate potential publication bias. A brief search of clinicaltrials.gov was conducted to identify any registered trials that had not been completed.
Results
Search results
A total of 613 records were identified in the searches, after duplicates were removed. Of these, 13 full-text articles were screened for eligibility, with five15–19 being suitable for inclusion as shown in Figure 1. Of the eight excluded studies, four were excluded primarily due to being the wrong study design or publication type,20–23 and four due to having the wrong population for inclusion in this review, with no paediatric forearm fractures.24–27
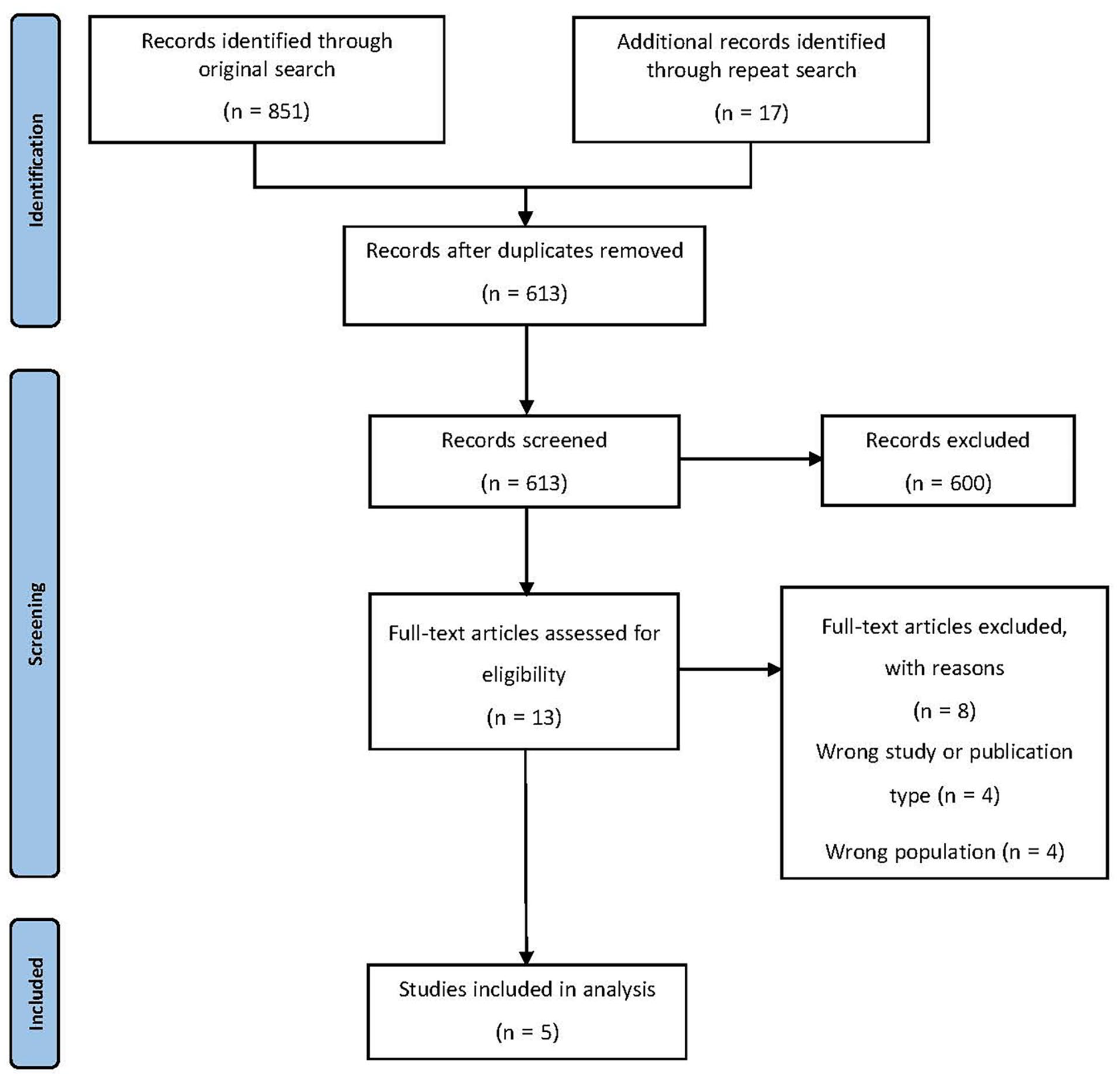
PRISMA flow diagram.
Study characteristics
A total of 255 paediatric patients with 399 forearm fractures were included in analyses, 159 of whom were treated using bioresorbable PLGA intramedullary nails. Control interventions included ESIN (n = 32), K-wires buried under the skin (n = 40) or non-buried K-wires (n = 24). Participants’ ages ranged from 4 to 15 years, and the mean age of participants across all included studies was 9.10 (SD 2.42). Where this was reported, most participants were male. Two of the included studies were controlled trials comparing BIN with ESIN in diaphyseal forearm fractures,15,16 two were retrospective multicentre observational trials18,19 and one of which compared BIN with K-wires 19 ; the final included study was observational comparing healing outcomes for BIN using magnetic resonance imaging. 17 Table 1 shows the characteristics of included studies.
The characteristics of included studies.
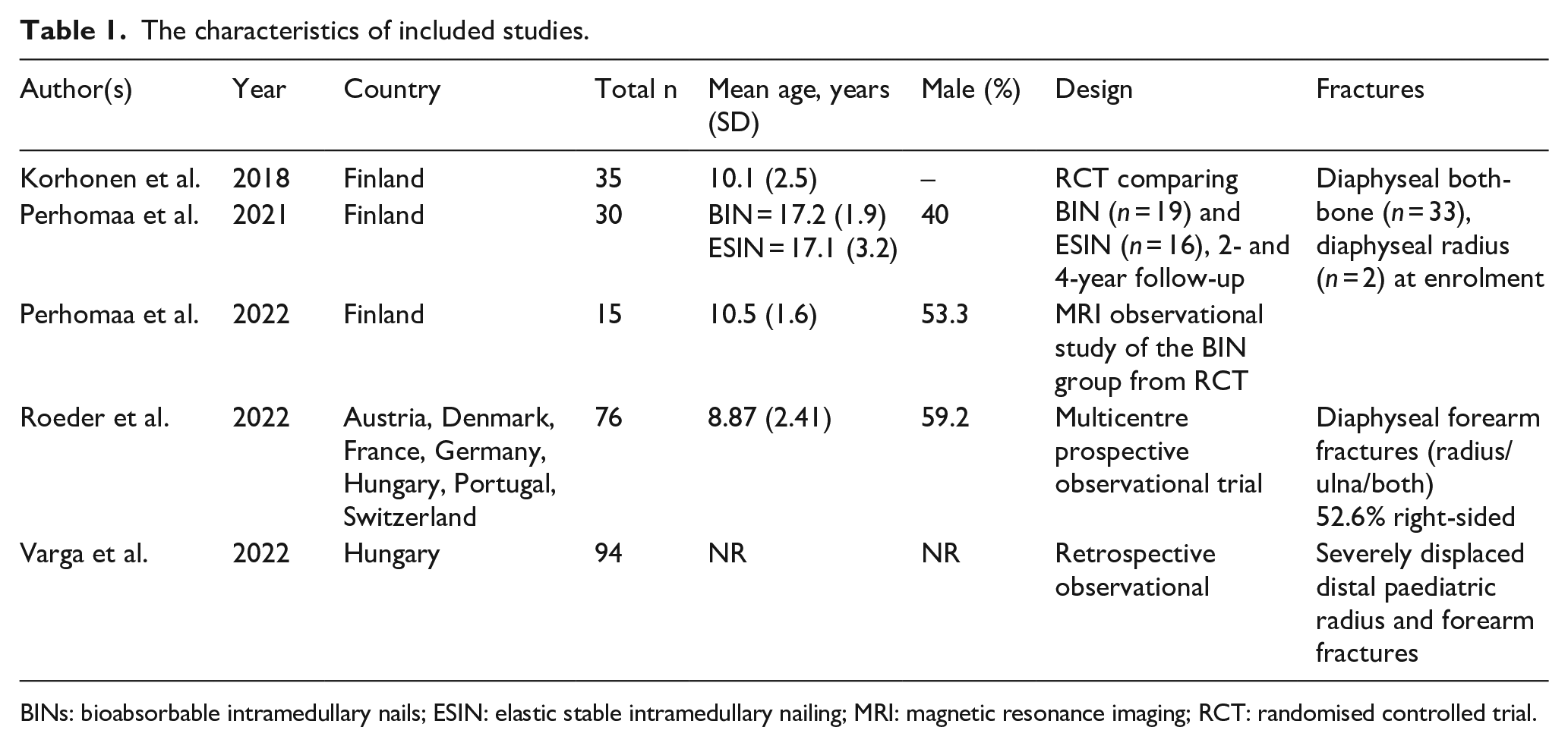
BINs: bioabsorbable intramedullary nails; ESIN: elastic stable intramedullary nailing; MRI: magnetic resonance imaging; RCT: randomised controlled trial.
Implants and surgical techniques used
The bioresorbable intramedullary nail used in all studies was the ActivaNail IM™ produced by Bioretec Ltd (Tampere, Finland): this implant is made of a PLGA polymer and has an osteoconductive β-tricalcium phosphate marker in the tip. 20 In the UK, the average selling price for these implants for 2025 is £375/implant (provided by company representative on 07 May 2025). The control groups in the RCTs used ESIN, with Synthes TEN™ implants by DePuy Synthes (West Chester, PA, USA). 15 These were removed 4–6 months postoperatively. Another study used one or two K-wires as a control intervention, which were either buried under the skin or left outside the skin; the specific K-wires used were not specified. 19 All patients’ operated limbs, regardless of implant used, were immobilised with a cast postoperatively.
Risk of bias and conflict of interest
Two controlled studies were assessed using the Cochrane Risk of Bias Tool 13 and found to have high risk of bias in only two domains, both of which related to blinding, which would be impractical due to the nature of the intervention. Three observational studies were assessed using the ROBINS Tool, 14 and found to have a moderate risk of bias overall.
One article did not report conflicts of interest 15 ; an author from one article reported being a shareholder for the company that produces the BIN implants used in all four studies, 18 and an author from two studies reported having consulted with the same company for surgical issues.16,17 Authors from the remaining article declared no conflicts of interest. 19
A search of clinicaltrials.gov found one registered trial with no published results, which had an unknown status and estimated completion date of October 2022 (NCT04848818). A further study was identified in the database search, which included results for eight paediatric patients with forearm fractures who underwent fixation using BIN; however, this study was not published in a peer-reviewed journal. 20
Findings
Fracture healing
The overall mean healing time for both BINs and traditional elastic nails (TENs) was 10.67 weeks (95% CI: 8.92–11.42) as reported in three studies,15–17 with no evidence of heterogeneity (I2 = 0%), as shown in Figure 2. Individual study results also indicated comparable radiological healing times for BINs and ESIN, with confidence intervals largely overlapping and p values suggesting no statistically significant group differences (Qb(1) = 0.00, p = 0.97).
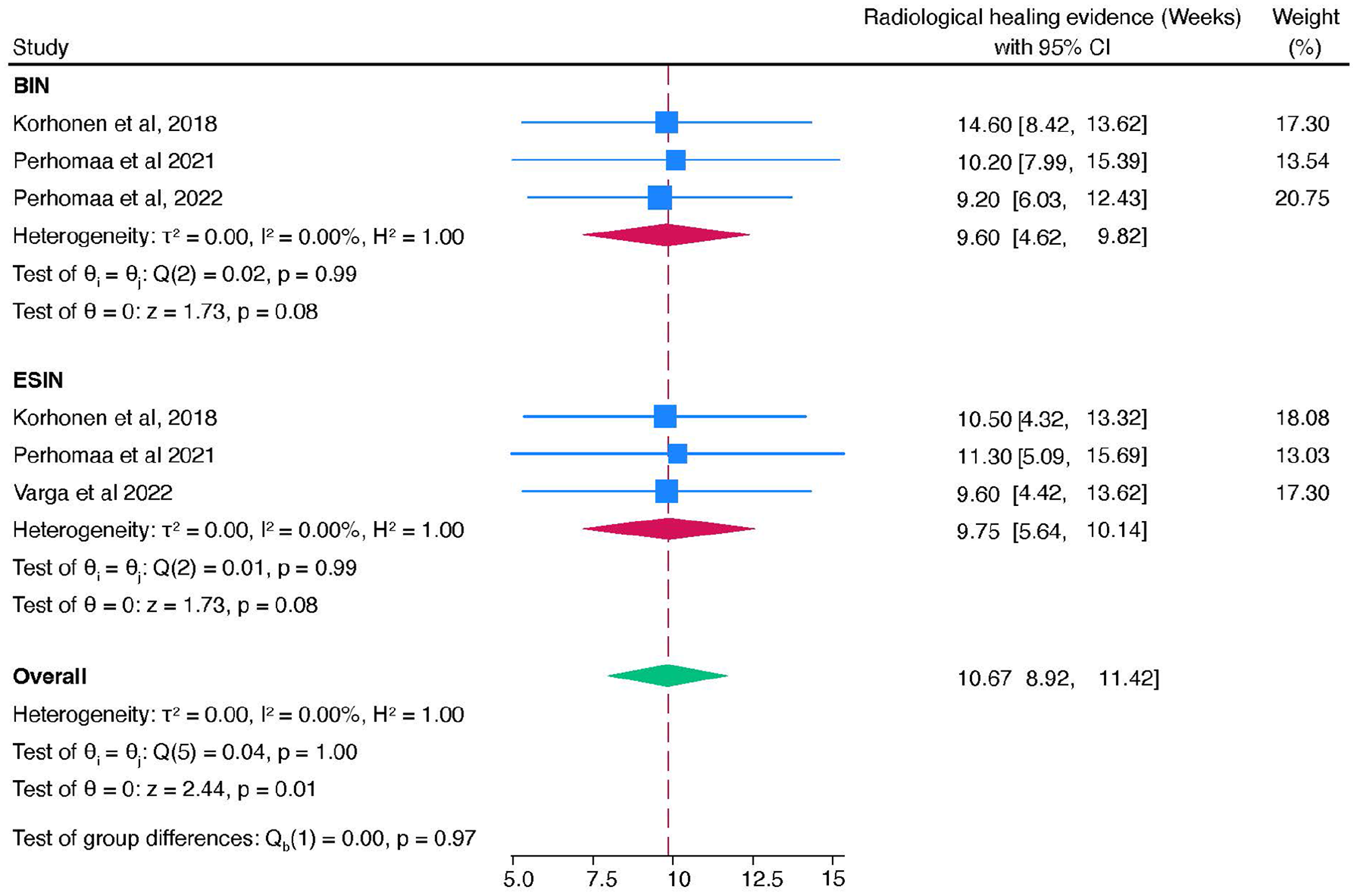
The forest plot of radiological healing time for BINs and ESINs.
Complication profiles
Of the 159 patients treated with BINs, nine experienced re-fractures. Four were due to implant failure, presenting as sudden pain without new trauma despite initially good functional and radiological results, all managed with plate and screw fixation. The remaining five resulted from new trauma after confirmed radiological healing, with one managed conservatively by cast immobilisation and the others with plate and screw fixation.
A meta-analysis comparing BINs and TENs showed no significant difference in complication rates, with a combined odds ratio of 0.39 (95% CI: 0.65–1.42) and minimal heterogeneity (I2 = 3.52%) from the included studies.15,16,19 Individual study odds ratios for BINs ranged from 0.39 to 0.85, while for TENs, they ranged from 0.65 to 1.42, as shown in Figure 3.
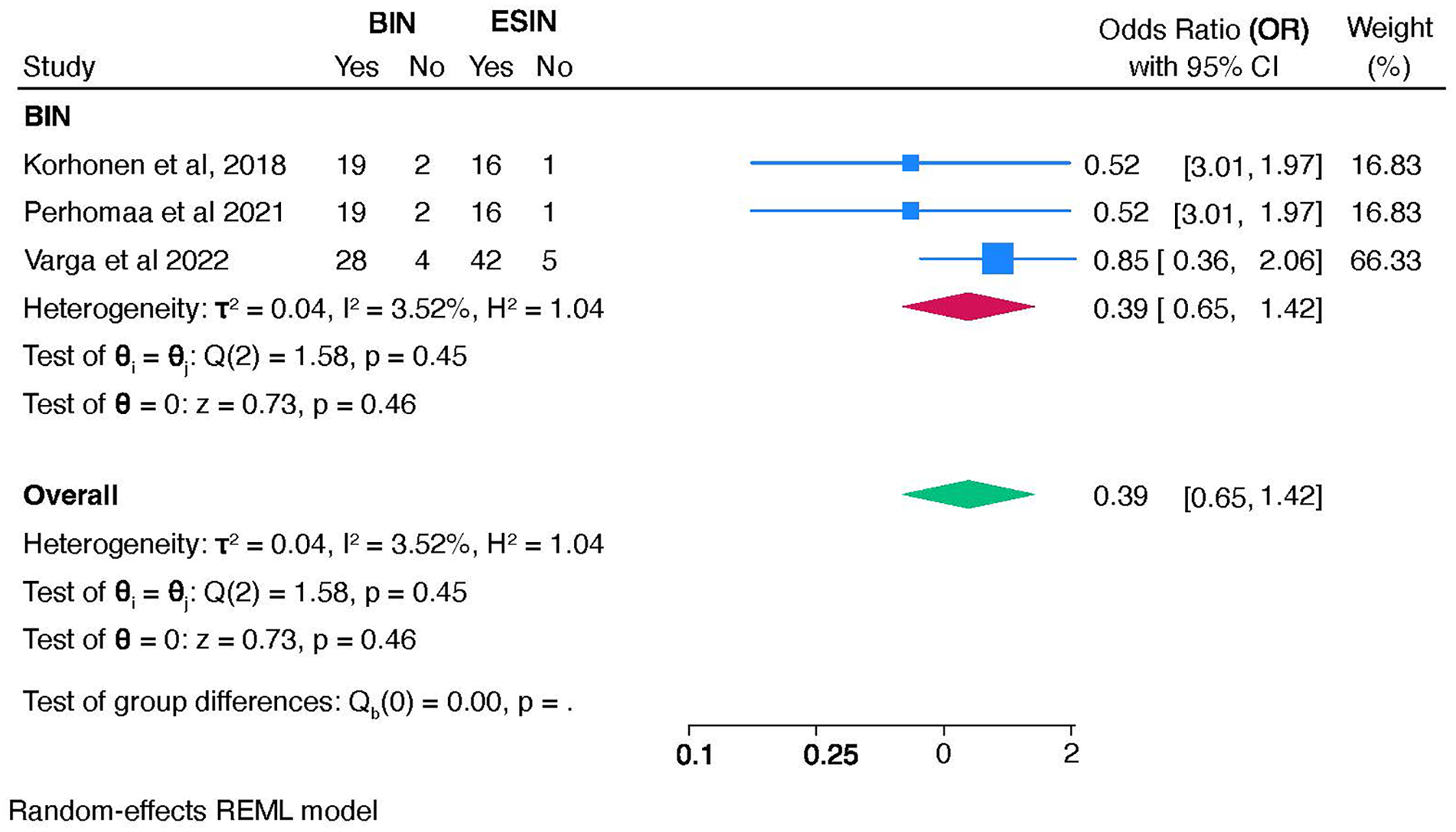
The forest plot of pooled complication rates for BINs and ESINs.
Fracture displacement
Fracture displacement rates were comparable between BINs and TENs, with an overall risk ratio of 1.08 (95% CI: 0.27–4.38) and no significant heterogeneity (I2 = 0.0%), indicating consistent findings across studies,15,16 as shown in Figure 4.
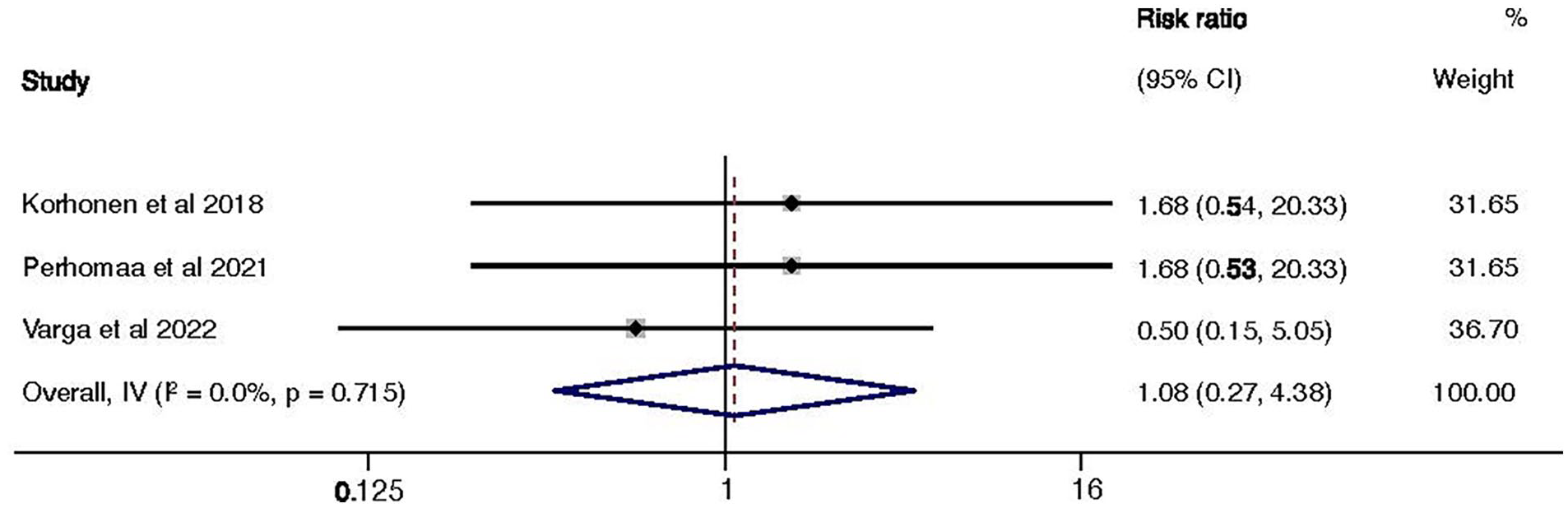
The forest plot of pooled fracture displacement rates for BINs and ESINs.
Re-operation rates
Re-operation rates were also similar, with BINs at 2.00% (95% CI: 1.12–3.21) and TENs at 3.00% (95% CI: 1.21–6.01). Both devices demonstrated low complication and re-operation rates, supporting BINs as a safe alternative that may eliminate the need for secondary implant removal.
Operative time
Two studies reported operative time.15,18 One study reported BIN to take an average of 58.9 min (±22.9 min) 18 ; in a RCT comparing BIN with ESIN, the mean operative time was 80 min for BIN and 53 min for ESIN (p = 0.014). 15
Length of hospital stay
Two studies reported length of hospital stay15,18: in an observational trial, the mean duration of hospital stay was 1.6 days (±0.8) 18 ; in an RCT comparing BIN with ESIN, there was no significant difference in length of hospital stay: the mean duration was 2.7 days for BIN and 2.58 days for ESIN (p = 0.2445). 15
Functional outcomes
Two studies evaluated functional outcomes following treatment with BINs and ESIN.15,16 One RCT found no significant differences in forearm, elbow or wrist range of motion (ROM) between groups. Another RCT, with a 6.8-year follow-up, reported significantly better ROM for BINs in pronation (78.5° versus 72.7°, p = 0.030), elbow flexion (153.8° versus 144.8°, p = 0.001) and wrist volar flexion (88.9° versus 82.7°, p = 0.023) compared to ESIN, with no differences in other ROM aspects. 16
Forearm rotation and flexion
A pooled meta-analysis of included studies15,16,19 showed slightly better forearm rotation with BINs (162.08°, 95% CI: 157.69°–166.31°) compared to TENs (158.19°, 95% CI: 153.20°–163.19°), as shown in Figure 5(a), though the overlapping confidence intervals suggest no significant difference. For forearm flexion, BINs had a mean of 152.43° (95% CI: 142.88°–161.98°) compared to 149.23° (95% CI: 142.96°–155.51°) for TENs, as shown in Figure 5(b), with minimal heterogeneity (I2 = 0%), indicating comparable outcomes.
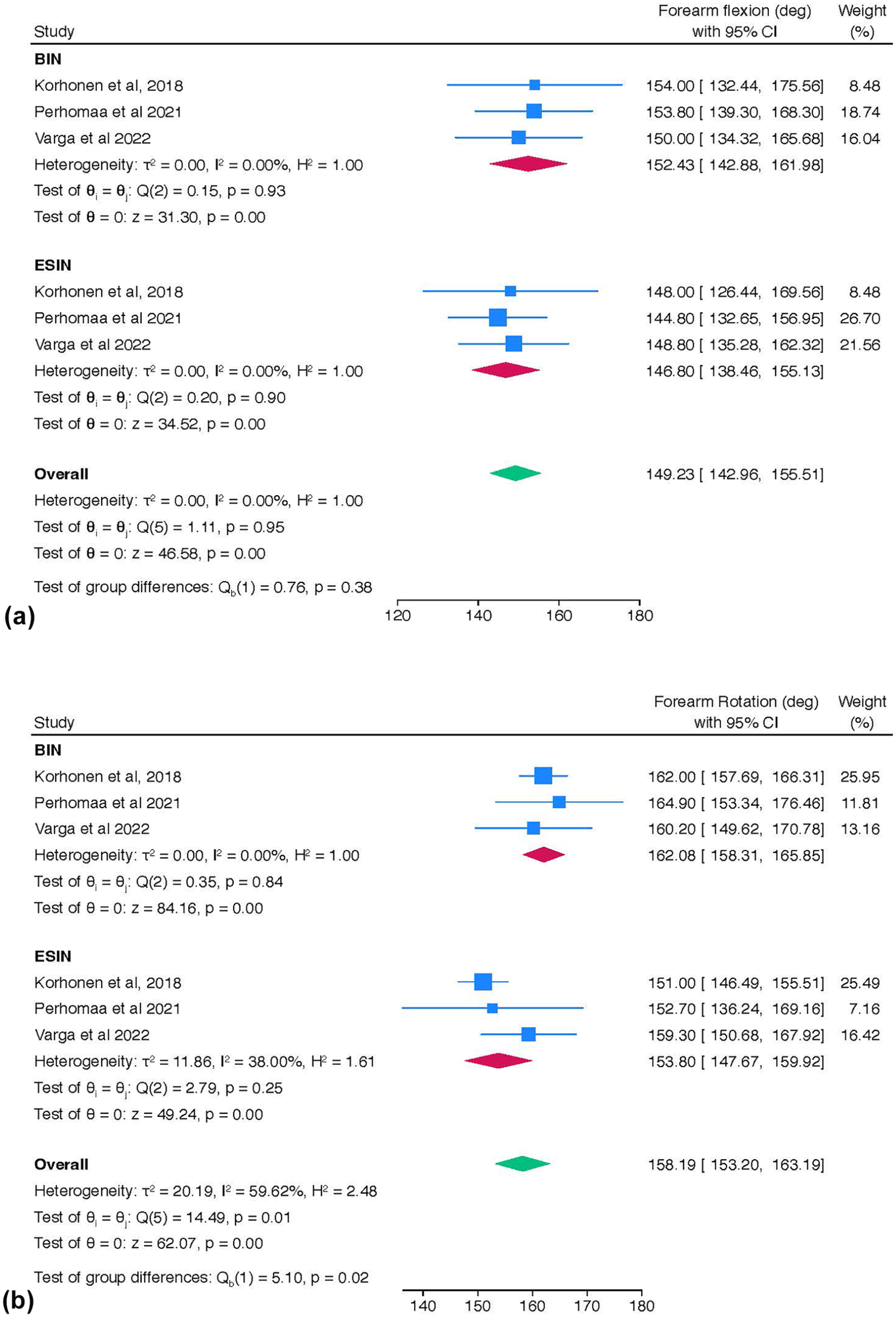
The forest plots of (a) forearm flexion comparing BINs and ESINs and (b) forearm rotation comparing BINs and ESINs.
Pain scores
Pain outcomes were similarly reported. At 2 years, one RCT noted pain in 3 of 16 ESIN patients compared to none in the BIN group, with greater postoperative pain scores for ESIN (VAS: 62.2 versus 40.9 mm), though differences were not statistically significant. 15 Another study reported pain in four ESIN patients and none in the BIN group at 4 years, which was also not statistically significant. 18 An observational study found no persistent pain or disability at 6 months in BIN or K-wire-treated patients but did not provide specific measurements. 19
Further reported complications included a superficial wound infection which was revised, with a fragment of the PLGA implant being found subcutaneously intraoperatively; a foreign body found at the fracture site at cast removal, with revision surgery subsequently being performed, removing a piece of PLGA implant; finally, a single patient was reported to have a lesion of the ramus dorsalis detected postoperatively. 16
Discussion
This systematic review included data from 255 paediatric patients with forearm fractures requiring surgical management, 159 of whom were treated using BIN. Overall, the results showed that BIN is a viable alternative in the management of these common injuries, offering comparable outcomes in terms of fracture healing, functional outcomes, complications and re-operation rates. The key advantage of BIN is that, by biodegrading over time, it obviates the need for secondary implant removal surgery, thus making it a potentially preferable option.
Functional outcomes were similar for BIN, ESIN and K-wires, including range of movement in elbow flexion, forearm rotation and wrist flexion, on the injured wide alone and compared with the uninjured side. There was no significant difference in postoperative pain levels, with no BIN patients reporting pain at follow-up.
Complication rates were comparable in the BIN group compared with current surgical methods. Of the 159 patients treated with BIN, there were nine reported re-fractures, five of which occurred secondary to a new traumatic injury after good bone healing had been previously demonstrated on plain film radiographs. Two patients required further surgery to revise the surgical wounds. Only five patients were reported to show secondary displacement at postoperative follow-up; however, all subsequently healed and remodelled satisfactorily without the need for further surgery. 18
All fractures treated with BINs demonstrated complete radiographic healing. Korhonen et al. reported full callus formation across all cortices in BIN patients and all but one patient in the ESIN group at 2 years, with no statistically significant difference between groups. 15 No residual dislocation was observed. Perhomaa et al. similarly found no significant differences in bone alignment or length between fractured and non-fractured bones in either group at 4 years. 17 The radiolucent tip of the BIN remained visible on radiographs but did not result in complications.
Secondary displacement was reported in some BIN-treated patients across observational trials.17,18 Varga et al. observed displacement in two BIN patients compared to 14 treated with K-wires; however, all fractures underwent complete radiological remodelling within 6 months without the need for reoperation. 19 Roeder et al. noted secondary displacement in 3 out of 76 BIN patients at 2 weeks, despite above-elbow casting, but no revision surgery was required, and all fractures were fully healed by the 12-week follow-up. 18
Paediatric forearm fractures are a common injury, with diaphyseal forearm fractures accounting for almost 55% of fractures requiring surgery in children. 4 Surgical management not only exposes a child to perioperative risks and complications but also places a burden on the healthcare system. Previous analyses of the cost-effectiveness of ESIN have shown metalwork removal to cost close to the same as the primary procedure; $632.76 for initial surgical management, $462.84 for metalwork removal. 7 Negating the need for removal of metalwork could potentially make BIN a more financially viable option. Further long-term studies are therefore warranted to explore this and assess the cost-effectiveness of BIN in clinical practice.
The findings of this review highlight another benefit of BINs: reducing psychological stress on paediatric patients and their families by eliminating the need for secondary implant removal surgery. 28 This avoids additional hospital visits, preoperative anxiety and disruptions to daily life, improving the overall treatment experience. Additionally, reducing the burden of care may free up healthcare resources and lower costs, making BINs a clinically effective and patient-centred alternative for managing paediatric forearm fractures. 7
This study is limited by a small body of evidence with variability in study designs, sample sizes and outcome measures. Future research should focus on long-term, large-scale RCTs to better evaluate the use of BINs against current fixation methods. Additionally, the included studies excluded key subgroups, such as patients with open fractures, multi-fragmentary fractures, significant soft tissue injuries, pathological fractures, metabolic bone diseases, systemic illnesses or fractures older than 7 days. Consequently, the findings of this review are not generalisable to these populations or injuries outside the specific demographics studied.
Conclusion
This study has shown that BINs are a viable alternative to ESIN for managing paediatric forearm fractures. BINs offer comparable outcomes in fracture healing, complication rates and re-operations, with the added benefit of biodegradability, removing the need for secondary implant removal. Functional outcomes, including ROM and pain, were similar or slightly better with BINs. While both groups experienced complications such as re-fractures and secondary displacement, all cases achieved complete radiographic healing. Further large-scale trials are needed to confirm these findings, assess cost-effectiveness and refine treatment protocols.
Supplemental Material
sj-docx-1-cho-10.1177_18632521251350854 – Supplemental material for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures
Supplemental material, sj-docx-1-cho-10.1177_18632521251350854 for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures by Florence Eastwood, Firas Raheman, Ghaith Al-Dairy, Maria Popescu, Clare Henney, Liz Hunwick and Pranai Buddhdev in Journal of Children's Orthopaedics
Supplemental Material
sj-docx-2-cho-10.1177_18632521251350854 – Supplemental material for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures
Supplemental material, sj-docx-2-cho-10.1177_18632521251350854 for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures by Florence Eastwood, Firas Raheman, Ghaith Al-Dairy, Maria Popescu, Clare Henney, Liz Hunwick and Pranai Buddhdev in Journal of Children's Orthopaedics
Supplemental Material
sj-docx-3-cho-10.1177_18632521251350854 – Supplemental material for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures
Supplemental material, sj-docx-3-cho-10.1177_18632521251350854 for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures by Florence Eastwood, Firas Raheman, Ghaith Al-Dairy, Maria Popescu, Clare Henney, Liz Hunwick and Pranai Buddhdev in Journal of Children's Orthopaedics
Supplemental Material
sj-docx-4-cho-10.1177_18632521251350854 – Supplemental material for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures
Supplemental material, sj-docx-4-cho-10.1177_18632521251350854 for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures by Florence Eastwood, Firas Raheman, Ghaith Al-Dairy, Maria Popescu, Clare Henney, Liz Hunwick and Pranai Buddhdev in Journal of Children's Orthopaedics
Supplemental Material
sj-docx-5-cho-10.1177_18632521251350854 – Supplemental material for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures
Supplemental material, sj-docx-5-cho-10.1177_18632521251350854 for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures by Florence Eastwood, Firas Raheman, Ghaith Al-Dairy, Maria Popescu, Clare Henney, Liz Hunwick and Pranai Buddhdev in Journal of Children's Orthopaedics
Supplemental Material
sj-docx-6-cho-10.1177_18632521251350854 – Supplemental material for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures
Supplemental material, sj-docx-6-cho-10.1177_18632521251350854 for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures by Florence Eastwood, Firas Raheman, Ghaith Al-Dairy, Maria Popescu, Clare Henney, Liz Hunwick and Pranai Buddhdev in Journal of Children's Orthopaedics
Supplemental Material
sj-docx-7-cho-10.1177_18632521251350854 – Supplemental material for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures
Supplemental material, sj-docx-7-cho-10.1177_18632521251350854 for Healing smarter: A systematic review and meta-analysis of bioresorbable implants for paediatric forearm fractures by Florence Eastwood, Firas Raheman, Ghaith Al-Dairy, Maria Popescu, Clare Henney, Liz Hunwick and Pranai Buddhdev in Journal of Children's Orthopaedics
Footnotes
Author contributions
PB, GA-D and FE designed the study, and GA-D wrote and registered the protocol; PB acted as supervisor. CH and LH conducted the search, and GA-D and FE conducted screening, with MP acting as the third reviewer. FE extracted data, and FE and FR analysed the data; FR conducted meta-analyses. FE and FR wrote, reviewed and edited the article, and FE, FR and PB approved the final article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
This systematic review and meta-analysis did not require ethical approval.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
