Abstract
The field of pediatric musculoskeletal imaging is undergoing significant advancements due to technological innovations and a growing emphasis on safety and patient-centered care. This review explores recent developments in imaging modalities such as advanced magnetic resonance imaging, ultrasound innovations, and artificial intelligence applications. Highlights include radiation dose-reduction techniques in radiography and computed tomography, enhanced diagnostic tools like contrast-enhanced ultrasound and ultra-high-frequency imaging, and the integration of artificial intelligence for pathology detection and workflow optimization. The adoption of advanced methods like whole-body magnetic resonance imaging and computed tomography-like magnetic resonance imaging sequences has improved diagnostic accuracy, minimized radiation exposure, and expanded the capabilities of noninvasive imaging. Emerging technologies, including photon-counting detector computed tomography and deep learning-based reconstructions, are transforming clinical practices by balancing precision and safety. Artificial intelligence applications are reshaping diagnostic approaches, automating complex assessments, and improving efficiency, although challenges such as external validation and limited scope persist. Functional imaging advancements, such as diffusion-weighted imaging and positron emission tomography-magnetic resonance imaging integration, are enhancing disease characterization and treatment planning. This review underscores the clinical impact of these innovations, emphasizing the need for standardized protocols, interdisciplinary collaboration, and continued research to address unmet needs in radiation safety and artificial intelligence integration. It aims to equip healthcare professionals with the knowledge to leverage these advancements for improved outcomes in pediatric musculoskeletal care.
Keywords
Introduction
The field of pediatric musculoskeletal imaging is advancing rapidly, driven by technological innovation, deeper insights into disease pathophysiology, and an evolving focus on patient-centered care. Imaging plays a pivotal role in diagnosing and managing a wide array of conditions affecting the pediatric musculoskeletal system, including congenital abnormalities, traumatic injuries, inflammatory disorders, and neoplastic diseases. Given the unique aspects of pediatric anatomy and disease presentation, imaging strategies tailored to this population are indispensable.
Recent developments in imaging modalities, such as advanced magnetic resonance imaging (MRI) techniques, novel ultrasound applications, and artificial intelligence (AI)-based tools, are reshaping the diagnostic landscape. These innovations not only enhance diagnostic accuracy but also prioritize safety by reducing radiation exposure and enabling noninvasive assessment. In addition, the incorporation of standardized imaging protocols and reporting systems, particularly for conditions like juvenile idiopathic arthritis and articular infections, is improving interobserver reliability and facilitating collaborative research.
This review highlights key advancements in pediatric musculoskeletal imaging, emphasizing their clinical impact and potential to improve outcomes in this vulnerable population. By exploring what’s new, we aim to provide radiologists, pediatricians, and healthcare professionals with an up-to-date perspective on the evolving capabilities and future directions in this dynamic field.
Radiography
Recent advancements in pediatric radiography focus on minimizing radiation exposure through advanced imaging techniques like digital radiography and dose-optimization protocols. Innovations include machine learning for enhanced image quality with lower radiation exposure doses and the development of age-specific guidelines for imaging indications. These improvements ensure accurate diagnosis while prioritizing the safety and unique physiological needs of children.
EOS imaging
EOS® imaging is an advanced technology that provides high-quality, low-dose biplanar radiography and three-dimensional (3D) reconstructions, making it particularly suited for pediatric musculoskeletal imaging.1,2 The system enables whole-body imaging, fast scans, and highly reproducible measurements, with applications extending to conditions like scoliosis and other spinal deformities.1,2
One of its most significant advantages is the substantial reduction in radiation exposure compared to conventional radiography and computed tomography (CT) scans.2,3
These ultra-low-dose options are critical for children requiring repeated imaging, such as during the ongoing assessment and follow-up of scoliosis, helping to minimize the long-term risks of radiation exposure.2,3
EOS is particularly valuable in pediatric settings due to its ability to provide reliable Cobb angle measurements and 3D reconstructions for evaluating spinal alignment.2,3
While standard-dose protocols remain the most accurate for detailed assessments, the reduced-dose options are ideal for preliminary screening and frequent follow-ups, offering a safer and more effective alternative for managing spinal and musculoskeletal conditions in children.1 –3
Ultrasound
Ultrasound (US) remains a cornerstone in pediatric musculoskeletal imaging due to its accessibility, absence of ionizing radiation, and capability for dynamic, real-time assessment.4,5
Recent technological advancements, such as contrast-enhanced ultrasound (CEUS), elastography, and ultra-high-frequency ultrasound (UHFUS), have significantly expanded the diagnostic potential of US. These innovations are transforming the evaluation and management of musculoskeletal disorders in the pediatric population.4 –8
Contrast-enhanced ultrasound
CEUS is an invasive ultrasound technique, which utilizes microbubble contrast agents confined to the vascular system to enhance visualization of tissue perfusion and vascular abnormalities.4,6,7 It provides superior detail compared to traditional grayscale and Doppler imaging in various pediatric musculoskeletal conditions.4,6
Juvenile idiopathic arthritis: Advances in juvenile idiopathic arthritis management have shifted toward early aggressive interventions. Although the use of CEUS in pediatric inflammatory arthropathies is less established than in adults, it may enable the detection of early synovial hypervascularity and monitoring of treatment responses more accurately than conventional methods. 6
Legg–Calvé–Perthes disease: In Legg–Calvé–Perthes disease, increased physeal vascularity is a poor prognostic factor, potentially leading to premature bony bridge formation and growth plate disruption. 6 While color/power Doppler US can demonstrate vascular flow in the deep capsular vessels of the femoral head, its utility for visualizing physeal and intraosseous vessels is limited by bone development stages. CEUS aids in visualizing physeal and intraosseous revascularization during healing, identifying children at risk of premature physeal closure, and correlating enhancement ratios with scintigraphic revascularization stages. This provides a radiation-free method for monitoring disease progression. 6 Emerging applications, such as 3D CEUS, may further improve anatomical visualization and prognostication. 6
Developmental hip dysplasia: For children requiring surgical hip reduction, intraoperatively, CEUS can detect decreased femoral head perfusion, enabling surgeons to adjust hip positioning and prevent necrosis. Studies show CEUS reliably detects vascular flow within the physis, epiphysis, and ossified nucleus, with findings comparable to post-reduction MRI. 6
Soft tissue masses: While pediatric studies on CEUS for soft tissue masses are lacking, its growing use suggests future utility in this field. CEUS may aid in the evaluation of morphological features, including size, margins, vascularity, and internal reflectivity, helping differentiate benign from aggressive masses and identify optimal biopsy sites while avoiding necrotic areas.6,7
The limitations of CEUS include operator variability, reproducibility challenges, and image quality affected by body habitus. 7 As a relatively new technique, further standardization, validation, and education are needed for its routine clinical integration. 4
Sonoelastography
Sonoelastography quantifies soft tissue elasticity noninvasively, with shear wave elastography (SWE) being particularly notable for its reproducibility and objectivity. 4 SWE measures tissue stiffness by analyzing shear wave speed, aiding in diagnosing and monitoring pediatric rheumatologic conditions. 4 Clinical applications include detecting skin stiffness in systemic sclerosis, correlating stiffness with tendon pain and dysfunction, 4 and identifying meniscal changes in obese children caused by mechanical stress. 8
Research in adults has demonstrated SWE’s utility in identifying abnormal muscle stiffness in inflammatory myopathies, warranting further pediatric studies. 4
Ultra-high-frequency ultrasound
UHFUS, with frequencies up to 70 MHz and resolutions as fine as 30 μm, offers exceptional detail for imaging superficial and small structures, such as soft-tissue lesions and vascular malformations. 5
UHFUS is particularly valuable for evaluating soft-tissue lesions, including pilomatricomas, dermoid cysts, lipomas, nodular fasciitis, and ganglion or synovial cysts, offering enhanced clarity and improved diagnostic confidence compared to conventional high-frequency ultrasound. 5
It can also aid in the detection of tiny phleboliths in vascular malformations and small cystic spaces in lymphatic malformations. 5
The primary limitation of UHFUS is its shallow penetration depth (up to 23.5 mm), restricting its use to superficial structures. In addition, UHFUS is sensitive to movement, with maneuverability and image quality often requiring a two-operator technique. 5
Computed tomography
CT has been a cornerstone of medical imaging since its introduction in the 1970s, recognized for its diagnostic accuracy, rapid acquisition times, widespread availability, and cost-effectiveness. 9 Despite these advantages, concerns about radiation exposure remain significant, as CT delivers higher doses of radiation compared to most other diagnostic imaging modalities.9 –11
Although CT accounts for only 17% of radiological procedures in the United States, it contributes nearly 50% of the collective effective radiation dose. This disproportionate contribution raises concerns about radiation-induced carcinogenesis, particularly in pediatric imaging. Children are more radiosensitive due to their smaller body size, rapidly dividing cells, and longer lifespan, making radiation dose reduction critical, especially in settings without dedicated pediatric CT protocols.9 –11
Large-scale studies have confirmed an increased cancer risk associated with CT radiation in children. 9 Research suggests that reducing CT doses by 20%–40% could potentially lower future cancer incidence by the same percentage. 9 These findings underscore the importance of adhering to the as low as reasonably achievable (ALARA) principle and implementing stringent radiation safety measures.9,12
Advanced reconstruction techniques
Efforts to minimize radiation exposure have driven advancements such as automated exposure control, lower tube voltage and current settings, high-pitch acquisition, and iterative reconstruction (IR) algorithms.9,11
IR algorithms are advanced image processing techniques used in pediatric CT imaging as a replacement for traditional filtered back projection (FBP). Unlike FBP, which reconstructs images in a single step, IR algorithms perform multiple computationally complex iterations, progressively refining the image through statistical modeling and noise reduction to enhance quality and reduce radiation dose. 10
Adaptive statistical iterative reconstruction (ASIR): ASIR is a software-based approach that employs multiple computational steps to improve image quality using a statistical model of noise. It has been shown to effectively reduce radiation doses for pediatric lower-extremity CT scans. 10 ASIR can decrease the CT dose index by 8% and the dose length product by 30%, although it may compromise image sharpness. 10
Model-based iterative reconstruction (MBIR): MBIR builds upon ASIR by offering enhanced noise reduction and low-contrast detectability. However, its computational intensity makes it time-consuming, limiting its application in routine clinical practice.10,11
Deep learning-based reconstruction: This emerging technology further improves image quality and processing speed, enabling radiation dose reductions while maintaining diagnostic accuracy. However, its efficacy in imaging high-contrast structures, such as bone, requires further investigation. 11
Similarly, combining low tube voltage settings with IR algorithms has proven effective in further decreasing radiation exposure while preserving adequate contrast resolution for musculoskeletal evaluations. Challenges such as increased image noise, metallic artifacts, and variability in scanner performance necessitate careful protocol optimization for musculoskeletal imaging. 9
Emerging technologies
Photon-counting detector CT (PCT): PCT represents a groundbreaking advancement in pediatric imaging. 11 By utilizing smaller detector pixels and individual photon counting, PCT achieves ultra-high-resolution imaging with reduced noise. 11 Its energy discrimination capability removes low-energy photons responsible for electronic noise, enabling the development of ultra-low-dose CT protocols, particularly advantageous for children. 11 Compared to traditional CT systems, PCT has demonstrated a 31%–47% reduction in radiation dose while maintaining or improving image quality in musculoskeletal examinations. 12 PCT also enhances spatial resolution and facilitates material-specific imaging, providing superior visualization of both bone and soft-tissue structures.11 –13 In addition, its rapid acquisition times often eliminate the need for sedation in younger patients, further improving safety. 11
The integration of PCT with deep learning-based image reconstruction holds great promise for further dose reductions and enhanced diagnostic accuracy. While studies in adults highlight the advantages of PCT, additional research is needed to validate its specific applications and benefits in pediatric musculoskeletal imaging. 12
Dose tracking and monitoring: Cloud-based dose-tracking systems are valuable tools for enhancing radiation safety by monitoring patient exposure and standardizing imaging protocols across institutions. These systems refine imaging practices but face challenges, including cost and data security concerns. 11
The integration of IR algorithms, PCT, and dose-tracking technologies exemplifies the balance between optimizing patient safety and achieving diagnostic precision. Continued research and innovation will further refine these technologies, expanding their clinical applicability and addressing operational challenges. 13
Magnetic resonance imaging
MRI remains a fundamental modality in pediatric musculoskeletal imaging due to its superior soft tissue contrast and lack of ionizing radiation. Recent advancements in MRI technology have further enhanced its role, particularly with the advent of whole-body MRI (WB-MRI) and CT-like MRI sequences.
Whole-body MRI
WB-MRI is an advanced imaging modality that allows comprehensive evaluation of the entire body in a single scan, providing both anatomical and functional data without radiation exposure. 14 Over the past two decades, its applications have expanded to encompass a wide range of pediatric musculoskeletal disorders, particularly multifocal and multisystem diseases requiring frequent follow-up, minimizing long-term health risks associated with ionizing radiation.14 –16
Technical innovations have significantly improved WB-MRIs feasibility in routine clinical practice. 16 Higher magnetic field strengths and sophisticated hardware, such as multi-channel surface coils and integrated body coils, have enhanced image quality and resolution. Optimized pulse sequences and image reconstruction algorithms have reduced scan times while maintaining diagnostic accuracy.16,17
While there is no standardized WB-MRI protocol, most scans include coronal short tau inversion recovery (STIR) and T1-weighted sequences. STIR is particularly sensitive to fluid and edema,14,15 while T1-weighted sequences provide detailed anatomical information. 17 Diffusion-weighted imaging (DWI) with background suppression is increasingly used for detecting bone lesions, though it requires careful interpretation to distinguish normal high-cellularity tissues from pathology. 17
Clinical applications of WB-MRI
WB-MRI is now employed for various pediatric conditions:
Chronic recurrent multifocal osteomyelitis (CRMO): WB-MRI helps in both diagnosing CRMO and in assessing the overall disease load and distribution (Figure 1). It is essential for detecting both symptomatic and asymptomatic bone and periarticular involvement, aiding in diagnosis and treatment monitoring. 15
Lymphoma: WB-MRI, particularly with DWI,14,15 is useful for staging and follow-up by identifying nodal and extranodal involvement as well as bone marrow infiltration. 16 It offers a radiation-free alternative to CT or positron emission tomography (PET)/CT, although PET/CT remains favored for differentiating benign from malignant lymphadenopathy. Emerging evidence suggests PET/MRI is a promising tool. 15
Metastasis: WB-MRI aids in detecting skip lesions and metastases in pediatric malignancies like osteosarcoma and Ewing sarcoma, crucial for staging and treatment planning.15,17 It has shown superior detection of skeletal metastases compared to conventional imaging (such as CT, conventional MR, bone scan, FDG-PET), though its role in extraskeletal metastasis detection remains debated. 17
Cancer predisposition syndrome surveillance: WB-MRI is a valuable screening tool for children with genetic syndromes like Li-Fraumeni syndrome, neurofibromatosis type 1&2, and Beckwith–Wiedemann syndrome, offering high diagnostic accuracy (100% sensitivity, 94% specificity, 25% positive predictive value, and 100% negative predictive value) 17 and eliminating radiation exposure.15,17 For example, in the largest study involving Li-Fraumeni syndrome, WB-MRI detected new malignant neoplasms in 7% of participants. 17
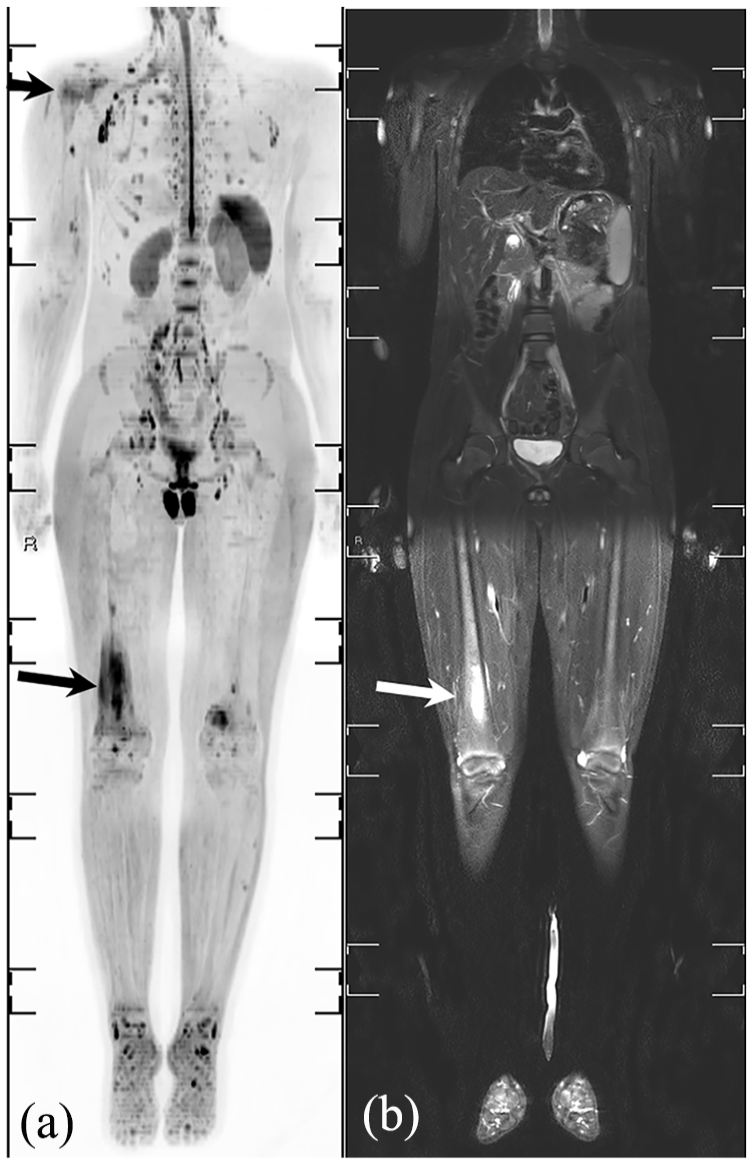
Whole-body MRI of a 13-year-old patient with CRMO. Coronal DWIBS (a) and T2-weighted with fat suppression (b) images demonstrating an abnormal signal representing osteitis in the proximal right humerus (short arrow on a), and in the distal right femoral diaphysis (long arrows on a and b).
The use of WB-MRI presents challenges such as long scan times, which may necessitate sedation in young children, increasing complexity and cost.14,16,17 Variability in protocols and a lack of standardized reporting further limit reproducibility and large-scale studies. 14 Cost, billing, and reimbursement remain significant issues, particularly in countries like the United States, where private insurance may cover WB-MRI but there is no consensus on a billing code for this study. 16 Furthermore, there is limited information on the reliability and validation of WB-MRI findings, underscoring the need for further research to establish its diagnostic consistency. 14 Research focuses on optimizing consistent protocols across institutions, exploring AI-based image reconstruction to shorten scan times,15 –17 and integrating PET/MRI for enhanced diagnostic accuracy.16,17
CT-like MRI sequences
MRI traditionally is limited in the evaluation of densely organized musculoskeletal tissues such as the bone cortex. New sequences like ultra-short echo time, zero echo time, and deep learning techniques provide images that closely resemble those of CT without the associated radiation. 11 These advancements allow detailed visualization of cortical bone and structural lesions, with high agreement between MRI and CT for detecting abnormalities like bone tumors and spinal defects. 11
The generation of synthetic CT-like images from MRI has expanded its utility in pediatric musculoskeletal imaging. Studies have demonstrated that CT-like MRI sequences are comparable to conventional CT for evaluating bone shape and cortical morphology in various anatomical regions. 11 These include the shoulder (e.g., identifying and measuring bony Bankart lesions (Figure 2) and Hill-Sachs defects), hip (e.g., femoroacetabular impingement syndrome), and sacroiliac joints (e.g., detecting erosions, sclerosis, and ankylosis, Figure 3). In addition, these sequences are effective for identifying pars defects and neural foraminal narrowing in the spine. 11
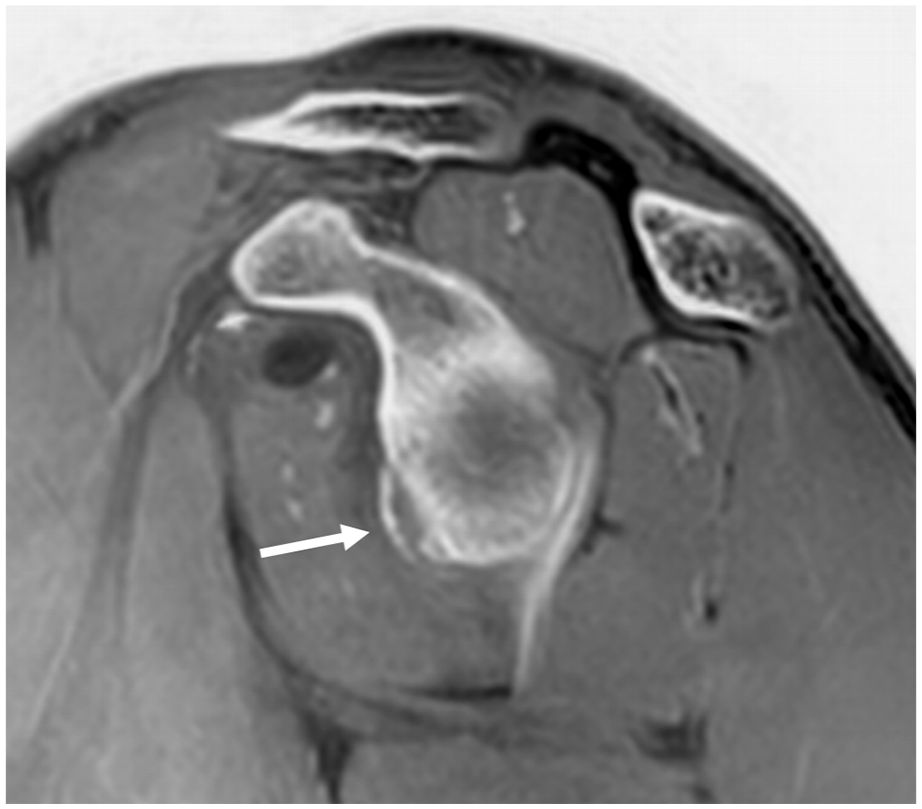
Synthetic CT-like sagittal image of the shoulder generated from a dedicated MR arthrogram sequence of a 17-year-old boy following anterior shoulder dislocation, illustrating a bony Bankart lesion in the anteroinferior aspect of the glenoid (arrow).
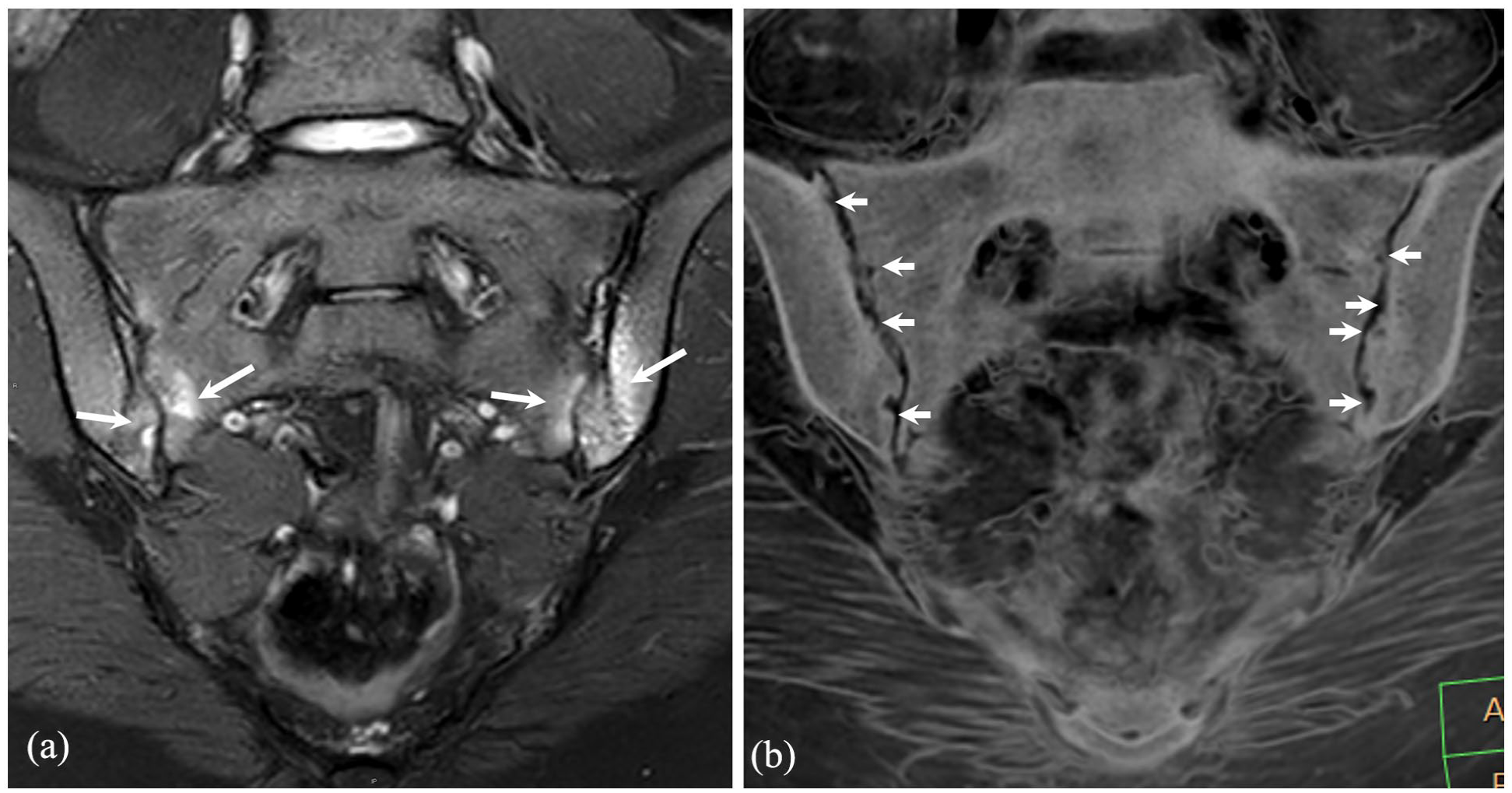
Semicoronal T2-weighted with fat suppression (a) and synthetic CT-like image generated from dedicated MR sequences (b) of a 15-year-old patient with ulcerative colitis and sacroiliitis. Bilateral extensive periarticular bone marrow edema is seen on the T2-W image (arrows on a) while bilateral multiple small erosions are observed on a CT-like image (arrows on b).
CT-like MRI has also been used as a substitute for conventional CT in craniofacial imaging and for characterizing both benign and malignant bone tumors. 11 There is a strong concordance between CT-like MRI images and conventional radiographs for evaluating destruction patterns and periosteal reactions and distinguishing between aggressive and nonaggressive tumors. These sequences also provide detailed insights into soft tissue extension and tumor architecture, further enhancing their clinical utility. 11
Emerging MRI applications
Innovative MRI techniques show promise in pediatric musculoskeletal imaging:
Quantitative imaging biomarkers: Techniques like diffusion tensor imaging and dynamic contrast-enhanced MRI provide insights into tissue properties and therapeutic response. 17
MR-guided high-intensity focused ultrasound: Used for pain palliation in metastatic bone lesions and treatment of desmoid tumors 17 with emerging applications in osteoid osteoma treatment. 17
Dynamic contrast-enhanced MRI: Valuable for assessing chemotherapy response with discrimination of viable tumor from necrotic one and distinguishing recurrent soft tissue sarcoma from postsurgical scarring. 17
Diffusion tensor imaging: Offers detailed evaluation of peripheral nerve tumors and their relationship to adjacent structures, aiding surgical planning. 17
Non-contrast perfusion imaging and MR spectroscopy (MRS): Techniques like arterial spin labeling and oxygen-enhanced MRI assess tumor perfusion and hypoxia, while MRS evaluates biochemical tissue properties, including choline metabolism in malignancies. 17
Perfusion MRI serves as an effective tool for evaluating femoral head hypoperfusion in the early stages of Legg–Calvé–Perthes disease. A strong correlation was identified between the extent of early-stage hypoperfusion and the severity of femoral head deformity at intermediate-term follow-up. These results highlight the critical role of early hypoperfusion assessment in predicting long-term outcomes. 18
These advancements highlight MRI’s evolving role in pediatric musculoskeletal imaging, with ongoing efforts to improve diagnostic accuracy, reduce scan times, and expand its clinical applications.
Artificial intelligence
AI is transforming radiology, particularly in pediatric and musculoskeletal imaging, with applications in image enhancement, measurements, and pathology detection. These advancements improve diagnostic accuracy and efficiency, especially for less experienced radiologists. However, challenges like reliance on retrospective data, small test cohorts, and limited external validation hinder widespread application. 19
In musculoskeletal imaging, AI supports three core tasks: detecting abnormalities, quantifying disease severity, and characterizing findings. By reducing cognitive workload, AI allows radiologists to focus on complex diagnostic tasks. 20
AI quantification applications
Bone age assessment in radiographs: Bone age, assessed via hand and wrist radiographs, indicates skeletal maturity using methods like Greulich–Pyle and Tanner–Whitehouse.21,22 These manual processes are time-intensive and variable. AI tools now automate bone age assessments, showing accuracy comparable to or better than clinicians in recent studies.21,22
Leg length discrepancy (LLD) and leg alignment measurement in radiographs: LLD, a condition affecting 40%–70% of the population, is best evaluated using standing AP radiographs. Angular deformities in children are assessed using parameters like the hip-knee-ankle angle (HKAA) from a full-length AP radiograph. AI algorithms for this assessment reduce measurement time and variability by analyzing anatomical landmarks like the femoral head and tibial plafond23,24 and automating HKAA calculations to enhance workflow efficiency and diagnostic consistency. 25
AI detection applications
Fracture detection in radiographs: Pediatric fractures, often challenging to diagnose due to unique bone structures, are a major source of diagnostic errors.26 –28 AI has proven effective in detecting subtle fractures and aiding radiologists, particularly in emergency settings (Figure 4).27,28 It enhances diagnostic accuracy for residents and reduces interpretation time, matching subspecialist performance in some cases. 26
Additional AI detection applications: AI technologies are demonstrating transformative potential in various medical imaging applications. In ultrasound diagnostics, AI aids in detecting developmental dysplasia of the hip, a common congenital condition diagnosed using Graf’s method, by automating key measurements and classifications, thereby reducing high inter-observer variability and improving clinical consistency. 29 Similarly, AI enhances lumbar lesion detection in SPECT (Single-Photon Emission Computed Tomography) imaging, particularly for pediatric athletes suffering from low back pain caused by stress injuries, where it has even outperformed experts in identifying small active lesions, enabling earlier diagnoses. 30 Moreover, in the context of bone malignancies, AI algorithms analyzing knee radiographs show promise in detecting suspicious lesions, offering a valuable tool for early and accurate tumor diagnosis in children. 31
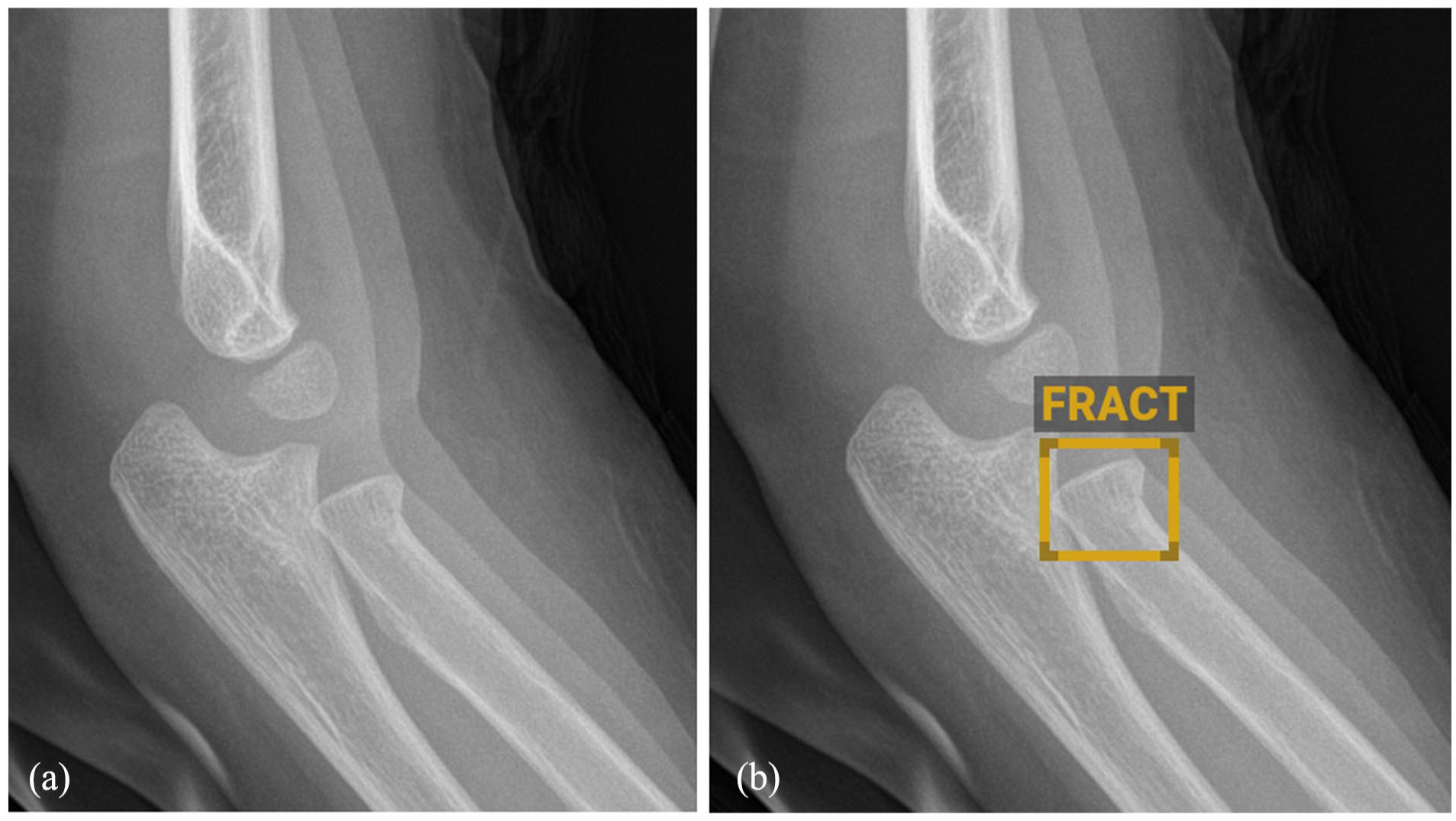
A lateral elbow radiograph (a) and an AI system evaluation (b) of the same radiograph of a 3.5-year-old girl following a fall on an outstretched hand showed a subtle proximal radius fracture highlighted with a box by the AI analysis.
AI characterization applications
Tumor and infection differentiation: Initial steps are being taken in applying AI to differentiate between conditions like Ewing sarcoma and osteomyelitis, which require distinct treatments and have different prognoses. While an AI algorithm leveraging radiographic imaging features has shown potential in distinguishing these conditions, there is currently no substantial evidence demonstrating its clinical contribution to managing musculoskeletal lesions. 32
Developing fields
Automatic bone segmentation in MRI: Segmentation serves as a core component of medical imaging, aiming to recognize and define the contours of meaningful anatomical regions. 33 Accurate bone segmentation in pediatric MRI remains challenging due to ongoing ossification and thin structures. A novel AI algorithm for multi-bone segmentation outperformed previous models and demonstrated promise for broader applications, including muscles and cartilage. This innovation could significantly improve diagnosis and treatment planning for pediatric musculoskeletal disorders. 33
Unmet needs
In pediatric musculoskeletal imaging, the need for radiation dose reduction remains a critical but relatively underexplored area compared to advancements achieved in other pediatric imaging fields such as abdominal, chest, or head CT. While AI-driven dose optimization has demonstrated significant potential—reducing radiation by up to 70% in modalities like CT and PET/MRI without compromising diagnostic quality—these innovations have primarily targeted non-musculoskeletal applications. 34 Pediatric musculoskeletal imaging lags behind, with limited research into AI solutions tailored for dose optimization in this subspecialty. Given children’s heightened sensitivity to ionizing radiation and the increasing use of imaging for musculoskeletal disorders, bridging this gap is essential to ensure safer and more effective diagnostic care. 34
AI has made strides in other medical fields, but its progress in musculoskeletal radiology is hindered by the complexity of conditions, subtle imaging abnormalities, and the need for biomechanical insight. A major limitation is AI’s narrow scope, as algorithms typically handle only a few diagnoses at a time. This contrasts with the broad range of musculoskeletal conditions, where identifying related injuries, like secondary fractures, often requires biomechanical understanding—an intuitive skill AI lacks. Musculoskeletal diagnosis also depends on recognizing how injuries in one area can affect others, a complex task human radiologists excel at but AI struggles with. 20
In addition, subtle abnormalities pose significant challenges for AI. Clinically critical injuries may present as minimal radiological findings, requiring the detection of just a few abnormal voxels among many—a task hindered by AI’s difficulty in handling low signal-to-noise ratios and the resolution loss during image processing. 20
Future efforts should focus on developing more refined AI systems that are tailored to diverse imaging modalities and a wider range of pathologies. Addressing current weaknesses, such as small sample sizes, lack of external validation, and limited clinician comparisons, is essential. Collaboration with clinical centers will also be critical to ensuring the practical applicability and seamless integration of these systems into real-world healthcare settings.
In pediatric musculoskeletal imaging, AI has the potential to significantly impact areas such as fracture detection, growth plate abnormalities, and rare skeletal disorders. Its integration can improve diagnostic accuracy, reduce variability, and provide critical support in under-resourced environments, particularly in time-sensitive and complex cases.
Conclusion
The field of pediatric musculoskeletal imaging is undergoing a transformative era, marked by innovative technologies and enhanced approaches prioritizing safety, accuracy, and efficiency. Advances in imaging modalities, including ultra-low-dose radiography, PCT, and whole-body MRI, have significantly improved diagnostic capabilities while minimizing radiation exposure. The integration of cutting-edge ultrasound techniques, such as CEUS and elastography, further broadens diagnostic horizons for complex conditions.
AI is reshaping diagnostic workflows by automating tasks, enhancing image interpretation, and offering novel tools for fracture detection, growth plate evaluation, and disease quantification. Despite these strides, challenges remain, particularly in the areas of standardization, validation, and widespread implementation. The need for interdisciplinary collaboration, robust research, and tailored protocols is paramount to address these gaps and fully harness the potential of these advancements.
Ultimately, these innovations empower healthcare providers to deliver more precise, safe, and patient-centered care, paving the way for improved outcomes in pediatric musculoskeletal imaging.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521251325122 – Supplemental material for What’s new in pediatric musculoskeletal imaging
Supplemental material, sj-pdf-1-cho-10.1177_18632521251325122 for What’s new in pediatric musculoskeletal imaging by Matan Kraus, Lihi Pertman and Iris Eshed in Journal of Children’s Orthopaedics
Footnotes
Author contributions
Matan Kraus: Validation, Investigation, Writing – Original draft.
Lihi Pertman: Validation, Investigation, Writing – Original draft.
Iris Eshed: Validation, Visualization, Writing – Reviewing and editing.
In accordance with guidelines for transparency, we disclose that OpenAI’s GPT language model was utilized in this manuscript solely for phrasing and improving textual clarity.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
There are no human participants in this article and informed consent is not required.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
