Abstract
Purpose:
To investigate robot-assisted percutaneous cannulated screw fixation for treating slipped capital femoral epiphysis, including acute, chronic, and acute-on-chronic slips. Our study included all stable and unstable slips.
Methods:
Thirty-one children with unilateral SCFE were treated from October 2019 to October 2021. All 31 patients were followed up for 12–36 months, with an average follow-up time of 24.56 ± 6.73 months. The femoral epiphysis was fixed with a percutaneous cannulated screw assisted by a surgical robot.
Results:
All 31 femoral head epiphyses underwent successful fixation in one attempt. The average operation time and bleeding were 98.25 ± 15.13 min and 21.65 ± 11.25 ml, respectively. The average distance between the actual and planned entry points was 1.13 ± 0.58 mm and 0.91 ± 0.72 mm in the anteroposterior (AP) and lateral views, respectively. The actual insertion trajectory deviated from the planned position by 3.61 ± 1.34° and 2.33 ± 1.32° in the AP and lateral views, respectively. The average fluoroscopy time was 6.56 ± 3.23 times per screw. The Non-Arthritic Hip Score improved from 28.53 ± 9.17 preoperatively to 67.34 ± 6.21 at the last follow-up (P < 0.05), and the Harris hip score improved from 46.47 ± 15.34 to 89.63 ± 7.52 (P < 0.05). The wounds healed without avascular necrosis or chondrolysis of the femoral head.
Conclusion:
Robot-assisted percutaneous cannulated screw fixation is effective for treating pediatric SCFE. Screw fixation was accurate and safe, and clinical outcomes were satisfactory.
Level of Evidence:
Level 4, Case Series.
Introduction
Slipped capital femoral epiphysis (SCFE) is a common disorder of the proximal femur in the adolescent population aged 8–15 years, affecting 1–10 per 10,000–20,000 children yearly.1 –3 SCFE is characterized by the displacement of the femoral epiphysis in relation to the femoral metaphysis due to weakness of the proximal femoral physis, with the slip usually occurring in a posterior and medial direction. 4 SCFE is associated with significant morbidity. The condition is typically classified based on stability, severity, and chronicity. Stability is determined by the patient’s ability to bear weight, with stable SCFE permitting ambulation, which unstable SCFE does not. 5 Severity is assessed using the Southwick angle, which measures the degree of epiphyseal displacement on a lateral radiograph. A slip is considered mild if the Southwick angle is less than 30°, moderate if it is between 30° and 50°, and severe if it exceeds 50°. 6 Chronicity is based on symptom duration: patients with symptoms for less than 3 weeks are classified as acute, those with symptoms for 3 weeks or more are considered chronic, and those with prolonged symptoms that have recently worsened are classified as acute-on-chronic. 7 This classification is essential for guiding treatment decisions.
Surgical intervention is the primary treatment for SCFE, with options for stable cases including fixation using a single screw or multiple screws, 8 which depends on the condition of the patient’s femoral neck. The modified Dunn procedure, through intraoperative assessment of femoral head perfusion and its relationship with postoperative single-photon emission computed tomography bone scan, has been shown to be safely implemented in stable SCFE while reducing femoral head avascular necrosis (AVN). 9 In patients with SCFE, the modified Dunn procedure significantly improves radiographic parameters, including the Southwick and alpha angles, and achieves better functional and clinical outcomes.10, 11 Despite this, many orthopedic surgeons still prefer in-situ fixation with a single central screw, which is the approach most commonly used in this study. For unstable slips, ultrasound can detect joint hemarthrosis or effusion, and urgent treatment involving open reduction, evacuation, and smooth K-wire fixation is a reliable option with a low risk of AVN. 12 The overarching goal of SCFE treatment is to stabilize the epiphysis, prevent further slip progression, and promote physeal fusion, with single screw fixation being the recommended method.8, 13
The ideal screw fixation for the epiphysis should be perpendicular to the physis in both the anteroposterior (AP) and lateral views through the center of the epiphysis, 14 achieving maximum acquisition without joint penetration. 15 Performing SCFE fixation is challenging owing to the complicated proximal femoral alignment in SCFE and the narrow safe zone available for screws.16, 17 In patients with unstable SCFE, gentle reduction is performed prior to fixation. Furthermore, multiple drilling attempts can compromise the integrity of the proximal femur by creating cavities in the femoral cortex, thereby increasing the risk of postoperative fracture. 18
Thus, precision instruments are required to improve the accuracy of screw implantation and reduce the number of drilling attempts. With robot assistance, preplanning the position of the screw on the platform is possible, and the robotic arm could help execute the treatment plan for SCFE. 19 A surgical robot reportedly achieves high operational accuracy.20, 21 With the assistance of the robotic system, we can plan a spatial center point where the AP and lateral views intersect, ensuring that the screw’s farthest point is 5 mm from the subchondral bone of the femoral head. Due to the robot’s precision, screws can be accurately placed in a single attempt in the vast majority of cases, which reduces radiation exposure from repeated imaging and minimizes bone damage from multiple fixation attempts.22, 23 This study is the first to present robot-assisted screw placement for SCFE.
Materials and methods
After obtaining institutional review board approval from Beijing Jishuitan Hospital, a retrospective review was conducted. As shown in Figure 1, we included 41 patients who underwent robot-assisted in-situ screw fixation for SCFE between October 2019 to October 2021. Children aged 8–18 years were eligible for enrollment but were excluded if they had bilateral SCFE, received previous surgical treatment, had a follow-up of < 1 year, had known or suspected underlying conditions (e.g., cerebral palsy, spina bifida, or osteogenesis imperfecta), or had an increased risk for SCFE, including renal disease, endocrinopathy, and history of growth hormone use. Ultimately, 31 patients were included in the study. The demographic and radiographic characteristics of these patients are shown in Table 1.
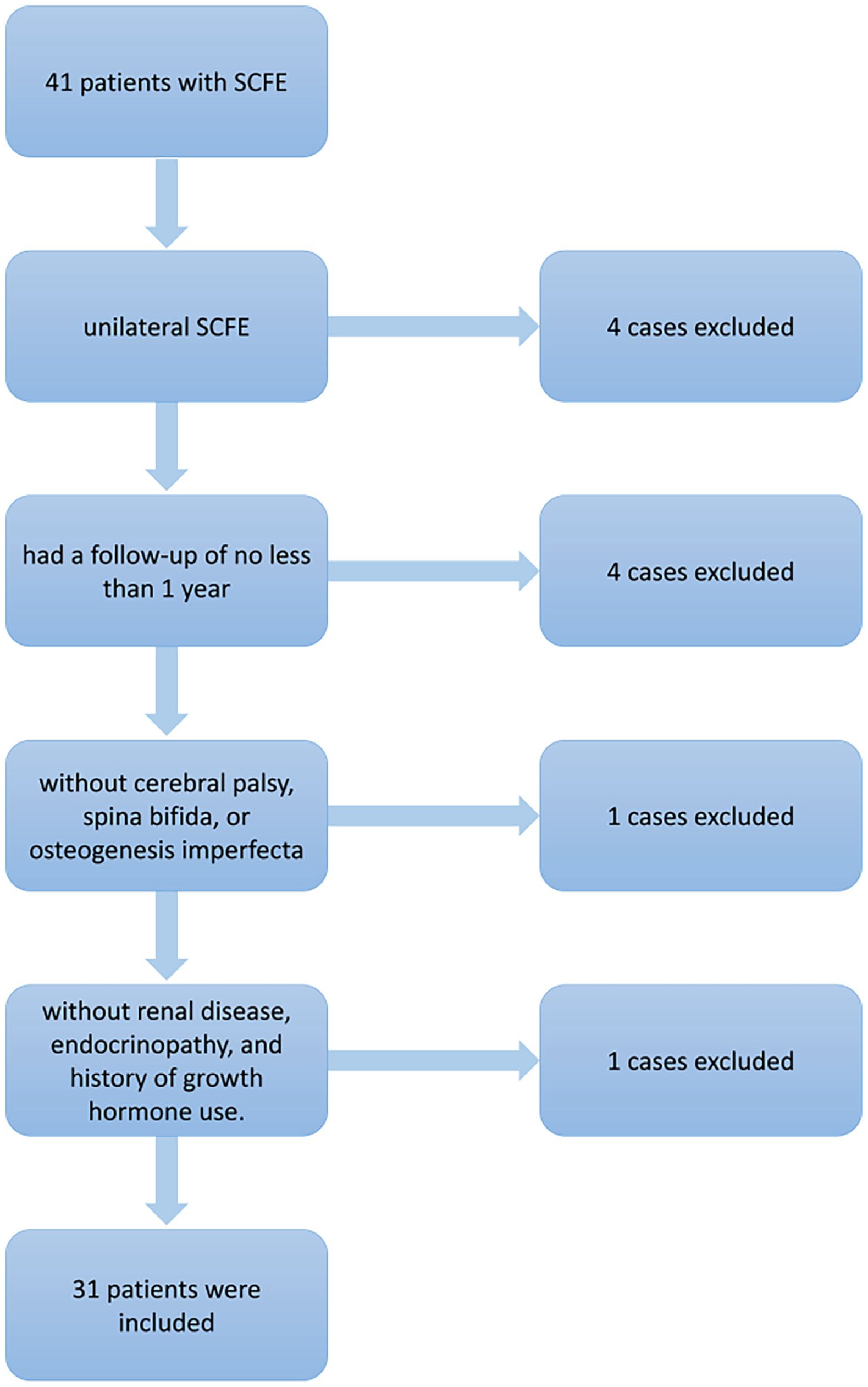
Flow diagram depicting the selection process of 31 of 41 patients for undergoing robot-assisted percutaneous cannulated screw fixation for treating slipped capital femoral epiphysis.
Diagram of the 41 patients who underwent robot-assisted screw fixation for SCFE.
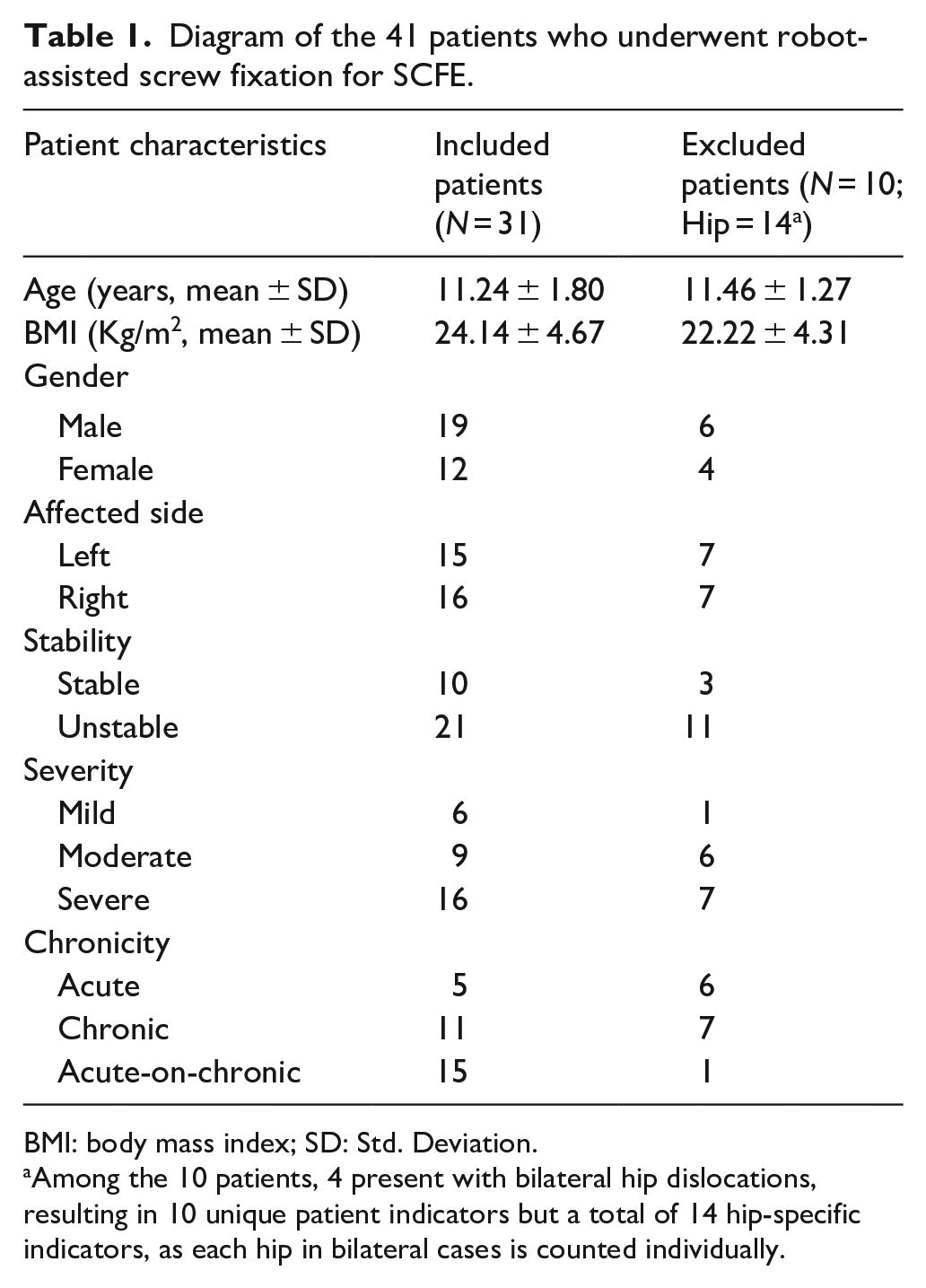
BMI: body mass index; SD: Std. Deviation.
Among the 10 patients, 4 present with bilateral hip dislocations, resulting in 10 unique patient indicators but a total of 14 hip-specific indicators, as each hip in bilateral cases is counted individually.
A new robotic system, TiRobot (TINAVI Medical Technologies, Beijing, China), assisted with screw insertion. The robot consists of a multi-degree-of-freedom robotic arm, surgical planning, controlling workstation, an optical tracking device, and positioning devices (Fig. 2). A C-arm X-ray system (Siemens, Munich, Germany) was used. After general anesthesia, the patients were placed on the orthopedic traction table in the supine position. The SCFE-affected limb was fixed on the traction table. For unstable SCFE, the limb was then gently pulled and internally rotated using the traction device, as shown in Figure 3B. This positioning is intended to reduce the femoral epiphysis. Following reduction, fluoroscopy was performed to confirm satisfactory alignment. The reduction process should be carried out as gently as possible to minimize iatrogenic injury. In contrast, for stable SCFE, screw fixation was performed directly using the TiRobot system without reduction.
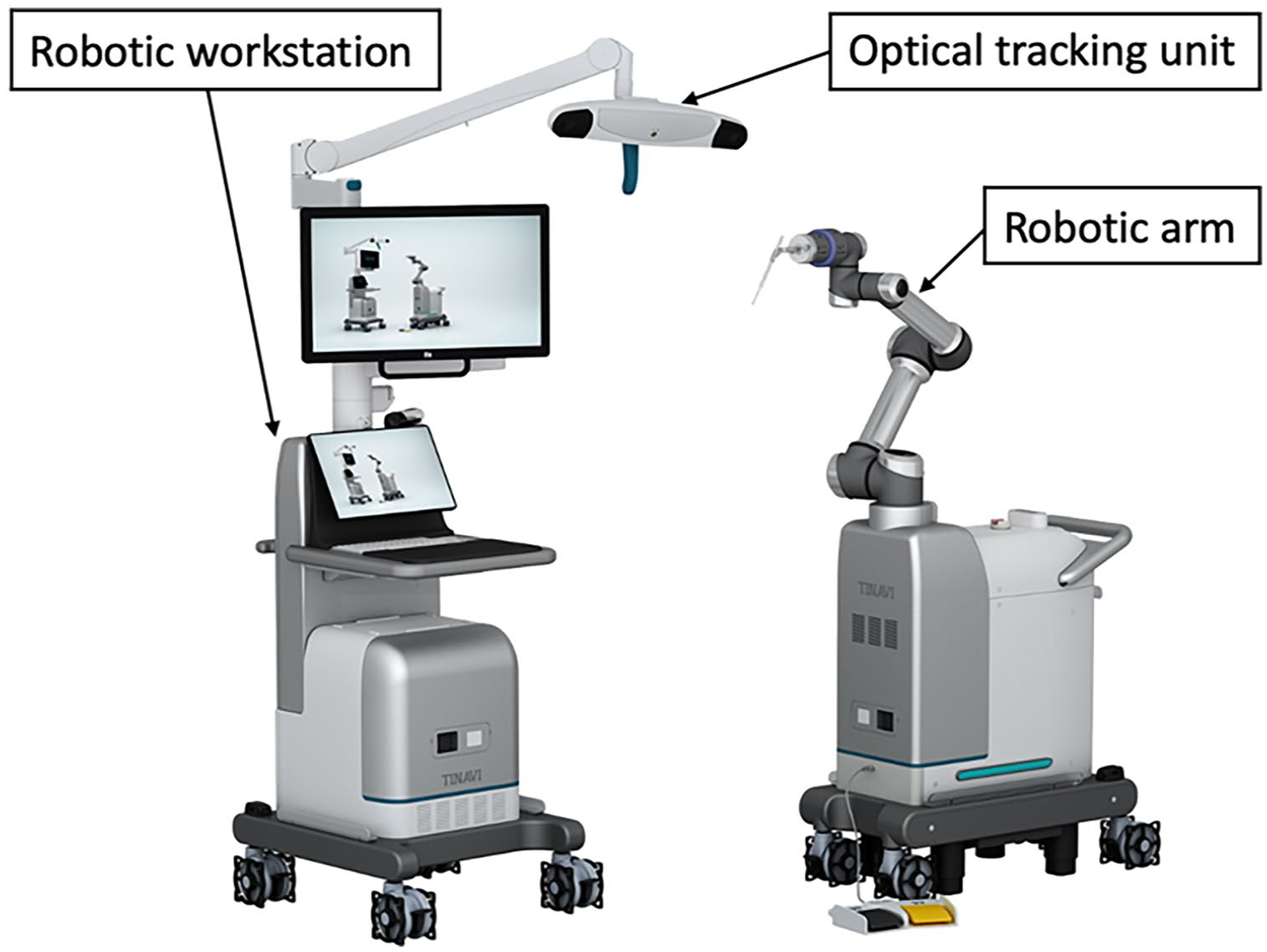
The TiRobot system comprised a robotic workstation, an optical tracking unit, and a robotic arm.
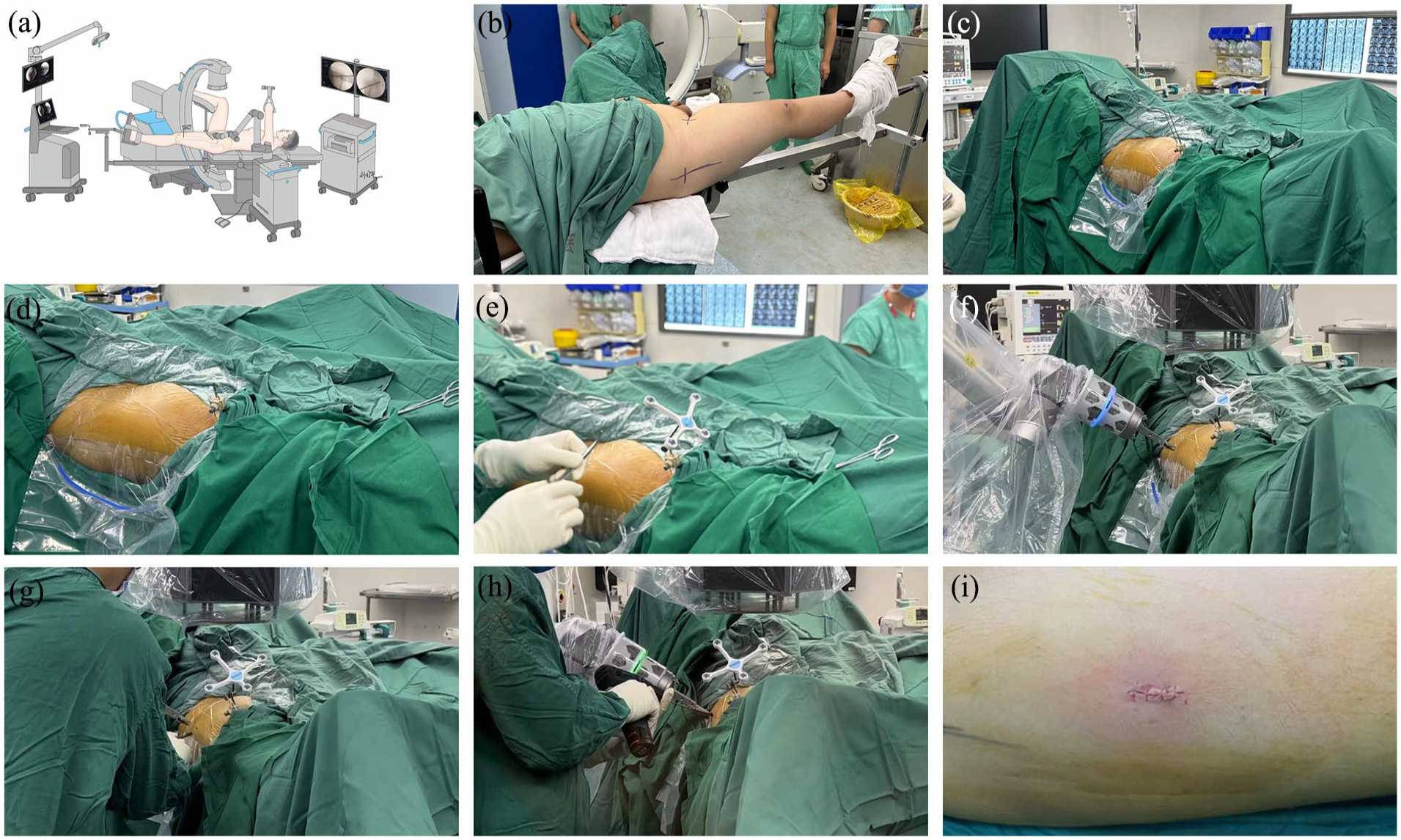
Workflow of the robotic system: a schematic diagram of this surgery procedure (a), patients were fixed on the traction table (b), two Kirschner wires were fixed to the distal femur percutaneously (c), the Kirschner wires were trimmed (d), the patient’s tracker was secured (e), the robotic arm was moved to the designated position (f), an incision was made (g), the Kirschner wires were inserted with robotic assistance (h), wound closure (i).
After routine disinfection and draping, the patient tracker was firmly fixed to the ipsilateral distal femur, with two 2-mm K-wires inserted percutaneously, as shown in Figure 3 ©. The AP and lateral views of the hip were obtained by the C-arm, with the patient positioned supine and the surgical team maintaining a safe distance from the radiation source. Ten locating points on the tracker were required for clear and distinguishable images in both views. The optical tracking unit, as shown in Figure 2, accurately identified the repositioned femoral head, due to these locating points being firmly fixed to the femoral shaft. The images were transferred to a robotic platform, as shown in Figure 4(b). On the robot platform, surgeons planned the screw placement, including the screw entry point, trajectory, diameter, length, and number of screws, based on the child’s femoral neck diameter and the type of slip. This was achieved using all previously acquired images without requiring further fluoroscopy.
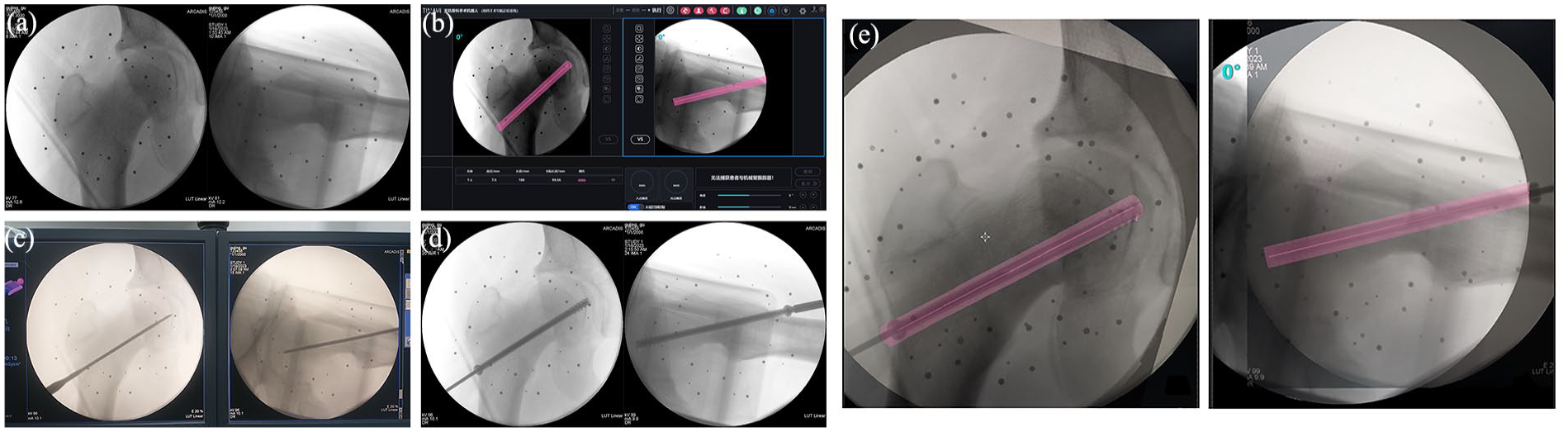
Anteroposterior and lateral views affected the femur (a), planned trajectory on the robotic workstation (b), a guidewire was inserted (c), a screw was inserted (d), comparison of the “planned” image of the hip with the “design” line (red), with the actual screw image as an indicator of accuracy (e).
After the screw placement plan was confirmed, the robotic arm was started, and it automatically moved to the corresponding position according to the design. The robotic arm provided a working channel; a sheath was inserted through the channel to the skin as shown in Figure 3(f). A 1-cm incision was made according to the sheath by a scalpel; a vascular forceps was used to separate the subcutaneous tissue until the periosteum, then the sheath was inserted into the periosteum without any soft tissue tension. A guidewire was drilled into the femur through the sheath, following the computer-planned path to precisely reach the femoral head epiphysis. If the channel deviated from the predetermined position during manipulation, an alarm was given by the robot. The position of the guide needle was confirmed by lateral and AP radiographs. One screw was inserted into the epiphysis along the guidewire, stopping approximately 5 mm from the subchondral bone of the femoral head. Final AP and lateral views were obtained for verification. The reference array was removed, and the wound was irrigated and closed, as shown in Figure 3(i).
The accuracy of the system was verified by superimposing the robot image onto an actual cannulated screw image (Figure 4(e)). The distance from the entry point of the screw to the post-instrumentation stage was compared with the planned exit point of the guidewire trajectory captured on the robotic display. The dispersion angles of the actual screws and planned guidewire trajectory on the posteroanterior and lateral images were also measured postoperatively. The measurements were recorded to determine the accuracy of the robotic system.
The intraoperative data included the number of guide needle passes made during the procedure (adjustment of the guide pin that created a new perforation in the proximal femur), fluoroscopy times (in both AP and lateral views), operation time, and bleeding.
All children were followed up postoperatively to note any potential complications, including slip progression, chondrolysis, AVN, and any complications that could be attributed to the distal femoral reference frame insertion.
Statistical analyses
Data were analyzed using SPSS (version 24.0; IBM, Armonk, New York, USA). Descriptive statistics were provided for all the variables. Student’s t-test was used to compare continuous variables with normal distributions. Statistical significance was set at <0.05.
Results
This technique was used in 31 patients (males, 19; females, 12; mean age, 11.24 ± 1.80 [range 8–13] years; mean body mass index, 24.14 ± 4.67 [range 21.31–29.17] kg/m2). Detailed demographic data are provided in Table 1. All screws were successfully inserted in one attempt, and no surgery was canceled because of technical problems. The average operation time was 98.25 ± 15.13 min, and the average bleeding was 21.65 ± 11.25 ml.
The average distance in the lateral view between the planned and actual screw entry points was 0.91 ± 0.72 mm. The average distance in the AP view between the actual and planned insertion points was 1.13 ± 0.58 mm; the actual entry point was cephalic compared to the planned point. The actual trajectory of the implant deviated from the planned trajectory by 3.61 ± 1.34° in the AP view and 2.33 ± 1.32° in the lateral view. The fluoroscopy time was 6.56 ± 3.23 images per screw, comprising an initial AP and lateral view for the image acquisition stage and additional images for verification. The average dose-area product (DPA) per screw was 0.7657 ± 0.5681 Gy·cm², leading to an average DPA per person of 5.02 ± 2.47 Gy·cm². Using the effective dose converting factor of 0.16 mSv/Gy per cm² for the femoral region, as described by McParland, 24 the average radiation dose was calculated to be 0.8032 ± 0.3952 mSv per screw.
The preoperation Non-Arthritic Hip Score was 28.53 ± 9.17, and it improved to 67.34 ± 6.21 at the final follow-up (P < 0.05). The Harris hip scores before the operation and at the last follow-up were 46.47 ± 15.34 and 89.63 ± 7.52, respectively (P < 0.05), with a significant difference.
No chondrolysis or AVN was observed. No morbidity related to percutaneous fixation of the patient’s tracking device, wound problems, infection, fractures, hip stiffness, or pain, were observed.
Discussion
Our study describes a robot-assisted technique for the simultaneous fixation of SCFE. As hypothesized, the robotic system had high operational accuracy, and all 31 cases successfully underwent screw insertion in a single attempt. The average distance between the actual and planned insertions was 1.13 ± 0.58 mm in the AP view and 0.91 ± 0.72 mm in the lateral view. The actual insertion trajectory deviated from the plan by an average of 3.61 ± 1.34° in the AP view and 2.33 ± 1.32° in the lateral view. Other details are shown in Table 2.
Summary of operative and postoperative outcomes.
AP: anteroposterior.
The distance in the AP view between the actual and planned insertion points
The angle of deviation between the actual implant and the planned trajectory in the AP view.
The angle of deviation between the actual implant and the planned trajectory in the lateral view.
SCFE fixation is technically demanding. The screw should be perpendicular to the epiphysis in both the AP and lateral views to prevent delayed physeal closure and progressive slippage, as noted by Morrissy. 14 Lavigne et al. 25 demonstrated that in a normal pediatric hip, the retinacular vessels are the predominant blood supply to the proximal femoral epiphysis; therefore, the central and inferior portions of the neck are considered safe zones for screw passage. Tokmakova et al. 17 reported a significantly higher rate (P < 0.001) of osteonecrosis when the screw position was further away from the center of the femoral head for the unstable SCFE. Therefore, the screw entry point should be anterior to or on the femoral neck to prevent posterior displacement of the epiphysis Ideally, the guidewire should be placed in a single pass to avoid creating multiple holes in the proximal femur, which increases the likelihood of postoperative fracture. 18
The complex geometry and narrow safe zone of the proximal femur in SCFE make optimal screw positioning challenging.16, 17 Surgeons rely on two-dimensional images from fluoroscopy, but this often offers only one plane, leading to difficulties in visualizing and correcting the instrument’s position across all planes. Adjusting the guide pin can prolong the operation, increasing X-ray exposure for both the patient and the operating team and potentially damaging the cancellous bone, which may compromise screw stability and increase fracture risk. The difficulty of proper screw placement in SCFE increases with the degree of slippage, challenging the surgeon’s spatial cognition. 26
Lateral and AP views are consistently available during robot assistance. This technique aids in accurate three-dimensional mapping of the screw route, especially for anatomically complex cases with limited safe zones. The robotic arm guides the surgeon to the correct entry point and minimizes skin incision and soft tissue dissection. The guide pin is inserted through a drill sleeve, aligning with the femoral head epiphysis in all views, reducing complications. The robotic system ensures correct estimation of the screw length and diameter, precise entry points, and trajectory, preventing joint penetration. It is especially advantageous for obese children, who are predominantly affected by SCFE.
In our study, robotic assistance ensured successful fixation in the first attempt for all patients, improving stability and reducing the risk of osteonecrosis. The system’s accuracy was high, with entry point errors < 2 mm and trajectory errors < 4°. Oetgen et al. 27 reported a navigation technology with a high accuracy of 5.3 ± 0.94 mm and a deviation from the planned trajectory of 4.9 ± 2.2°. The current robotic system offers superior accuracy by eliminating eye-hand coordination errors. He et al. 28 found that the same robotic system for adult femoral neck screws significantly reduced intraoperative fluoroscopy time, drilling attempts, and operation time compared to conventional methods.
As shown in Table 2, the new technique significantly reduced radiation exposure. The mean fluoroscopy time was minimized, requiring only four to six images. The average radiation dose was 0.8032 ± 0.3952 mSv, which is significantly lower compared to traditional procedures, where the average dose is 1.4 ± 1.6 mSv. 29 This reduction in radiation is crucial given the adolescent age of the patients. The amount of radiation delivered to the human pelvis within 1 min of fluoroscopy is 40 mSV, equivalent to 250 chest radiographs. 30 Therefore, we believe this to be an important advantage of the system.
To our knowledge, this is the first study to introduce a robotic technology for SCFE. The positive impact of this technology is that it shows promise in improving accuracy and reducing radiation. However, the duration of the procedure and the time under anesthesia may be prolonged. Complications such as guide wire advancement into the joint during overdrilling and screw insertion into the joint may be caused by the robotic arm. The other two Kirschner pins may also cause postoperative complications. However, these complications were not found in our study. Our study has certain limitations; namely, it involves a small number of initial cases, only one treatment group, and is not a randomized controlled trial. For these reasons, the study could not effectively compare the robotic approach with the traditional “freehand” method. Future studies with larger sample sizes, a comparison group, and randomized controls are needed to confirm these results.
This study does not imply that all SCFE fixation cases warrant robotic assistance. While the robotic system may improve the accuracy of screw placement and minimize intraoperative fluoroscopy, the high cost, limited availability, and lack of long-term evidence on effectiveness and safety have limited its widespread application. 31
Supplemental Material
sj-pdf-1-cho-10.1177_18632521251319987 – Supplemental material for Robot-assisted percutaneous cannulated screw fixation in the treatment of slipped capital femoral epiphysis
Supplemental material, sj-pdf-1-cho-10.1177_18632521251319987 for Robot-assisted percutaneous cannulated screw fixation in the treatment of slipped capital femoral epiphysis by Yunfeng Xu, Gang Fu, Chao Feng, Lin Huang, Yuan Ma, Yucheng Zhang, Yuan Zhou, Xiao Luo, Ming Lu, Jie Yang, Yukun Wang, Xuemin Lv, Xieyuan Jiang and Zheng Yang in Journal of Children’s Orthopaedics
Footnotes
Author contributions
YX (Contribution: study design, performed measurements, statistical analysis, manuscript preparation). GF (Contribution: performed measurements, manuscript preparation). CF (Contribution: statistical analysis, and manuscript preparation). LH, YZ, YZ, XL, QL (Contribution: performed measurements). ML, JY, YW, YM (Contribution: manuscript preparation). XL, XJ, ZY (Contribution: study design)
Data availability statement
Data stored on hospital intranets is not available for free and open access at the moment. Sorry about that.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Supported by Beijing Natural Science Foundation (L212041, L222060), Beijing JST Research Funding (ZR-202222), and Beijing Jishuitan Orthopaedic Robot Engineering Reasearch Center Co.,LTD(GCZX202207)
Ethics approval statement
Approval No. of Ethics Committee: [K2023] No. [336]-00
Approval institution: Ethics Committee(seal)of Beijing Jishuitan Hospital
Patient consent statement
All the patients are aware and supportive of our research.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
