Abstract
Purpose:
Spinal muscular atrophy type 1 has a devastating natural course and presents a severe course marked by scoliosis and hip subluxation in nonambulatory patients. Nusinersen, Food and Drug Administration-approved spinal muscular atrophy therapy, extends survival and enhances motor function. However, its influence on spinal and hip deformities remains unclear.
Methods:
In a retrospective study, 29 spinal muscular atrophy type 1 patients born between 2017 and 2021, confirmed by genetic testing, treated with intrathecal nusinersen, and had registered to the national electronic health database were included. Demographics, age at the first nusinersen dose, total administrations, and Children’s of Philadelphia Infant Test of Neuromuscular Disorders scores were collected. Radiological assessments included parasol rib deformity, scoliosis, pelvic obliquity, and hip subluxation.
Results:
Mean age was 3.7 ± 1.1 (range, 2–6), and average number of intrathecal nusinersen administration was 8.9 ± 2.9 (range, 4–19). There was a significant correlation between Children’s of Philadelphia Infant Test of Neuromuscular Disorders score and the number of nusinersen administration (r = 0.539, p = 0.05). The correlation between Children’s of Philadelphia Infant Test of Neuromuscular Disorders score and patient age (r = 0.361) or the time of first nusinersen dose (r = 0.39) was not significant (p = 0.076 and p = 0.054, respectively). While 93.1% had scoliosis, 69% had pelvic obliquity, and 60.7% had hip subluxation, these conditions showed no significant association with patient age, total nusinersen administrations, age at the first dose, or Children’s of Philadelphia Infant Test of Neuromuscular Disorders scores.
Conclusion:
Disease-modifying therapy provides significant improvements in overall survival and motor function in spinal muscular atrophy type 1. However, progressive spine deformity and hip subluxation still remain significant problems in the majority of cases which would potentially need to be addressed.
Introduction
Spinal muscular atrophy (SMA) is an autosomal recessive genetic disorder that leads to neuronal degeneration of the anterior horn cells in the spinal cord.1,2 Homozygous deletion or mutation in the survival motor neuron 1 (SMN1) gene causes decreased survival motor neuron (SMN) protein expression. The resulting lack of SMN protein leads to neuronal degeneration in the central nervous system. Disease severity of SMA is strongly influenced by the copy number of closely related survival motor neuron 2 (SMN2) gene, which differs only in five base pairs from the SMN1 gene. SMN2 is a disease modifier of SMA phenotype, and the copy number of SMN2 inversely correlates with disease severity.3 –5
The estimated incidence of all types of SMA is approximately 1 in 6000 to 10,000 live births.6 –9 The classification of SMA is based on the patient’s age of onset and achievement of motor milestones. SMA type 1 (Werdnig–Hoffmann disease) is the most common subtype which constitutes about 60% of all SMA patients10,11 which has a devastating natural course. Diagnosis is usually made in the first 6 months of life. Besides having significant decline in the motor functions caused by severe neuromuscular weakness, there is also respiratory insufficiency due to intercostal muscle paralysis as well as swallowing and feeding difficulties caused by bulbar motor neuron involvement.12 –14 If left untreated, patients with SMA type 1 never acquire head control or sit independently, and they rarely survive to their second birthday. 15 Nonambulatory SMA patients often develop scoliosis and hip subluxation or dislocation, but orthopedic management of SMA type 1 patients has been rarely discussed in the literature due to the very limited survival.16 –23
Although comprehensive pulmonary and nutritional management has made some positive impact on survival, historically there was no real cure for SMA until recently.24 –27 Over the past decade, development of the disease-modifying therapy provided a significant improvement to patient prognosis. The first therapy for SMA, nusinersen (Spinraza®), has been approved by the Food and Drug Administration (FDA) in the United States in 2016 28 and by the European Medicines Agency (EMA) in Europe in 2017. 29 Nusinersen is an antisense oligonucleotide intrathecal therapy that enhances the inclusion of exon 7 into the mRNA transcript from the SMN2 gene, thus resulting in full-length SMN protein production. 30
As shown by the previous studies, intrathecal nusinersen treatment not only prolonged patient survival but also improvements in motor function and respiratory function were reported.31 –33 However, the impact of nusinersen therapy on the natural course of musculoskeletal deformities in SMA type 1 is still unclear. The aim of this study is to ascertain the prevalence of scoliosis, parasol rib deformity, pelvic obliquity, and hip subluxation in patients with SMA type 1 who underwent nusinersen treatment.
Methods
Study design
This study was conducted after receiving approval from the Republic of Turkiye Ministry of Health with a waiver of informed consent for retrospective data analysis (ID: 95741342-020/27112019). All citizens in Turkiye have a unique civil registration number, which is used in all health care contacts and enables complete connection between the health care registries. All patient records are kept on an electronic database since 2016 (E-Nabiz). 34 Starting from 2017, nusinersen has been reimbursed by the Turkish National Health Insurance Fund for SMA type 1 with certain criteria on a case-by-case basis. In this study, the search was done between January 2017 and December 2021, based on the E-Nabiz database using the International Classification of Disease (ICD)-10 codes, G12.0 (infantile SMA, type 1 Werdnig–Hoffman), G12.1 (other inherited SMA), G12.2 (SMA and related syndromes), G12.8 (other spinal muscular atrophies and related syndromes), and G12.9 (SMA, unspecified).
To be eligible to participate in this study, a patient had to meet all of the following inclusion criteria: (1) diagnosis of SMA type 1, (2) diagnosis confirmed by genetic testing, (3) born between 2017 and 2021, (4) had received intrathecal nusinersen (Spinraza) therapy, (5) had registered to the e-Nabiz national electronic health database, and (6) being alive during the database search. A patient having any of the following exclusion criteria had been excluded from participating in this study: (1) had <2 years of radiological follow-up, (2) having inadequate clinical information or suboptimal radiographs in the electronic database, (3) received the first nusinersen dose older than 6 months of age, and (4) received SMA treatment other than nusinersen onasemnogene abeparvovec-xioi Zolengsma,risdiplam Evrysdi during their course of follow-up.
Medical record review
Demographic parameters including age, gender, SMA characteristics, time of initial diagnosis, and duration of follow-up were obtained. Patient records were also reviewed for genetic test confirmation, age at the first nusinersen dose, total number of intrathecal nusinersen administration, status of having tracheostomy, and feeding route. Assessment of motor function of the patients was performed by reviewing The Children’s Hospital of Philadelphia Infant Test of Neuromuscular Disorders (CHOP-INTEND) scores, which have been obtained from patient’s electronic records. 35 The CHOP-INTEND is a reliable measure to quantify motor skills in patients with SMA type 1 with a score between 0 and 64. It has been developed and validated to be used on children aged from 3 months to 4 years, yet not limited to this age range.
Radiological evaluation
Radiological evaluation was done for each patient by assessing their chest, whole spine, and pelvis radiographs in the electronic database. All radiographic measurements were performed by a radiologist. Measured radiographic parameters included major coronal spinal deformity angle (using the Cobb method), pelvic obliquity, hip subluxation (using the head migration index (MI)), and the presence of parasol rib deformity. Cobb angle >10° is considered as scoliosis for the coronal spinal deformity measurement.36,37 The coronal pelvic obliquity (CPO) angle was defined as the angle between a line connecting the top of both iliac crests and the horizontal line was measured on anteroposterior radiographs. 38 CPO angle >5° is considered as pelvic obliquity. Femoral head MI >40% in the AP pelvis X-ray is considered as the threshold for hip subluxation. 39 Parasol rib deformity was defined as marked down sloping and collapse of the ribs leading to a narrowed chest wall which was noted in the spine X-ray. 40
Trends of change in the Cobb angle (ΔCobb), pelvic obliquity (ΔPO), and femoral head MI (ΔMI) were also calculated by dividing the deformity magnitudes in the last follow-up to the duration of radiological follow-up.
Statistical analysis
Descriptive statistics was presented as mean ± standard deviation or (median, interquartile range (IQR)) according to the assumption of normal distribution for quantitative variables and frequency (%) for qualitative variables. The normality assumption of quantitative data was evaluated with the Shapiro–Wilk test. Demographic and clinical data of SMA patients were evaluated with the Whitney U test. Relationships between categorical data were evaluated with Fisher’s exact test and Spearman’s Rho test. Statistical significance level was accepted as 0.05, and Statistical Package for Social Sciences (SPSS, version 25.0, Chicago, IL) was used for the analysis.
Results
A total of 119 patients with SMA type 1 born between 2017 and 2021 and received nusinersen therapy were registered in the e-Nabiz. Among them, 31 patients were dead at the time of the data collection (mortality rate: 26%), and 26 patients with inadequate medical records in the electronic database were excluded. Sixty-two patients were eligible for this study according to our inclusion criteria, of which 29 patients were enrolled after applying the exclusion criteria (Figure 1). Demographic and other clinical data of the patients are summarized in Table 1. The mean age was 3.7 ± 1.1 years (range, 2–6). Male-to-female ratio was 1.1. The mean number of intrathecal nusinersen administration was 8.9 ± 2.9 (range, 4–19). The mean age of first nusinersen was 2.7 ± 1.1 months (range, between 1–6 months). The follow-up period ranged between 2 and 6 years, with a mean of 3.3 ± 1.1 years. Parasol rib deformity was noted in 27 patients (93.1%) in the last follow-up X-ray. All patients had tracheostomy cannula initially, whereas 12 patients (37.9%) had tracheostomy closure during follow-up. Twenty-two patients had gastric feeding tube (75.9%). There was no difference in the total number of nusinersen administrations or the time of administration of the first nusinersen dose between patients with or without parasol rib deformity, tracheostomy cannula, or gastric feeding tube (Tables 2 and 3).
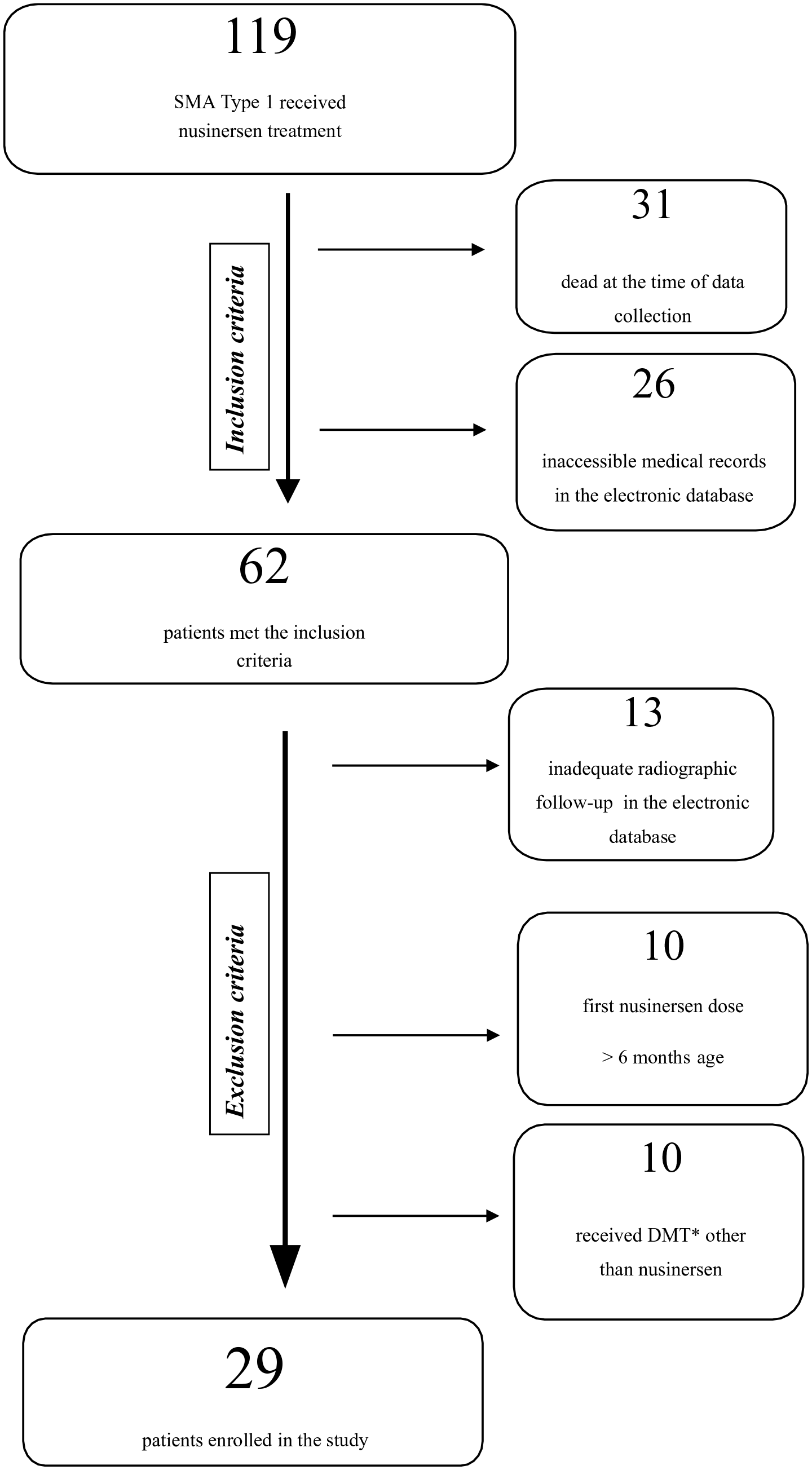
Flow chart documenting for inclusion and exclusion criteria, and the number of patients included in the study population. DMT: disease-modifying therapy.
Summary of the demographic and clinical parameters of the patients.

SD: standard deviation.
The effect of the time of first intrathecal nusinersen administration on the outcome variables.
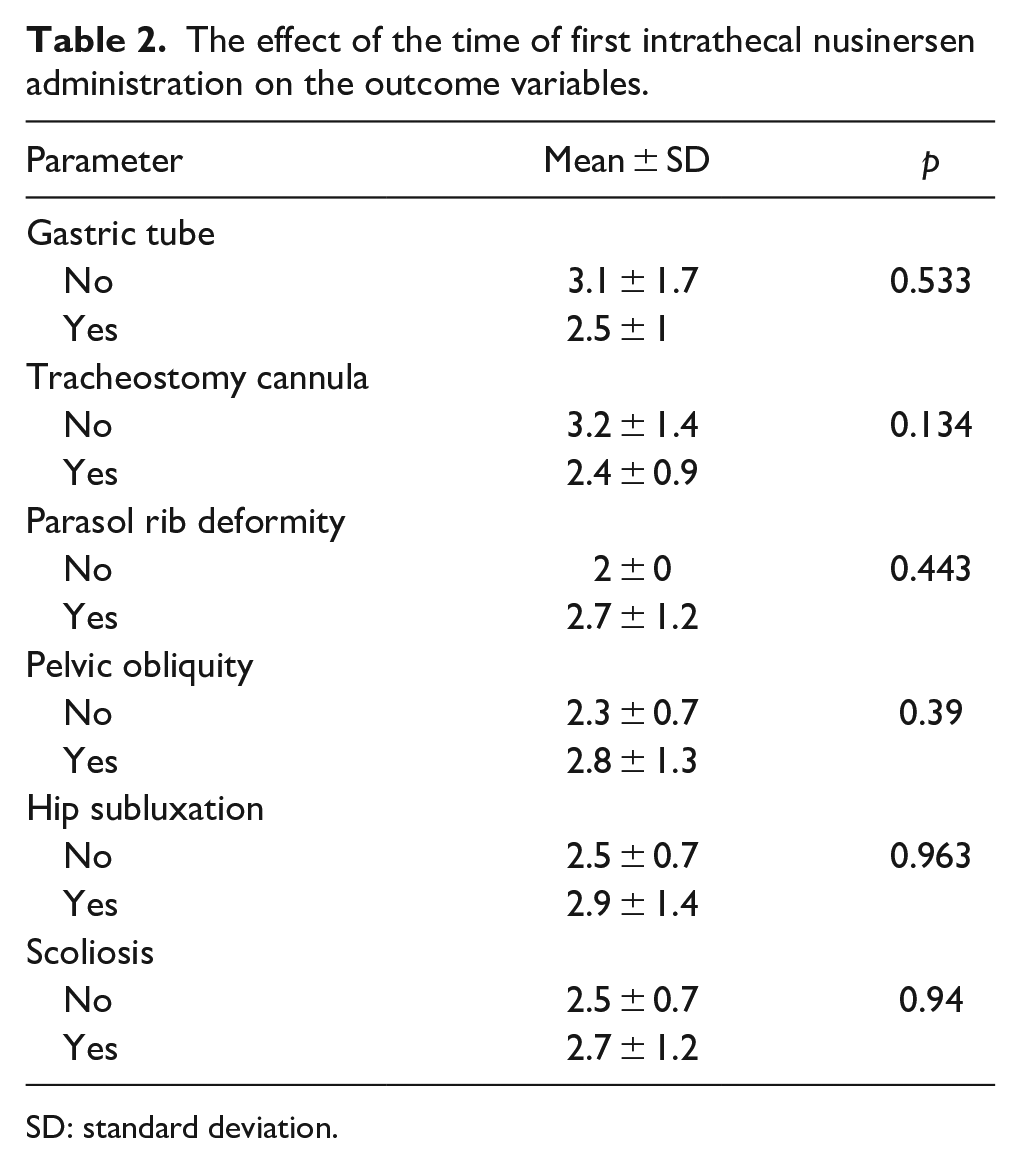
SD: standard deviation.
The effect of the total number of intrathecal nusinersen administration on the outcome variables.
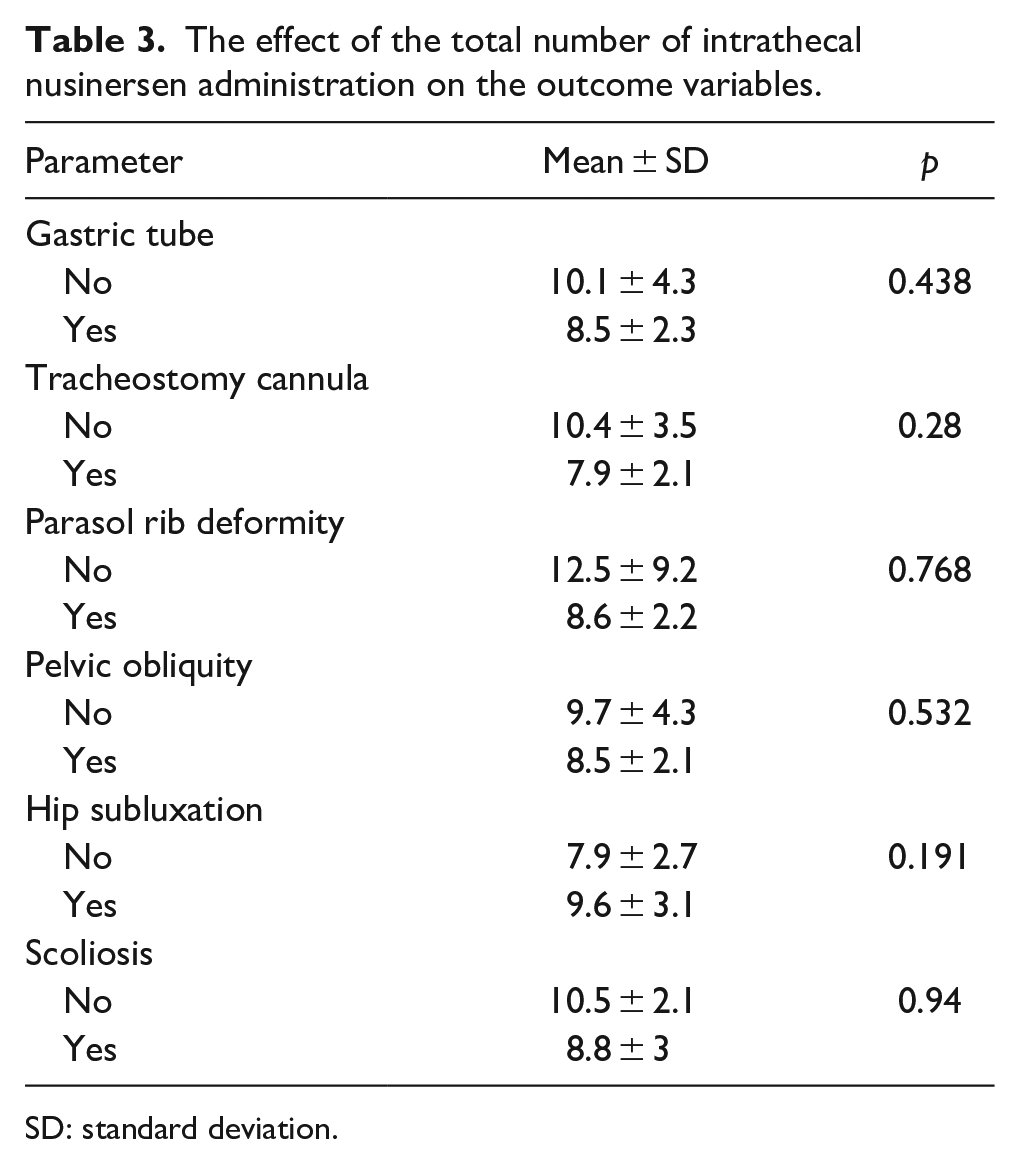
SD: standard deviation.
Motor function
The mean CHOP-INTEND score of the patients was 45.2 ± 11.7 (range, 25–62) at the last follow-up. There was a positive correlation between the CHOP-INTEND score and the number of nusinersen administration which was statistically significant (r = 0.539, p = 0.05). However, there was no statistically significant correlation between CHOP-INTEND score and patient age (r = 0.361) or the time of first nusinersen dose (r = 0.39; p = 0.076 and p = 0.054, respectively).
Scoliosis
All patients had nonstandardized anterior–posterior spine radiographs in the supine position, whereas no patient had lateral spine radiographs. Twenty-seven patients (93.1%) had scoliosis (Figure 2). Scoliosis developed before 2 years of age in all those patients. The mean Cobb angle was 39.3° ± 19.9° (range, 14–88) at the last follow-up. ΔCobb was 12.7° ± 7.2° per year (range, 3.5–27) in patients with scoliosis. A single long c-shaped curve was noted in 16 patients (59.3%), whereas a double thoracolumbar curve pattern was seen in 11 patients (40.7%). Relationships between ΔCobb in patients with scoliosis and age (r = −0.325, p = 0.098), CHOP-INTEND scores (r = −0.036, p = 0.87), age at the first nusinersen dose (r = −0.09, p = 0.65), or total number of nusinersen administration (r = 0.075, p = 0.711) were not statistically significant.
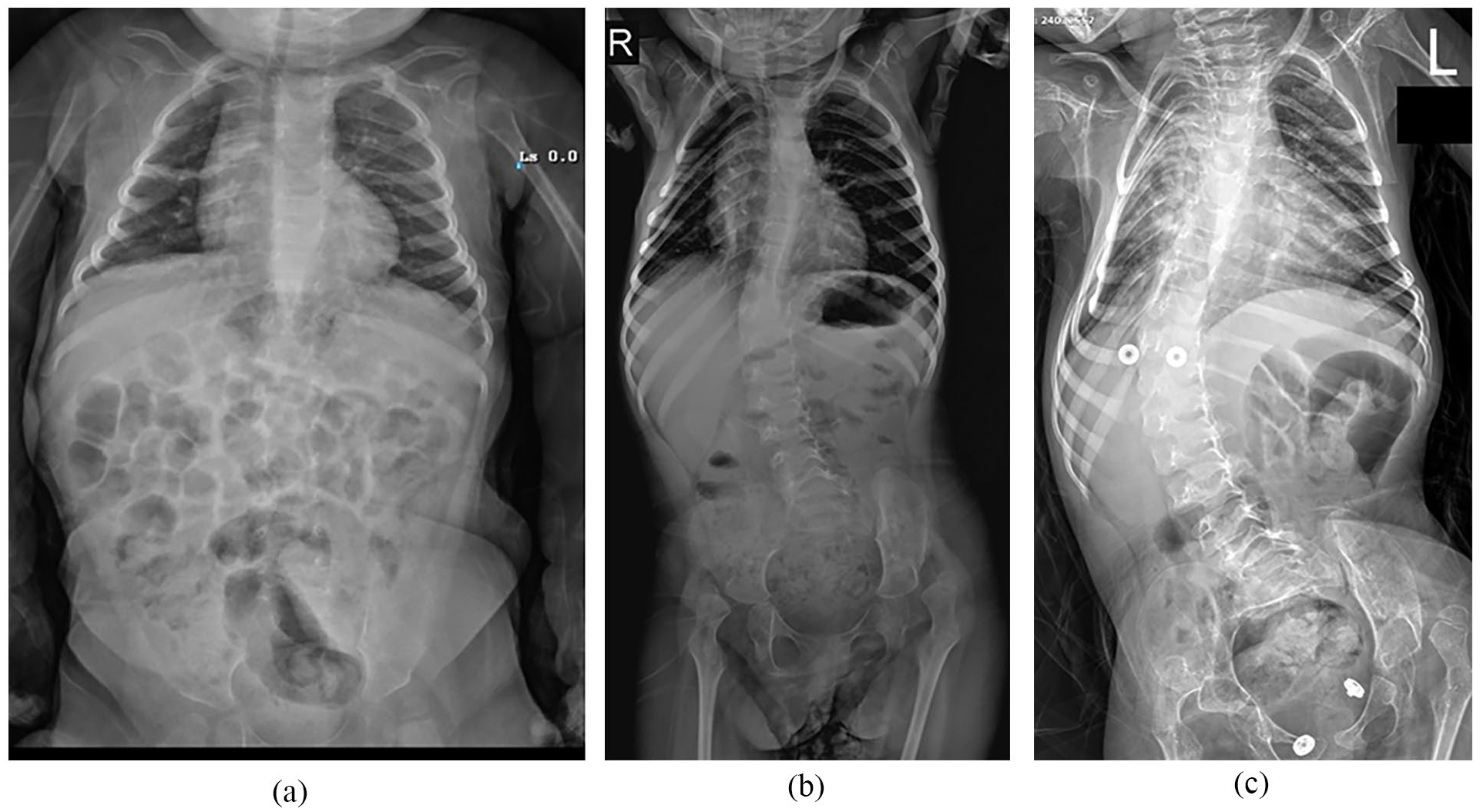
A 6-year-old girl with SMA type 1. The first nusinersen dose has administered at 5 months of age. (a) All X-rays were taken at supine position. Initial X-ray was taken when she was 1 year old. Note that there is no scoliosis, pelvic obliquity, or hip subluxation; (b) X-ray at 4 years of age revealed that she had scoliosis with a Cobb angle of 25° and pelvic obliquity of 12°. Bilateral hip dislocation was also noted. There is no parasol rib deformity; (c) X-ray was taken when she was 6 years. She had nine intrathecal nusinersen administrations. Cobb angle is 48°, and pelvic obliquity is 19°. Parasol rib deformity is noted.
Pelvic obliquity
Nine patients had no pelvic obliquity (31%), whereas 20 patients had pelvic obliquity (69%) with a mean value of 12.1 ± 5.5 (range, 5–22). Pelvic obliquity was always associated with concomitant scoliosis deformity. ΔPO was 4.1° ± 2.6° per year (range, 1.4–10) in patients with pelvic obliquity. The distribution of patient age was same across patients with or without pelvic obliquity (p = 0.835). There was no difference between patients with and without pelvic obliquity in terms of total number of nusinersen administration or the time of first nusinersen dose (Tables 2 and 3). The distribution of the CHOP-INTEND scores in patients with or without pelvic obliquity was similar (p = 0.238). Relationships between ΔPO in patients with pelvic obliquity and age (r = −0.401, p = 0.08), CHOP-INTEND scores (r = −0.362, p = 0.153), age at the first nusinersen dose (r = −0.126, p = 0.597), or total number of nusinersen administration (r = 0.235, p = 0.318) were not statistically significant.
Hip status
Seventeen patients had hip subluxation (60.7%) with a mean MI of 83.4 ± 21.8 (range, 40–100). ΔMI was 31.3° ± 13.6° per year (range, 10–50). The distribution of patient age was similar across patients with or without hip subluxation (p = 0.487). Again, there was no difference between patients with or without hip subluxation in terms of total number of nusinersen administration or age at the first nusinersen dose (Tables 2 and 3). The distribution of the CHOP-INTEND scores in patients with or without hip subluxation were not significantly different (p = 0.285). Relationships between ΔMI in patients with hip subluxation and age (r = −0.428, p = 0.086), CHOP-INTEND scores (r = −0.417, p = 0.138), age at the first nusinersen dose (r = 0.060, p = 0.818), or total number of nusinersen administration (r = −0.419, p = 0.094) were not significant.
Discussion
Before the new treatment era, SMA type 1 patients had a tendency to deteriorate progressively over time with extremely reduced survival rates. In the last decade, introduction of the disease-modifying therapy has been a real breakthrough. Novel therapeutic agents combined with other comprehensive multidisciplinary medical approaches extended the life expectancy in this devastating condition.41 –43 Our findings were in line with the previous studies which showed that nusinersen is effective in terms of improving survival and motor function in SMA type 1. We noticed a significant positive correlation between the total number of nusinersen administration and CHOP-INTEND scores at the last follow-up. All of the patients had the first nusinersen dose before 6 months of age, and we did not observe any significant correlation between CHOP-INTEND scores and age at the first nusinersen dose. Previous studies reported that SMA type 1 patients who started nusinersen earlier than 7 months had better motor function improvements compared with older children.44,45
Scoliosis almost invariably develops with an onset before 2 years of age in the natural course of untreated SMA type 1. 23 In our study, scoliosis emerged in 93.1% of the patients. All those patients developed scoliosis before 2 years of age, and the most common pattern was a single c-type curve. The orthopedic literature is scarce regarding scoliosis development and progression in SMA type 1 patients who received disease-modifying therapy. Al Amrani et al. 46 reported that all seven patients with SMA type 1 who received nusinersen developed scoliosis by 12 months of age with an average progression of 2.3° per month. Soini et al. 37 recently published a retrospective study of 16 SMA type 1 and 2 children treated with gene therapy with onasemnogene abeparvovec. Eleven patients had other disease-modifying treatments including nusinersen and risdiplam. They reported that 10 patients had significant kyphosis and 9 had significant scoliosis. In another recent observational study of nine pediatric patients with SMA types 1 and 2 treated with onasemnogene abeparvovec, Stettner et al. 47 reported that 67% of their patients developed scoliosis after 1-year follow-up.
Scoliosis progressed by 11.7° per year in our cohort. To our knowledge, there are no published data about the scoliosis progression rate in untreated SMA type 1 patients. On the contrary, previous studies reported 5° to 15° per year scoliosis progression rate in SMA type 2.48,49 Thus, scoliosis progression rate in our cohort of patients with SMA type 1 had nusinersen was similar to the natural course of SMA type 2 patients. We did not notice any relationship between scoliosis progression rate and patient age, CHOP-INTEND scores, age at the first nusinersen dose, or total number of nusinersen administration. We also observed pelvic obliquity in 69% of the patients which was always associated with concomitant scoliosis. To our knowledge, there are no published data about the rate of pelvic obliquity in SMA type 1 patients who either untreated or received disease-modifying therapy.
Hip subluxation is another primary orthopedic concern in SMA. Previous studies showed that hip subluxation is more common in nonambulatory patients (types 1 and 2).26,50 In a review of 136 hips, Hanna et al. assessed the frequency of hip subluxation and the prevalence of hip pain among nonambulatory children with SMA types 1 and 2. 20 They reported that all patients with SMA type 1 and 97% of the patients with SMA type 2 without disease-modifying therapy developed radiographic hip pathology. As far as we know, our study is the only one to date which describes the prevalence of hip subluxation in patients with SMA type 1 who underwent nusinersen treatment. Sixty-one percent of our patients developed hip subluxation with a MI >40%. We did not observe any relationship between development or progression of hip subluxation and the distribution of patient age, total number of nusinersen administration, age at the first nusinersen dose, or CHOP-INTEND scores.
The main limitation of this study is the small number of patients which could be explained by the rarity of the disease. Another notable limitation is the absence of a control group of untreated SMA type 1 patients to compare with the patients treated with nusinersen. However, we believe that leaving newly diagnosed SMA type 1 patients untreated in the new treatment era would be ethically unacceptable. In addition, sitting (or standing) AP or lateral spine radiographs were not available; therefore, our analyses for spine deformity were based on nonstandardized AP supine X-rays. We recognize that all patients with scoliosis might actually have more severe spinal deformity than we appreciated in the supine X-rays. In addition, we were unable to make any implication about the sagittal plane with the current data.
Prior to the introduction of disease-modifying therapies, surgical management of musculoskeletal deformities was rarely discussed as a possible treatment option in SMA type 1 due to their very limited survival. In line with the previous studies, our results also showed that nusinersen treatment improved overall survival and motor function in SMA type 1 patients as compared with the devastating natural history of the disease. Our results also revealed that progressive spine deformity and hip subluxation still remain significant problems in the majority of cases. A recent study reported that early onset spinal deformity in SMA type 1 can be safely treated using surgical methods with comparable complication rates to SMA type 2. 51 Presumably, the number of SMA type 1 patients seeking care for their musculoskeletal conditions will noticeably grow in the near future. This is a consequence of the altered natural history with modern treatment methods in a formerly fatal disease. The improvement but not a normalization of motor functions in patients with SMA type 1 receiving disease-modifying therapy would probably expose the spine and the hip joints to forces which may not be controlled and thus lead to secondary orthopedic problems. We believe this information should be a part of the initial family counseling prior to the start of disease-modifying therapies. As for the orthopedic surgeon perspective, significant improvements in overall survival and motor function in the disease-modifying therapy era should encourage us to reconsider our intentional decision to neglect spine deformity and hip subluxation in SMA type 1.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521241235028 – Supplemental material for Nusinersen therapy changed the natural course of spinal muscular atrophy type 1: What about spine and hip?
Supplemental material, sj-pdf-1-cho-10.1177_18632521241235028 for Nusinersen therapy changed the natural course of spinal muscular atrophy type 1: What about spine and hip? by Niyazi Erdem Yasar, Guzelali Ozdemir, Elif Uzun Ata, Mustafa Okan Ayvali, Naim Ata, Mahir Ulgu, Ebru Dumlupınar, Suayip Birinci, Izzet Bingol and Senol Bekmez in Journal of Children’s Orthopaedics
Footnotes
Acknowledgements
All authors had made appropriate contributions to the conception and design of the study.
Author contributions
S.B., N.E.Y., and G.O. contributed to design of the study, development of checklists, interpretation of data, writing of the article, and drafting and revision of the article. M.U. contributed to acquisition of data and co-writing of the article. M.O.A., N.A., E.U.A., and S.B. contributed to the acquisition and analysis of the data. S.B., I.B., and S.B. contributed to the interpretation of the data and design of the research. E.D. contributed to statistical analysis. All authors critically revised the manuscript, agreed to be fully accountable for ensuring the integrity and accuracy of the work, and read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
This study was conducted according to the Declaration of Helsinki and received approval from the Turkish Ministry of Health with a waiver of informed consent for retrospective data analysis and health information privacy law ( ID: 95741342-020/27112019).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Guarantor
S.B.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
