Abstract
Purpose:
To evaluate the safety of growth-friendly instrumentation for early-onset scoliosis (EOS) in patients with spinal muscular atrophy (SMA) type 1 who received disease-modifying treatment (DMT) and analyze short-term efficacy.
Methods:
Retrospective search was conducted between 2017 and 2023. Patients with genetically confirmed SMA type 1 who were surgically treated for spinal deformity and receiving DMTs (nusinersen, risdiplam, or onasemnogene abeparvovec) were included. SMA types 2 and 3 and patients who do not receive DMTs were excluded. Clinical and radiographic data were collected at preoperative, postoperative, and latest follow-up visits.
Results:
Twenty-eight patients (mean follow-up: 16 months (range 2–41)) were included. The mean age at surgery was 60 months (range 29–96). Fifteen were treated with dual magnetically controlled growing rods (MCGR), four with unilateral MCGR and a contralateral guided growth system, three with Vertical Expandable Prosthetic Titanium Rib (VEPTR®) implants, five with self-distracting systems, and one with traditional dual growing rods. The mean amount of correction was 57% (44°± 17) for scoliosis and 83% (13°± 11) for pelvic obliquity. The mean T1-12 height gain during surgery was 31 mm (±16 mm), while the mean T1 S1 height gain was 51 mm (±24 mm), and instrumented growth was observed during follow-up. Five patients (18%) developed six serious adverse events: three surgical site infections, two anchor failures, and one rod fracture, and all required unplanned reoperations. No neurologic complication, difficulty during nusinersen injections, or respiratory decline was recorded.
Conclusion:
We report that spinal deformity in this population can be safely treated with growth-friendly instrumentation, with similar complication rates when compared with SMA type 2.
Introduction
Spinal muscular atrophy (SMA) is a hereditary neuromuscular disorder that is characterized by degeneration and loss of lower motor neurons, which results in progressive muscle atrophy and weakness. 1 The gene, survival motor neuron 1 (SMN1) responsible for this condition is located on chromosome 5q13, and it was first identified in 1995. 2 In addition to possible unrevealed duties in motor neurons, the SMN protein is required for the correct assembly of small nuclear ribonucleoprotein (snRNP) complexes and mRNA splicing in the cell. 3 The severity of clinical manifestations are closely related to the number of copies of the SMN2 gene, and an inverse correlation can be observed. 4 Traditionally, this disorder has been classified into types 1 to 3, with type 1 having the highest newborn incidence and the worst prognosis. More recently, the classification has been expanded to include types 0 and 4, which are the intrauterine-onset and adult-onset types, respectively. 4
The natural course of SMA has improved since the development of disease-modifying treatment (DMT) options, such as mRNA splicing modifiers (nusinersen and risdiplam) and gene therapies (onasemnogene abeparvovec). 5 Nusinersen and risdiplam act by increasing the levels of SMN2 through different mechanisms, and onasemnogene abeparvovec is a gene therapy that is delivered via intravenous infusion, which uses a viral vector to introduce new functioning copies of SMN1 into the cell. Nusinersen is delivered intrathecally, while risdiplam can be taken orally. Both these splicing modifiers require continuous usage, and onasemnogene abeparvovec has the advantage of being single-use. 5
Early-onset scoliosis (EOS) is one of the most common orthopedic manifestations in children with SMA, and as a historical cohort reports, virtually all nonambulatory patients develop scoliosis of varying degrees. 6 Due to expected poor natural history with very high mortality within the first 2 years of life, patients with SMA type 1 were rarely seen as candidates for surgery, and their spinal deformities were either not seen or not treated. For patients with a better prognosis, however, lifetime risk of requiring surgical treatment is very high, reaching up to 84% for SMA type 2. 7 As with EOS of any etiology, allowing spinal growth and pulmonary development should be the main principles of treatment, and growth-friendly instrumentation must be preferred whenever possible.
Growth-friendly instrumentation has been applied safely to patients with SMA type 2, and good results have been reported.8 –11 Studies on DMT-treated type 1 patients showed longer overall survival with better motor development and respiratory function,5,12 but scoliosis is still a major problem, and reported progression rates are very high. 13 It is now clear that patients with SMA type 1 will require more attention by surgeons in the DMT era, and to our knowledge, there is currently no study in the literature focusing on the treatment of EOS in patients with SMA type 1 treated with DMTs. In this study, we aimed to evaluate the safety of growth-friendly instrumentation in patients with SMA type 1 who received DMTs and to analyze the efficacy of growth-friendly instrumentation in the short term.
Materials and methods
The study was approved by the institutional ethical boards of each of the contributing centers.
The study is designed as a retrospective case series among European Paediatric Orthopaedic Society (EPOS) Spine Study Group members. Databases of seven different institutions were retrospectively reviewed between May 2017 and July 2023. Inclusion criteria were patients with genetically confirmed diagnosis of SMA type 1, surgically treated for spinal deformity with growth-friendly instrumentation methods, and receiving DMTs starting before index surgery. Exclusion criteria were patients with SMA types 2 and 3, received nonoperative treatment for EOS, not receiving DMTs, or DMTs started after the index surgery.
In addition to demographic variables, clinical variables (type of DMT used, night-time ventilation requirement, condition of the hips before and after surgery), surgical variables (estimated blood loss, surgical time, hospital and intensive care unit (ICU) stays, method of growth-friendly instrumentation used, levels of instrumentation, complications), radiographic variables (major curve scoliosis angle, pelvic obliquity, T2-12 kyphosis, L1 S1 lordosis, T1-12 and T1 S1 heights), and the status of the hips were also documented. Complications were reported if considered a serious adverse event, (i.e. necessitating reoperation or prolonged hospital stay) as rates and yearly rates per patient (complication/patient/year). Unplanned returns to the operating room (UPROR) were documented. Radiographic measurements were made on sitting full spine anteroposterior and lateral radiographs taken before the surgery (preoperative), immediately after the surgery (postoperative), and at the latest follow-up visit. Spinal heights were measured as the perpendicular distance between the two parallel lines that are drawn passing through the centers of the chosen endplates. 14 All the measurements were done by experienced pediatric orthopaedic surgeons at each institute.
Statistical analyses were performed using the software package SPSS (IBM Corp. Released 2015. IBM SPSS Statistics for Mac OS, Version 23.0, Armonk, NY). Descriptive statistics are presented as means and standard deviations/ranges. Dependent variables were compared by using the paired samples t-test of Wilcoxon signed rank test. Threshold for statistical significance was designated as p < 0.05.
Results
Twenty-eight patients (M/F = 12/16) were included in the study, with a mean follow-up of 16 months (range 2–41). The mean age at surgery was 61 months (range 29–96), and the mean age at DMT initiation was 12 months (range 1–58). Treatment with DMTs was started with nusinersen in all 28 of the patients, while four of them later switched to risdiplam, and another four switched to onasemnogene abeparvovec.
Growth-friendly instrumentation methods were used in all patients: Fifteen were treated with dual magnetically controlled growing rods (MCGR), four with unilateral MCGR in combination with a contralateral guided growth system 15 (Figure 1), three with Vertical Expandable Prosthetic Titanium Rib (VEPTR®) implants, five with self-distracting systems, and one with traditional dual growing rods.16,17 In four patients, instrumentation ended at L5 (Figure 2), while 24 patients were primarily fixed to the pelvis.
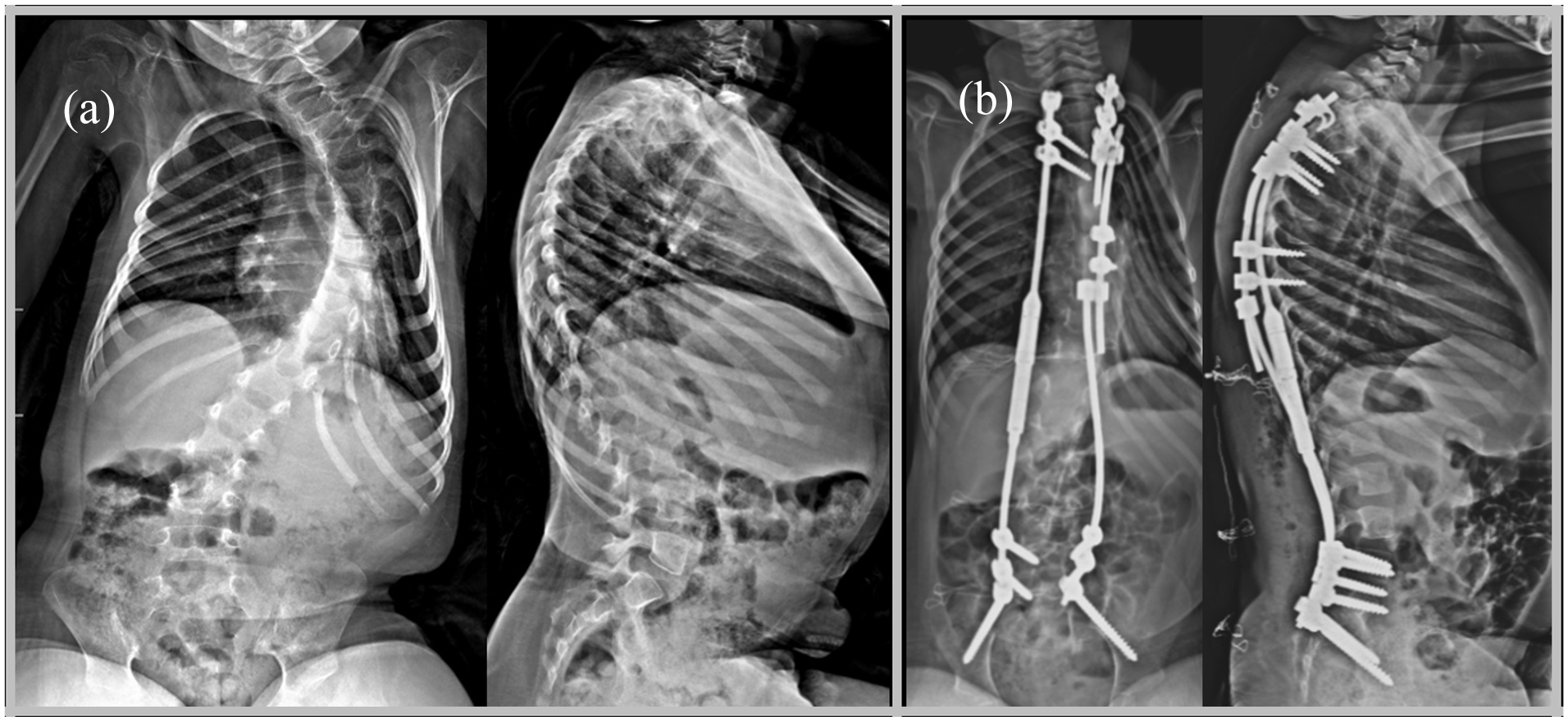
Preoperative (a) and postoperative (b) radiographs of a 76-month-old female patient, treated with magnetically controlled growing rods (MCGRs) and gliding rod combination technique.
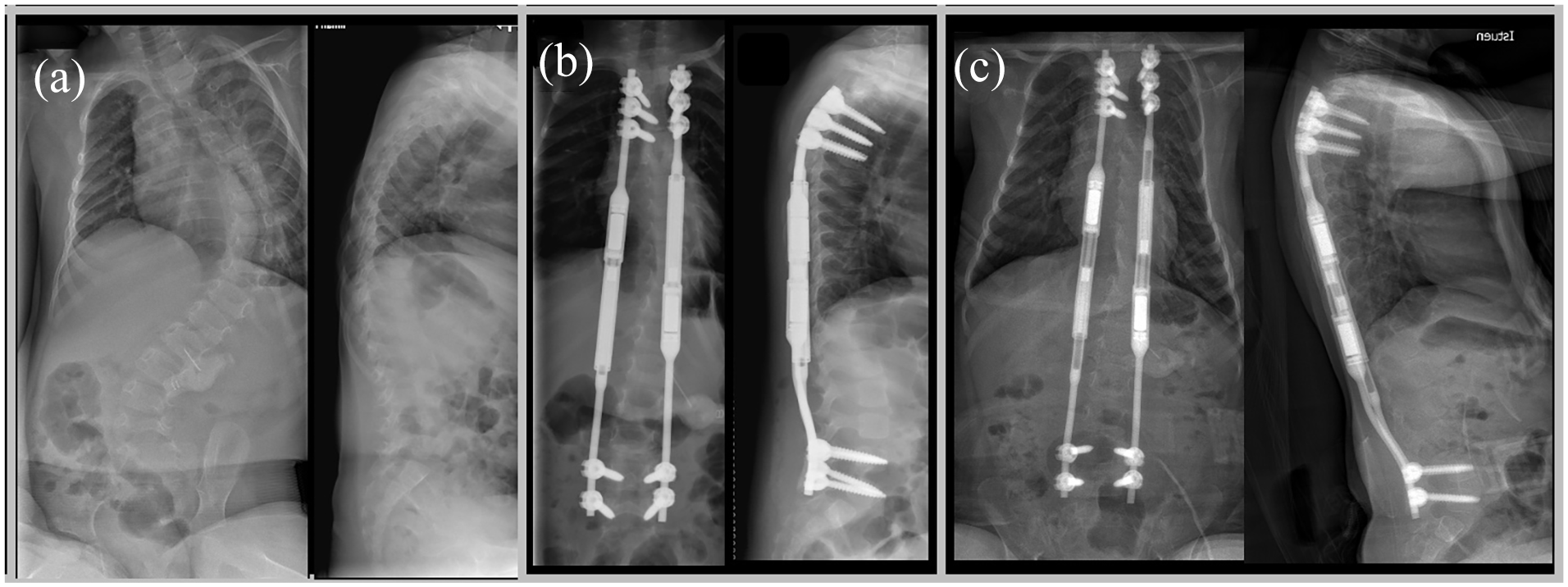
A 30-month old-female patient who was treated with dual magnetically controlled growing rods (MCGRs) between T2 and L5. Preoperative (a), postoperative (b), and 18-month follow-up (c) radiographs show successfully maintained spinal and pelvic balance.
Preoperatively, 14 (50%) patients exhibited subluxation in at least one of the hips, seven (25%) exhibited dislocations, and one (4%) had already undergone surgical treatment for bilateral hip dislocations. None of these patients had hip pain or required immediate surgical treatment for the hips at the time of the spine surgery, or afterwards during the follow-up.
The mean surgical time was 157 min (range 92–340), and estimated blood loss was 219 ml (range 50–400). The mean hospital stay duration was 5.6 days (range 3–13), while 18 patients (64%) required additional ICU care for 1.9 days (range 1–7) after the surgery.
Radiographic measurements are listed in Table 1. The mean amount of major curve correction after surgery was 57% (44°± 17), and pelvic obliquity correction was 83% (13°± 11). Major scoliotic curve and pelvic obliquity were corrected significantly by surgery (p < 0.001 for each). Pelvic obliquity did not increase during the follow-up (mean 23 months, range 12–36) in patients with a distal instrumentation level of L5. Seventeen out of 28 patients had a preoperative T2-12 kyphosis of over 70°, with a mean value of 89°± 12°, and it was reduced to 43°± 10° postoperatively. The latest follow-up mean thoracic kyphosis of this subgroup was 45°± 13°.
Radiographic measurements of the study population.
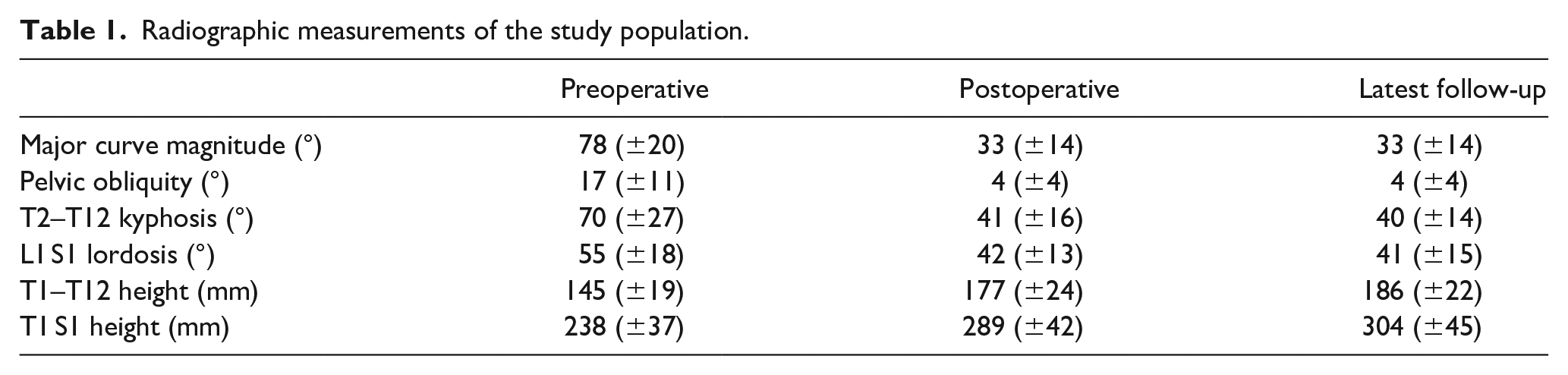
The mean amount of T1–12 height gained during surgery was 31 mm (±16 mm) while the mean T1 S1 height gain was 51 mm (±24 mm). Sixteen patients had a follow-up of ≥12 months, and in this subgroup, yearly T1–T12 height gain was 6.5 (±3.5) mm, while for T1 S1, it was 11.5 (±9.2) mm.
Five patients (18%) developed a total of six complications, with a rate of 0.16 complications/patient/year. The complications occurred after a mean period of 7 months (range 1–12) after the index surgery. Three deep surgical site infections (SSI), two proximal anchor failures, and one rod fracture were documented. All six complications required UPRORs, while in a patient with SSI, two surgical debridements were undertaken; therefore, a total of seven UPRORs and a rate of 0.19 UPRORs/patient/year were reported. In addition to these complications, one patient with a self-distracting system required a surgical intervention for implant exchange because the system reached its maximum distraction capacity of 5 cm. The complication rate within 90 days of the initial surgery was 7% (two SSIs), and within 30 days of initial surgery, it was 4% (one SSI).
No neurologic complications were recorded, and none of the patients experienced any difficulty during intrathecal injections for nusinersen after the instrumentation. Twenty-four patients were using night-time noninvasive ventilation devices before the surgery, and none required invasive ventilation assistance or tracheostomies during the follow-up. At the time of the latest follow-up, all patients were following their growth-friendly treatment course as planned.
Discussion
Treatment of EOS in patients with SMA depends on many factors including, but not limited to, disease severity, curve magnitude, and progression rate. Most of the studies focus on SMA types 2 and 3 as they are less affected by the disease and seem more amenable to surgery. However, it is well known that more severely affected patients (e.g. more pronounced affection of the axial muscles and more severely affected motor capabilities) develop scoliosis earlier and tend to progress at a higher rate. 6 Therefore, patients with SMA type 1 possess the real challenge as they almost invariably develop scoliosis within the first year of life and have a mean progression rate of 2.3/month which is considerably higher than the reported 5.6/year rate for SMA type 2.13,18 Owing to DMTs, these patients have now started seeking care in spine clinics more often, and the literature on their treatment options is sparse.
DMTs have changed the clinical phenotypes for all types of SMA, and the most dramatic improvement was seen in patients with SMA type 1. 19 When treatment is started very early, especially in the presymptomatic phase, prominent improvements can be expected. 20 However, newborn or prenatal screening is not universally available, and treatment is usually started after the onset of symptoms. In this case, patients with SMA type 1 can be expected to follow a clinical course more similar to the type 2 disease. 5 These assumptions can only go so far in describing the characteristics of spinal deformities in this patient group, as the recent studies suggest a different profile. Soini et al. 21 analyzed an SMA type 1 predominant group of patients after treatment with onasemnogene abeparvovec and reported a kyphosis-predominant deformity, which is not a generally accepted feature for SMA type 2. Another study reported that despite the functional improvements seen with nusinersen treatment, the effects were more prominent in the appendicular skeleton, and this lack of effect in the axial skeleton might have caused a 100% scoliosis rate in the first year of life. 13 On the contrary, another study on a larger group of patients reported a more prominent increase in the axial and proximal motor function scores. 22 As evident by these reports, DMT-treated SMA patients are a heterogenous population, and more data are necessary to acquire a better understanding of their deformity characteristics.
EOS of neuromuscular origin is mostly managed by growth-friendly surgical techniques to preserve spinal growth and pulmonary functions, and the same principles are applied in SMA. These treatments have been safely applied to patients with SMA type 2.8 –11,23 Lorenz et al. 11 prospectively analyzed a cohort of 17 MCGR-treated children with SMA type 2 with a minimum follow-up of 4 years and reported 13 complications of which eight required unplanned surgeries and a patient-related complication rate of 41%. For purposes of comparison, this can be converted into a rate of 0.17 complications/patient/year, and 0.1 UPRORs/patient/year, which are similar to the numbers we report. Another retrospective study on 66 patients reported a complication rate of 24% for MCGRs and 45% for VEPTRs. It must be emphasized that the hyperkyphotic deformity that is seen in our cohort may be causing a predisposition toward implant-related complications: A study on unplanned surgeries during EOS treatment showed that the general rate of 25% for neuromuscular EOS rises dramatically to 39% in patients with a hyperkyphotic deformity. 24 With increased follow-up, complication rate of our cohort will inevitably rise. Patients must be closely monitored for implant-related mechanical complications, anchor failure and proximal junctional kyphosis.
As the primary outcome of our study was on safety, it is worthy of note that none of our patients experienced any major medical or nontreatable complications during the initial hospital stay or the following 90 days. A 2011 study on 15 patients with SMA reported worsened respiratory status in two patients and intraoperative cardiac arrest in one patient. 8 These major complications have not occurred in our cohort, neither in the other recent studies.10,11,23 In the last decade, not only DMTs but also the improvements in general medical care of these patients certainly played an important role in making surgery a safer option.
Complications and readmissions within the first 90 days after the initial surgery is a well-known quality measure for surgical procedures and reflects on the safety, feasibility, and cost-effectiveness of that intervention.25 –27 Especially in patients with neuromuscular disorders such as SMA, the severity of the disease itself will cause frequent medical problems, including mortality. 28 For this reason, longer follow-ups are very likely to be eventful. 29 To assess the safety of an intervention, serious adverse events within the first 90 days postoperatively can be considered more likely related with the procedure than the disease itself. In this aspect, our study is reassuring for the safety of growth-friendly instrumentation in this patient group.
Additional respiratory support was not needed in any of the patients, and we observed that the pulmonary effects of growth-friendly instrumentation in the short term are manageable. However, studies focusing on the pulmonary functions have noted that percentage of predicted forced vital capacity (FVC) tends to decrease in these patients after growth-friendly instrumentation.10,30 It is important to remind that these studies mostly report on patients not receiving DMTs, and pulmonary function in this group is negatively affected not only by the scoliotic deformity but also by the gradually weakening respiratory muscles. DMTs may help in preventing the latter, and surgery may have a more permanent positive effect. More follow-up and detailed pulmonary function analyses are obviously needed.
Growth-friendly instrumentation systems have been shown to control the spinal deformity effectively, both in SMA and the general EOS population.8,9,11,31 We have achieved 57% correction of the main scoliotic curve after surgery, which is in line with the literature as the reported rates in patients with SMA generally range between 40% and 60%.8,11,23 We also observed that after the mean follow-up of 16 months, major curve correction was preserved. Short-segment apical fusions can also be considered in patients with larger curves, as they achieved successful deformity control in select patients with SMA type 2. 32 Hyperkyphosis was a prominent feature in our cohort, similar to the report by Soini et al. 21 This is not pointed out by other series on patients with SMA type 2 disease, so it may be a unique feature for DMT-treated SMA type 1 patients and needs further studies with larger cohorts to confirm. We successfully managed to reduce the mean thoracic kyphosis from 70° to 41° on the latest follow-up, and we advise due diligence on this matter to prevent high rates of implant-related problems. Pelvic obliquity is another very important component of the deformity, which was corrected at a mean of 83%. All patients in our study group achieved good sitting balance after surgery, which remained stable at the latest follow-up, including the four patients with a distal instrumented level of L5.
In addition to the retrospective design and small patient cohort, the short follow-up period is a major limitation of this study. Growth-friendly treatment is known for high complication and reoperation rates, and with longer follow-up, complication rates will most likely increase. However, we believe that short-term observations are relevant to demonstrate the safety and feasibility of this procedure in this vulnerable patient population. Heterogeneity of our cohort by means of different devices is both an advantage as to show real-world data and a limitation as different devices have different mechanisms of action.
A growing number of SMA type 1 patients are seeking care in spine clinics after the introduction of DMTs, and the number of surgeries performed on these patients is expected to increase. To our knowledge, this is the first study in the literature focusing on the safety and efficacy of growth-friendly instrumentation on patients with SMA type 1 and EOS receiving DMTs. We report that early-onset spinal deformity in this population can be safely treated with growth-friendly instrumentation, with similar early complication rates and deformity control ability when compared with SMA type 2.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521231214780 – Supplemental material for Safety and efficacy of growth-friendly instrumentation for early-onset scoliosis in patients with spinal muscular atrophy type 1 in the disease-modifying treatment era
Supplemental material, sj-pdf-1-cho-10.1177_18632521231214780 for Safety and efficacy of growth-friendly instrumentation for early-onset scoliosis in patients with spinal muscular atrophy type 1 in the disease-modifying treatment era by Riza Mert Cetik, Dror Ovadia, Kiril Mladenov, Moyo C Kruyt, Ilkka Helenius, Matti Ahonen, Daniel Studer and Muharrem Yazici in Journal of Children’s Orthopaedics
Footnotes
Author contributions
RMC: Data analysis, manuscript drafting, final approval. DO: Data acquisition, reviewing of the manuscript, final approval. KM: Data acquisition, reviewing of the manuscript, final approval. MCK: Data acquisition and interpretation, manuscript drafting, final approval. IH: Conceptualization, data acquisition, reviewing of the manuscript, final approval. MA: Data acquisition and interpretation, reviewing of the manuscript, final approval. DS: Data acquisition and interpretation, reviewing of the manuscript, final approval. MY: Conceptualization, supervision, data acquisition, and final approval.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: IH reports receiving grants to study group (Finnish Pediatric Research Foundation), research grants to institution (Medtronic, Stryker, Nuvasive and Cerapedics), consultant fees (Medtronic), payment for lectures (Nuvasive), and support for traveling expenses (Medtronic). MA reports receiving research grants (Finnish Pediatric Research Foundation). MCK reports ownership (Cresco Spine) and inventor rights (Spring distraction system). DO reports reimbursement of expenses for a lecture (Nuvasive). MY reports holding the position of Secretary in the Pediatric Spine Foundation.
Ethical statements
The study was approved by the Ankara Etlik City Hospital Ethical Committee. Address: Varlık Mah., Halil Sezai Erkut Cd., 06010 Yenimahalle, Ankara/TURKEY. Approval number: AEŞH-EK1-2023-264. Approval date: 12.07.2023
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
All the patients and their legal representatives included in this study have given written informed consent.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
