Abstract
Background:
Patient-reported outcome measures have become crucial in the clinical evaluation of patients. Appropriate selection, in a young population, of the instrument is vital to providing evidence-based patient-centered healthcare. This systematic review applies the COnsensus-based Standards for selection of health Measurement INstruments methodology to provide a critically appraised overview of patient-reported outcome measures targeted at pediatric orthopedic patients with lower limb impairment.
Method:
A systematic search of electronic databases was performed to identify original studies reporting the development and/or validation of patient-reported outcome measures evaluating children with impairment of the lower extremity. Data extraction, quality assessment, and risk of bias evaluation were performed following the COnsensus-based Standards for selection of health Measurement INstruments guidelines and Preferred Reporting Items for Systematic reviews and Meta-Analyses statement.
Results:
A total of 6919 articles were screened. Thirty-three studies were included, reporting evidence on the measurement properties of 13 different patient-reported outcome measures and 20 translations. Four studies reported on content validity and patient-reported outcome measure development. The methodological quality of studies on structural validity, content validity, or patient-reported outcome measure development was mostly rated as “doubtful” or “very good.” The quality of evidence on measurement properties varied noticeably, with most studies needing to perform improve their methodological quality to justify their results.
Conclusion:
This review provides an extensive overview of all available patient-reported outcome measures for patients with lower extremity impairment within pediatric orthopedics. We cautiously advise the use of four patient-reported outcome measures. However, the scarce availability of research on content validity and patient-reported outcome measure development highlights an area for future research endeavors to improve our knowledge on the currently available patient-reported outcome measures.
Level of evidence:
Diagnostic level 1
Keywords
Introduction
A patient-reported outcome (PRO) is an informational statement coming directly from patients describing their mental and physical well-being over time without the clinicians’ interpretation. 1 Measuring these outcomes accurately requires a validated patient-reported outcome measure (PROM). 1 Clinicians and researchers can, through PROMs, evaluate a patient’s health status and track changes over time.2,3 PROMs are specifically developed to evaluate the patient within a predefined study population and require validation studies to provide adequate scientific justification for proper usage. A recent study by Arguelles et al. 4 revealed improper use of the majority of PROMs in pediatric orthopedic research. These results describe the adverse effects of using PROMs without proper validation studies, consequently influencing the findings of researchers and clinicians using these pediatric orthopedic PROMs.
The selection of a PROM is vital to its effectiveness. Factors essential to the proper selection and application of a PROM are the study population and disease for which the PROM was developed and subsequently validated. Therefore, it is imperative that clinicians and researchers are provided with validated PROMs. Traumatic injuries/impairments of the extremities are among the most common pediatric diagnoses.5,6 Meanwhile, clinical follow-up through survival-based outcomes has transitioned to clinical follow-up using PROMs.7,8 The need for proper use of validated PROMs has become more apparent because of this transition and increased incidence of injury.
This study is part of a research collaboration that aims to provide a comprehensive overview of PROMs used in pediatric orthopedics, evaluate the methodological quality of the respective validation studies, and formulate a recommendation on proper PROMs selection. A comprehensive overview of PROMs used for evaluating upper extremity problems has been previously published as part of this joint effort. 9 This review will provide an extensive overview of PROMs used in orthopedic patients with a lower extremity impairment.
Methods and materials
A detailed study protocol has been previously published. 10
Design: This systematic review was performed in accordance with the 10-step procedure within the updated COnsensus-based Standards for selection of health Measurements INstruments (COSMIN) risk of bias guidelines.11 –13 This systematic review adhered to the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) statement when reporting the synthesized evidence and data. 14
Pre-registration: This study was pre-registered in PROSPERO with registration number: CRD42021287323.
Search strategy: The electronic database of PubMed and Embase were systematically searched to identify all relevant and current studies between 1 January 2000 and 1 December 2022. A clinical librarian was consulted to design a search string for both search engines to improve sensitivity and ensure proper identification of all relevant studies. The search was restricted to English and/or Dutch studies using a language filter. The search strings for each database can be found in greater detail in Appendix 1. In addition, a validated pediatric study search filter (by Leclercq et al. 15 ) and two validated data filters (by Terwee et al. 16 ) were used to enhance sensitivity.
Eligibility criteria: Articles were considered eligible for inclusion if the following criteria were met: the full-text article of the original study was available and must describe the development and/or the evaluation of one or more measurement properties of a generic and/or disease-specific patient-reported and/or proxy-reported questionnaire of any language, in a population consisting of children (0–18 years old) with an impairment of the lower extremity. The exclusion criteria were as follows: a study using PROMs as an outcome measurement instrument or a PROM in a study population consisting of children with prosthetics.
Study selection: Study selection was split into two phases: a screening selection through title and abstract followed by a careful selection through full text. Two reviewers (T.F.F.S. and J.P.R.K.) independently identified eligible articles, and disagreements were resolved by a third reviewer (C.J.A.v.B.).
Data extraction and appraisal: The data of all included studies were extracted and underwent qualitative appraisal using the COSMIN methodology for qualitative evaluation of PROMs. 11 More detailed information on the taxonomy, guideline, and checklists can be found in studies by Mokkink et al.,17,18 Terwee et al., 19 and Prinsen et al. 11
Evaluation of the methodological quality: The methodological quality of all included studies assessing the following measurement properties were rated with the COSMIN Risk of bias checklist; 12 validity (structural validity, hypotheses testing for construct validity and cross-cultural validity), reliability (internal consistency, reliability, and measurement error), and/or responsiveness of a PROM. To evaluate the overall methodological quality, the study was rated on a 4-point scale: “very good,” “adequate,” “doubtful,” or “inadequate.” After this “the worst score counts” principle was applied to determine the study’s overall methodological quality on the PROM’s psychometric properties.
Data extraction: The data on the patient characteristics of the included study populations, the characteristics of the included PROMs, and the evidence within each study on a psychometric property were extracted using the predefined tables within the COSMIN risk of bias checklist. 11
Assessment of psychometric properties: A set of predefined criteria 11 was constructed to standardize the rating of the studies on measurement properties. The results gathered from each individual study were rated as “sufficient (+),” “insufficient (-),” or “indeterminate (?)” accordingly.
Evidence synthesis: The evaluation of the methodological quality and the assessment of the studies on psychometric properties were performed by two reviewers (T.F.F.S. and J.P.R.S.) independently. If consensus could not be reached, an additional reviewer (C.J.A.v.B. and/or I.N.S.) was consulted. To ensure an evaluation and assessment of high quality, both reviewers upheld the predefined criteria corresponding to the COSMIN risk of bias checklist. The inter-rater agreement was considered appropriate when reviewers reached >80% agreement, a value proposed by the COSMIN development group. 20
Results
General characteristics of included studies and instruments: After duplicate removal (n = 2792), a total of 6919 original studies were identified. After careful selection using the predefined inclusion and exclusion criteria, 33 original studies were included. The PRISMA flow diagram describing the selection process is shown in Figure 1. The inter-rater agreement was calculated to be 87.8%, which exceeded the predefined minimum of 80% and was therefore deemed appropriate.
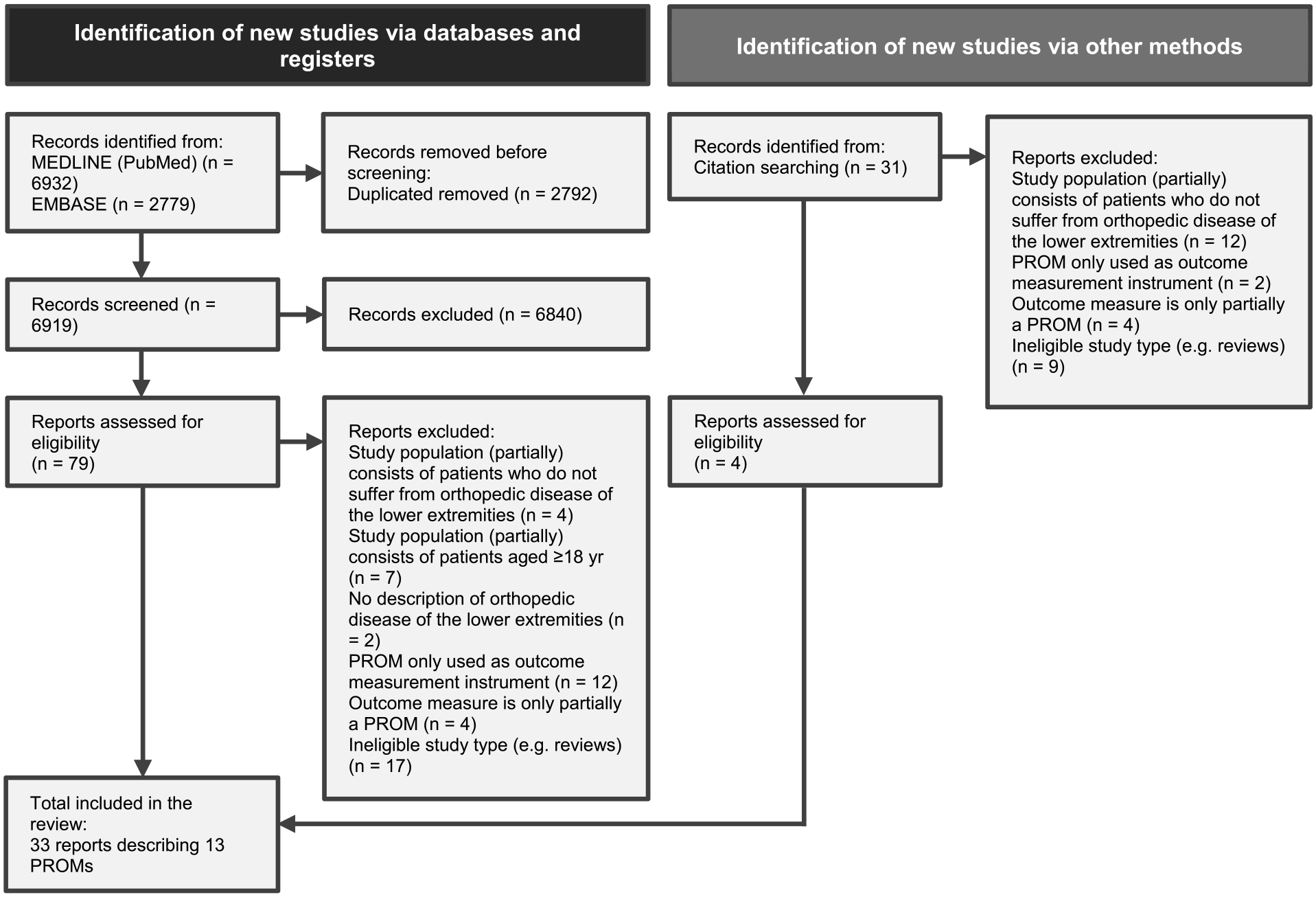
PRISMA flowchart detailing the article selection process with the inclusion and exclusion criteria.
The key characteristics of the included studies and their study populations are described in Table 1. The 33 studies reported evidence on 13 individual PROMs and 20 translations. The least evaluated psychometric property was the responsiveness of the instrument (n = 1). The most evaluated psychometric property was the construct validity (n = 26). The detailed characteristics of the included measurement instruments can be found in Table 2.
Characteristics of the included studies.
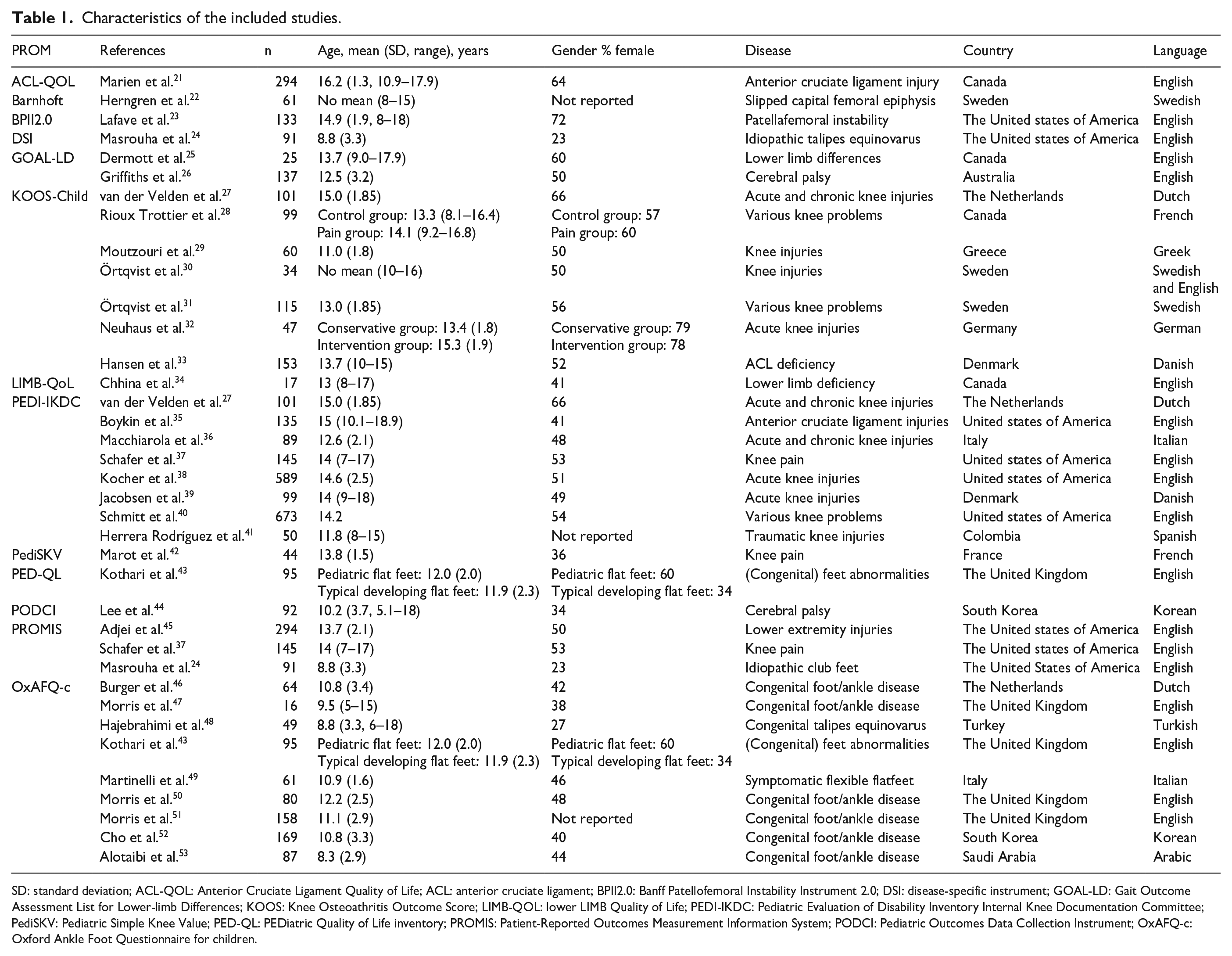
SD: standard deviation; ACL-QOL: Anterior Cruciate Ligament Quality of Life; ACL: anterior cruciate ligament; BPII2.0: Banff Patellofemoral Instability Instrument 2.0; DSI: disease-specific instrument; GOAL-LD: Gait Outcome Assessment List for Lower-limb Differences; KOOS: Knee Osteoathritis Outcome Score; LIMB-QOL: lower LIMB Quality of Life; PEDI-IKDC: Pediatric Evaluation of Disability Inventory Internal Knee Documentation Committee; PediSKV: Pediatric Simple Knee Value; PED-QL: PEDiatric Quality of Life inventory; PROMIS: Patient-Reported Outcomes Measurement Information System; PODCI: Pediatric Outcomes Data Collection Instrument; OxAFQ-c: Oxford Ankle Foot Questionnaire for children.
Characteristics of the included PROMs.
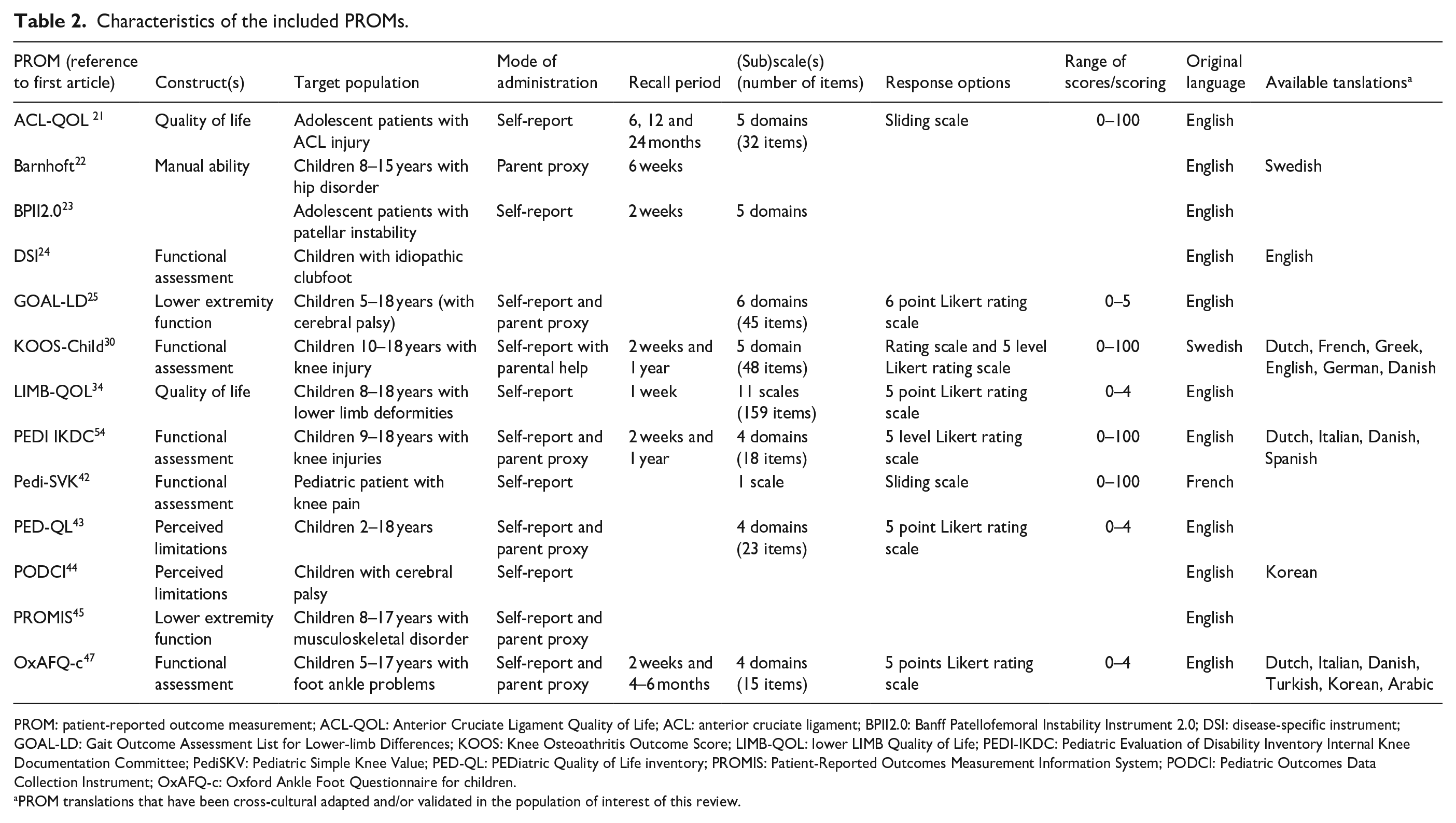
PROM: patient-reported outcome measurement; ACL-QOL: Anterior Cruciate Ligament Quality of Life; ACL: anterior cruciate ligament; BPII2.0: Banff Patellofemoral Instability Instrument 2.0; DSI: disease-specific instrument; GOAL-LD: Gait Outcome Assessment List for Lower-limb Differences; KOOS: Knee Osteoathritis Outcome Score; LIMB-QOL: lower LIMB Quality of Life; PEDI-IKDC: Pediatric Evaluation of Disability Inventory Internal Knee Documentation Committee; PediSKV: Pediatric Simple Knee Value; PED-QL: PEDiatric Quality of Life inventory; PROMIS: Patient-Reported Outcomes Measurement Information System; PODCI: Pediatric Outcomes Data Collection Instrument; OxAFQ-c: Oxford Ankle Foot Questionnaire for children.
PROM translations that have been cross-cultural adapted and/or validated in the population of interest of this review.
Synthesized evidence: The results of the methodological quality assessment of the included studies, according to the COSMIN guidelines, can be found in Table 3. In addition, a comprehensive summary of the pooled results per psychometric properties reported for each study on an original PROM, according to COSMIN guidelines and GRADE approach, can be found in Table 4.
Methodological quality and ratings of measurements properties of the included PROMs.

PROM: patient-reported outcome measurement; ACL-QOL: Anterior Cruciate Ligament Quality of Life; ACL: anterior cruciate ligament; BPII2.0: Banff Patellofemoral Instability Instrument 2.0, DSI: disease-specific instrument; GOAL-LD: Gait Outcome Assessment List for Lower-limb Differences; KOOS: Knee Osteoathritis Outcome Score; LIMB-QOL: lower LIMB Quality of Life; PEDI-IKDC: Pediatric Evaluation of Disability Inventory Internal Knee Documentation Committee; PediSKV: Pediatric Simple Knee Value; PED-QL: PEDiatric Quality of Life inventory; PROMIS: Patient-Reported Outcomes Measurement Information System; PODCI: Pediatric Outcomes Data Collection Instrument; OxAFQ-c: Oxford Ankle Foot Questionnaire for children.
Synthesized evidence.
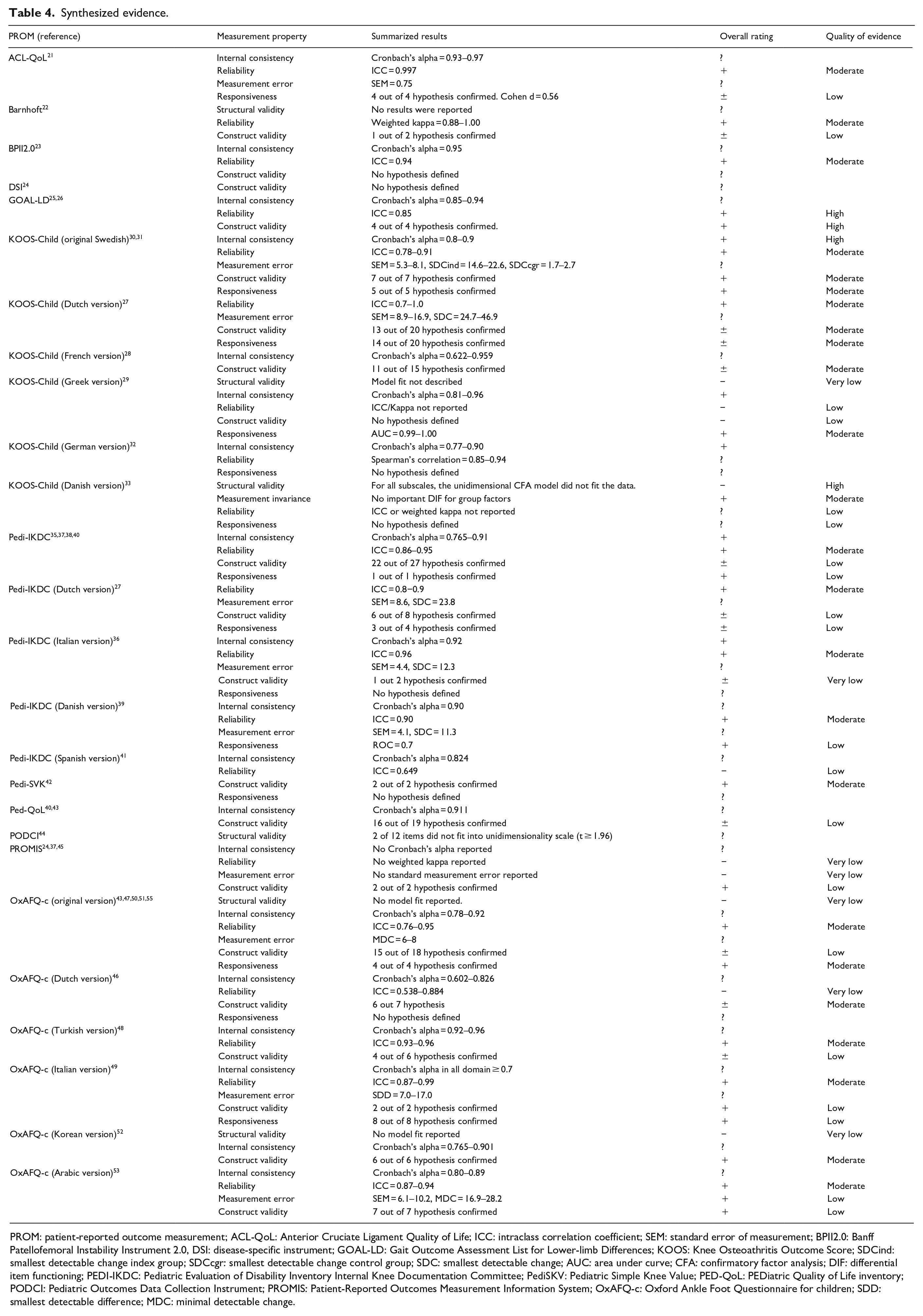
PROM: patient-reported outcome measurement; ACL-QoL: Anterior Cruciate Ligament Quality of Life; ICC: intraclass correlation coefficient; SEM: standard error of measurement; BPII2.0: Banff Patellofemoral Instability Instrument 2.0, DSI: disease-specific instrument; GOAL-LD: Gait Outcome Assessment List for Lower-limb Differences; KOOS: Knee Osteoathritis Outcome Score; SDCind: smallest detectable change index group; SDCcgr: smallest detectable change control group; SDC: smallest detectable change; AUC: area under curve; CFA: confirmatory factor analysis; DIF: differential item functioning; PEDI-IKDC: Pediatric Evaluation of Disability Inventory Internal Knee Documentation Committee; PediSKV: Pediatric Simple Knee Value; PED-QoL: PEDiatric Quality of Life inventory; PODCI: Pediatric Outcomes Data Collection Instrument; PROMIS: Patient-Reported Outcomes Measurement Information System; OxAFQ-c: Oxford Ankle Foot Questionnaire for children; SDD: smallest detectable difference; MDC: minimal detectable change.
Content validity and PROM development: Four studies described content validity and PROM development. To determine the methodological quality of PROM development, a study should report on the comprehensibility and comprehensiveness of the instrument. Two studies25,30 reported on these parameters and were rated as “adequate.” Two studies34,47 reported on these parameters and were rated as “very good.”
Structural validity: The structural validity was evaluated for six PROMs.22,29,33,44,51,52 Three studies reported on the structural validity of a cultural adaption of the original PROMs.29,44,52 All studies 22,29,33,44,51,52 failed to report on the important aspects of the item-related theory or Rasch analyses and did not describe the model fit. The results of the PROMs did not meet the COSMIN criteria for good measurement properties.
Internal consistency: To provide a correct analysis on the quality of the internal consistency within a PROM, the instrument must show at least low-quality evidence of its structural validity. Accordingly, the results of the analyses on internal consistency for 19 PROMs were reported as “indeterminate” due to insufficient evidence on the structural validity studies of these PROMs. The original version of the KOOS-Child questionnaire 30 and the original version of the OxAFQ-c 47 were the only two studies that both showed an adequate quality of evidence for structural validity and were therefore rated.
Reliability: Twenty-one studies21 –23,26,27,29,31 –33,36,38,39,41,45,46,48,49 –51,53 reported on the test–retest reliability of the instrument. Four studies reported no intraclass correlation coefficient (ICC) values or ICC values ranging below <0.7, thus demonstrating insufficient evidence on the quality of the test–retest reliability of the instrument.29,41,45,46
Measurement error: Eleven studies reported the measurement error of their respective PROM.21,27,31,33,36,39,45,49,50,53 None of the PROMs included in these analyses had previously published information on the minimal important change. Therefore, all included data on measurement error were rated as “indeterminate.”
Discussion
This is the first systematic review to provide an extensive overview of the available evidence on the psychometric properties of PROMs used to evaluate pediatric patients with an impairment of the lower extremity. This systematic review is a continuation of the joined effort to provide a comprehensive overview of the evidence on the psychometric properties of PROMs used for both upper and lower extremities.9,10 With the use of the updated COSMIN methodology, to ensure a high-quality assessment, a total of 6919 publications were screened, resulting in 13 PROMs reporting a total of 112 psychometric properties. The results of this study increase the knowledge on proper use of PROMs through an evidence-based review and extensive recommendations.
Given the data currently available and our corresponding analyses for both upper and lower extremities, which together constitute the current evidence on psychometric properties of pediatric orthopedic PROMs, a lack of sufficient quality evidence becomes unmistakable. Consequently, our extensive overview demonstrates that the use of the instruments included in this review and currently used in pediatric orthopedics for evaluating lower extremity impairment is not sufficiently supported. At least a low-quality evidence on content validity and internal consistency, combined with at least adequate methodological quality of the study itself, is essential to justify proper usage of a PROM in current clinical practice and for research purposes. 11 Therefore, more studies should have provided sufficient evidence to support the structural validity of the instruments and have provided more evidence on content validity and internal consistency.
The most commonly researched PROMs, and commonly used in current practice, are the KOOS-Child, OxAFQ-c, and Pedi-IKDC. Other PROMs (ACL-QoL, Barnhoft, BPII 2.0, DSI, Pedi-SVK, Ped-Qol, PODCI, PROMIS) have shown at least adequate evidence on methodological quality of their studied psychometric properties; however, these PROMs have no evidence on the quality of content validity and PROM development. During our analyses, four PROMs were supported with evidence on PROM development and/or content validity. The OxAFQ-c, LIMB-Q, and GOAL-LD25,34,47 provided adequate quality of evidence on content validity, the most important psychometric property when determining whether or not the instrument has the ability to detect how well an instrument covers all relevant parts of the construct it aims to measure. The KOOS-Child provided very good evidence on the respective PROM development but no evidence concerning content validity. Our analyses showed none of the PROMs were tested for the same population and disease in two different individual studies. Because of the strengths of the studies on the KOOS-Child, OxAFQ-c, and GOAL-LD, we could formulate a very cautious positive advice on the use of these PROM. The PEDI-IKCD has no supportive evidence on the development of the PROM; however, studies provided quality evidence on the psychometric properties, we could also cautiously advice using the PEDI-IKCD.
For clinicians and researchers using the instrument, it is imperative to know whether the instrument is fitted to test the study population at hand. The use of each instrument should therefore be validated for a specific patient population and it should be clearly stated for what disease or disability the PROM has been validated. The patient and disease-specific characteristics of the included study populations were, in general, not described in extensive detail; the severity of the disease was mostly left out. The applicability of an instrument to be tested on a specific population is therefore lessened, especially combined with the overall very low quality of provided evidence for the suspected population in which the instrument is tested in the original study.
The COSMIN methodology makes a review on psychometric properties of PROMs a standardized process. This systematic approach strengthens the objective results gathered. However, using the COSMIN checklist requires subjective input and judgment by the reviewer. To address this potential source of selection and information bias, two (and sometimes three) reviewers independently extracted, gathered, and evaluated the data according to the COSMIN guidelines after which these results were shared, pooled, and discussed. Throughout the evaluation on the methodological quality of the studies, the “worst score counts” principles was applied in multiple instances. The incidental subjective input from the reviewers combined with a “worst score counts” principles could lead to an underestimate of the true quality and quantity of the evidence available. This is especially important in pediatric orthopedic PROMS, where research is scarce and not widely known or available.
In conclusion, a comprehensive overview was given of PROMs for pediatric patients with lower extremity impairment. We cautiously advice on the use of the KOOS-Child, OxAFQ-c, and GOAL-LD PROM because of their relevant evidence on PROMs development and content validity with supportive evidence on the psychometric properties. The Pedi-IKCD could also, cautiously, be used in practice due to good quality of evidence on the psychometric properties; however, no studies support the content validity or PROM development of this PROM. Noticeably, the absence of content validity–“only” studies on the PROMs accentuates the knowledge gap in proper PROMs usage, as no study has provided any evidence that the PROMs reflect the construct they intend to measure. These results are similar for PROMs used for pediatric patients with upper extremity impairments. Future efforts must be made to develop high-quality and well-validated patient-reported outcomes measurement questionnaire for the pediatric orthopedic patient. A summarized overview of the PROMs which can, cautiously, be used in daily clinical practice and research can be found in Table 5.
Summary of the PROMs which have shown enough evidence to be used in daily clinical practice and for research purposes.
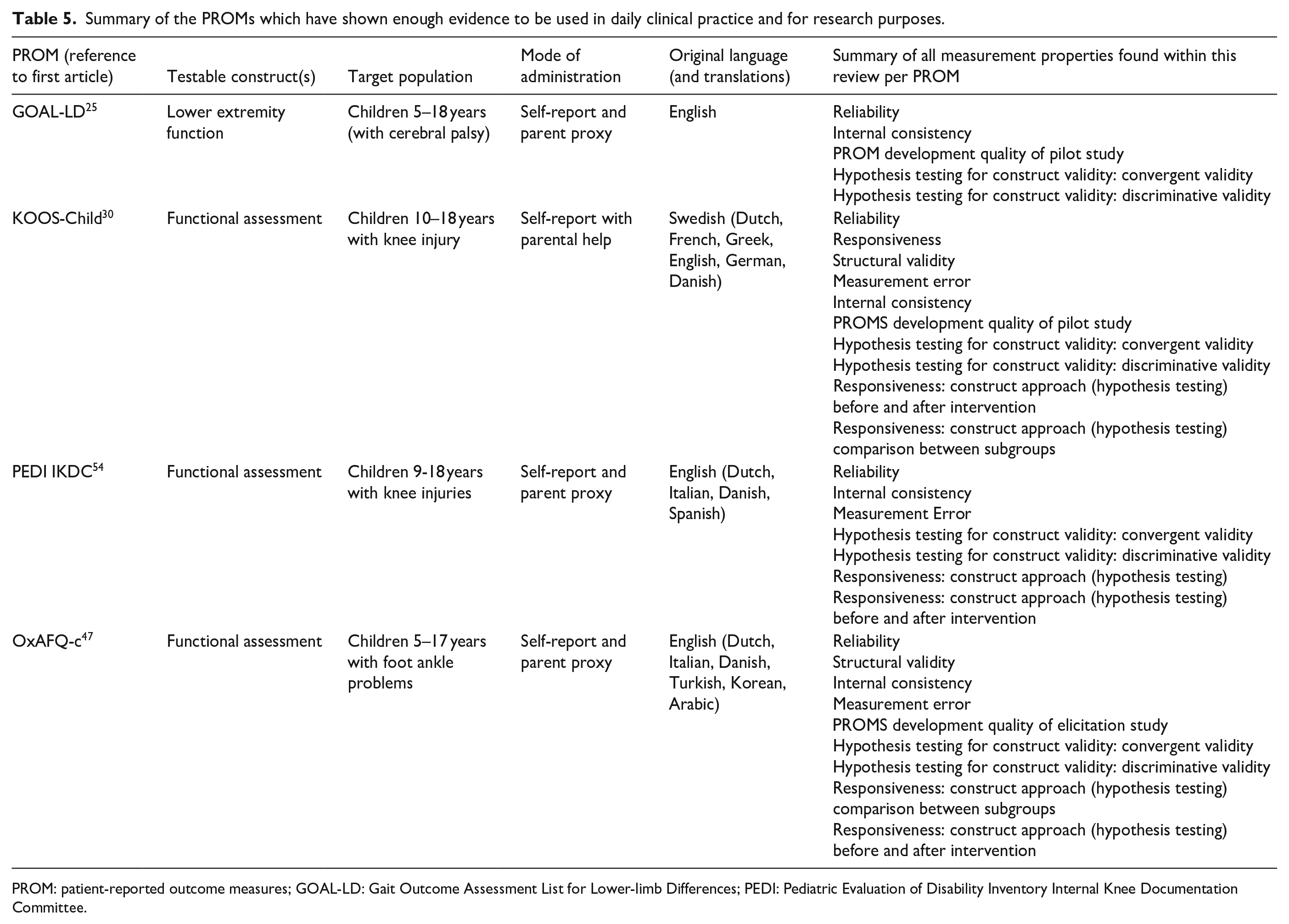
PROM: patient-reported outcome measures; GOAL-LD: Gait Outcome Assessment List for Lower-limb Differences; PEDI: Pediatric Evaluation of Disability Inventory Internal Knee Documentation Committee.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521231207081 – Supplemental material for Quality of patient- and proxy-reported outcomes for children with impairment of the lower extremity: A systematic review using the COnsensus-based Standards for selection of health Measurement INstruments methodology
Supplemental material, sj-pdf-1-cho-10.1177_18632521231207081 for Quality of patient- and proxy-reported outcomes for children with impairment of the lower extremity: A systematic review using the COnsensus-based Standards for selection of health Measurement INstruments methodology by Tim FF Saris, JP Ruben Kalle, Inger N Sierevelt, Denise Eygendaal and Christiaan JA van Bergen in Journal of Children’s Orthopaedics
Supplemental Material
sj-pdf-2-cho-10.1177_18632521231207081 – Supplemental material for Quality of patient- and proxy-reported outcomes for children with impairment of the lower extremity: A systematic review using the COnsensus-based Standards for selection of health Measurement INstruments methodology
Supplemental material, sj-pdf-2-cho-10.1177_18632521231207081 for Quality of patient- and proxy-reported outcomes for children with impairment of the lower extremity: A systematic review using the COnsensus-based Standards for selection of health Measurement INstruments methodology by Tim FF Saris, JP Ruben Kalle, Inger N Sierevelt, Denise Eygendaal and Christiaan JA van Bergen in Journal of Children’s Orthopaedics
Footnotes
Acknowledgements
A special thanks to Paulien H. Wiersma MSc, faculty liaison medical sciences (University Library Utrecht, UMC Utrecht), for helping to create our search strings.
Authors’ note
All listed authors have contributed to the design and analysis of the decision rule, commented on the drafts, and approved the final manuscript and agree with being accountable for all of its aspects and with its submission to Journal of Children’s Orthopaedics all in accordance with the ICMJE guidelines.
Author contributions
In the process of creating the manuscript, the five authors fulfilled the following roles and tasks:
1. TFFS Main author and first reviewer; design of the review, constructing and executing the search strategy, screening publications, analysis and interpretation of data, conception, and writing of the manuscript.
2. JPRK Second reviewer; constructing and executing the search strategy, screening publications, analysis, and interpretation of data.
3. INS Second supervisor; providing critical revisions and help with interpretation of data.
4. DE Third supervisor; provided orthopedic and scientific expertise and critical revisions to the study protocol.
5. C.J.A.v.B. First supervisor; conception and design of the review, provided orthopedic and scientific expertise and critical revisions to the study protocol.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
There are no human participants in this article and informed consent is not applicable.
Consent for publication
Not applicable.
Availability of data and materials
All data analyzed during this study are included in this article and its supplementary files.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
