Abstract
Background:
Proximal femoral and/or pelvic osteotomies (PFPOs) can be indicated for a multitude of hip pathologies in (often asymptomatic) children, to prevent future hip problems. These procedures can result in significant blood loss. Tranexamic acid (TXA) is an antifibrinolytic agent routinely administered in adult trauma and joint reconstruction surgery to reduce blood loss. TXA is also registered for use in children and reported safe and beneficial for pediatric trauma, cardiac, and spinal surgery. However, for pediatric orthopedics, particularly for PFPOs, the available evidence is limited. Therefore, the current trial will investigate the potential reducing effect of preoperative TXA on intraoperative blood loss in pediatric PFPOs.
Methods:
In this single-center, double-blind, randomized placebo-controlled trial, we aim to include 180 participants aged from 1 to 18 years undergoing PFPOs for any indication at our institution. Participants will be randomized to receive either TXA or placebo (saline) during anesthetic induction. The primary outcome is intraoperative estimated blood loss (mL/kg), which is determined gravimetrically. Secondary outcomes include the percentage of patients with excessive blood loss (>20 mL/kg), procedure time and hospital stay, and postoperative hemoglobin level changes.
Discussion:
This will be the first prospective study investigating the effect of preoperative TXA on intraoperative blood loss in pediatric PFPOs. Its results will help to determine whether it would be advisable to adopt preoperative TXA as a standard medication to minimize blood loss and prevent complications in this vulnerable population.
Trial registration:
EudraCT: 2022-002384-30. Prospectively registered on September 26, 2022.
Introduction
Proximal femoral and/or pelvic osteotomies (PFPOs) are surgical procedures aiming to realign incongruent hip joints. Children undergoing PFPOs are often asymptomatic but are treated to maintain walking function or sitting balance or to prevent future pain and loss of function. 1 The range of indications for PFPOs is wide, including developmental dysplasia of the hip (DDH), blood flow problems (e.g. Perthes disease), sequalae of trauma, and neuromuscular conditions. 1 PFPOs can lead to significant blood loss. Average blood loss in pediatric patients ranges from 159 mL to 369 mL in proximal femoral osteotomies2,3 and 200 mL to 971 mL in combined (proximal femoral and pelvic) osteotomies.4,5 Blood product transfusion is reported in 9.8%–61.5% of cases.2,3,5 Substantial blood loss and transfusions are associated with increased length of hospital stay, higher mortality, pneumonia, and other adverse events.6,7 To prevent substantial blood loss and transfusions, as well as associated complications, it is essential to evaluate potential preventive measures.
Antifibrinolytic agents such as tranexamic acid (TXA) have been reported to reduce intraoperative blood loss. 8 TXA is a synthetic derivate of lysine and binds to plasminogen to prevent plasmin activation and thus degradation of blood cloths. In adults, TXA administration is associated with reduced blood loss and mortality in trauma patients and with less blood loss and transfusion rates in arthroplasty, spinal surgery, and osteotomies.9 –12 In addition, the use of TXA is reported safe, with few adverse events. 13 Consequently, TXA is a part of standard care for adult trauma and joint reconstruction surgery in the Netherlands and other countries.
Considering pediatrics, TXA use has been reported beneficial and safe for trauma, cardiac, and spinal surgeries.14 –16 As solid evidence and guidelines for pediatric orthopedic indications are not available, TXA is applied ad hoc and on a variable basis in clinical practice, following the physician’s preference. In a recent retrospective analysis at our institution, an association of TXA use with reduced intraoperative blood loss was found in children undergoing PFPOs. 17 Other retrospective studies examining the effect of various TXA regimes on intraoperative blood loss in pediatric PFPOs for several neuromuscular conditions, so excluding DDH, have yielded heterogeneous findings.2 –5 Since high-quality evidence is lacking, well-designed randomized controlled trials (RCTs) should investigate the effect of TXA on blood loss in PFPOs to confirm or reject previous low-quality findings.
Therefore, we will perform a double-blind randomized placebo-controlled trial in which we will investigate the effect of preoperative TXA on intraoperative blood loss during PFPOs in children. Our primary aim is to determine whether preoperative TXA administration is associated with a reduced amount of intraoperative blood loss compared to placebo. Second, we aim to assess whether TXA reduces excessive intraoperative blood loss (>20 mL/kg), procedure time, and hospital stay and the decrease in postoperative hemoglobin levels, compared to placebo. Furthermore, we will investigate what patient and treatment characteristics besides TXA are associated with intraoperative blood loss. We hypothesize that preoperative TXA administration will result in less intraoperative blood loss, fewer patients with excessive intraoperative blood loss, shorter procedure time and hospital stay, and a lower decrease in postoperative hemoglobin levels than placebo.
Methods
Trial design
We will conduct a single-center, double-blind, parallel, superiority, randomized placebo-controlled trial. This study protocol follows the recommendations of the Standard Protocol Items: Recommendations for Interventional Trials (SPIRIT) statement 18 for clinical trial protocols (Supplement 1). The trial will be conducted at the Erasmus MC-Sophia Children’s Hospital in Rotterdam, The Netherlands, where six orthopedic surgeons perform PFPOs. The protocol follows the World Health Organization’s guidelines, is registered at the European Union Clinical Trial Register (EudraCT: 2022-002384-30), and approved by the local ethics committee (MEC-2022-0598).
Participants
We aim to include 180 participants aged from 1 to 18 years undergoing PFPOs at our institution for any indication. Treating physicians are asked to select and screen patients at the outpatient clinic, using the following exclusion criteria:
Contraindications for TXA (e.g. known allergy to TXA, severe renal insufficiency);
active use of alternative (anti)fibrinolytics;
diagnosed bleeding or coagulation disorder;
medical history of thromboembolic complications;
use of hormonal contraception;
or insufficient Dutch language skills of the patient and/or caretakers.
Procedures
Eligible patients and/or their caretaker(s) will be informed about the study at the outpatient clinic by their pediatric orthopedic surgeon. The physician will provide them with informed consent form(s) (Supplement 2). The research coordinator will contact the patient and/or caretaker(s) after a minimum of 3 days to answer additional questions. If the patient and/or caretaker(s) (depending on the age and competency of the patient) are willing to participate, they return the signed informed consent form to the research coordinator at a return visit or on the day of admission. The research coordinator will co-sign and provide the participant and/or caretaker(s) a copy. Subjects can withdraw their consent at any time for any reason, without any consequences.
After inclusion, baseline demographics will be collected from the electronic patient file and entered into the encoded web-based database Castor EDC. 19 After baseline data collection, randomization will take place. Before surgery, during anesthetic induction, patients will receive their allocated study medication. Preoperative blood tests will be performed during induction, including hemoglobin levels, as part of the usual clinical care. The surgery will take place according to usual care, during which the surgical team will determine the estimated blood loss (EBL), while blinded for randomization. Postoperative care conforms to usual care as well, including capillary blood testing for hemoglobin levels, on the first postoperative day. Hospital discharge is generally at postoperative days one to three. Data will be collected preoperatively and during surgery and hospital admission only. After hospital discharge, the study procedures end. Patients will be treated further following standard clinical care. Complications will be recorded until 6 weeks postoperatively.
Interventions
Children will undergo usual care treatment, except for the administration of study medication. Participants will receive either a preoperative bolus of TXA (Mylan) or placebo during anesthetic induction (15–30 minutes before incision). A patient randomized to the TXA group will receive an intravenous bolus of 15 mg/kg in a 0.9% saline solution of 15 mL, preoperatively, while the placebo bolus consists of 15 mL of 0.9% saline only.
Surgical teams are allowed to use an additional escape bolus of TXA during surgery should this be deemed necessary by the surgical team, for example, in case of excessive intraoperative blood loss. The maximum registered dosage of TXA in children is 25 mg/kg per day. Hence, the permissible dosage of extra intraoperative TXA bolus is 10 mg/kg for all participants (TXA and placebo group). If the surgical team considers more than 10 mg/kg TXA to be necessary, an unblinding procedure will be initiated. In these cases, only in the placebo group, an additional 15 mg/kg TXA would be allowed.
Randomization and blinding
In this trial, pediatric patients who undergo PFPO are randomly assigned to the TXA or placebo group, in a 1:1 ratio. The allocation sequence will be generated by Castor. 19 Randomization is stratified for single (unilateral, pelvis, or femur) versus multiple (pelvis and femur and/or bilateral) osteotomies. Block randomization with variable block sizes (2, 4, and 6) is used to prevent predictability.
The randomized group allocation is concealed from the researchers, surgical team, and patients. Only the trial pharmacy will be notified of the study group allocation through Castor. They will prepare either a placebo or TXA solution on the day of surgery, with identical packaging and labeling. In the case of a placebo, the product will contain a similar volume as would be the case with TXA, so the researchers, surgical team, patients, and caretakers will remain blinded. The pharmacy transports the study medication to the operating theater, where the anesthesiologist will administer it intravenously during induction. In case of unblinding, the surgical team will contact the trial pharmacy by phone, after which they are provided with the assigned study group. Hence, in this case, only the surgical team will be unblinded; researchers and patients remain blinded for group allocation. Reasons for unblinding and the amount of additionally administered TXA will be registered.
Outcome measures
The schedule of enrollment, interventions, and outcome assessments is presented in Table 1. The primary outcome is intraoperative EBL corrected for the patient’s weight (mL/kg). EBL is calculated using the gravimetric method. 20 Through this method, intraoperative blood loss is deduced by weighing the surgical materials contaminated with blood and subtracting the dry weights and rinse liquid. By summing up the measured weight of the blood and estimating the amount (mL) of mixed liquid (blood, rinse liquid) in the suction container, the blood loss is calculated with a conversion of 1 g = 1 mL of blood. This method of blood loss estimation is practical, requires minimal training, and is standard procedure at our institution. 21 A meta-analysis on this technique found a correlation of 0.77 (95% CI 0.61–0.93) to a validated reference. 20 The gravimetric method is therefore a reliable technique feasible in clinical practice.
Schedule of enrollment, interventions, and outcome assessments.
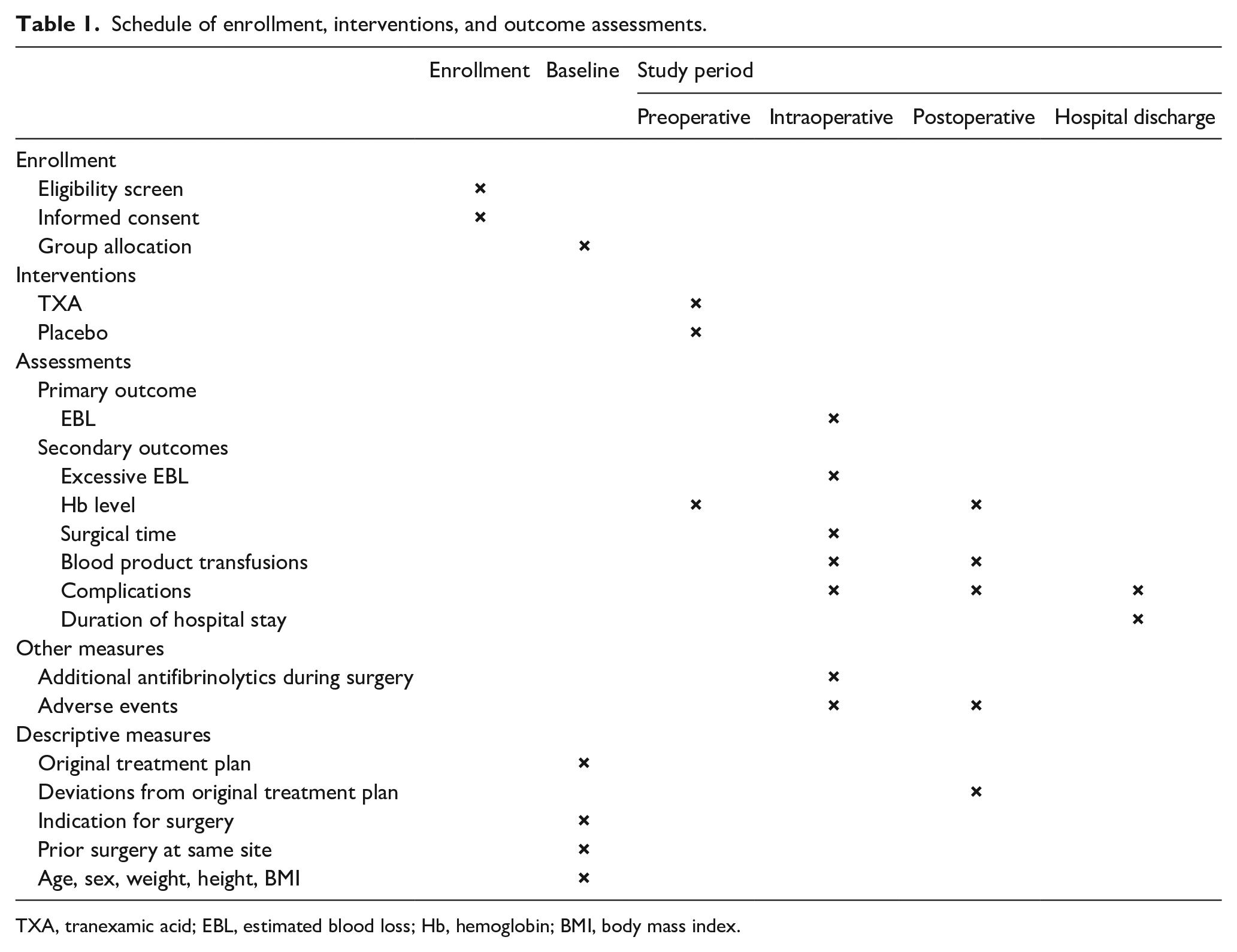
TXA, tranexamic acid; EBL, estimated blood loss; Hb, hemoglobin; BMI, body mass index.
Secondary study outcomes are:
Postoperative hemoglobin level and change thereof between preoperative and postoperative measurements (mmol/L);
excessive blood loss (defined as EBL > 20 mL/kg; yes/no);
type and volume of blood products administered (mL);
surgical time (minutes);
duration of hospital stay (days);
and complications during hospital stay or during the first 6 postoperative weeks.
Other parameters that will be collected are:
Demographic data (sex, age, length, weight, body mass index (BMI));
indication for surgery (e.g. DDH, neuromuscular condition, trauma);
treatment characteristics (type of osteotomy, pelvic and/or femur, side, unilateral or bilateral);
prior surgery at the same site (yes or no);
and antifibrinolytic agents administered intraoperatively (including TXA).
Data management
Data will be extracted from electronic patient records and stored online in Castor. 19 In Castor, each patient is assigned a unique study-related ID code. This ID code is not directly associated with the subject, and the database will therefore only hold pseudonymized patient data. Within the Castor database, restrictions are built in for numerical variables so that entered values are within realistic ranges. Before analyses, a research team member will perform a quality check of the data in Castor. After data from all participants have been collected and checked, the research coordinator will extract the data from Castor into a statistical package file for data analyses and extract a copy into a Microsoft Excel file (.csv) for long-term storage. Participant files will be kept in storage at the coordinating center for a period of 15 years after study completion (last participant’s hospital discharge).
Sample size
Sample size calculations were performed with the aid of a statistician from the Department of Biostatistics at our institution. Calculations were performed using the primary outcome, EBL. In the aforementioned retrospective study, mean EBL without TXA administration (n = 269) was 200 ± 151 mL, 17 which was recalculated to 7.63 ± 5.19 mL/kg. In the preoperative TXA group (n = 20), average blood loss was 152 ± 110 mL. For this trial, we defined a 25% reduction in blood loss as a clinically relevant difference, which equals a reduction of 50 mL or 1.9 mL/kg. Using Altman’s nomogram with a standardized difference, a power of 0.8, and α = 0.05, a total of 180 participants are needed. Similarly, the required sample size is equal to 180 with the formulae of Rosner, 22 based on a one-sided hypothesis and the same assumptions as the first calculation. Given the nature of the study design, that is, mostly usual care and no long follow-up period, we do not expect loss to follow-up.
Statistical methods
Descriptive analyses will be performed on baseline data and outcomes. The results will be expressed as means and standard deviations, or medians with interquartile ranges in case of non-normal distribution. Categorical data will be expressed in frequencies and percentages.
Data will be primarily analyzed according to intention-to-treat principle and secondarily compared by per protocol analysis. The primary analysis, a multivariate linear regression, will assess the association of preoperative TXA use (TXA versus placebo) with EBL. Stratification factors used for randomization (single versus multiple osteotomies) and potential confounders to EBL (sex, BMI, surgical time, and prior surgery at the same site) will be entered as covariates. We will check the following model assumptions: linearity, multicollinearity, homoscedasticity, and normality and independence of residuals in the linear regression model. Exploratory subgroup analyses will be performed based on etiology (DDH, secondary hip dysplasia (e.g. neuromuscular disease, Down syndrome), and hip incongruencies (e.g. blood flow problems, trauma, and infection)). Similar analyses will be performed for the secondary outcomes. Statistical significance is defined as p < .05.
Following recommendations of Jakobsen et al., 23 if the proportion of missing data exceeds 10%, missing outcome data will be imputed using multiple imputation methodology. In addition, the sensitivity of the missing data at random assumption will be investigated. If a patient decides to revoke informed consent, data collected up to that point will be used in the data analysis.
Monitoring
Monitoring will be conducted following the Netherlands Federation of University medical centers guidelines. 24 Right before the first inclusion, the initiation visit will take place. The second visit will be performed after 10 inclusions are completed, and the third after 90 inclusions. Finally, a close-out visit will take place after the last participant’s hospital discharge. In addition, monitoring will take place at least once a year. Should inclusion be delayed, extra monitoring visits will be planned accordingly.
Harms
The physicians report all adverse events. This will include information about any adverse events that participants believe may be related to the trial intervention, including their nature, how long they lasted, and what action, if any, was taken. Known possible side effects of TXA use are nausea, vomiting, or diarrhea. In fewer cases, allergic skin reactions and convulsions are reported. 25 However, in these cases, higher dosages of TXA were administered than those in our protocol, or other ways of administration were used. Therefore, we do not expect to encounter these effects in our trial. The Central Committee on Research Involving Human Subjects and local medical ethics committee will be notified of any serious adverse events. In these cases, the advice of the committee will be followed accordingly.
Discussion
Substantial blood loss during pediatric hip osteotomies can lead to various complications, some of which may be serious. Therefore, it is crucial to implement preventive measures. TXA, an antifibrinolytic agent, has demonstrated its potential in reducing intraoperative blood loss and is used as part of standard care in several adult patient populations, including trauma and orthopedics. However, the evidence for TXA use in pediatric orthopedics is limited, including for PFPOs with its potentially substantial blood loss. Therefore, a double-blind, randomized, placebo-controlled trial will be performed, which will be the first employing this robust methodological approach to investigate the effect of preoperative TXA against placebo for reducing intraoperative blood loss during pediatric PFPOs.
The double-blind RCT design allows for minimizing bias and confounding variables, as neither the patients nor the healthcare providers involved in the study will be aware of the treatment allocation. By correcting the trial’s primary outcome, intraoperative EBL, for the patient’s weight, differences in body size due to the wide age range of the included children, and therefore their total blood volume, are accounted for. 26 In addition to EBL, the hemoglobin level changes, number of cases with excessive blood loss, type and amount of blood product transfusions, and related complications will be evaluated in this trial. All outcomes combined will provide a complete image of the possible reducing effect of preoperative TXA on blood loss in pediatric PFPOs.
It is important to acknowledge potential limitations in our study protocol. First, despite randomization and blinding procedures, there is a possibility of unavoidable confounders or biases. For example, the surgical team may be able to identify the treatment group based on certain intraoperative observations. Stratification, block randomization, and multivariate analyses will be conducted to address confounders and enhance the validity of our results. Second, our chosen intraoperative blood loss quantification method is both practical and reproducible, offering an accurate and reliable EBL assessment in our study. Although more precise methods theoretically exist, they are not feasible in clinical practice due to high costs or burdens they impose on caretakers and/or patients.20,21 Therefore, the gravimetric method is the most suitable approach for our study, striking a balance between reliability and feasibility.
Despite these possible limitations, this study protocol presents a comprehensive and well-designed approach to evaluating the effect of a preoperative TXA bolus on intraoperative blood loss in pediatric PFPOs. The robust methodology, including randomization and blinding, enhances the internal validity of the study. Because PFPOs for all indications treated by multiple physicians in a high-volume hospital will be included, the results of this study will be generalizable to other pediatric patients that undergo PFPOs.
We are committed to disseminating our findings widely to maximize their impact and benefit to patients. Our study results will be published in a peer-reviewed journal, presented at national and international conferences, and incorporated into local guidelines and practice recommendations. If our study indeed demonstrates a significant and clinically relevant reduction in intraoperative blood loss and/or related complications through preoperative TXA administration, it will provide valuable evidence supporting its implementation as a preventive measure in pediatric PFPOs. In that case, given the reported safety and low cost of TXA, adopting it as a standard medication to minimize blood loss and prevent complications in this vulnerable population would be advisable.
Supplemental Material
sj-pdf-1-cho-10.1177_18632521231199518 – Supplemental material for Effect of tranexamic acid on intraoperative blood loss in pediatric osteotomies around the hip: Study protocol for a double-blind randomized placebo-controlled trial
Supplemental material, sj-pdf-1-cho-10.1177_18632521231199518 for Effect of tranexamic acid on intraoperative blood loss in pediatric osteotomies around the hip: Study protocol for a double-blind randomized placebo-controlled trial by Hilde W van Kouswijk, Jaap J Tolk, Christian PE van Bommel, Max Reijman, Dagmar RJ Kempink and Pieter B de Witte in Journal of Children’s Orthopaedics
Supplemental Material
sj-pdf-2-cho-10.1177_18632521231199518 – Supplemental material for Effect of tranexamic acid on intraoperative blood loss in pediatric osteotomies around the hip: Study protocol for a double-blind randomized placebo-controlled trial
Supplemental material, sj-pdf-2-cho-10.1177_18632521231199518 for Effect of tranexamic acid on intraoperative blood loss in pediatric osteotomies around the hip: Study protocol for a double-blind randomized placebo-controlled trial by Hilde W van Kouswijk, Jaap J Tolk, Christian PE van Bommel, Max Reijman, Dagmar RJ Kempink and Pieter B de Witte in Journal of Children’s Orthopaedics
Supplemental Material
sj-pdf-3-cho-10.1177_18632521231199518 – Supplemental material for Effect of tranexamic acid on intraoperative blood loss in pediatric osteotomies around the hip: Study protocol for a double-blind randomized placebo-controlled trial
Supplemental material, sj-pdf-3-cho-10.1177_18632521231199518 for Effect of tranexamic acid on intraoperative blood loss in pediatric osteotomies around the hip: Study protocol for a double-blind randomized placebo-controlled trial by Hilde W van Kouswijk, Jaap J Tolk, Christian PE van Bommel, Max Reijman, Dagmar RJ Kempink and Pieter B de Witte in Journal of Children’s Orthopaedics
Footnotes
Acknowledgements
We thank Sara Baart (Department of Biostatistics, Erasmus MC) for her help with the setup of the statistical analyses.
Author contributions
HWvK drafted and revised the manuscript. JJT, CPEvB, MR, DRJK, and PBdW designed the original study protocol and revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Sophia Fund (2021-WAR22-48/WO), JKF Fund (20210053), and EPOS Research Grant 2022. Funding bodies played no role in the design of the study or the collection, analysis, and interpretation of data and in writing the manuscript.
Ethics approval
The Medical Ethics Committee of the Erasmus University Medical Center approved the protocol of this trial (MEC-2022-0598).
Availability of data and materials
The data sets generated and analyzed during the described trial will be available from the corresponding author upon reasonable request after the results are published.
Trial status
Study inclusion has started in March 2023. Final results are anticipated mid-2026, and the authors aim to report the findings shortly thereafter.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
