Abstract
Trehalose is a disaccharide molecule consisting of two molecules of glucose. Industrially, trehalose is derived from corn starch and utilized as a drug. This study aims to examine whether the integration of nanoparticle-encapsulated trehalose to the Ice-Free Cryopreservation (IFC) method for preserving heart valves has better cell viability, benefits to protect the extracellular matrix (ECM), and reduce immune response after storage. For the experiment to be carried out, we obtained materials, and the procedures were carried out in the following manner. The initial step was the preparation of hydroxyapatite nanoparticles, followed by precipitation to acquire Apatite colloidal suspensions. Animals were obtained, and their tissue isolation and grouping were done ethically. All samples were then divided into four groups, Control group, Conventional Frozen Cryopreservation (CFC) group, IFC group, and IFC + T (IFC with the addition of 0.2 M nanoparticle-encapsulated Trehalose) group. Histological analysis was carried out via H&E staining, ECM components were stained with Modified Weigert staining, and the Gomori Ammonia method was used to stain reticular fibers. Alamar Blue assay was utilized to assess cell viability. Hemocompatibility was evaluated, and samples were processed for immunohistochemistry (TNFα and IL-10). Hemocompatibility was quantified using Terminal Complement Complex (TCC) and Neutrophil elastase (NE) as an indicator. The results of the H&E staining revealed less formation of extracellular ice crystals and intracellular vacuoles in the IFC + T group compared with all other groups. The CFC group’s cell viability showed better viability than the IFC group, but the highest viability was exhibited in the IFC + T group (70.96 ± 2.53, P < 0.0001, n = 6). In immunohistochemistry, TNFα levels were lowest in both IFC and IFC + T group, and IL-10 expression had significantly reduced in IFC and IFC + T group. The results suggested that the nanoparticle encapsulated trehalose did not show significant hemocompatibility issues on the cryopreserved heart valves.
Introduction
The use of human biological valves known as allograft heart valves (AHV) has significant advantages over the traditional xenografts, especially in terms of hemodynamic response and durability. The preservation of these allogeneic heart valves requires high-level care to ensure the retention of cell integrity before clinical use. Cryopreservation has been the method of preservation for the various types of tissues, but the issue of ice crystal formation, which has damaging effects on the integrity of the cell, has often raised challenges. 1,2 Consequently, it became necessary to get an alternative technique that would maintain cellular integrity and functions. In essence, the need for more favorable preservation methods led to the invention of the Ice-Free Cryopreservation (IFC) alternative. 3 Among the major challenges of Frozen Cryopreservation (FC), as a standard biobank in the storage and transportation of allogeneic heart valves, are the limited retention of cellular integrity due to the presence of ice crystals as well as poor hemocompatibility. Thus, it leads to high incidences of immune responses and inflammation. 4,5 Brockbank et al. 3 found that “Ice-Free Cryopreservation of heart valve transplants at −80°C prevents ice formation, tissue-glass cracking and preserves ECM integrity which results in minimal inflammation and improved hemodynamics in allogeneic juvenile sheep.” Therefore, this alternative has been favored over the traditional cryopreservation, which requires expensive specialized equipment to preserve cells at −135°C while still maintaining their integrity. 6 This approach is referred to as vitrification. Vitrification involves ultra-fast freezing that induces cell solidification and the formation of a vitreous solution (a glass-like formation of water) that surrounds the allografts and preserves them without ice crystallization.
VS83 (Vitrification Solution 83) protocol as a method of IFC of allogeneic heart valves is preferred over the VS55 (Vitrification Solution 55) protocol 1. Since the latter only allows for small samples due to the application of rapid cooling and warming, the method may not work with large samples. 7,8 In contrast, VS83 allows for long-term preservation of relatively large samples at −80°C, with better maintenance of the cellular matrix, among other advantages. The need to preserve cell viability, protect the ECM, and prevent deleterious immune response led to the use of trehalose, a disaccharide of two glucose molecules. 9,10 Eroglu et al. 11 note that “the introduction of low concentrations of intracellular trehalose can greatly improve mammalian cell’s survival during cryopreservation.” This observation is emphasized by Garcıa de Castro and Tunnacliffe, 12 who note that trehalose has a positive effect on preserving cellular morphology in cryopreservation. The addition of this sugar acts as an intracellular cryoprotective agent. The purpose of using this disaccharide is to achieve retention of cell viability after thawing. 13,14 Sugars are effective in preserving post-thaw viability since they enable the balancing of outer cell membranes and proteins. In fact, microorganisms are known to accumulate disaccharides that protect them from being damaged by freezing in cold regions or when put in frozen environments. 15 As a result, trehalose is used in the allografting of human valves due to its ability to induce this favorable outcome in clinical practice.
Practitioners are also concerned about the hemocompatibility of the allogeneic valves since immune responses can significantly affect the success of the clinical application. The challenge of immune-induced inflammation has led to concerns over the possibility of trehalose triggering such a reaction. Since the sugar has significant positive effects on IFC preservation as a major cryoprotective agent, it is crucial to study its impact on hemocompatibility.
Therefore, the purpose of this paper is to determine the impact of nanoparticle-encapsulated trehalose on the hemocompatibility of allogeneic valves in the VS83 protocol; and to specifically investigate whether the disaccharide induces an immune response and whether it reduces immunogenic responses post-thawing. The study also seeks to determine whether nanoparticle-encapsulated trehalose leads to improved cell viability and maintains the integrity of the ECM in IFC techniques for heart valve preservation. The first hypothesis being, the use of nanoparticle-encapsulated trehalose in vitrification of allogeneic heart valves promotes cellular viability and protects the ECM structures. The second hypothesis being, the intracellular uptake of nanoparticle-encapsulated trehalose does not trigger any significant hemocompatibility activity in the VS83 vitrification of allogeneic heart valves.
Materials and methods
Animals and chemicals
Eighty-four male Wistar rats were bought from the Qingdao University Laboratory Animal Center. Calcium nitrate, 2-aminoethylphosphate (AEP), and dimethyl sulfoxide (DMSO) were obtained from Sigma-Aldrich, Santa Clara, CA, USA. Di-ammonium hydrogen phosphate and sodium hexametaphosphate (HMP) were acquired from Macklin, Shanghai, China. Concentrated ammonia and cellulose membrane (molecular weight cut-off: 6000–8000 Da), glucose, and a magnetic stirrer were acquired from the Central Laboratory of the Affiliated Hospital of Qingdao University.
All cryoprotectants were bought from Sigma, Santa Clara, CA, USA. Sterile polyethylene bags were purchased from Fisher Scientific, Pittsburg, PA, USA. As depicted by Brockbank et al., 7 the Euro-Collins solutions and final vitrification solutions containing 4.65 mol/L formamide, 4.65 mol/L DMSO, and 3.31 mol/L 1,2-propanediol were prepared. Chemicals of the highest purity and deionized water were utilized for the making of all cryopreservation solutions and were stored at 4°C for up to about a month after preparation.
Trehalose was bought from Solarbio, Beijing, China; Dulbecco’s modified Eagle’s medium (DMEM) and Fetal Bovine Serum (FBS) were acquired from Gibco, Grand Island, NY, USA. Phosphate-Buffered Saline (PBS) was obtained from HyClone, USA. Alamar Blue was purchased from Solarbio, Beijing, China.
Immunohistochemistry kit was purchased from Beijing Soledad Symbol Technologies Ltd. Improved Weigert Elastic fiber staining kit, Improved Gomori Ammonia Silver, Reticular Fiber Staining Kit, IL-10, and TNFα antibody kit were obtained from Solarbio, China. All kits were stored as per the manufacturer’s instruction before use.
Rat Thrombin/Antithrombin Complex Elisa Kit, Rat TCC C5b-9 (Terminal Complement Complex C5b-9) Elisa kit, Rat NE/ELA2(Neutrophil Elastase/Elastase-2) Elisa Kit, and Rat βTG/PBP/CXCL7/NAP2 (Beta-Thromboglobulin) Elisa Kit were acquired from Elabscience, China.
Preparation of hydroxyapatite nanoparticles
Apatite colloidal suspensions were obtained by precipitation method as follows: Precipitation of calcium nitrate and di-ammonium hydrogen phosphate was carried out in deionized water, at pH 9.5 in the presence of 2-aminoethylphosphate NH3 + -CH2-CH2-O-P(O) (O−)2 or “AEP” for agglomeration control to deliver bioinspired non-stoichiometric nanocrystalline apatite Ca10−x(PO4)6− x (HPO4, CO3)x(OH)2−x. The molar proportion of AEP: Ca in the reacting medium was set to 1:1. 6.25 mL of calcium nitrate tetrahydrate Ca (NO3)2.4H2O (4.87 mmol) forms Solution A, 6.25 mL of AEP (4.87 mmol) forms Solution B and 12.5 mL of di-ammonium hydrogen phosphate (1.62 mmol) forms Solution C. The reaction was carried out using reagent graded chemicals. Solution D was created by the consistent mixing of Solution A and B. A couple of drops of concentrated ammonia were incorporated to alter the acidic pH of both Solutions D and C to 9.5. Mixing C and D permitted precipitation. The suspension was then kept in an oven preset at 100 o C for 16 h to mature. 16
The colloid (25 mL) was cleansed by dialysis as previously reported. 16 –18 A tubular cellulose membrane (15 cm length, 3 cm diameter, 6000–8000 Da cut-off) was hydrated preliminarily by contact with deionized water. 25 ml of the suspension was presented by clasping the lower end, and the second end was clipped 7 ml above the fluid dimension loaded up with air. The film was introduced in 800 ml of dialysis medium by suspending vertically under consistent stirring. The dialysis medium was frequently replaced with a new one after 4, 8, 22, and 26 h. Sodium hexametaphosphate (NaPO3) 6 or “HMP” was added to the cleansed colloids to ameliorate dispersibility and to bring the suspension to the physiological pH level. Glucose (15 mM) was added to the colloidal NP (Nanoparticle) to avert uncontrolled aggregation and support the NP redispersion in the medium. Thus, safeguarding molecule estimate distribution preceding freeze-drying for storage facilitation. Each freeze-dried colloid was reconstituted with deionized water to achieve the final NP concentration of 2.25 mg/mL and sifted through a 0.2 µm syringe-filter before the commencement of experiments. HMP-stabilized apatite NP promotes the trehalose permeation because of the positive and negative charge distribution around the particles, which leads to interactions with the cell membrane. 16 The vitrification solution employed in the preservation and the Euro-Collins solution used in the washing step was supplemented with 2.25mg/ml NP and 0.2 M Trehalose to facilitate the permeation of trehalose.
Animals, tissue isolation and grouping
Pentobarbital (5 mg/100 g body weight) was administered as general anesthesia into male Wistar rats by intraperitoneal injection. The aortic valve with the affixed aorta and the myocardium under the valves were extracted. The tissues were washed gently with PBS, drenched, and maintained in ice-cold DMEM containing 120 μg/mL Lincomycin HCl, 50 μL/mL Vancomycin HCl and 124 μg/mL Polymyxin B sulfate for 24 h at 4°C. All indicated procedures were performed under a sterile environment utilizing aseptic techniques. All animals received humane care according to the international guidelines. The study protocol was approved by the Animal Ethics Committee at Qingdao University. Efforts were made to minimize suffering and the number of animals used.
All samples were arbitrarily allotted to one of four groups(A-D). Group A, Samples incubated with antibiotics, which acclimatized as a control group (n = 21); Group B, samples exposed to conventional freezing cryopreservation (CFC) (n = 21); Group C, samples treated with the Ice-free cryopreservation (IFC) technique with VS83 formulation (n = 21); and Group D, samples treated via the IFC with the additament of 0.2 M nanoparticle-encapsulated Trehalose (IFC + T) to both vitrification solution and Euro-Collins solution in washout steps (n = 21).
Conventional freezing cryopreservation (CFC)
Tissues exposed to CFC were transferred into polyethylene cryo-vials containing DMEM with 10% DMSO and 10% FBS and stored at 4°C for 30 mins. Vials were then put into a mechanical storage freezer and started the controlled rate freezing at 1°C per minute to −80°C. In the wake of being sustained at −80°C for a minimum of 24 h, samples were stored in the vapor phase above liquid nitrogen. After a 2-month storage time, singular vials were amassed and defrosted in a 37°C water bath. Tissues were taken out when all ice had vanished. The elution of the cryoprotectant was accomplished by consequently washing in DMEM with 0.5 M mannitol, DMEM with 0.25 M mannitol, and determinately in DMEM without mannitol for 5 min each at 4°C. Tissues were then stored quickly on ice in DMEM until further use. 19 –22
Ice-free cryopreservation (IFC)
The IFC procedure was completed, as explained before. 23 Heart valves exposed to IFC were put in sterile polyethylene bags with 80 mL of an 83% 12.6 molar cryoprotectant formulation (VS83) in the Eurocollins solution (4.65 mol/L formamide, 4.65 mol/L DMSO and 3.31 mol/L 1,2-propanediol). Vitrification solution utilized in Group D contained 0.2 M nanoparticle-encapsulated Trehalose (Trehalose and nanoparticle were added with the vitrification solution as per the concentration and mixed). After incubation for 6 hours at room temperature, the cooling procedure was accomplished by putting the bag containing the valves in a pre-cooled methyl butane bath (∼ −130°C) for 10–15 mins. The bags containing the valves were then stored at −80°C for about 2 months. Warming of the tissue was performed by submersion in a 37°C water bath for 1–2 min until the liquid turned out to be clear. The five-step washing procedure was followed to dilute the cryopreservation agents in a 4°C cold Euro-Collins solution. Each step endured for 5 mins and, eventually, tissue submersion in DMEM. 0.2 M nanoparticle-encapsulated trehalose was commixed with the Euro-Collins solution for the washout step employed in Group D. Tissues were then transferred into a 37°C incubator with 5% CO2 for tissue culture or were utilized for detection immediately.
Histological analysis via H&E staining
Routine H&E (Hematoxylin & Eosin) staining was used to describe the changes in cellular composition and tissue architecture. The staining was performed immediately after rewarming. Tissue sections were fixed straightforward in 12.5% formaldehyde solution, embedded in paraffin, cut into subsequent sections of 5 μm. The sections were stained with Hematoxylin-Eosin solution and observed under a light microscope.
Evaluation of ECM components and elastic fibers
ECM components were observed by staining with Modified Weigert staining. Samples were embedded in paraffin, dewaxed, oxidized with Weigert oxidant, and washed remotely. Then bleached with Weigert bleach, rinsed with water, stained with Weigert resorcinol fuchsin solution, acid differentiation, Van Gieson (VG) dyeing solution, rapid differentiation with 95% ethanol, and dehydrated. Sections were sealed and observed under a light microscope.
Gomori Ammonia method was used to stain reticular fibers. Samples were fixed in 10% formaldehyde solution, embedded in paraffin, cut into serial sections of 4 μm. Then oxidized with Gomori oxidant, bleached with oxalic acid, dyed with ammonium ferric sulfate, Gomori, and silver ammonia solution, reduced with Gomori reducing agent and dehydrated. Sections were sealed and observed under a light microscope.
Assessment of cell viability
Viability evaluations were initiated within an hour of completion of the rewarming and cryoprotectant elution protocol. Cell viability was resolved using an AlamarBlue Assay. This metabolic assay incorporates a water-soluble fluorometric viability oxidation-reduction pointer that fluoresces and changes color in light of the chemical reduction of the growth medium. 24 Tissue samples (containing the leaflets, muscle band, and the artery) were incubated for 3 h (37°C, 5% CO2). At that point, the tissue was cut into 3 mm3 pieces, set into a 96-well microtiter plate, and 100 µL of culture medium with 10% Alamar Blue solution was included. The microtiter plate was incubated for 4 h, and an equivalent amount of the supernatant solution was transferred into another 96-well plate and analyzed utilizing a microtiter plate spectrofluorometer (excitation 550 nm, emission 590 nm). The information was standardized to the dry load of the tissue and expressed as fluorescence intensity per mg dry weight of the tissue. The fluorescence intensity of the untreated group was utilized as a standard for comparison.
Determination of hemocompatibility
Tepe et al. 25 published the evaluation of thrombogenic and immunogenic potential in detail. Hemocompatibility of valve surfaces was tested with whole blood acquired from healthy rats employing heart puncture. Blood was gathered in heparin pre-coated monovettes. Three valves in each group were incubated with 12 ml blood under consistent movement for 1 h at 37°C. The accompanying solvent proteins were resolved by the producer’s protocol. 20,26
2.4 ml blood from each group was collected into an EDTA (Ethylenediaminetetraacetic acid)- monovette. Monovettes were placed on ice for centrifugation at 2500 × g for 20 min at 4°C. 800 µL of the supernatant was utilized for the determination of TCC C5b-9.
2.8 ml of blood from each group was collected in a Citrate-monovette. Monovettes were centrifuged at 3000 rpm for 15 min at room temperature. 600µl supernatant was utilized for thrombin-antithrombin (TAT) complex and neutrophil elastase (NE) determination.
5.4 ml of blood from each group was incorporated into 0.3 ml of CTAD (Citrate-Theophylline, Adenosine, Dipyridamole) anticoagulant monovettes. Tubes were set on ice for 15 min. Following centrifuging at 2500 × g for 20 min at 4°C, 1.4 ml plasma from the center portion was aspirated and transferred to an incipient neutral monovette. After reiterated centrifugation, 200 µL plasma was utilized for the determination of β-Thromboglobulin (β-TG).
By subtracting the background fluorescence intensity obtained from the control wells without the tissues, the fluorescence intensity was determined.
Immunohistochemical analysis of interleukin-10 (IL-10) and tumor necrosis factor-alpha (TNFα)
Immunohistochemistry analysis was done as previously described by Jing et al., and Jammal et al. 27,28 Briefly, the samples were fixed on the slide, sectioned, stained with a primary antibody, counterstained with secondary antibody, dehydrated, hyalinized, and the slides were sealed. For the staining of both IL-10 and TNFα, the primary and secondary antibody titer was 1:200 and 1:50, respectively. Image Pro-Plus Software quantitatively examined the optical density of IL-10 and TNFα positive granules and calculated the mean optical density.
Statistical analysis
Experimental data were handled by means of GraphPad Prism 6.0. Statistical analyses were completed utilizing a t-test or one-way analysis of variance. Mean, and standard deviation (SD) were used. Statistical significance was considered with a p-value lower than 0.05 (P < 0.05).
Results
Morphological features after thawing
Histological sections of the aortic valve showed clear cellular structures and an orderly cell arrangement below the endothelium in all the groups. Nevertheless, some pathological differences were defined as follows in the CFC group: some tissue areas had wider gaps and minor disorganization in the arrangement of cells. These changes may be due to the formation of ice during the cooling and rewarming process. On the contrary, extracellular ice crystals and intracellular vacuole development were lower in the IFC group and the lowest in the IFC + T group (Figure 1).
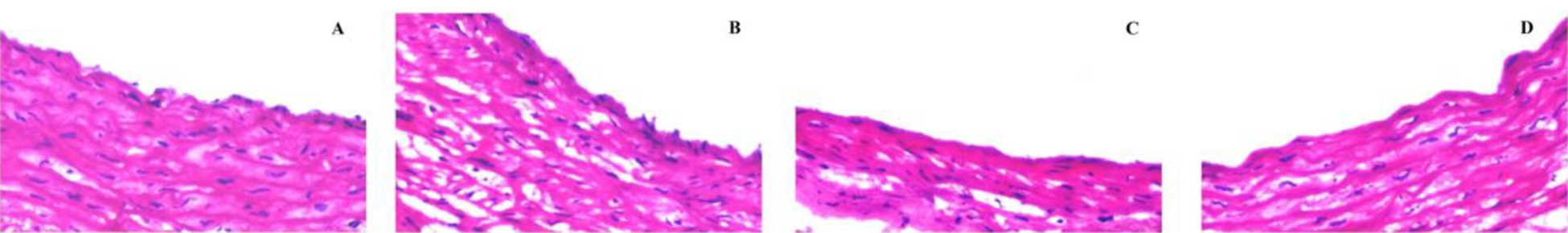
Representative light-microscopic images of samples stained with Hematoxylin and eosin (H&E) (original magnification ×40). A: control group; B: CFC group; C: IFC group; D: IFC + 0.2 M nanoparticle-encapsulated Trehalose (IFC + T) group.
Improved Weigert Elastic fiber staining demonstrated ECM components such as elastin, collagen, and muscle fiber. Modified Gomori ammonia staining was used for the observation of reticular fibers. The results revealed excellent ECM components in the control group; some broken elastic fibers and homogeneous collagen structures were present in certain areas of the CFC group, which could be related to ice formation during processing. No unusual elastic fibers were found in samples from the IFC group. The IFC + T group indicated that ECM is superior to the CFC and IFC groups. The elastic fibers in the control group are complete and constant. Pathological deterioration was found in the CFC group, which displayed more degraded and highly degenerated elastic fibers than in other groups. There was no significant difference between the IFC and the IFC + T groups. Elastin and collagen had similar structures in each group and were qualitatively stronger in the IFC and IFC + T groups. The preservation and demarcation of elastin and collagen fibers in the IFC + T group have, therefore, been enhanced by the addition of nanoparticle (Figures 2 and 3).
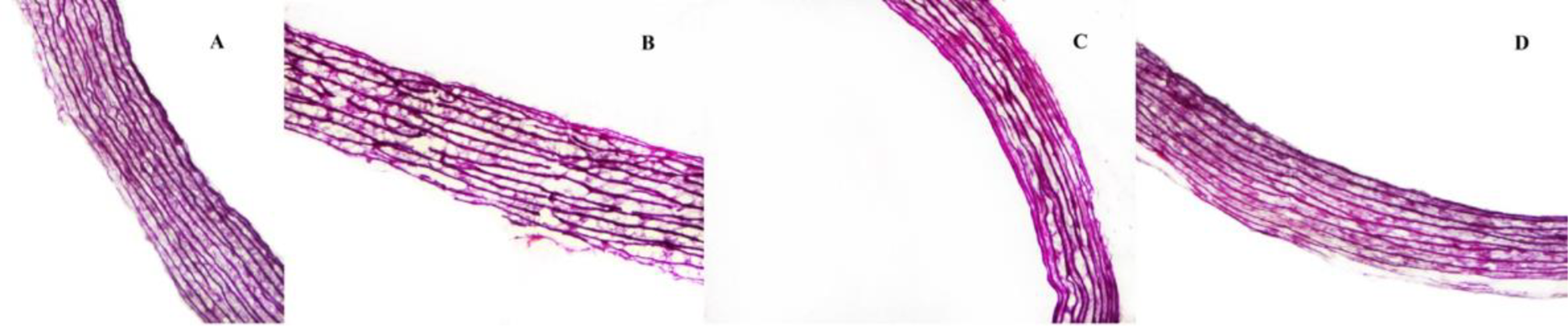
Representative light-microscopic images of the stained specimens. Modified Weigert Elastic Fibers stained for the examination of ECM (original magnification ×40). A: control group; B: CFC group; C: IFC group; D: IFC + 0.2 M nanoparticle-encapsulated Trehalose (IFC + T) group.
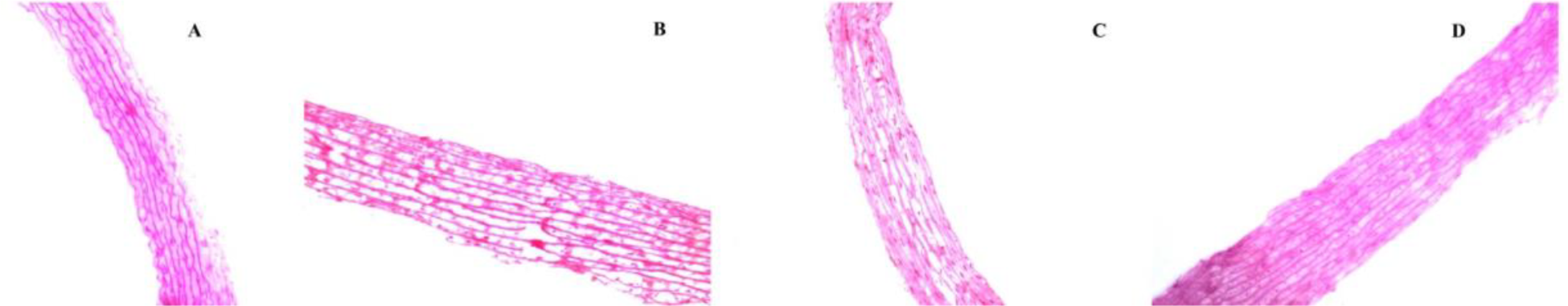
Modified Gomori ammonia-stained specimens showing elastic fibers (original magnification × 40). A: control group; B: CFC group; C: IFC group; D: IFC + 0.2 M nanoparticle-encapsulated Trehalose (IFC + T) group.
Cell viability
Cell viability is an important indicator of quality control. The viability assessment of aortic valve tissues in the various cryopreservation methods has been standardized to the control group (Figure 4). According to statistical comparisons, the data on cell viability of the three cryopreserved groups versus the control group showed a significant loss of viability, with the IFC + T group showing the least damage. The control group exhibited statistically more viability than the frozen samples using the CFC or IFC method (96.75 ± 0.22, n = 6, p < 0.05). Both the CFC (62.73 ± 1.01, n = 6, p < 0.05) and IFC groups (50.71 ± 1.29, n = 6, p < 0.05) demonstrated significantly less viability relative to the control group, which could be due to ice forming during the cooling and rewarming process. However, the IFC + T group (70.96 ± 2.53, P < 0.0001, n = 6) displayed higher viability than both the CFC and IFC groups, which may be due to the delivery of trehalose to the cells that was supported by nanoparticle encapsulation. Nanoparticle encapsulated trehalose reduced the amount of ice formation, thus protecting cells from damage.
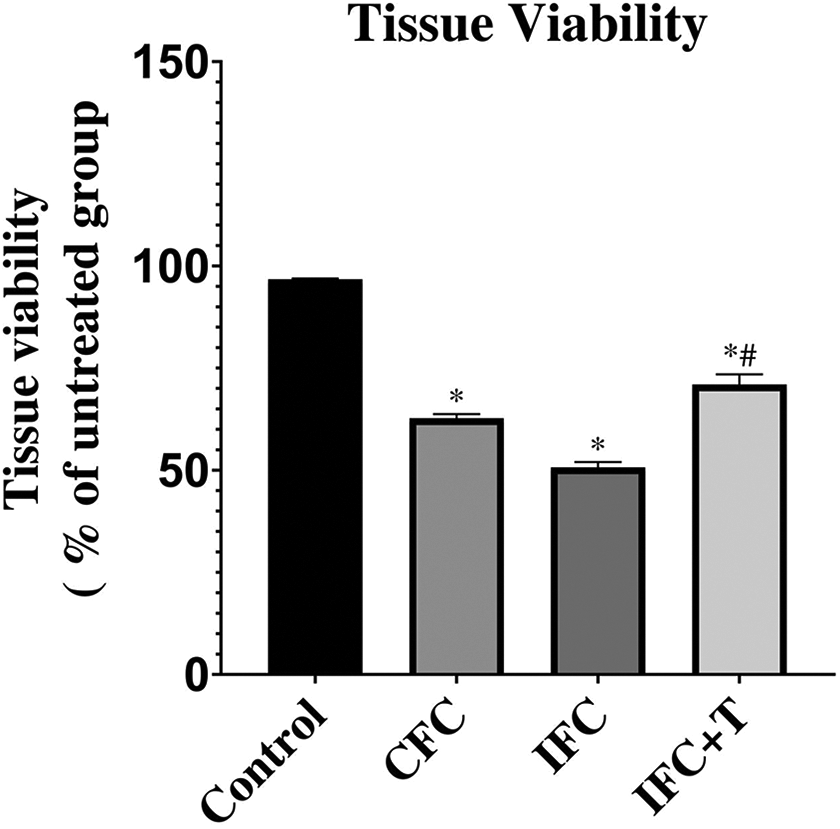
Cell viability in aortic valve tissues from different groups. Viability was measured using the Alamar Blue, and data collected from each group were normalized with respect to the control group. All data were expressed as the mean ± SD. (*P < 0.05 versus control group, #P < 0.0001versus IFC group, n = 6).
Assessment of hemocompatibility
Coagulation or inflammatory mechanisms are determined by soluble protein markers, which are measured in plasma after incubation of cryopreserved valves in relation to fresh blood samples. Coagulation activation and thrombin generation can be assessed through a TAT complex formation assessment. Thus, β-TG and TAT concentrations in blood plasma were measured after incubation of tissue samples to assess the potential for thrombogenesis. 29 The control group and the CFC or IFC or IFC + T group had a noticeable contrast to β-TG (P < 0.05, n = 6), but the IFC and IFC + T had no significant difference (P > 0.05, n = 6) (Figure 5a). Notable discrepancy between the control group and the CFC group (p < 0.05, n = 6), and no relevant differentiation between IFC or IFC + T group (p > 0.05, n = 6) has been found. Findings of hemocompatibility favor the use of nanoparticle encapsulated trehalose mediated preservation (Figure 5b).
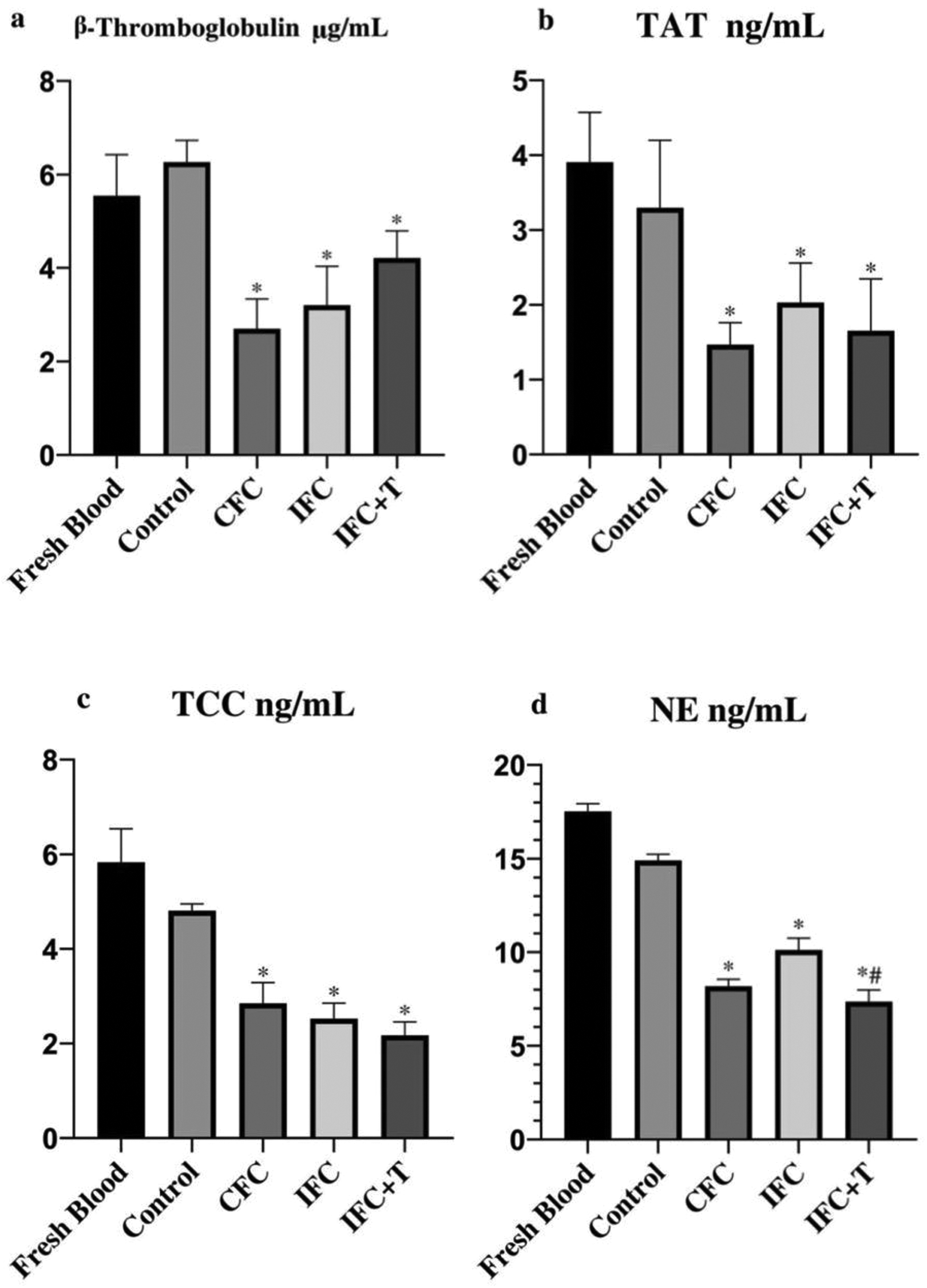
Diagram of hemocompatibility analysis: concentrations of thrombogenic markers beta-thromboglobulin (β-TG, a) and thrombin-antithrombin complex (TAT, b), inflammation marker Terminal complement complex (TCC, c) and Neutrophil elastase (NE, d) after incubation of tissues with whole rat blood for 1 hour (*P < 0.05 versus control group, #P < 0.05versus IFC group, n = 6).
Immunogenicity was quantified using the TCC and NE as markers. TCC is formed by assembling C5 through C9 after the activation of the complementary system, either by the classical, lectin, or alternative pathway. TCC is a complement to the activation level indicator and facilitates coagulation. 30 TCC contrast between all groups was remarkable when compared to the control group (p < 0.05, n = 6), but the difference between the IFC and the IFC + T groups was not sufficiently high (p > 0.05, n = 6) (Figure 5c). NE is a potent serine protease that plays a key role in the inflammatory response and during coagulation as a primary protective cell. It is commonly used to measure granulocyte activity during inflammation, and higher levels of NE mediate tissue degradation. NE demonstrated a significant difference when all the cryopreserved groups were compared with the control group and, likewise, between IFC and IFC + T (p < 0.05, n = 6) (Figure 5d).
Immunohistochemistry
Numerous cytokines modulate immune reactions, which are an interlinked series of events. 31 For immune homeostasis maintenance, the balance between TNFα and IL-10 is essential. TNFα, a biologically active pro-inflammatory cytokine whose expression is tightly regulated, and when produced in high levels is consistent with uncontrolled inflammation. The anti-inflammatory cytokine IL-10 neutralizes inflammation by decreasing TNFα production. 32 The Control group (57.77 ± 2.54) exhibited the highest levels of TNFα release, while the TNFα levels were most reduced in both IFC and IFC + T group (23.50 ± 3.47 and 23.69 ± 0.94) (P < 0.01, n = 6) (Figures 6 and 7). IFC and IFC + T groups have not shown marked distinction among them. A significant increase in IL-10 secretion was observed in the CFC group (87.07 ± 7.96) when compared with the control group (78.71 ± 3.20). IL-10 expression had significantly reduced in IFC and IFC + T group (57.37 ± 8.67 and 67.06 ± 2.63) (Figure 8), respectively, as compared with the control group and CFC group, and there was a huge distinction between these two groups (P < 0.05, n = 6). Immunohistochemical analysis images are shown in (Figure 9).
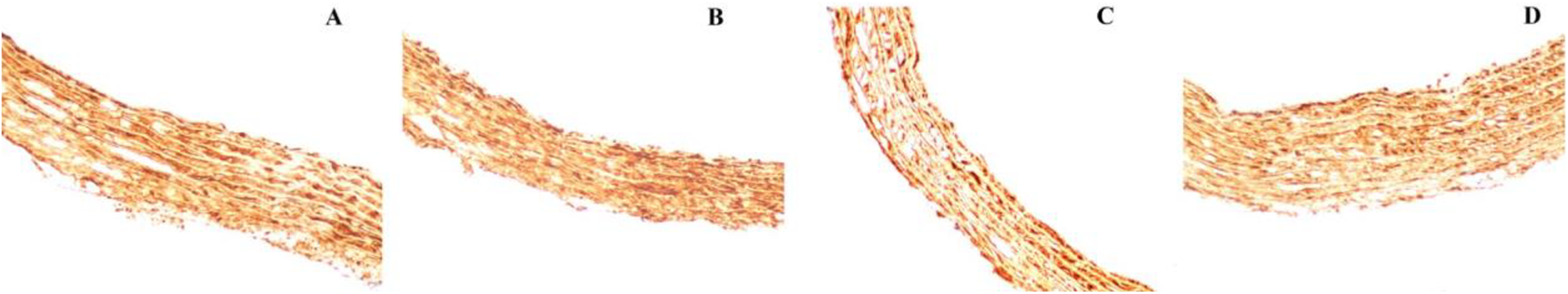
Representative images following immunohistochemical staining for IL-10 (original magnification × 40). A: control group; B: CFC group; C: IFC group; D: IFC + 0.2 M nanoparticle-encapsulated Trehalose (IFC + T) group.
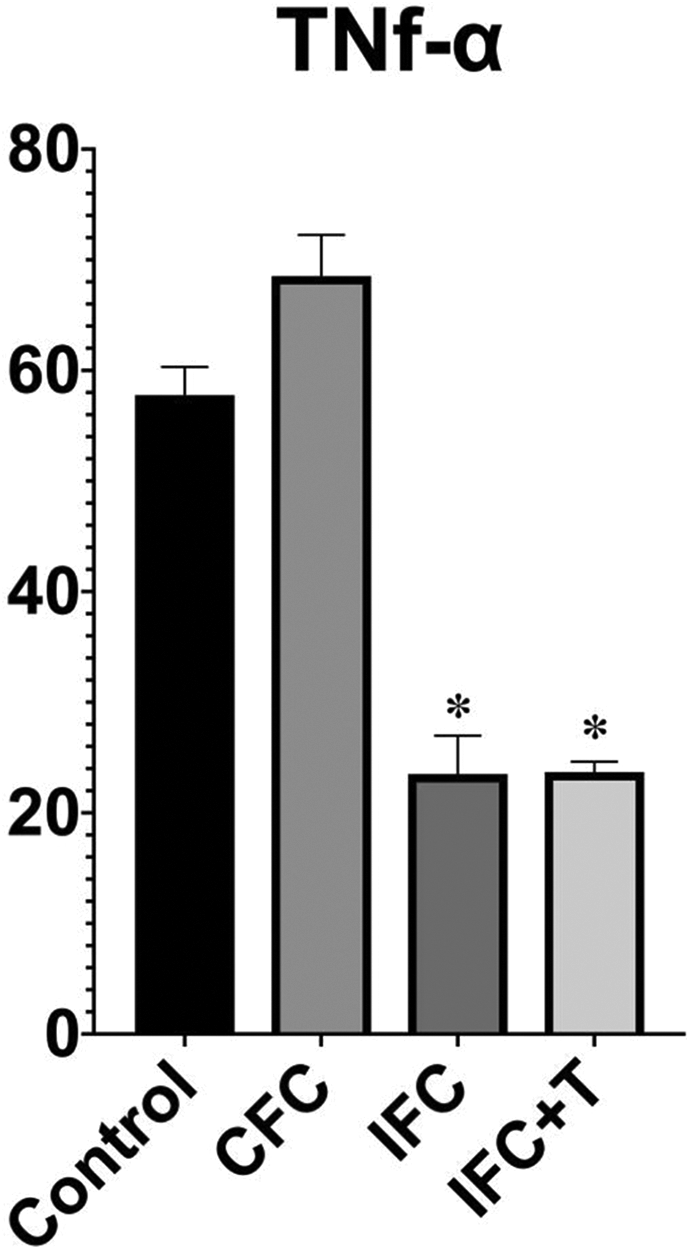
Optical density values of TNF-α analyzed using image pro plus software. (*P < 0.05 versus control group, n = 6).
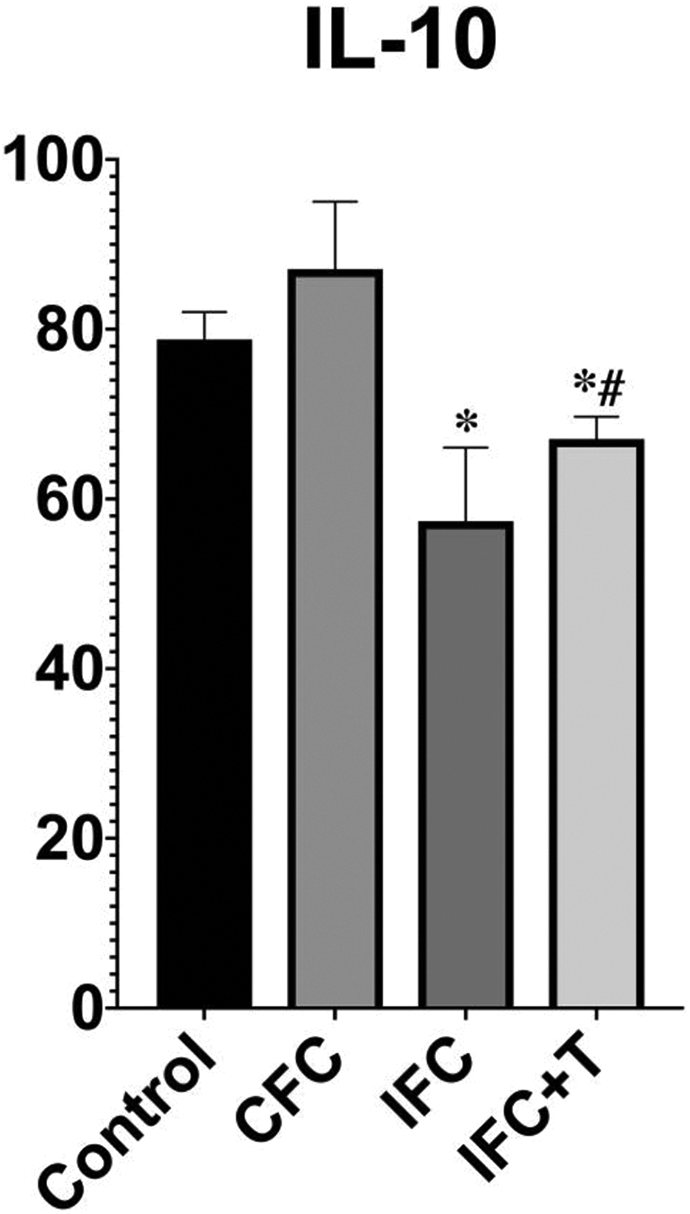
Optical density values of IL-10 analyzed using image pro plus software. (*P < 0.05 versus control group, #P < 0.05versus IFC group, n = 6).
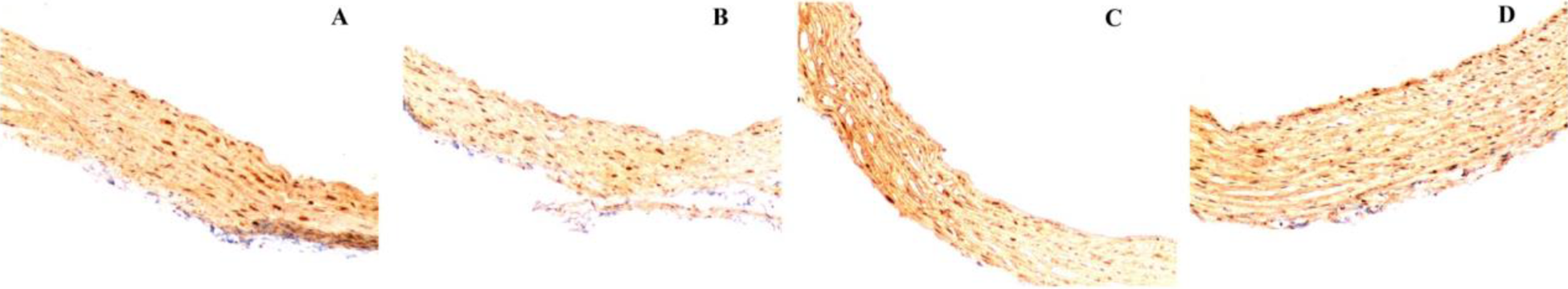
Representative images following immunohistochemical staining for TNFα (original magnification × 40). A: control group; B: CFC group; C: IFC group; D: IFC + 0.2 M nanoparticle-encapsulated Trehalose (IFC + T) group.
Discussion
Cellular deterioration of allogeneic heart valves after transplantation is a major concern in cryopreservation practice. Conventional frozen cryopreservation (CFC) demonstrates intracellular ice formation, which causes cell damage after thawing. The ECM structures of the heart valves, including collagen, elastic fibers, proteoglycans, and glycoproteins, significantly deteriorate due to the effect of ice. 33 –36 As a result, the incipient vitrification method was developed to overcome the challenges experienced in CFC. The primary advantage of this protocol is the absence of ice formation that causes cell damage in conventional cryopreservation. The use of VS83 has been shown to result in excellent retention of ECM integrity, which was assessed using multiphoton-induced autofluorescence and second harmonic generated (SHG) imaging. 37 To enhance the maintenance of cell integrity, nanoparticle-encapsulated trehalose is used in the VS83 protocol, with significantly positive outcomes. Numerous studies have focused on the impact of trehalose as a sugar on the cellular integrity and hemocompatibility of allogeneic heart valves. But the effect of nanoparticle-encapsulation has not received as much attention. Therefore, our research sought to explore the effect of nanoparticle-encapsulated trehalose in vitrification protocols.
In our study, the use of trehalose encapsulated with nanoparticles exhibited less ice formation as compared to the IFC group. However, there were no significant differences in ECM preservation between IFC and IFC + T groups. Heart valve components such as elastic fibers were not damaged in either of the cases. Therefore, the study proved that the IFC vitrification method is ideal for the preservation of cell morphology even before the introduction of trehalose. Cell viability was among the most crucial items of focus in our study due to its significance in cryopreservation. Our attention to this aspect was influenced by existing literature, which shows that intracellular uptake of trehalose leads to cell viability of 83% in mammalian cells, such as the human coronary artery endothelial cells (HCAECs), which is significantly higher than what is possible with DMSO. 38 Our study findings confirmed the literature observations; the post-warming viability of cells was better in the VS83 protocol with trehalose as compared to both CFC and IFC without the disaccharide. This outcome underlined the importance of adding trehalose to vitrification protocols to maintain cellular integrity and functions in cryopreserved heart valve tissue.
The use of trehalose in IFC protocols has proven to be effective in preserving cell viability after thawing. Eroglu et al. 11 found that low concentrations of the disaccharide (0.2 M) led to the long-term survival of mammalian cells after thawing, with 70% of human keratinocytes and 80% of the 3T3 fibroblasts used in the study remaining viable for a considerable length of time. Therefore, the researchers concluded that using sugars as additives for cryoprotective purposes could be a successful widespread practice. Specifically, trehalose prevents the degradation of cellular proteins during the freeze-thaw protocols. 39 The use of this sugar as a cryopreservation agent has been effective in promoting the desired results to a considerable extent. 40,41 In applying this agent for cellular protective purposes, it is imperative that the right concentrations are used depending on the cell type; different cells require specific concentrations of the disaccharide. For instance, human hematopoietic cells require 0.06 M, while human ES cells need 0.2 M, epidermal cells—0.4 M, and fetal skin cells—0.5 M. 40,42 –45 Kang and Shen 46 made similar conclusions on the effectiveness of trehalose in improving skin graft integrity by cryopreservation. Nanoparticle encapsulation with trehalose improves the cryopreservation of human adipose-derived stem cells, where trehalose was excellently distributed within the cell, therefore, enhancing the preservation of plasma membrane and the morphology of cells; in this case, the outcome was high viability. 9 In some cases, trehalose may need to be combined with another agent for the preferred results. For example, for the cryopreservation of RBCs, survival improved with the inclusion of apatite NP in addition to trehalose. 16,38 These instances indicate that trehalose is significantly effective in preserving the integrity and viability of cells and cellular composition during cryopreservation and after thawing.
In practice, encapsulating trehalose with a nanoparticle and using it inVS83 protocol leads to less ice crystallization. Diverse studies have demonstrated that this quality of disaccharides acts as better cryoprotective agents than other substances. For instance, Mantri et al. 47 found that the addition of 0.2 M trehalose demonstrated superior effects on the preservation of hematopoietic cells as compared to the use of 10% DMSO alone. The authors concluded that trehalose could be used to replace higher concentrations of DMSO, where combining the disaccharide with 2% DMSO would have more preferable results than a 5% DMSO only. A study by Liu et al. 48 supports the use of reduced concentrations of DMSO in cryopreservation protocols in the presence of trehalose or polyethylene glycol. These findings further affirm the usefulness of trehalose in preserving cell viability.
Another focus for our study was the impact of trehalose on hemocompatibility of the allogeneic heart valve in the VS83 protocol. Inflammatory reactions in allografting affect the success of the procedure in clinical practice. Previous studies have indicated that vitrification results in significantly fewer hemocompatibility problems as compared to conventional cryopreservation methods. 20 As aforementioned, the hemocompatibility of cryopreserved allogeneic valves after thawing is a major challenge in allografting. Poor hemocompatibility is a major issue with mechanical heart valves (MHV), which requires recipients to live on anticoagulants throughout life. 49,50 With tissue engineering, better options may be realized, with the most favorable hemodynamics, thus eliminating reliance on anticoagulant therapy. However, the full realization of this goal may take a considerably long time. As a result, the best current option is the tissue heart valve grafting, which, unfortunately, is inhibited by progressive degeneration due to inflammation and calcification. 51 Immunogenicity associated with allografting is mediated by various pro-inflammatory cytokines and other immune agents. For instance, PMN (Polymorphonuculear)-Elastase is a biochemical marker that influences the stimulation of pathologic granulocytes. 52 By triggering granulocyte activity, NE plays a significant role in inflammation. 26 Additionally, the complement pathway activation leads to the TCC generation through the assembly of C5 through C9. 26 Another process in the inflammation is the thrombotic activity, which occurs through agents such as β- TG, platelet factor 4, and thrombin, and antithrombin-III. 26,53
Moreover, activated monocytes, which are referred to as macrophages, produce cytokines, including TNFα, which is among the pro-inflammatory cytokines that contribute to hemocompatibility reactions, while IL-10 counters the activity of TNFα. Elevated levels of TNFα lead to the secondary activation of the immune system. 54 These agents induce immune reactivity that can severely damage allogeneic valves and impede successful grafting.
Lupinetti et al. 55 note that aortic valve allografts demonstrate high immunogenicity, although they do not exhibit marked constriction of endothelial cells or considerable presentation of endothelial surface antigens. This challenge necessitates the application of techniques that may impede immune reactivity. However, in the VS83 vitrification protocol, excellent results are observed in the matrix preservation and retention of the relevant cellular properties. The results show a reduced risk of immune reactivity in vivo and the absence of any significant allogeneic immune response in the recipients. 7,56 Similar observations are made by Seifert et al., 57 who found that the production of pro-inflammatory cytokines was considerably reduced in the IFC technique. Thus, indicating that the method is a promising cryopreservation alternative due to the low immunogenicity of heart valves. Such outcomes further highlight the advantage of using IFC methods, specifically vitrification, for mammalian cells’ preservations, particularly allogeneic heart valves.
Since trehalose has been excellent in preserving the viability of cells, its impact on hemocompatibility needs to be assessed. The success of this molecule in promoting cell integrity and viability in vitrification protocol means that it could receive widespread use. Thus, it becomes a part of the standard operating procedures in cryopreservation of allogeneic heart valves. As a result, concerns arise with regards to its potential in inducing immune reactivity or affecting the hemodynamics of the allograft tissues. Specifically, our study sought to understand the impact of incorporating trehalose encapsulated with nanoparticles in IFC. It is generally expected that the formulation does not induce inflammatory response; nano-size aquasome preparations are known to exhibit favorable immunological outcomes. 58,59 In fact, Rao et al. 9 found that the use of trehalose encapsulated with nanoparticles in cryopreservation ensures mammalian cells’ high viability without impairing cellular function. The difference between our study and most of the previous work is that we focused on the effect of nanoparticle-encapsulating trehalose, whereas others paid attention to trehalose without encapsulation.
Our study did not show any significant indicators of trehalose-induced hemo-incompatibility that would trigger the production of immunogenic and thrombogenic agents. Furthermore, immunohistochemistry results indicated a significant reduction in the release of TNFα for the IFC + T sample as compared to the CFC and control. As aforementioned, TNFα is an inflammatory cytokine whose production leads to secondary activation of the immune system. 60 Similarly, the concentrations of IL-10 were near normal for IFC + T in contrast to IFC and CFC in relation to the control group; this observation indicated that there would be mild inflammatory reactions with no significant effect on tissue immunogenicity for this group. As a result, our research showed that the uptake of nanoparticle-encapsulated trehalose by cells of allogeneic heart valves preserved through the vitrification method significantly reduces immunogenicity. Thus, proving to be promising for the clinical application of IFC in allografting.
Conclusion
Our research demonstrated that the use of trehalose encapsulated with nanoparticles in the VS83 protocol leads to a significant reduction in ice formation, which protects cells from damage. As a result, the cellular morphology is maintained after thawing, thus preserving the tissues’ functioning. This outcome means that incorporating nanoparticle-encapsulated trehalose in vitrification procedures is crucial for cellular viability. Before the advent of the IFC, CFC entailed considerable intracellular ice formation, which often resulted in deleterious effects on the cell structure and functioning. Additionally, CFC required expensive equipment to store and transport the cells at −135°C. Therefore, vitrification resulted in more efficient tissue preservation in addition to reduced cell damage. The inclusion of trehalose in VS83 formulations has been advised by literature findings, which indicate that certain disaccharides improve cellular integrity in freezing-thawing processes.
Moreover, vitrification has been shown to have considerably fewer hemocompatibility issues, thus eliminating major concerns over immune-induced inflammation. Our study findings confirmed these observations, with significantly better outcomes for the IFC + T samples as compared to IFC and CFC. However, IFC still scored better for immunogenicity as compared to CFC. Therefore, the intracellular uptake of trehalose has a positive effect on hemocompatibility in the VS83 protocol. However, the immune modulation mechanism of our incipient protocol is still obscure and needs further evaluation. Therefore, our study findings are promising for clinical application in the future, but currently, the topic needs further exploration.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
