Abstract
In this study, Arabic gum-capped ZnO NPs (ZnO NPs-Cap) were synthesized via a simple precipitation method, and a comparative study was performed with uncapped ZnO. The synthesized nanoparticles (NPs) were characterized using UV–visible (UV-vis) spectroscopy, Fourier-transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), and Energy-dispersive X-ray spectroscopy (EDS) coupled with scanning electron microscopy (SEM). The absorption peak of ZnO NPs-Cap appeared at 366.5 nm, and the calculated band gap energy was 3.38 eV. Antibacterial activity was assessed against gram positive and negative foodborne pathogens. The minimum inhibitory concentration (MIC) and the minimum bactericidal concentration (MBC) of ZnO NPs and ZnO NPs-Cap against S. aureus, Bacillus cereus, E. coli and S. typhimurium bacterial strains revealed a clear superiority of ZnO NPs-Cap than uncoated ZnO. Time–kill assays performed at different concentrations and times has demonstrated a time-dependent reduction in bacterial viability for both formulations. The present study demonstrates that Arabic gum capping enhances the dispersion, stability, and antibacterial efficacy of ZnO NPs. The findings also provide useful insight into the biological activity and potential eco-toxicological implications of biopolymer-modified ZnO nanomaterials.
1. Introduction
Nanoparticles (NP) have been extensively applied in various fields. 1 Matei et al., 2008], and their unique properties have drawn widespread scientific interest. Among the differences from their bulk-size counterparts, 2 NPs have a larger surface area, which increases the probability that they will interact with the bio-organic surface found on viable cell surfaces. 3 Inorganic metal oxide NPs, such as ZnO, TiO2, MgO, and SiO2, appear toxic to biological systems and are thus chosen for their potential as therapeutic, diagnostic, and antimicrobial agents.4–8 As inorganic oxide NPs have less toxicity and heat resistance than traditional organic nanoparticles, their use as an antimicrobial agent has been effective against resistant strains of microbial pathogens. In addition to these advantages, they provide human cells with a small amount of mineral element that, despite its size, exhibits strong activity.9–11
Zinc oxide (ZnO) is well-known for its chemical and physical stability, antimicrobial activity against certain microorganisms and catalytic activity. In addition, ZnO can absorb ultraviolet and infrared in an intensive state; hence, it has been extensively applied in semiconductors, sensors, solar cells, and transparent electrodes. 1
Moreover, nanocomposite of ZnO NPs might have adverse impacts on many aspects of food and agricultural systems. In particular, the antimicrobial activity of ZnO NPs against foodborne pathogens might be an alternative choice that offers cost- effectiveness and safer for human consumption. This is particularly important in view of the increased number of using antibiotic-resistant strains of different bacteria in the environment and food processing.3,12 At present, Many additives are commonly added as antibacterial, antioxidant agents, flavor compounds, and other functional nutrients to enhance the performance of edible coatings. 13
Although several studies have investigated the antibacterial activity of ZnO NPs, limited research has focused on ZnO NPs incorporated with biopolymers such as Arabic gum. Therefore, this study aims to conduct a comparative evaluation of uncapped ZnO NPs and Arabic gum-capped ZnO NPs (ZnONPs-Cap) against selected foodborne pathogens, including both Gram-positive and Gram-negative bacteria, to assess their antibacterial performance in detail. This work also seeks to explore the potential of biopolymer-based ZnO nanocomposites as promising antimicrobial agents for controlling pathogenic bacteria in contaminated foods during storage, processing, and production.
2. Materials and methods
2.1. Synthesis of ZnO NPs
ZnO NPs were synthesized through a modification of reported protocol in this study.
14
Briefly, a simple method of ZnO NPs formation using zinc nitrate hexahydratem (Zn(NO3)2·6H2O) as the zinc precursor and sodium hydroxide (NaOH) as the precipitating agent has been reported here. In a typical procedure, 100 mL of 1 M Zn(NO3)2·6H2O solution (molecular weight 297.49 g mol-1) was prepared in double-distilled water under continuous stirring at room temperature. Separately, 30 mL of 5 M NaOH solution was prepared by dissolving NaOH pellets (molecular weight 40 g mol-1) in double-distilled water with vigorous stirring. The NaOH solution was then added dropwise to the zinc nitrate solution under constant magnetic stirring at a rate of approximately 1 mL min-1 to control the nucleation process. The addition continued until the pH of the reaction mixture reached around 10–11, leading to the formation of a dense white precipitate of zinc hydroxide, Zn(OH)2. The resulting suspension was stirred for an additional 1 hour at room temperature to ensure complete precipitation and homogeneity, followed by heating at 70–80 °C for 2 hours to promote the in-situ dehydration of Zn(OH)2 into ZnO NPs according to the following reaction:
After the reaction, the white precipitate was separated by centrifugation at 8000 rpm for 10 minutes, then washed several times with deionized water and ethanol alternately to remove residual chloride and sodium ions. The washed sample was then dried overnight in a hot-air oven at 90 °C, yielding a fine white ZnO nanopowder. To improve the crystallinity, the dried sample was calcined at 400 °C for 4 hours in a muffle furnace, which converted the amorphous hydroxide into crystalline wurtzite ZnO NPs. 14
2.2. Synthesis of ZnO NPs-Cap
After the synthesis of ZnO NPs (13 g), encapsulation was carried out using Arabic gum. A 5% (w/v) Arabic gum solution was first prepared by dissolving 5 g of Arabic gum in 95 mL of boiled distilled water under continuous stirring until a clear and homogeneous solution was obtained and then cooled to room temperature. The synthesized ZnO NPs were gradually added to the cooled Arabic gum solution while stirring with a magnetic stirrer for 15–30 minutes to ensure uniform wetting and dispersion. The resulting mixture was then transferred to a homogenizer (21 rpm, 10 min) to further improve the uniformity and reduce particle agglomeration, followed by treatment in an ultrasonic bath or probe sonication at medium power for 15–30 minutes. Ahmed et al. [2025] 15 has reported that sonication effectively prevented NPs aggregation and promoted a uniform coating of the NPs with Arabic gum, thereby enhancing stability and minimizing further aggregation. After sonication, the encapsulated ZnO NPs were cooled to room temperature and stored in airtight containers until use.
2.3. Characterization of ZnO NPs and ZnO NPs-Cap
The synthesized ZnO NPs and ZnO NPs-Cap samples were characterized using a variety of analytical techniques. The optical properties were examined by UV-visible/near-infrared (UV-vis/NIR) spectroscopy (Jasco V-770, Japan). Fourier transform infrared spectroscopy (FTIR, Perkin-Elmer FTIR-Spectrum BX, USA) was employed to identify the functional groups present in the samples. The morphology and particle size distribution were studied using transmission electron microscopy (TEM, JEOL JEM-1400 Plus, Japan). Elemental composition was confirmed through energy dispersive spectroscopy (EDS) coupled with scanning electron microscopy (SEM, JSM-7610F, Japan). X-ray diffraction (XRD) patterns were recorded with a D8 Advance diffractometer (Bruker, USA) equipped with a graphite monochromator and CuKα radiation source (λ = 1.5418 Å), operated at 45 kV and 40 mA with a shape factor of K = 0.9. The average crystallite size (D) was calculated using the Scherrer equation: D= Kλ/βcosθ
Where λ is the X-ray wavelength,θis the Bragg angle,βis the full width at half maximum (FWHM) of the diffraction peak, and K is the crystallite-shape factor.
The hydrodynamic diameter and polydispersity index (PDI) of the colloidal NPs were determined by dynamic light scattering (DLS) using a Zetasizer (ZEN3600, HT Laser, Malvern Nano Series, UK. Additionally, Zeta potential analysis was conducted using Zetasizer Advance (ZSU3305, Malvern Nano Series, Instruments, UK).
2.4. Antibacterial assessment
This experiment followed the method proposed by Nosaka et al., 1998 16 with some modifications. ZnO NPs and ZnO NPs-Cap at different concentrations (30, 40, 50, and 60 mg/ml) were dissolved in ethyl alcohol and sterilized distilled water, and then the samples were carefully mixed in an ultrasound device for 30 hours. Staph. aureus ATCC 8739, B. cereus ATCC 10876, Escherichia coli ATCC 25922, and Salmonella typhimurium ATCC 14028 were obtained from American-type culture collection. All these strains were grown aerobically in nutrient broth for 24 hours at 37° C before being used as target organisms. The density of the bacterial strain was adjusted to an optimal density of 0.5 McFarland standards.
2.5. Minimum inhibitory concentration assay (MIC)
The MIC for the different concentrations of ZnO NPs and ZnO NPs-Cap (30–60 mg/ml) was determined by the disc borer (well) method followed for antimicrobial activity. Colonies of main was overnight activated in nutrient broth medium (24 h) until 0.5 McFarland turbidity (corresponding to∼105 CFU mL−1 for Staph. aureus and B. cereus and ∼ 104 for E. coli and S. typhimurium). Muller-Hinton agar medium was used to determine antibacterial activity. The respective strains of bacteria were swabbed (sterile cotton swabs) in susceptible media. The agar disc diffusion method can be performed using an 8 mm diameter cork borer to create four wells in the agar. One hundred µl of each concentration (30–60 mg/ml) was put in the well. One well in the middle of the petri dish, which served as the control, contained 100 µl ethyl alcohol or water, and a duplicate for each petri dish was performed. The plates were incubated at 37 °C for 24 hours, and the inhibition zone was measured and calculated.
2.6. Minimum bactericidal concentrations
The minimum bactericidal concentrations (MBC) of ZnO NPs and ZnO NPs-Cap were carried out on four tested bacterial strains at a concentration that began at 60 mg/ml dissolved in ethyl alcohol or water, then diluted in Muller-Hinton broth to 30, 15, 7.5, and 3.75 mg/ml. One hundred µl of main overnight activated bacteria (corresponding to∼105 CFU mL−1 for Staph. aureus and B. cereus and ∼ 104 for E. coli and S. typhimuriu) was inoculated into every tube and incubated at 37 °C for 24 hours. In comparison, the positive control of each microbe grew in Muller-Hinton broth alone. After the incubation period (24 h), 10 μl of each tube at different dilutions was subcultured on petri dishes containing Muller-Hinton agar (Oxoid, CM0337). After a 24-hour incubation period, the MBC was determined and defined as the lowest concentration that inhibited visible growth of the subculture.
2.7. Time–kill curves
The method proposed by Jensen et al., 2020 17 with a small modification was followed, and cultures of the four tested bacterial strains were grown overnight at 37 °C in Muller- Hinton broth and diluted to reach the 0.5 McFarland turbidity as previously described (corresponding to∼105 CFU mL−1 for Staph. aureus and B. cereus and ∼ 104 for E. coli and S. typhimurium). One hundred μl of bacterial suspensions were adjusted to different concentrations, namely, 30, 15, 7.5, and 3.125 mg mL−1, of ZnO NPs and ZnO NPs- Cap to a final volume of 50 mL, and then incubated with aeration (150 rpm) at 37 °C. Cell counts were determined via tenfold serial dilution on Muller-Hinton agar every hour for the first 4 hours and then after 12 hours and 24 hours. Experiments were conducted in duplicate. A time–kill curve is conventionally plotted with time on the x- axis and the total viable count (or log10 CFU/mL) on the y-axis.
Data were expressed as mean± standard deviation (SD). Data were analyzed using an analysis of variance (ANOVA) using SPSS software, Version 16. Significance was expressed at the 5% level.
3. Results and discussion
3.1. UV-Vis absorption analysis of ZnO NPs and ZnO NPs-Cap
The UV-vis absorption spectra of the synthesized ZnO NPs and ZnO NPs–Cap are presented in Figure 1. The pristine ZnO NPs show a sharp absorption peak at approximately 350 nm, which is characteristic of the intrinsic band-to-band transition of ZnO due to electron excitation from the valence band to the conduction band.
18
The optical band gap, calculated using the relation: UV–vis absorption spectra of pristine ZnONPs and Arabic gum-encapsulated ZnONPs (ZnONPs Cap), showing the optical absorption behavior of both samples.
3.2. FTIR analysis of ZnO NPs and ZnO NPs-Cap
The FTIR spectra ZnO NPs, Arabic gum, and ZnO NPs–Cap are presented in Figure 2. The spectrum of pure ZnO NPs exhibits a prominent absorption band at ∼416 cm-1, which corresponds to the stretching vibration of the Zn–O bond, confirming the successful formation of ZnO NPs.20,21 The broad band observed at ∼3448 cm-1 is attributed to the O–Hstretching vibration of surface-adsorbed water molecules and hydroxyl groups, while the peaks around 1381 cm-1 and 1002 cm-1 correspond to the bending vibrations of O–H and C–O stretching, respectively, which may arise from residual species or absorbed CO2.21,22 The FTIR spectrum of Arabic gum shows characteristic bands associated with its polysaccharide structure. The broad absorption at ∼3410 cm-1 is due to the O–H stretching vibration of hydroxyl groups, while the peaks at 2924 cm-1 and 1620 cm-1 correspond to the C–H stretching and C=O stretching vibrations of carboxyl and carbonyl groups, respectively. Additional peaks at 1072 cm-1 and 1419 cm-1 are assigned to the C–O–C and C–O stretching vibrations of glycosidic linkages within the gum matrix.21,22 In the case of ZnO NPs-Cap, the spectrum retains the major functional group bands of Arabic gum with slight shifts and changes in intensity, indicating the interaction between ZnO NPs and gum biomolecules. The broad O–H band appears at ∼3425 cm-1, while the peaks at 2924 cm-1, 1612 cm-1, 1404 cm-1, and 1072 cm-1 correspond to C–H, C=O, C–O, and C–O–C stretching vibrations, respectively. Notably, the Zn–O stretching vibration shifts slightly from 416 cm-1 in the pure ZnO NPs to 470 cm-1 in the capped sample, suggesting coordination or electrostatic interaction between the ZnO surface and the hydroxyl or carboxyl groups of Arabic gum.
22
These spectral changes confirm the successful encapsulation and stabilization of ZnO NPs by Arabic gum through surface functionalization, which enhances NP dispersion and prevents agglomeration.21,22 FTIR spectra of ZnONPs synthesized via precipitation, Arabic gum-encapsulated ZnONPs (ZnONPs-Cap), and Arabic gum.
3.3. X-ray diffraction patterns of ZnO NPs and ZnO NPs-Cap
The crystalline phases of the synthesized ZnO NPs and ZnO NPs-Cap were confirmed by XRD, as presented in Figure 3. Both samples exhibited characteristic diffraction peaks at 2θ values of 31.7°, 34.4°, 36.2°, 47.5°, 56.6°, 62.8°, 66.3°, 67.9°, and 69.1°, which correspond to the (100), (002), (101), (102), (110), (103), (200), (112), and (201) planes of the hexagonal wurtzite ZnO structure (JCPDS card no. 36-1451). The absence of additional impurity peaks indicates that the synthesized materials are phase-pure ZnO NPs. The average crystallite sizes, calculated using the Debye–Scherrer equation, were found to be 23 nm for ZnO NPs and 19 nm for ZnO NPs-Cap. The smaller crystallite size of ZnO NPs-Cap demonstrates the stabilizing effect of Arabic gum, which likely restricted crystal growth and prevented agglomeration by providing surface functional groups that interact with the ZnO nuclei.
19
In contrast, the uncapped ZnO NPs exhibited sharper and more intense peaks, confirming relatively higher crystallinity and larger crystallite dimensions. This observation is consistent with previous studies, which reported that biopolymer capping agents such as Arabic gum, starch, or cellulose derivatives act as growth-controlling agents, thereby reducing NP size while enhancing colloidal stability, as also reported by.21,23 These findings indicate that Arabic gum plays a crucial role in tailoring the structural properties of ZnO NPs, producing smaller and more stable crystallites suitable for photocatalytic, biomedical, and environmental applications.21,24 XRD patterns of synthesized ZnONPs and Arabic gum-encapsulated ZnONPs (ZnONPs-Cap).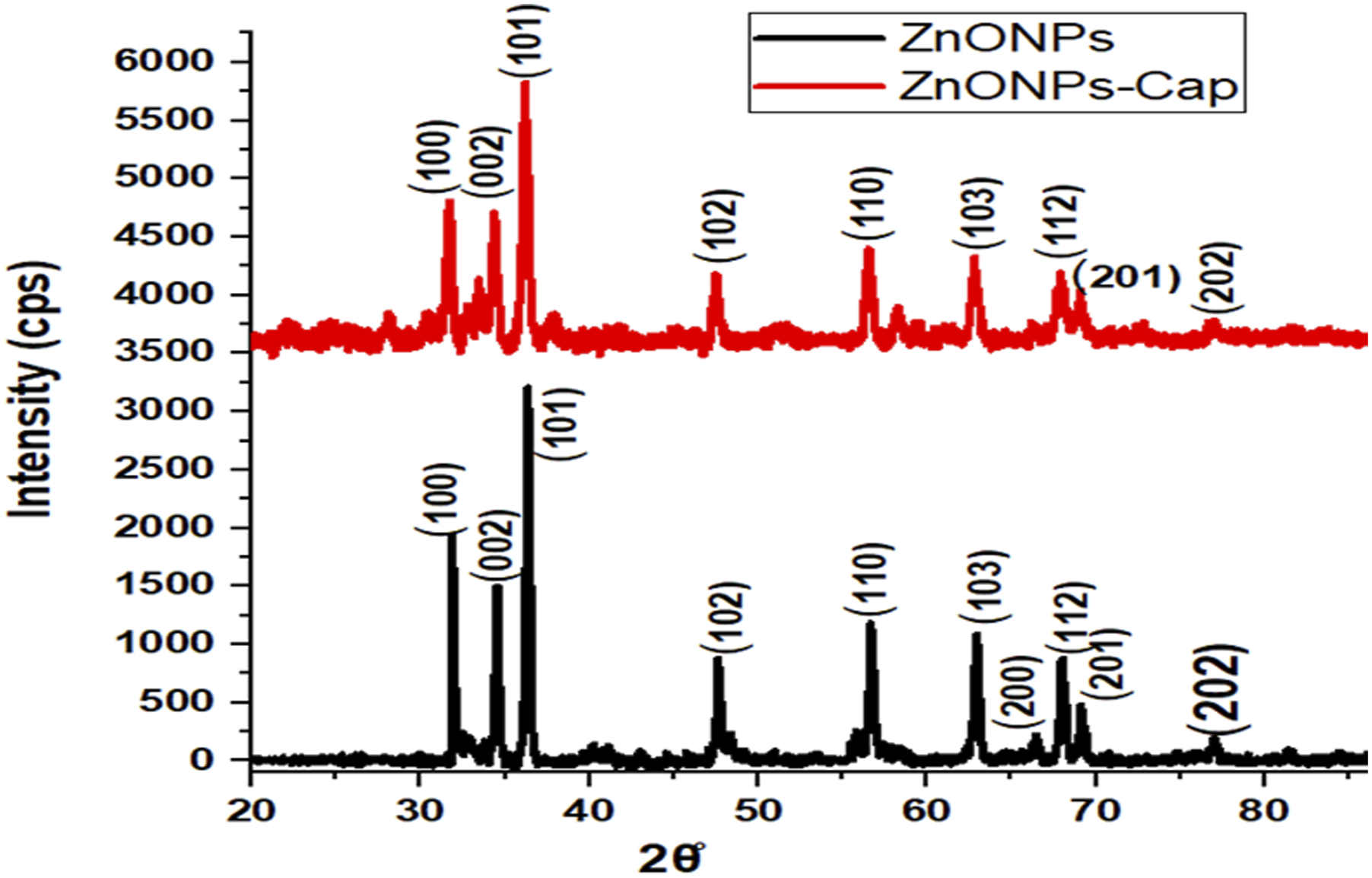
3.4. Particle size distribution by DLS of ZnO NPs and ZnO NPs-Cap
DLS measurements revealed that the pristine ZnO NPs exhibited a Z-average diameter of 54.49 nm with a PDI of approximately 0.3, indicating a moderately uniform size distribution and satisfactory colloidal stability with limited aggregation. In contrast, the ZnO NPs-Cap showed superior colloidal stability, characterized by a sharply defined monomodal peak centered at 241.2 nm and a significantly reduced PDI of 0.1 (Figure 4(a)). The narrow size distribution and lower PDI clearly demonstrate the effectiveness of Arabic gum in providing steric stabilization through surface adsorption of its hydroxyl and carboxyl functional groups, which prevent NP agglomeration by introducing repulsive barriers between particles. The slightly larger hydrodynamic diameter observed for ZnO NPs-Cap compared with the uncapped sample arises from the organic coating layer of Arabic gum surrounding the ZnO cores, increasing the overall particle size detected by DLS. Similar findings were reported by Pauzi et al., 2020
25
; Kora et al., 2025,
26
who demonstrated that biopolymer encapsulation substantially enhances NP stability and homogeneity by mitigating the destabilizing effects of high surface energy and residual synthetic impurities.25,26 DLS measurements of Arabic gum-encapsulated ZnONPs (ZnONPs-Cap) showing (a) the average hydrodynamic diameter (d. nm) and (b) the zeta potential distribution.
The synthesized dispersion samples exhibited a negative surface charge in both cases, with zeta potential values of −38.22 mV for pristine ZnO NPs and −25.12 mV for ZnO NPs-Cap (Figure 4(b)). The highly negative value for the uncapped ZnO NPs indicates strong electrostatic repulsion between particles, placing them within the highly stable colloidal regime, consistent with their limited aggregation. Upon capping, the absolute magnitude of the zeta potential decreased (became less negative) because Arabic gum adsorbed onto the ZnO surface and partially screened the surface charge. 27 The partial protonation of hydroxyl and carboxyl groups in the biopolymer, along with counter-ion condensation, likely compressed the electrical double layer surrounding the NPs. Although this reduction in zeta potential would, according to Derjaguin–Landau–Verwey–Overbeek (DLVO) theory, weaken purely electrostatic stabilization, the adsorbed polysaccharide shell introduces additional steric and hydration barriers that effectively prevent close particle approach. 28 This steric stabilization explains the excellent colloidal stability and the low PDI observed by DLS for ZnO NPs-Cap, despite their smaller (less negative) zeta potential value.
3.5. TEM of ZnO NPs and ZnO NPs-Cap
The morphological characteristics of the synthesized ZnO NPs and ZnO NPs-Cap were examined using TEM image analysis, as shown in Figure 5 (a) and (b), respectively. The uncapped ZnO NPs (Figure 4(a))) exhibit irregularly shaped aggregates composed of nearly spherical to slightly hexagonal nanoparticles with an average size in the nanometer range. The particles show partial agglomeration, which can be attributed to strong van der Waals forces and the high surface energy of bare ZnO nanoparticles. The well-defined contrast between the dark particle regions and the bright background indicates the crystalline nature of the ZnO, consistent with the wurtzite structure confirmed by XRD analysis, as has been reported by Alwash et al. 2025.
21
In contrast, the ZnO NPs-Cap (Figure 4(b))) displays a more dispersed morphology, where the NPs are embedded within or surrounded by a lighter-contrast organic matrix corresponding to the gum coating. The presence of this polymeric layer contributes to a slight increase in the apparent particle size observed under TEM.
29
The smoother boundaries and reduced particle clustering in the capped sample demonstrate that Arabic gum effectively acts as a steric stabilizer, minimizing aggregation by forming a thin organic shell around the ZnO cores. This encapsulating layer improves NP dispersion and surface passivation, which correlates well with the enhanced colloidal stability and lower PDI observed from DLS measurements.
22
TEM micrographs of (A) uncapped ZnONPs and (B) Arabic gum-encapsulated ZnONPs (ZnONPs-Cap).
3.6. Elemental composition analysis by EDS
The EDS spectrum of the uncapped ZnO NPs (Figure 6(a))) shows the expected Zn and O signals with intense Zn Kα/Kβ peaks at ∼8.6/9.6 keV and an O K peak near ∼0.52 keV, confirming ZnO formation. Semi-quantitative analysis gives 88.94 wt% Zn (66.30 at%) and 11.06 wt% O (33.70 at%). The Zn:O atomic ratio (∼1.97:1) is slightly Zn- rich, a common outcome in EDS because heavier elements are over-represented and surface oxygen can be underestimated; it may also reflect modest oxygen-vacancy content. These peak positions and assignments match standard EDS signatures reported for ZnO.30,31 EDS spectral elemental analysis of (a) un-capped ZnONPs and (b) Arabic gum-encapsulated ZnONPs (ZnONPs-Cap).
For the ZnO NPs-Cap sample (Figure 6(b), Zn remains prominent (Zn K at ∼8.6 keV), but the spectrum also contains strong C K and O K signals and trace K K and Ca K lines, with the composition C 21.76 wt% (44.26 at%), O 22.92 wt% (34.99 at%), K 0.14 wt%, Ca 0.17 wt%, and Zn 55.01 wt% (20.56 at%). The increased C and O, together with K and Ca traces, are consistent with the polysaccharide nature of Arabic gum, which contains potassium and calcium salts of arabinogalactan/protein units; an organic shell also attenuates the Zn signal from the core, lowering its apparent wt% relative to the uncapped powder, as also reported by Prasad et al. (2022). 32 Comparatively, the capped spectrum evidences successful surface functionalization/encapsulation: (i) Zn peaks persist, confirming the inorganic core; (ii) polymer-related elements (C, O) and minor K/Ca appear, consistent with the gum matrix; and (iii) the relative reduction of Zn wt% reflects signal attenuation by the organic coating and smaller interaction volume in the near-surface region probed by EDS. Such compositional signatures and interpretations for gum-modified ZnO agree with recent reports on Arabic gum-assisted ZnO and ZnO NPs-Cap. 21
3.7. Antibacterial evaluation
MIC determination of ZnONPs and ZnONPs-Cap on some foodborne pathogens (mean zone of inhibition (mm)2).

A: Alcohol; W: Water; -: no effect on bacterial growth.
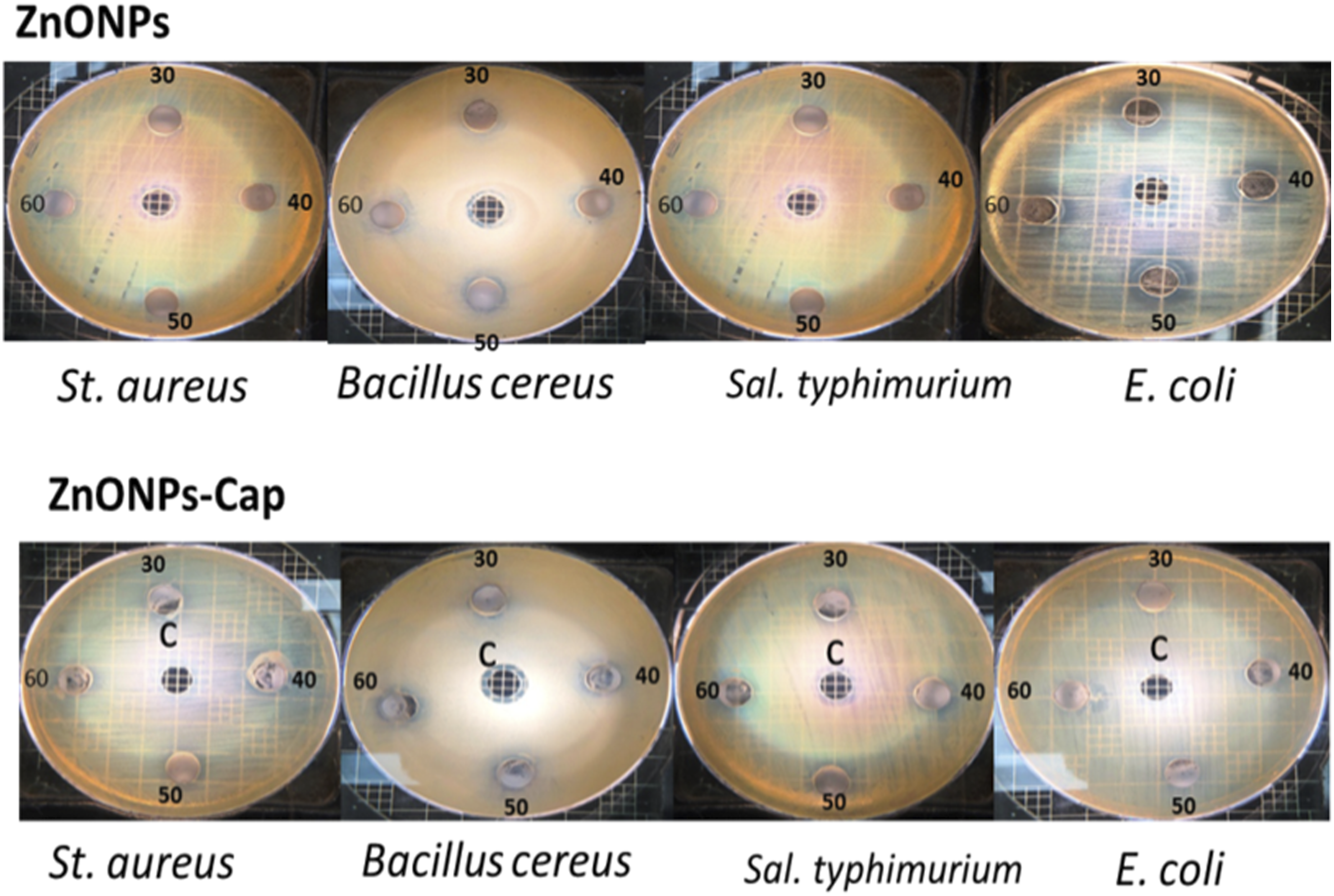
Antimicrobial activity of ZnONPs and ZnONPs-Cap at concentrations of 60, 50, 40, and 30 mg/mL dissolved in ethyl alcohol. C: Negative control (ethyl alcohol). The highest zone of inhibition noticed for ZnONPs-Cap against S. aureus was 20 mm² at 60 mg/ml, compared with only 16 mm² for ZnONPs. Similar effect ZnONPs-Cap against Bacillus cereus was 15 mm² vs. 14 mm² for ZnONPs.The MIC against E. coli and S. typhimurium was 14 mm² and 12 mm² at 60 mg/ml of ZnONPs-Cap and ZnONPs, respectively.
Both Zinc Oxide Nanoparticles (ZnO NPs and ZnO NPs-Cap) dispersed in ethanol or as part of an ethanolic mixture generally exhibit higher antibacterial efficacy against both gram-positive and gram-negative bacteria compared to aqueous (water) suspensions. The superior performance is largely due to better dispersion, reduced agglomeration, and synergistic effects with the alcohol solvent. Ethanol acts as a better dispersant, keeping the particles separated and maximizing their surface contact with bacteria.
These findings indicate that NPs coating enhances biological performance by improving stability, reducing aggregation, increasing surface area, and facilitating cellular penetration. These results are consistent with previous studies confirming that ZnO surface modification significantly increases antibacterial activity compared with the unmodified material.37–39 Accordingly, ZnO NPs-Cap, coated with Arabic gum—a commonly used encapsulating material known for its excellent emulsifying and film-forming properties, 40 exhibit improved functional characteristics. Beyond its encapsulation ability, Arabic gum also serves as an effective stabilizer, enhancing product texture and extending shelf life. 41 The documented health benefits of Arabic gum include its role as a source of soluble dietary fiber, and its potential biological effects, 37 further underscore its versatility.
Many authors have proposed that the antibacterial activity of ZnO nanoparticles (ZnO NPs) arises from multiple synergistic mechanisms. These include disruption of the bacterial cell membrane, increased permeability, and leakage of cytoplasmic contents. ZnO NPs also generate reactive oxygen species (ROS), which induce oxidative damage to lipids, proteins, and DNA, disrupt electron transport processes, and promote the release of Zn2+ ions. These combined effects lead to cellular dysfunction and ultimately bacterial cell death. Furthermore, the interaction between ZnO NPs and the bacterial cell wall plays a critical role in their toxicity.
42
Brayner et al.
43
reported that ZnO NPs cause significant damage to the bacterial cell wall of Escherichia coli, leading to membrane disorganization and increased permeability. This facilitates the accumulation of nanoparticles on the cell surface and their internalization into the bacterial cells, thereby enhancing their antibacterial effect (Figure 8). da Silva et al. 2019,
44
noticed the same effect against S. aureus, with holes created in the cell membrane after contact with ZnO NPs. Authors investigated the effect of Arabic gum-ZnO NPs on S. aureus and E. coli as antibacterial properties. The author reported that the MBC values of Arabic gum-ZnO NPs for S. aureus and E. coli were 31.25 μg/mL and 62.5 μg/mL, and 62.5 μg/mL, and 125 µg/mL, respectively.
25
A greater-than-50% toxicity to biofilm for both S. aureus and E. coli was observed when a high concentration (500 μg/mL) was used. Hence, Arabic gum ZnO NPs are thought to represent a new-generation, natural antibacterial agent. Schematic diagram illustrating the antibacterial mechanism of ZnO NPs on bacterial cells.
ZnO NPs and ZnO NPs-Cap dissolved in water did not yield any suitable results against any of the bacterial strains tested. Some authors have made similar observations and mentioned that ZnO NPs’ instability in water limits their biological applications, considering that biomolecules such as DNA, proteins, and peptides are present in aqueous media.
Hence, the surface of ZnO NPs must be modified to enhance the distribution of the molecules. We dissolved ZnO NPs in two other chemicals, namely, ethyl alcohol and dimethyl sulphoxide (DMSO), and both yielded good reactions against the bacterial strains tested. However, we prefer to use ethyl if needed for applications to food in the future. For these reasons, different strategies have been suggested in an attempt to change the surface of ZnO NPs to render them stable in water. 45
The attributes outlined above make Arabic gum a highly suitable component for the encapsulation of bioactive molecules to enhance their stability and bioavailability. Consequently, ZnO NPs-Cap emerge as an effective nanomaterial for microbial and food-related applications that require high inhibitory potential. This suggestion is in agreement with previous reports showing that NP surface modification—through coating with biopolymeric or bioactive compounds—can enhance antibacterial activity by improving interaction with the bacterial cell membrane, increasing the release of active ions and reducing aggregation that limits biological performance.37,46 ZnO NPs- Cap demonstrated clear superiority over ZnO NPs, rendering them a promising candidate due to their strong and consistent antimicrobial activity against both Gram-positive and Gram-negative bacteria.
MBC of selected natural extracts of ZnONPs and ZnONPs-Cap on some foodborne pathogens (mg/mL).

A: Alcohol, W: Water; - : No bacterial growth; +: Bacterial growth; ±: Bacteriostatic/Bactericide.
3.8. Time–kill curve
The reduction in bacterial count (cfu/ml) to 3 log decrease, of the killing of 99.9% over a specific time, is referred to as bactericidal.
48
The killing occurred with a constant logarithmic phase through a time–kill; 90% of the kill carried out at 6 hours is equivalent to a 99.9% kill at 24 hours.
49
In our study, the kill rate was determined as a viable count at every one hour for the first 4 hours, then after 12 and 24 hours. Figures 9-12 present the time–kill curve to evaluate the effect of ZnO NPs-Cap and ZnO NPs on the viability counts of four bacterial strains at different concentrations (30, 15, 7.5, and 3.75 mg/ml) and for 1, 2, 3, 4, 12, and 24 hours of growth. It appears that ZnO NPs-Cap and ZnO NPs at all the concentrations were effective on three bacterial strains (S. aureus, E. coli, and S. typhimurium), and the growth decreased gradually until it could not be counted on the culture media. Hence, we can conclude that both ZnO NPs-Cap and ZnO NPs had a bactericidal effect on the three strains. Time-kill curve for S. aureus at different concentrations of ZnO NPs and ZnO NPs-Cap (mg/ml). Time-kill curve for Bacillus cereus at different concentrations of ZnO NPs and ZnO NPs-Cap (mg/ml). Time-kill curve for Salmonella typhimurium at different concentrations of ZnO NPs and ZnO NPs-Cap (mg/ml). Time-kill curve for E. coli at different concentrations of ZnO NPs and ZnO NPs-Cap (mg/ml).



It further appears that ZnO NPs at 30 mg/ml have a bactericidal effect on B. cereus. The viable count decreased to non-countable after 4 hours of growing; then, no growth occurred until the end of the growth period (24 h). At 30 mg/ml of ZnO NPs-Cap, the total viable count decreased to 2 1og 10 cfu/ml after 4 hours of growth and reached a steady state in number by the end of the period. At 15 mg, the total viable bacteria were about 3 log10 after 4 hours of growth and decreased to about 2 Log10 at the end of the growth period. At 7.5 mg/ml, a decrease was seen at 4 hours; the total viable count was 3.2 Log10, which decreased to about 2.5 Log 10 at the end of the growth period (24 h). At 3.125 mg/ml, the total viable count was about 4 Log10 after 4 hours, which decreased to 3 Log 10 at the end of the growth period (24 h). Thus, ZnO NPs-Cap also had a bactericidal effect against these kinds of bacteria, although the variability in the reduction of viability in the bacteria may be due to the spore-forming state that helps it resist abnormal growth conditions and adapt to circumstances.
Zhang et al. (2015) 49 assess the effect of ZnO NPs at different concentrations (0, 0.6, 3, 15, 60, 240, and 480 mg/ml) on the viability of E. coli (∼ 3x107 cfu/ml) and growth at 37°C in water for a time interval. They found that E. coli changed its viability compared with the control after 24 hours. ZnO NPs led to an obvious decline in the bacterial viability, with an increased concentration and longer time, proving that ZnO has an antibacterial activity. The reduction in viability of E. coli ranged between 1-log10 at a concentration of 0.6 mg/ml over 12–24 hours. As the concentration increased from 3 to 60 mg/ml, the viability of cells reduced to 4.5-log10 and was almost eliminated when the time was increased to 24 hours. A further increase was noticed at a concentration of ZnO NPs to 240 and 480 mg/L, which induced faster bacteria elimination after 12 hours of treatment.
4. Conclusions
It can be concluded that the XRD, UV-vis, and SEM analyses confirmed that both uncapped ZnONPs and ZnONPs-Cap were successfully synthesized, with an average crystallite sizes of approximately 23 nm and 19 nm, respectively. The antibacterial activity of both formulations appears to involve a common mechanism based on zinc ion interaction with the bacterial cell membrane, leading to membrane damage and subsequent cell death. This effect was enhanced in the ZnO NPs-Cap, suggesting that the biopolymer coating improves NP stability and interaction with bacterial cells. ZnO NPs-Cap demonstrated superior antibacterial performance compared to uncapped ZnO, particularly against the Gram-positive bacterium S. aureus and the Gram-negative bacteria E. coli and S. typhimurium. A concentration of 30 mg/ml of ZnO NPs-Cap resulted in rapid and effective bacterial inactivation, achieving nearly complete growth inhibition. The antibacterial efficacy of both ZnO NPs and ZnO NPs-Cap was strongly dependent on NPs concentration, exposure time, dispersion medium, and bacterial species. These findings highlight the potential application of Arabic gum–ZnO NPs as promising antimicrobial agents for food safety and biomedical use.
Footnotes
Acknowledgments
The authors would like to extend their sincere appreciation to the Ongoing Research Funding Program, (ORF-2026-589), King Saud University, Riyadh, Saudi Arabia, for supporting this work.
ORCID iDs
Author contributions
Declaration of conflicting interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data Availability Statement
Data will be made available on request.


