Abstract
The synthesis, optical properties, theoretical calculations, and antibacterial activities of a series of new red heterocyclic dyes derived from imidazo[1,2-a]pyridine are presented. 3-(Hydroxyimino)imidazo[1,2-a]pyridin-2(3H)-ylidene)-1-arylethanones are obtained from the reaction of 3-nitroimidazo[1,2-a]pyridine with substituted acetophenone derivatives in good yields (65%–72%). The structures are confirmed by spectral and analytical data, and the optical properties of the dyes are characterized by spectrophotometry. Density functional theory calculations are performed to provide the optimized geometries and relevant frontier orbitals. Calculated electronic absorption spectra are also obtained by the time-dependent density functional theory method. Moreover, the antibacterial activities (minimum inhibitory concentration) of the new dyes against Gram-positive and Gram-negative bacterial species are determined (minimum inhibitory concentration: 5–200 μg mL−1).

Introduction
Heterocyclic dyes have wide applications in the dyestuff industries.1,2 The use of these dyes in the electronic industry, for example, as colorimetric sensors, nonlinear optical (NLO) devices, and liquid crystalline displays (LCDs), has been investigated, while their use as potential sensitizers for photodynamic therapy (PDT) has attracted much attention. 3 In addition, they have been evaluated and employed as optoelectronic devices, 4 photoconductors, 5 sensitizers, 6 biomedical probes, 7 photo/catalysts, 8 for solar-energy utilizations, 9 and so on. Furthermore, heterocyclic dyes have been widely used in the preparation of disperse dyes with outstanding dischargeability on cellulose acetate. 10
Imidazo[1,2-a]pyridines, as an important type of bicyclic nitrogen heterocycle, have been used for the production of dyes and some valuable drugs. They exhibit interesting biological properties such as antiviral, 11 anticancer, 12 anxiolytic, 13 antimalarial, 14 hypnotic, 15 antiprotozoal, 16 and anti-inflammatory 17 activities, which has rendered this ring system an attractive target. Recently, imidazo[1,2-a]pyridine scaffolds became of interest as dye and fluorescent compounds.18–20 In some case, imidazo[1,2-a]pyridines with suitable functionalization can use as acid–base indicators. 21
Based on these aspects and in continuation of our previous studies on the synthesis of new heterocyclic dyes,18–23 we have synthesized three new heterocyclic dyes in good yields from the reaction of 3-nitroimidazo[1,2-a]pyridine with substituted acetophenone derivatives in a basic medium. The optical properties, density functional theory (DFT)/time-dependent density functional theory (TD-DFT) calculations, and antibacterial activity of the dyes have also been examined.
Results and discussion
Synthesis and structures of the dyes 3a–c
In order to the synthesis new dyes, the precursor 3-nitroimidazo[1,2-a]pyridine (

Synthesis of new dyes
The structural assignments of compounds
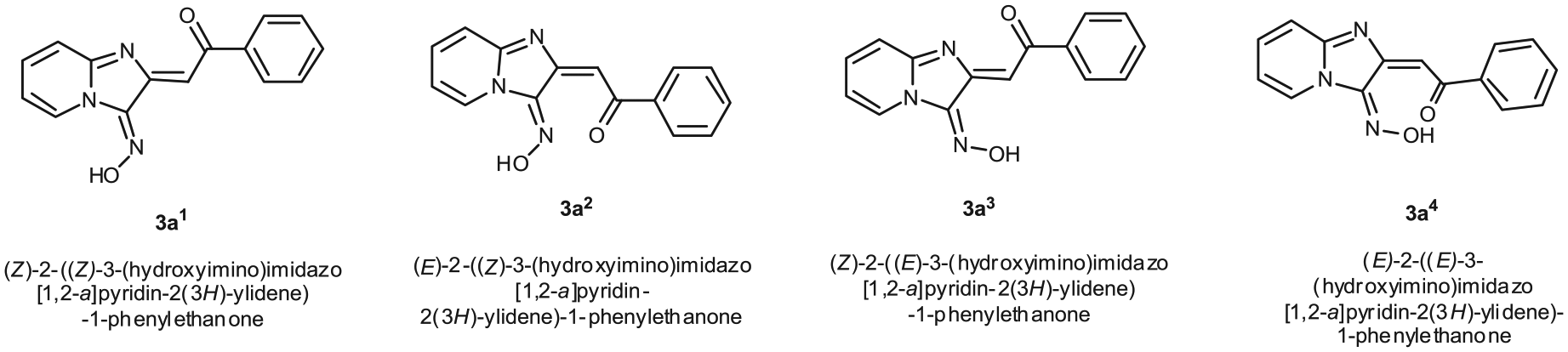
Structures of the isomers of

A plausible reaction mechanism for the formation of
We made a several attempts to isolate the isomers by classical methods, but they could not be isolated from each other. This implies that the isomers can convert into each other. Recently, we performed DFT calculations by using the B3LYP hybrid functional and the 6-311++G(d,p) basis set to determine the most reasonable mechanism for the formation of 3H-imidazo[4,5-a]acridines.
27
According to these results, conversion of the isomers into each other can be facilitated by their tautomerization into intermediate

Optimized geometries of the isomers of
Energies (kJ mol−1) of the isomers of
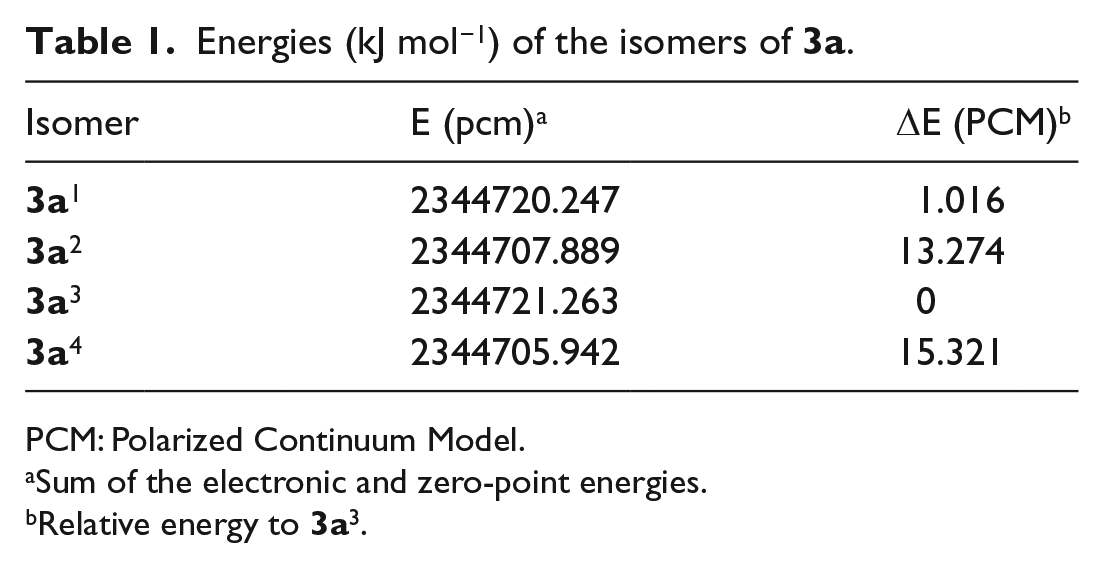
PCM: Polarized Continuum Model.
Sum of the electronic and zero-point energies.
Relative energy to
The data from the NOESY experiment on compound
The percentage of isomers can be easily obtained from integrations. Similarly, the percentage of each isomer in compounds
The detailed results of each reaction including the yields and percentage of isomers in compounds

Optical properties
The new dyes

Visible absorption spectra of dyes
Spectroscopic data for
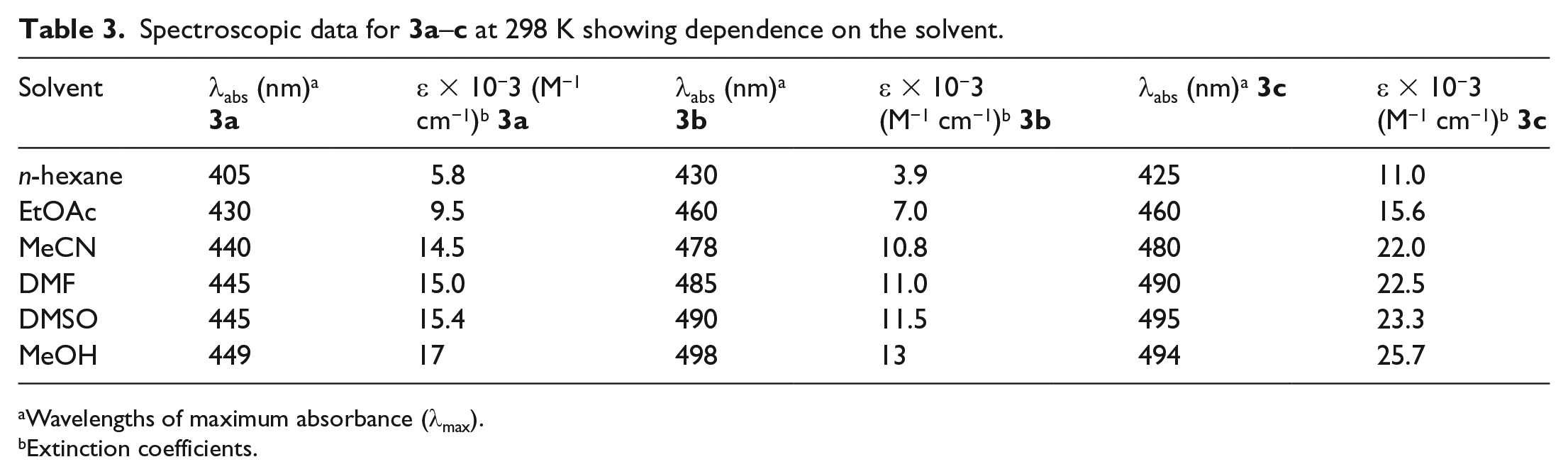
Wavelengths of maximum absorbance (λmax).
Extinction coefficients.
The absorption spectra of dyes
Antibacterial activity (MIC, μg mL−1) of references and compounds

Staphylococcus aureus (methicillin-resistant S. aureus).
Bacillus subtilis.
Pseudomonas aeruginosa.
Escherichia coli.
The color intensity of the dyes

Some mesomeric structures of
The energy difference between the HOMO and LUMO frontier orbitals is one of the important characteristics of molecules, which has a determining role in such cases as electric properties, electronic spectra, and photochemical reactions. The HOMO and LUMO maps of

The HOMO (bottom) and LUMO (up) frontier orbitals of the dyes
The calculated electronic absorption spectra were also obtained by the time-dependent density functional theory (TD-DFT) method. The TD-DFT electronic spectra calculations on of
The calculated electronic absorption spectra of non-isolable isomers

The calculated electronic absorption spectra of isomers
Antibacterial studies
The antibacterial activity of new dyes
As shown in Table 4, compounds
Conclusion
In conclusion, we have synthesized three new red dyes in good yields via nucleophilic substitution of the hydrogen of 3-nitroimidazo[1,2-a]pyridine with different acetophenone derivatives in basic methanol solution. The 1H NMR spectra of the products revealed that the dyes exist as four non-isolable isomers. Although data from NOESY experiments did not allow assignment of the OH groups of the four isomers, the DFT-calculated chemical shifts of compounds
Further investigations into the scope and application of these new dyes are in progress and will be reported soon.
Experimental
Materials
All reagents and solvents used in this work were purchased from Merck. The microorganisms Bacillus subtilis ATCC 6633, Pseudomonas aeruginosa ATCC 27853, and Escherichia coli ATCC 25922 were purchased from the Pasteur Institute of Iran and MRSA was isolated from different specimens which were referred to the Microbiological Laboratory of the Ghaem Hospital of the Medical University of Mashhad, Iran, and its methicillin resistance was tested according to the national committee for clinical laboratory standards (NCCLS) guidelines.
32
All solvents were dried according to standard procedures. Compound
Equipment
Absorption spectra were recorded on a Varian Cary 50-bio UV-Vis spectrophotometer. UV-Vis scans were recorded from 200 to 1000 nm. Melting points were measured on an Electro thermal type-9100 melting-point apparatus. The IR (as KBr disks) spectra were obtained on a Tensor 27 spectrometer and only noteworthy absorptions are listed. The 13C NMR (100 MHz), 1H NMR (400 MHz) and NOESY spectra were recorded on a Bruker Avance DRX-400 Fouriertransform spectrometer in DMSO-d6. Chemical shifts are reported in parts per million downfield from tetramethylsilane (TMS) as the internal standard; coupling constants (J) are given in hertz. The mass spectra were recorded on a Varian Mat, CH-7 at 70 eV. Elemental analysis was performed on a Thermo Finnigan Flash EA microanalyzer. All measurements were carried out at room temperature.
Computational methods
The DFT calculations were performed with the Gaussian 98 software package 33 by using the B3LYP hybrid functional 34 and the 6-311++G (d,p) basis set. The geometries of the compounds were fully optimized in MeOH solution.
Here, one of self-consistent reaction field methods, the sophisticated Polarized Continuum Model (PCM) 35 was used for investigation of the solvent effects. The PCM calculations were performed in MeOH, and zero-point corrections were considered to obtain the energies. Based on the optimized geometries and using time-dependent density functional theory (TD-DFT)36–38 methods, the electronic spectra of the compounds were predicted.
The 1H NMR chemical shifts of the compounds were predicted with respect to TMS. Here, the gauge-independent atomic orbital method (GIAO) was used for prediction of DFT nuclear shielding. 39
Synthesis of 3a –c from 1 and 2a–c
3-Nitroimidazo[1,2-a]pyridine (
3-(Hydroxyimino)imidazo[1,2-a]pyridin-2(3H)-ylidene-1-phenylethanone
1-(4-Bromophenyl)-2-(3-(hydroxyimino)imidazo[1,2-a]pyridin-2(3H)-ylidene)ethanone
3-(Hydroxyimino)imidazo[1,2-a]pyridin-2(3H)-ylidene)-1-(4-nitrophenyl)ethanone
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We express our sincere gratitude to the Research Office, Mashhad Branch, Islamic Azad University, Mashhad, Iran, for financial support of this work.
