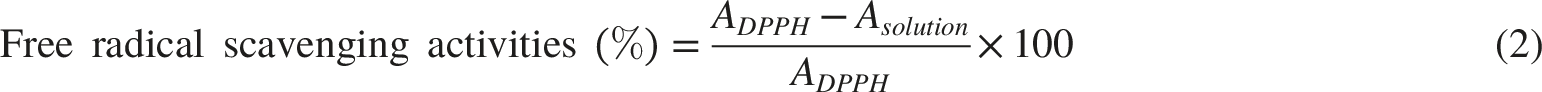Abstract
Lignin, a renewable aromatic polymer found in lignocellulosic biomass, has several potential uses due to its antioxidant, antibacterial, and UV-absorbing qualities. The aim of this work was to extract soda lignin for sunscreen, antioxidant, and antibacterial properties from Oxytenanthera abyssinic (Ethiopian lowland bamboo) stems using an alkaline treatment. A 1, 4-dioxane-based nanoprecipitation technique was then used to create lignin nanoparticles produced from O. abyssinica (OA-LNPs) via a self-assembly process. The extraction procedure was optimized with temperature (23–120°C), time (1–24 hours) and NaOH concentration (5–15%). 1, 4-Dioxane-based nanoprecipitation was used to synthesis OA-LNPs which were then examined using SEM, EDX UV-Vis, FTIR and 1HNMR for morphology size zeta potential and chemical structure. Antioxidant activity using DPPH assays, sun protection factor (SPF) measurements in commercial lotions and antibacterial testing against Gram-positive (S. aureus and S. epidermidis) and Gram-negative (E. coli and K. pneumoniae) strains were among the functional evaluations. Up to 294 mg/g of soda lignin were produced by the extraction. OA-LNPs had a spherical shape size ranging from 65 to 135 nm (mean 115 nm) a zeta potential of -30. 1 mV and no sulfur was found. They also retained phenolic and aromatic functionalities. The antioxidant activity of OA-LNPs was 11. 47 µg/mL which was higher than that of soda lignin (131.1 µg/mL). Due to UV absorption by phenolic groups SPF evaluations showed improvements in commercial lotions up to 114.8±1.3 when OA-LNPs were added. Antibacterial testing showed moderate efficacy against the tested strains with inhibition zones ranging from 12.43± 0.90 to 28.8± 1.11 mm. With better bioavailability and multifunctionality than Soda lignin (SL) these results highlight the environmentally friendly valorization of lignin from underutilized biomass and position OA-LNPs as promising candidates for sustainable applications in food packaging cosmetics and biomedicine.
1. Introduction
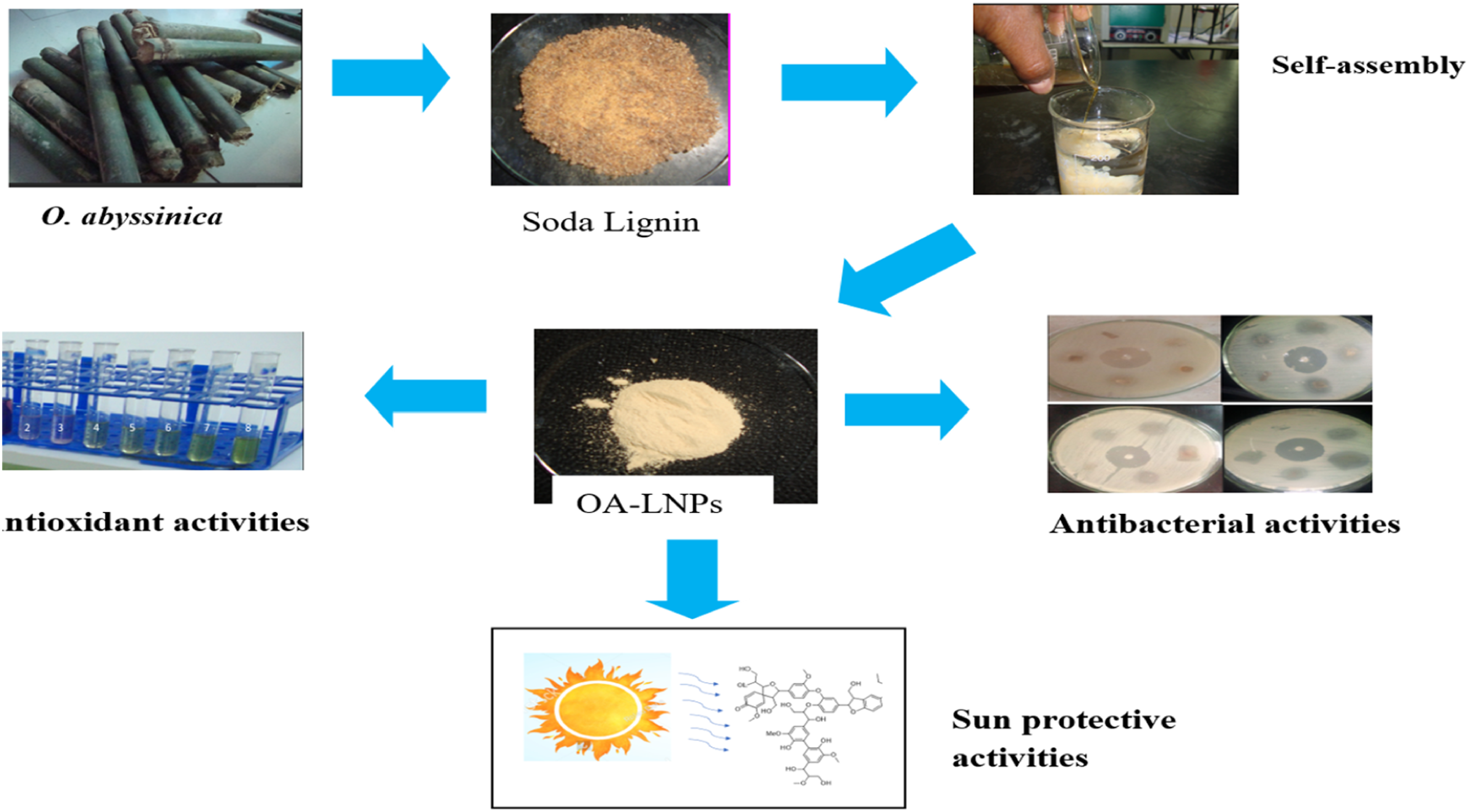
Lignin, a complex polyphenolic polymer abundant in plant cell walls, represents one of the most plentiful renewable resources on Earth, accounting for up to 30% of the organic carbon in the biosphere. 1 Because of its antioxidant, antimicrobial and UV-absorbing qualities lignin—which was once thought of as a waste byproduct of the pulp and paper industry—has drawn more attention for its potential in sustainable materials science. 2 The extraction and valorization of lignin from non-woody sources, such as bamboo, offer promising avenues for developing eco-friendly nanomaterials. 3 Due to its high lignin content and quick regeneration Oxytenanthera abyssinica a fast-growing bamboo species native to Africa that is widely grown for its versatility in construction and crafts makes an excellent feedstock for lignin 4 . The goal of this research is to extract lignin from O. abyssinica to create nanoparticles meeting the need for biodegradable substitutes for use worldwide.
By utilizing lignins amphiphilic properties to create stable nanostructures without the need for harsh chemical modifications the synthesis of lignin nanoparticles (LNPs) via self-assembly techniques has become a viable and efficient method. 5 Self-assembly typically involves dissolving lignin in a suitable solvent followed by controlled precipitation or solvent exchange, resulting in nanoparticles with tunable sizes and morphologies. 6 Because of its capacity to promote uniform dissolution and subsequent aggregation into nanoparticles 1, 4-dioxane a cyclic ether with low toxicity and high solvency for lignin has been used more frequently among different solvents. 7 Compared to traditional nanoparticle synthesis methods that depend on hazardous reagents self-assembly techniques minimize environmental impact while preserving the intrinsic functional groups of lignin. 8 By employing 1,4-dioxane in the self-assembly of LNPs from O. abyssinica lignin, this research aims to optimize a scalable, solvent-based process that yields monodisperse nanoparticles suitable for multifunctional applications.
The resultant LNPs are perfect for a variety of applications such as wound dressings, food packaging and personal care items because they have three desirable qualities: antibacterial antioxidant and sunscreen functionalities. 1 The phenolic structures in lignin provide a natural substitute for synthetic antibiotics in the face of growing antimicrobial resistance by rupturing microbial cell membranes and preventing the formation of biofilms. 9 The ability of lignins hydroxyl and methoxy groups to scavenge radicals gives it antioxidant qualities that shield biological systems from oxidative stress. 10 Furthermore LNPs are positioned as environmentally friendly UV blockers because the UV-absorbing chromophores in lignin filter harmful UV radiation offering effective sunscreen protection. 11 These diverse characteristics highlight Os potential. LNPs produced from abyssinica to support long-term environmental and health solutions.
The majority of lignin nanoparticle (LNP) preparation methods rely on costly or hazardous solvents like THF, DMSO and DMF which results in low yields, low solubility and uneven nanoparticle distribution. 12 Dioxanes polar aprotic nature effectively dissolves lignin into homogeneous solutions resulting in nanoparticles with narrow size distributions and high monodispersed through solvent-shifting techniques that precipitate uniform spheres.12,13 Additionally the method produces high yields of up to 80–90% without the use of harsh chemicals making it economical for industrial scale-up. 14 Additionally, because lignin is hydrophobic, LNPs made in this manner have better colloidal stability in aqueous media and can be functionalized with surfactants for biomedical applications like antioxidant carriers or cancer treatments.
This study investigates the self-assembly synthesis of lignin nanoparticles from O. abyssinica using 1,4-dioxane, with a focus on characterizing their physicochemical properties and evaluating their antibacterial, antioxidant, and sunscreen efficacies. By optimizing synthesis parameters such as solvent concentration, temperature, and lignin-to-solvent ratios, we aim to produce LNPs with enhanced stability and bioactivity. The novelty lies in utilizing an underutilized bamboo species as a lignin source and demonstrating the versatility of 1,4-dioxane-mediated self-assembly for multifunctional nanomaterials. Ultimately, this work seeks to bridge the gap between lignin’s abundance and its practical application, promoting a circular economy through the valorization of agricultural residues into high-value products.
2. Materials and methods
2.1. Plant collection
Three years aged O. abyssinica plants was obtained from Pawe woreda, which is found in Metekel zone of Benishangul-Gumuz regional state, west part of Ethiopia. The place was found 1120 meters above sea level with latitude of 11°12′ and 11°21′ and longitude 36°20′ and 36°32′ longitude. Plants materials were selected randomly based on TAPPI standards 2002. 15 The collection site was on land that is neither protected, private, nor indigenous. Appropriate prior informed consent was secured from relevant stakeholders. On the basis of a letter provide from Debre Tabor University, a permit was acquired from the stakeholders to collect the plant materials. The plant material was authenticated by Mr. Wogayew from the Department of Biology and Biodiversity Management, College of Natural Sciences, Addis Ababa University (AAU), using standard taxonomic keys from the Flora of Ethiopia and Eritrea and by comparison with authenticated reference specimens. A voucher specimen was deposited at the AAU Herbarium under voucher number 4271. The use of plant materials in the present study complies with all applicable institutional, national, and international regulations, including the guidelines of the International Union for Conservation of Nature (IUCN). 16
2.2. Sample preparation
Chips of the plants are produced from the stem part of the O. abyssinica using a grinder machine and exposed for sun light more than two weeks to be dry. With the use of an electric grain mill, dried O. abyssinica chips were crushed, and then allowed to pass through a 40-mesh filter before being caught by a 60-mesh screen. Powdered raw O. abyssinica was placed at room temperature with plastic bag labeled as “ROA” for further experiments.
2.3. Dewaxing and aqueous sodium hydroxide treatment
Using Soxhlet extraction, dewaxing was performed by using 2:1 ratio combination of benzene to ethanol as solvent. Ten grams of the ROA were utilized for six hours to get rid of waxes, resins, and other extractives. To extract pectin, hemicellulose, and lignin as black liquor via vacuum filtration, dewaxed O. abyssinica was subjected to alkaline treatment under varying conditions to optimize lignin yield. Sodium hydroxide concentrations of 7%, 13%, and 20% were applied at temperatures of 23, 100, 110, and 120 °C for reaction times of 1, 2, 3, and 24 h, respectively. with a fiber to solvent ratio of 1:20. The collected black liquor was kept for the determination of total recovered lignin (OASL). While the pretreated O. abyssinica fiber (OAF) was air-dried at room temperature (25 °C) and washed with tap water until the pH of the final washing water reached 6 to 7. 17
2.4. Separation of soda Lignin from O. abyssinica
Lignin was isolated from the black liquor of an alkali pretreatment that was optimized to reduce the lignin content in the solid or increase the lignin content in filtrate. By adding H2SO4 at a weight-to-weight ratio of 98% to the alkaline liquor, the pH was reduced to 2 (Figure 1). Sulfuric acid solution (0.2 mol/L) was used to adjust the pH of the filtrate to 2-3 under vigorous agitation (700 rpm) in a magnetic stirrer for 15 minutes at room temperature.
18
In the system, a precipitation was seen to form and the color changed from black to brown. The residual solid was rinsed with deionized water till pH 6-7, collected in Petri dishes, and dried at 60 °C for 12 hours in the oven after the suspension had been filtered under vacuum. Total recovered lignin (SL) was calculated according to the formula given in equation (1).
19
Three kinds of lotions collected from Debre Tabor town cosmetics shops after diluted with water (green solvent).
2.5. Preparation of OA- LNPs using 1,4-dioxane nanoprecipitation method
For 1,4-dioxane nanoprecipitation method, we used the methods used by Zhang et al. (2013) and Tian et al. (2015)20,21 with some modification. In a Soxhlet extractor, the soda lignin was purified using a 9:1 v/v solution of 1,4-dioxane and water. The dark, clear solution was centrifuged after 2 hours of refluxing, and then it was added to ice water with a pH of 2.0 to cause precipitation. The precipitate was centrifuged, after which it was twice rinsed with ethanol and three times with deionized water. After freeze drying lignin nanoparticle (OA-LNPs) was obtained.
2.6. Testing and characterization
The characterization of OA-LNPs involved multiple analytical techniques to assess their physical, chemical, and morphological properties. Dynamic light scattering (DLS) was employed using a Malvern Zetasizer Nano apparatus to determine the average hydrodynamic diameter and zeta-potential, with samples diluted in deionized water to a concentration of 500 µg/mL. UV-vis spectroscopy was conducted on a Shimadzu UV-160 spectrophotometer, adapting a method from Bu et al. (2011), where 5 mg of OA-LNPs were dissolved in 10 mL of 0.1 mol/L NaOH, followed by diluting a 1 mL aliquot to 10 mL with deionized water, and measuring absorbance from 200 to 800 nm. OA-LNP production was examined using Fourier Transform Infrared (FTIR) spectroscopy which scanned from 4000 to 500 cm-1 on KBr pellets using an Agilent Cary 660 instrument with 4 cm-1 resolution. After drying diluted suspensions on silicon slices scanning electron microscopy (SEM) with energy-dispersive X-ray (EDX) analysis was carried out at 2. 0 kV accelerating voltage and room temperature to investigate morphologies and elemental composition. Additionally, proton nuclear magnetic resonance (1H NMR) spectra were acquired using a Bruker Avance III 500 MHz spectrometer, dissolving approximately 10 mg of purified OA-LNPs powder in 0.7 mL of DMSO-d6, with a 90° pulse flip angle, 2 s acquisition time, and measurements at 27.5°C to elucidate the chemical structure.
2.7. Antioxidant activities of OA-LNPs
The antioxidant activities of OA-LNPs were performed based on DPPH radical scavenging activities. OA-LNPs ability to scavenge free radicals was ascertained using the stable radical DPPH technique. 1 mL of OA-LNPs at different concentrations of 25.0, 50.0, 75.0, 100.0, and 125.0 µg/mL was mixed with 3 mL of newly made DPPH (0.1 mM in methanol) solution and carefully vortexed. After that, the mixture sat at room temperature (23 oC) in the dark for 30 munities. The absorbance of the samples was measured using UV-Vis spectrophotometer at 517 nm. Methanol was used as the blank solution, and DPPH was utilized as the control. Ascorbic acid (AA) is being used as a positive control. Equation (2) was performed to obtain the percentage of inhibition for the free radical scavenging activity. The antiradical activity is measured by calculating the percentage inhibition of DPPH radical.
ADPPH and Asolution denote DPPH absorbance at 517 nm before and after adding OA-LNPs.
2.8. Determination of sun protective factors
Three widely used and commercially available human hand and body lotions, including Lotion A (LA), Lotion B (LB), and Lotion C (LC) (Figure 1) purchased from a local market in Debre Tabor, Ethiopia. For determination of SPF, 0.2 g of each LA, LB and LC lotion was added separately in a 100-mL volumetric flask, diluted to 15 mL with green solvent (water). Then, from these flasks, 2 mL of the lotions were taken and mix with OA-LNPs (100 µg/mL) to form a OA-LNPs-LA, OA-LNPs-LB and OA-LNPs-LC formulated lotion. The OA-LNPs, and the commercially available cosmetics (lotions) without OA-LNPs were used as a control. Cotton was used to filter the final solution. Following a 15-minute incubation period, the solution was measured for absorbance in the wavelength range of 290 - 320 nm at 5 nm intervals. The Mansur equation Equation (3) was then used. Triple-checked experiments and the resulting values were utilized to compute SPF.
The Mansur equation is used to determine the SPF of different formulations. Sayre et al. (1989)
22
developed the equation as follows:
2.9. Antimicrobial activity
Testing for antibiotic susceptibility involves the use of Kirby-Bauer disk diffusion methods. The swab was streaked over a sterile agar surface to inoculate different Gram-positive (S. aureus and K. pneumonia) and Gram-negative (E. coli and P. aeruginosa) bacteria. Whatman No. 1 sterile disks, measuring 6 mm, were dipped in plant-mediated OA-LNPs-RVFE. The antibacterial properties of OA-LNPs-RVFE and the plant’s crude extracts were compared. These disks will be sterilely air-dried before being put onto the MHA (Mueller Hinton Agar plates) seeded top layer and cultured for twenty-four hours at 37 °C. diameters (mm) of the bacterial inhibitory zones less the reported disk diameter. 23
2.10. Statistical analysis
The values were expressed using the means ± standard error of the means. Using SPSS/20 software, a one-way analysis of variance was employed to do a statistical analysis of the data. A p-value of less than 0.5 was considered statistically significant.
3. Result and discussion
3.1. Delignification process
Delignification of O. abyssinica was achieved using two separate treatments. Wax, resin, and extractives were removed using a soxhlet extraction process in the first step. In the second step alkali treatment was carried out to prepare the black liquor. The cellulosic fibers are released during this process, while the majority of the lignin and degradation byproducts from many of the hemicelluloses that were originally present in the biomass are typically incinerated in a recovery process to create the black liquor. 24 The black liquor contain a mixture of hemicellulose, inorganic chemical, and lignin residues. 25
3.2. Lignin extraction via aqueous NaOH treatment
The effect of concentration of NaOH, temperature and reaction time on the value of soda lignin.

The principal light-absorbing components of lignin are unsaturated functional groups, which give the substance brownish to black color. Aromatic rings, conjugated carbonyl groups and carbon-carbon double bonds, are some of these groups.26,27 The black liquor’s color was changed as a result of the lignin removal. Figure 2 shows that the liquid was initially dark brown (pH = 13.1) but became clear as a result of the pH lowering. The liquor’s color changed to a light yellow at the lowest pH level (2.0). The chromophoric functional groups such as phenolic hydroxyl, hydroperoxy radicals, carboxylic acids, carbonyl groups, and quinones, among others, produced during the decomposition of lignin, give the black liquor its dark color.
28
The isolation of soda lignin using H2SO4 at different pH value and preparation of OA-LNPs using self-assembly process.
Using the ultrasonication method, OA-LNPs (Figure 2) were synthesized in 1,4-dioxane as the solvent, followed by a self-assembly process induced by the gradual addition of water as an antisolvent. Ultrasonication facilitated the uniform dispersion and breakdown of lignin aggregates, promoting the formation of nanosized particles. The introduction of water reduced the solubility of lignin, triggering self-assembly through intermolecular interactions and resulting in stable, well-defined lignin nanoparticles.
3.3. Zeta-potential of lignin particles
The electrostatic interaction between the particles was determined by their zeta-potential, which measures the surface charge of the particles.
29
This reveals important facts regarding a colloidal stability of particles, and is very important when studying nanoparticles preparation.
30
High absolute value Zeta-potential measurements show that the particles have enough electrical double layer repulsion between the particles, which prevents them from aggregating.
31
LNPs often have negative zeta-potential values because of the negatively charged phenols and, to some extent, the adsorption of hydroxyl ions.31–33 The relatively high negative zeta potential keeps the particles from sticking together and creates strong electrical double-layer repulsion. This lets the nanoparticles’ zeta potential signal the electrical surface properties of the particle. So there was no particular aggregation, and the lignin nanoparticle dispersion was very stable in pure water.
34
Figure 3 depicts the average zeta potential of LNPs. At a pH of 2.01, LNPs had the highest absolute value zeta potential (-30.1 mV). This is because lignin nanoparticles of smaller sizes contain more negatively charged phenolic compounds. The current studies absolute value of LNPs zeta potential was higher than the values discovered by Matsakas et al. (2020).
35
They discovered that the Birch and Spruce samples had zeta potential values between -14. 0 and -31. 6 mV and -11. 5 and -31. 4 mV respectively. In the work of Yaqoob et al. (2021),
36
a lower absolute value zeta potential (-25.0 to -32.0 mV) was also found. Salami et al. (2017)
37
produced well-dispersed lignin nanoparticles with a zeta potential of -31. 6 mV which is a similar absolute value. Zeta potential value of LNPs.
3.5. Scanning electron microscopy (SEM) analysis
Scanning electron microscopy (SEM) was used to analyze the surface morphologies of lignin nanoparticles (OA-LNPs) representative images are shown in Figure 4(a) and (b). The overall shape and distribution of the LNPs were confirmed by these SEM images which also showed their morphological structures. In particular the particle size distribution as a function of nanoparticle percentage is shown in Figure 4(b) which demonstrates the spherical morphology of the LNPs. With a mean size of roughly 115 nm the particle sizes varied from a minimum of 65 nm to a maximum of 135. Because it encourages greater colloidal stability higher surface area-to-volume ratios and improved dispersibility in a variety of media—all crucial for applications in drug delivery composites and environmental remediation—this spherical shape is beneficial. Because of carefully regulated synthesis conditions that reduce aggregation and improve performance in specific applications the size range seen here is comparatively narrow and monodisperse. SEM image of four different OA-LNPS (a) and their size distribution (b).
These results both agree with and deviate from the body of existing literature when compared to other studies frequently because of different preparation techniques. For example, Li et al. (2016) 38 used nanoprecipitation to create spherical LNPs with sizes ranging from 10 to 100 nm which is smaller than what we found because nanoprecipitation involves quick solvent shifts that favor nucleation over growth resulting in finer particles with less aggregation. Tian et al. (2017) 39 used a dialysis technique with dimethyl sulfoxide (DMSO) as the solvent to create spherical LNPs about 200 nm in size. The slower diffusion rates during dialysis which permit particle coalescence and growth led to larger particles. As an alternative Lievonen et al. (2016) 33 used a solvent exchange method to create LNPs with irregular shapes between 80 and 360 nm. This method can introduce polydispersity and non-uniform shapes by encouraging uneven precipitation and aggregation during phase separation. In a similar finding Wang et al. (2020), 40 purified lignin using self-assemble to produced spherical nanoparticles when certain conditions are met such as the right pH and solvent polarity which promote hydrophobic interactions and reduce surface energy producing uniform spherical forms that are preferred for consistent functional properties.
3.6. Energy-dispersive X-ray spectroscopy (EDX)
As seen in Figure 5 additional elemental analysis was carried out using energy-dispersive X-ray spectroscopy (EDX). The EDX profile confirmed the organic polymeric nature of lignin derived from plant biomass by showing the presence of carbon (65.4 %) and oxygen (34. 6%) with a small oxygen peak (probably a trace element or artifact). Crucially Figure 5 data showed that the synthesized LNPs are sulfur-free which is a major advantage because sulfur contaminants—typically from sulfite pulping processes—can impair biodegradability introduce toxicity and compromise thermal stability. According to Iravani et al. (2020)
14
the lack of sulfur makes these LNPs more suitable for high-value applications like bioplastics and composites where purity guarantees better mechanical properties environmental safety and compatibility with other materials. Additionally, by eliminating the need for extra purification processes and supporting environmentally friendly manufacturing objectives this sulfur-free composition promotes sustainable uses. EDX peaks for four OA-LNPs.
3.7. Ultraviolet-visible (UV-vis) spectrophotometry
The UV-Vis absorbance of four LNPS extracted from O. abyssinica was conducted using UV- vis instrument (UV-160, Shimadzu Co., Kyoto, Japan) at a wave length range of 200 to 800 nm. For checking the presence of pure lignin nanoparticles, a wavelength of 280 nm could be used due to its excessive number of phenols. As seen on Figure 6, LNPS samples had maximum absorption at around 280 nm which indicated the presence of aromatic rings/non-conjugated phenolic groups in the lignin structure.41,42 The UV spectra of LNPs, at 281 nm corresponding to the π→π* electronic transition in the aromatic ring of unconjugated phenolic groups or guaiacyl (G) units. Moreover, absorption at this wavelength is also indicative of free and etherified hydroxyl group.
43
Absorption at wavelength 215 nm (Figure 6) indicates the π→π* electronic transition in the aromatic ring. The absorption band above 300nm indicates the presence of conjugated structure present with the aromatic moieties.44,45 The broad shoulder band of the samples at the maximum wavelength of about 360 nm is due to the associated hydroxycinnamic acids (such as ferulic and p-coumaric acids).
46
UV-vis absorbance peaks of four different OA-LNPs.
3.8. Fourier transform infrared (FTIR)
Fourier Transform Infrared (FTIR) spectroscopy identifies functional groups in materials by detecting vibrational frequencies of chemical bonds, typically reported in wavenumbers (cm-1). Lignin, a complex aromatic polymer from plant cell walls, exhibits characteristic peaks related to its phenolic, aliphatic, and carbonyl components. Figure 7 showed the FT-IR spectra of OA-LNPs. The spectrum clearly indicated the presence of various functional groups such as aromatic skeletal vibrations due to guaiacyl group, carbonyl stretching-unconjugated ketone, aromatic C-H in plane deformation and aromatic C-H out of plane, hydrogen bonded, and carboxyl group, etc. The hydroxyl groups in phenolic and aliphatic structures are responsible for the broad band at 3410-3460 cm-1, and the bands that center around 2936 and 2838 cm-1 are primarily caused by CH stretching in aromatic methoxyl groups as well as in methyl and methylene groups of side chains. Carbonyl stretching unconjugated ketones and carbonyl groups are responsible for a small peak at 1698 cm-1. A sharp peak at 1594 cm-1, 1513 cm-1, and 1459 cm-1 represents aromatic skeletal vibrations. The vibrations characteristic of the guaiacyl unit (1226 cm-1) are visible in the spectra of OA-LNPs samples.47–49 The bands (syringyl and guaiacyl) shown at 1134 and 1119 cm-1 are caused by ether stretching.
50
FTIR Peak at 1100–1000 cm-1 associated with C-O-C stretching vibrations from ether linkages, particularly those in β-O-4 structures, which are common in lignin, as well as C-H bending modes from aliphatic groups. It often appears as a broad band in FTIR spectra. The broad, intense band at 3400–3200 cm-1 corresponds to O-H stretching from abundant phenolic and aliphatic hydroxyl groups, essential for reactivity and hydrogen bonding. Adjacent to this, the 3000–3100 cm-1 region shows C-H stretching in aromatic rings. Carbonyl groups from oxidation appear at 1700–1750 cm-1, potentially more pronounced in LNPs due to processing, influencing biodegradability and antioxidant capabilities, while their absence points to preserved lignin integrity. The core aromatic signatures at 1600–1500 cm-1, featuring sub-peaks around 1590 and 1505 cm-1 for C=C vibrations, allow differentiation of guaiacyl (G) and syringyl (S) units through intensity ratios, confirming aromatic integrity crucial for UV-blocking properties. Aliphatic contributions emerge at 1450–1400 cm-1 via C-H bending in methyl and methylene groups, often as a doublet that reflects lignin type and may diminish in processed LNPs from degradation. Phenolic and ether linkages dominate 1300–1200 cm-1, with syringyl peaks at ∼1320–1330 cm-1 and guaiacyl at ∼1260–1270 cm-1 revealing methoxylation levels. FT-IR spectra of OA-LNPs sample.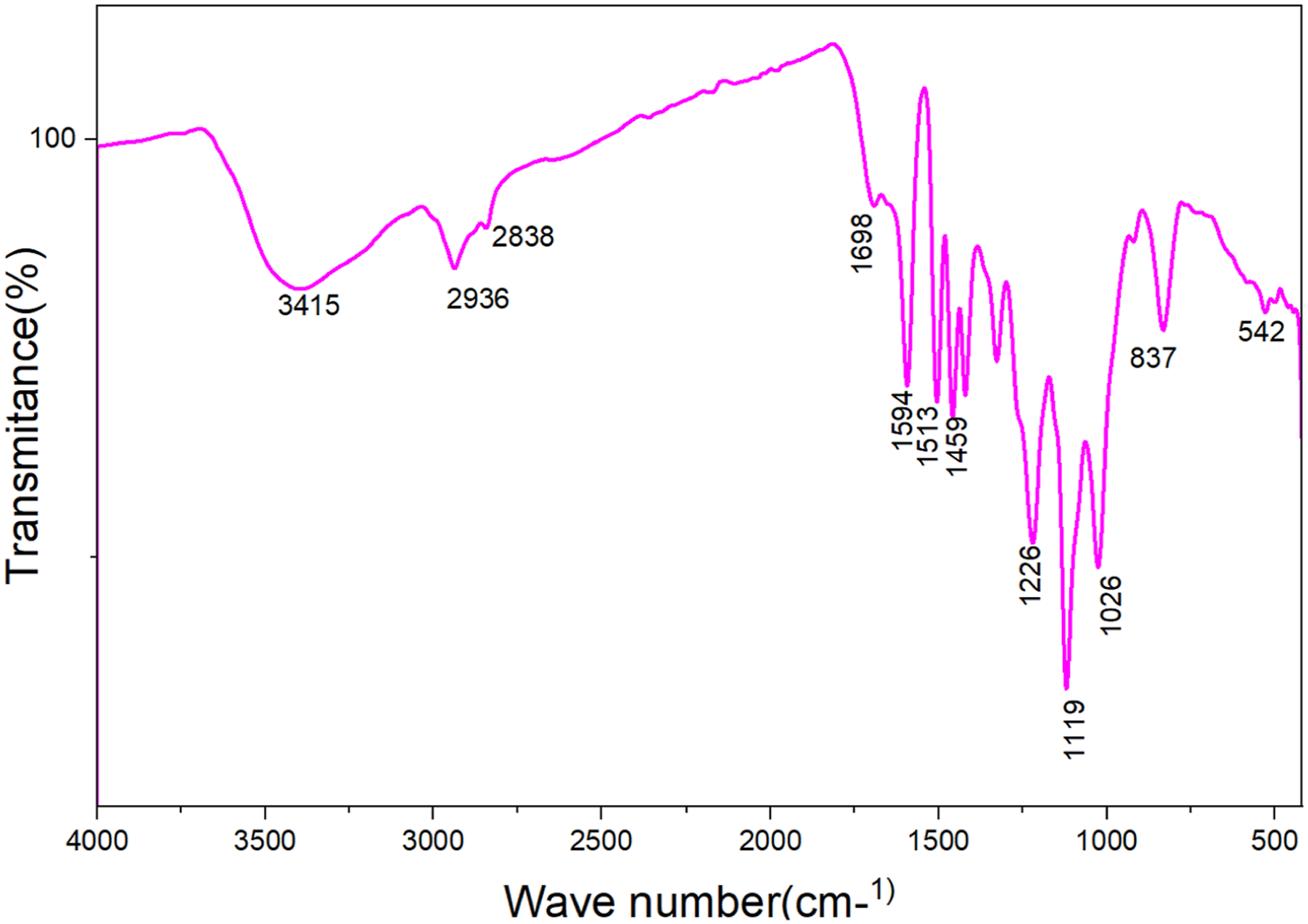
3.9. Proton nuclear magnetic resonance (1H NMR)
Figure 8 shows the 1H NMR spectrum of lignin nanoparticles (OA-LNPs). The peaks at 6.7 ppm and 6.9 ppm correspond to the G2 and G6 positions of guaiacyl units, respectively, which are prevalent in softwood lignin.
51
A broad signal around 7.3–7.6 ppm is attributed to p-hydroxyphenyl units in OA-LNPs.52,53 Signals from 6.2 to 7.5 ppm are associated with aromatic protons or phenolic hydroxyl groups. Predominant phenolic groups not conjugated with carbonyl groups appear in the range of δ 8.0–9.4. Specifically, large signals at δ 8.99 and 8.76 are assigned to phenolic groups in phenylcoumaran and arylglycerol β-O-4 structures, respectively.
54
Few aliphatic proton peaks are observed in the 4.2–5.6 ppm range. Peaks between 0.8 and 2 ppm indicate the aliphatic region, linked to various methyl and methylene groups (C–CH2–C, –CH3). Aliphatic and phenolic hydroxyl groups are represented by peaks at 1.6–2.1 ppm and 2.1–2.45 ppm, respectively.55,56 The aliphatic and phenolic hydroxyls, respectively, are represented as peaks at 1.6-2.1 ppm and 2.1-2.45 ppm, respectively.
56
The peak at 2.5 ppm is due to the DMSO-d6 solvent. Aliphatic hydroxyl groups appear in the 3.2–3.6 ppm range.57,58 Proton signals from 3.5 to 4.1 ppm arise from methoxyl groups (-OCH3) directly attached to the benzene ring, resulting from lignin degradation fragments.56,59 Peaks in the 3–4 ppm range relate to protons in ether linkages (C–O–C).55,59 FITR peaks of OA-LNPs.
3.10. Test sun protection activities
All year long regardless of the climate in which we live we must protect our skin even on overcast days. Anybodys skin can be harmed by UV radiation regardless of skin tone. Regular application of sunscreen is necessary. 60 Three ultraviolet (UV) wavelength ranges of electromagnetic radiation are emitted by the Sun. The three UV wave length radiations are UV-A, UV-B and UV-C. UV-A radiation at medium wavelengths (UV-A 290–320 nm) and longer wavelengths (UV-B 320–400 nm) reach the Earth’s surface while the atmosphere absorbs the shortest wavelengths (UV-C 100–290 nm). While UV-A penetrates the skin more deeply excessive UV-B exposure results in sunburn. 61 DNA damage and cancer are caused by both UV-A and UV-B. 62 Due to the presence of various chromophores and auxochromic groups lignin exhibits a wide absorption range in the UV region with a maximum absorbance at 283 nm. 63 The capacity of a sunscreen lotion to absorb light in the range of 290 to 400 nm helps shield skin from sunburn and other harmful effects. 64 Sunscreen functions by absorbing reflecting or scattering ultraviolet (UV) light in the 290–400 nm range which includes UV-A (320–400 nm associated with skin aging and deeper tissue damage) and UV-B (290–320 nm causing sunburn and DNA damage). But not all sunscreens block both to guarantee UV-A protection as well look for broad-spectrum labels. 65 The SPF is a figure that indicates how effective a sunscreen product by measuring its absorbance with the range. 64
The SPF values of All samples LA, LB, LC, AO-LNPs-LA, AO-LNPs-LB and AO-LNPs-LC fell between 5.05 and 114.8 (Figure 9). OA-LNPs-LC has a maximum absorbance and observed SPF value of 114.8 ± 1.3 that is higher than all samples. This is most likely because sample OA-LNPs-LC contains more sunscreen material overall than the other samples. Sample LB has fewer sunscreens (SPF = 4.13±1.6) than sample OA-LNPs-LB (SPF = 74.9 ± 0.9). Sample LA contains fewer sunscreens compared to OA-LNPs- LA (SPF = 102.8 ± 1.5). Sample LC contain fewer sunscreen potential than sample OA-LNPs- LC (SPF = 114.8 ±1.3). It was discovered that adding OA-LNPs to LA, LB, and LC increased by a value of 97.75, 70.75, and 108.32 respectively. The above data showed that, the presence of lignin in the form of nanoparticles increase the capacity of the three lotions to absorb sun light. This is because lignin contains a lot of aromatic phenolic structures which have powerful UV-absorbing chromophores. When lignin is transformed into nanoparticles the lotion matrixs surface area and dispersion are greatly increased enabling more efficient interaction with UV light.
11
Furthermore the conjugated aromatic rings π–π transitions in lignin nanoparticles effectively absorb UV-A and UV-B radiation enhancing the lotions overall sunscreen performance.
66
However, main characteristics of that make OA-LNPs excellent for sun screening are its smaller particle size.
67
As indicated in Figure 7, LNPs contains phenolic groups that strongly absorb UVB (280–320 nm) and UVA (320–400 nm) radiation, acting as a physical and chemical barrier. This absorption prevents UV photons from penetrating the skin, reducing erythema (sunburn) and DNA damage. Studies show LNPs can achieve SPF values of 10–50 or higher.
68
Exposure to UV generates reactive oxygen species (ROS) that cause oxidative stress and skin aging. Lignin’s polyphenolic structure scavenges free radicals, neutralizing ROS and protecting cellular components. This dual action (UV blocking + antioxidant defense) makes LNPs more effective than lignin alone, which has lower solubility and efficacy. In addition to this a smaller particle size work better for a UV absorber. Synthesized nanoparticles may be a preferable option for use. The SPF value of different samples of OA-LNPs.
3.11. Antioxidant activity of OA-LNPs
The antioxidant properties of the green-synthesized OA-LNPs was evaluated by measuring the decrease in DPPH absorbance at 517 nm, which reflects their capacity to scavenge free radicals generated by DPPH.
69
The observed shift from blue at the beginning to yellow at the end showed the nanoparticles’ antioxidant activities. In this study, the DPPH technique was used to measure the antioxidant activity of phytochemical-assisted biosynthesized OA-LNPs.
70
Methanol was used as a solvent, and DPPH was used as a free radical, which gave a violet color to methanol. When biosynthesized OA-LNPs and leaf extract are combined in an alcoholic solution, the violet color of DPPH is reduced; if the antioxidant property is high, then it changes to yellow after 30 minutes. Alcoholic solutions were prepared by combining ascorbic acid (AA), OA-LNPs and soda lignin (SL), with concentrations of OA-LNPs adjusted to 25, 50, 75, 100, and 125 µg/mL, respectively. The resulting solutions were then labeled as OA-LNPs1, OA-LNPs2, OA-LNPs3, OA-LNPs4, and OA-LNPs5, respectively (Figure 10(b)). An equal quantity of DPPH was then added for the same duration. The percentage inhibition of DPPH for ascorbic acid (AA), SL, and OA-LNPs samples at different concentrations is shown in Figure 10(a). The percentage inhibition for biosynthesized OA-LNPs were 50.6±2.2 for OA-LNPs1 and 94.7±1.8 for OA-LNPs5; however, for SL the percentage inhibition was 17.8±1.1 at 25 µg/mL and 54.6±0.21 at 100 µg/mL, which are less than those of OA-LNPs samples. According to Figure 10, the inhibition percentages for AA at comparable concentrations were 67.0±1.5 and 97.3±0.8, respectively. The results presented in Figure 10(a) clearly indicate that the antioxidant activity of OA-LNPs is concentration-dependent, with higher concentrations exhibiting greater activity. Lignin shows antioxidant activity due to the presence of the phenolic pharmacophore, which is able to scavenge reactive radical species with the formation of highly stabilized mesomeric forms.
10
The phenolic structure of lignin provides a readily available means of quenching radicals and yielding a stabilized phenoxy radical.
71
The high antioxidant properties of lignin nanoparticles can be explained by the higher density of phenolic and carboxylic groups on the surface of LNPs with respect to the native polymer,
72
as well as by the occurrence of favorable electron transfer processes between the ordered π-π stacked aromatic moieties.
73
Antioxidant power of OA-LNPs, SL and AA at (25, 50, 75, 100 and 125 μg/mL) (a) and colour change observed during DPPH free radical scavenging activities (b). Where 1= DPPH, 2= SL, 3 = AA, 4= OA-LNs5, 5= OA-LNPs4, 6= OA-LNPs3, 7= OA-NPs2 and 8 = OA-LNPs1 at 125 μg/mL.
As observed in Figure 10(b), the color intensity of different samples varies. DPPH exhibits a deep blue color after adding OA-LNPs, SL, and AA; the deep color of DPPH gradually fades and changes to yellow, as observed in Figure 10(b). This demonstrated that the DPPH free radicals were efficiently scavenged by the antioxidant compounds in OA-LNPs, SL and AA changing them into their non-radical yellow-colored form. Since electron donation from the antioxidants reduces DPPH radicals—a common mechanism in radical scavenging tests—this color shift signifies the presence of strong antioxidant activity. The degree of fading provides quantitative proof of the antioxidants capacity to inhibit free radicals and correlates with their potency.
Figure 11 shows the IC50 value for biosynthesized OA-LNPs samples. A lower IC50 value indicates better antioxidant activity against DPPH free radicals. Our results showed that soda lignin had an IC50 value of 131.1 μg/mL, which is significantly higher than the biosynthesized OA-LNPs, with an IC50 value of 11.47 μg/mL, however, the IC50 value for ascorbic acid was 4.6 μg/mL as shown in Figure 11. These results indicate that the biosynthesized OA-LNPs possess markedly enhanced antioxidant activity compared to soda lignin and exhibit antioxidant performance comparable to that of the standard antioxidant, ascorbic acid. Biosynthesized OA-LNPs have better antioxidant properties than soda lignin. This is because of Lignin nanoparticles have higher antioxidant activity than lignin because their nanoscale size results in a larger surface-area-to-volume ratio, which increases the accessibility of their antioxidant functional groups, particularly phenolic hydroxyl groups. This larger surface area allows for more interactions with free radicals, leading to enhanced antioxidant properties. In comparison, other studies a comparable IC50 value of 10.38 µg/mL was obtained from lignin nanoparticle derived from Egyptian cotton cultivar byproducts.
74
Furthermore, in DPPH scavenging tests lignin nanoparticles made from sugarcane bagasse showed an IC50 value of 28. 5 µg/mL suggesting a lower level of antioxidant activity than the biosynthesized OA-LNPs.
75
Furthermore, lignin nanoparticles prepared from rice straw showed an IC50 value of 45.2 µg/mL in similar antioxidant evaluations,
76
and those derived from wheat straw had an IC50 of 62.1 µg/mL (Kumar et al., 2021). These findings further highlight the enhanced antioxidant efficacy of OA-LNPs relative to other lignin-based nanomaterials. IC50 value for biosynthesized OA-LNPs.
3.12. Antibacterial activity
E. coli, K. pneumoniae, S. aureus and S. epidermidis are the four bacterial strains against which the antibacterial activities of OA-LNPs at four different concentrations—OA-LNPs1 OA-LNPs2 OA-LNPs3 and OA-LNPs4—are displayed in the table. The findings which were obtained using a disk diffusion assay are presented as mean inhibition zone diameters (in mm) with standard deviations. Tetracycline (TTC) a common antibiotic used as a control exhibits consistent 30 mm zones in every bacterium suggesting high baseline activity (Figure 12 and Table 3). Disc diffusion methods for evaluating the antibacterial activities of OA-LNPs1, OA-LNPs2, OA-LNPs3, and OA-LNPs4 against E. coli, K. pneumonia, S. aureus, and S. epidermidis. Antibacterial activities of lignin nanoparticles (OA-LNPs1, OA-LNPs2, OA-LNPs3, and OA-LNPs4) against four different bacteria species E. coli, K. pneumonia, S. aureus, S. epidermidis. The mean value ± SD was utilized after more than three examinations of each data set. Subscriptions a and b have values in the same column that differ significantly (P ≤ 0.05).

All lignin nanoparticles (LNPs) demonstrate antibacterial activity, with inhibition zones varying from 12.43 mm (OA-LNPs1 against S. aureus) to 28.80 mm (OA-LNPs3 against K. pneumoniae), though these zones are generally smaller than those of the tetracycline control (TTC), indicating comparatively lower potency. E. coli, inhibition zones are consistently moderate across all OA-LNPs (16.20–19.81 mm). For K. pneumoniae, OA-LNPs1 exhibits lower activity (17.43 mm), while OA-LNPs2, OA-LNPs3 and OA-LNPs4 show significantly higher zones in rage of 25.35 to 28.80 mm with minimal variation among them. S. aureus susceptibility is lowest with OA-LNPs1 (12.43 mm), but progressively increases with OA-LNPs2, LNPs3 and LNPs4 with value in the range between 19.11 to 26.71 mm, hinting at enhanced potency through variant modifications. Similarly, against S. epidermidis, OA-LNPs1 and OA-LNPs2 yield comparable, lower zones (17.10–17.55 mm), whereas OA-LNPs3 and OA-LNPs4 are more effective (21.88–23.94 mm), with OA-LNPs4 demonstrating the strongest overall performance.
In terms of antibacterial potency and spectrum, the reported inhibition zones of 12–29 mm against Gram-negative (E. coli, K. pneumoniae) and Gram-positive (S. aureus, S. epidermidis) bacteria, though less effective than TTC, mirror moderate results from 77 (zones of 10–15 mm via solvent-exchange LNPs) and 78 (MIC values of 0.5–2 mg/mL for kraft LNPs), yet some enhanced activities, like 79 silver-functionalized LNPs achieving up to 25 mm zones against S. aureus. Strain-specific differences show Gram-negative bacteria exhibiting lower susceptibility due to outer membranes, consistent with this study’s zones (∼16–28 mm), while 80 noted stronger effects against Gram-positive strains like S. aureus, aligning with OA-LNPs’ up to 26.71 mm performance. Table 3 shows that in all bacterial species the antioxidant activities of soda lignin (SL) are lower than those of lignin nanoparticles (OA-LNPs). OA-LNPs have a high surface area-to-volume ratio and are nanoscale particles usually measuring 115 nm. Greater contact with bacterial cell walls is made possible by this which promotes membrane integrity disruption intracellular content leakage and growth inhibition. Additionally increased surface area increases the release of bioactive substances. Phenolic acids) that have antibacterial properties. 81 SL on the other hand has a lower surface area and is a bulk micron-sized material (typically irregular aggregates from soda pulping). Because of this it cant interact with bacteria as well which makes inhibition zones smaller. 82
4. Conclusion
Through the extraction of SL and its subsequent conversion into lignin nanoparticles (OA-LNPs) using a dioxane-based nanoprecipitation technique this study effectively illustrated the valorization of underutilized O. abyssinica biomass. As confirmed by SEM, EDX UV-Vis, FTIR and 1H NMR analyses the synthesized OA-LNPs had a spherical morphology with a mean particle size of 115 nm (ranging from 65 to 135 nm) a zeta potential of -30. 1 mV and a sulfur-free composition. The optimized alkaline extraction produced up to 294 mg/g of soda lignin. Functional analyses showed that OA-LNPs had greater multifunctionality than bulk soda lignin. OA-LNPs had a much lower IC50 value of 11. 47 µg/mL compared to 131. 1 µg/mL for soda lignin when antioxidant activity was measured using DPPH assays. This was explained by the nanoparticles greater surface area and phenolic group accessibility. Sun protection factor (SPF) measurements indicated substantial improvements in commercial lotions reaching up to 114. 8 when OA-LNPs were incorporated due to effective UV absorption by phenolic groups. With inhibition zones ranging from 12 to 29 mm antibacterial testing showed moderate efficacy against both Gram-positive (S. aureus and S. epidermidis) and Gram-negative (E. coli and K. pneumoniae) strains. This was probably due to membrane disruption and the release of bioactive compounds. These results highlight the potential of O. abyssinica as good raw material to produced lignin nanoparticle using 1,4-dioxine for the application of antioxidant, antibacterial and sun screening properties. To further confirm their industrial viability and increase their usefulness in cutting-edge biomedical and environmental applications future research should examine scalability long-term stability and in vivo toxicity evaluations.
Footnotes
Acknowledgments
The author would like to express sincere gratitude to Debre Tabor University for providing the necessary chemicals and laboratory facilities that enabled the successful completion of this research during the period 2025/2026 G.C.
Ethical considerations
O. abyssinica plants were selected randomly from West part of Ethiopia, Metekel Zone, Pawi Woreda based on TAPPI standards 2002, from non-protected, non-private, and non-indigenous area. The use of plant materials in the present study complies with all applicable institutional, national, and international regulations, including the guidelines of the International Union for Conservation of Nature (IUCN). The plant was taxonomically identified by Mr. Wogayew of the College of Natural Sciences, Department of Biology and Biodiversity Management at AAU using taxonomic keys from the Flora of Ethiopia and Eritrea, and a voucher specimen was deposited at Addis Ababa Univerity Herbarium (Voucher No. 4271).
Author contributions
Limenew Abate Worku contributed to the investigation, methodology, and software aspects of the study and writing the manuscript.
Funding
No financial support was provided to the authors for the research, authorship, or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Datasets generated and/or analysed during this study are available in the paper.