Abstract
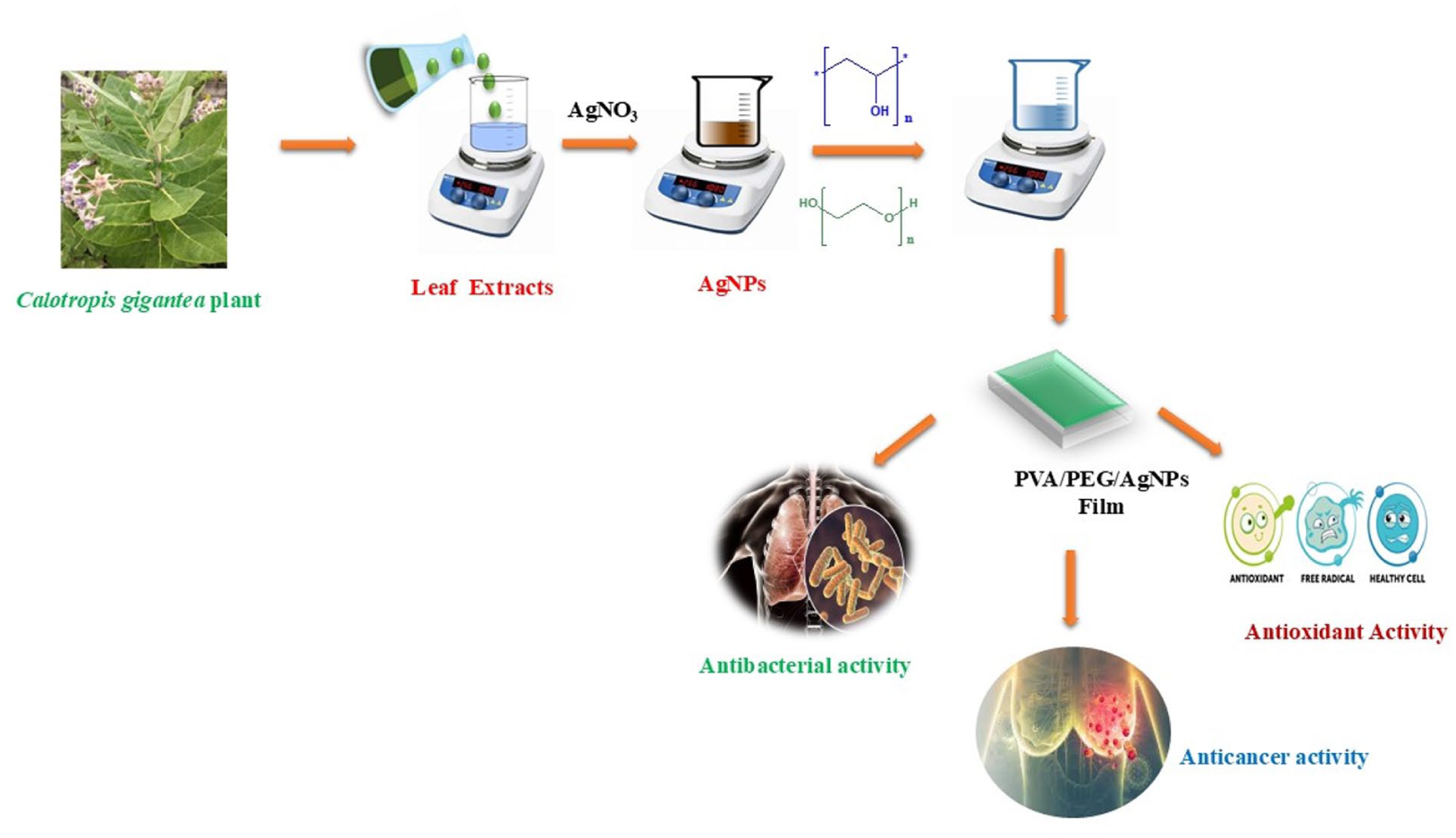
The Indian Medicinal plant Calotropis gigantea leaf extract was incorporated in silver nanoparticle with the added polymeric matrix of PVA and PEG. Thus, Calotropis gigantea was used as capping agent in silver nanocomposite, and hence, a thin film was synthesized by solvent casting method. The morphology of the as-prepared thin film was confirmed by XRD, FESEM and EDAX studies. The presence of functional groups of Calotropis gigantea and silver nanocomposite was well-established in FTIR report. Optical property of as synthesized thin film was confirmed by UV-Vis analysis. Antibacterial studies of the PVA/PEG films comprising AgNPs show a potent antibacterial effect against Gram-negative bacterial strains by Agar-Agar well-diffusion method. Antioxidant activities were carried out through four different assays: DPPH, SOD, NO and H2O2. Cytotoxic studies were carried out with these synthesized PVA/PEG films containing AgNPs using MTT assay on MCF-7 cells. The synthesized thin film with an IC50 value of 43.29 (μg/ml) will show a promising result in in vitro studies.
Introduction
The development of effective antimicrobial agents for pharmaceutical and biomedical applications has garnered significant attention due to rising bacteria resistance to numerous antibiotics and bactericides.1–4 Silver nanoparticles have been found to possess remarkable biocidal properties against several kinds of germs and viruses and fungi, effectively combating 650 different types. Moreover, these nanoparticles can inhibit the growth of resistant microbial strains even at extremely low concentrations, while maintaining a low level of toxicity towards mammals. Nevertheless, the primary concerns that need to be tackled are the controlled release and prevention of agglomeration. The entrapment of Ag nanoparticles on different substrates and matrices may offer a solution to these challenges, ultimately guaranteeing a continuous release of Ag.5–10
Polymers are considered the primary materials of interest in this scenario due to their adaptable structures containing appropriate functional groups along extended polymeric chains. These characteristics enable the integration and fixation of nanoparticles, resulting in the formation of nanocomposites. PVA/PEG offer promising solutions for film-forming polymers in biomedical applications which hold AgNPs or NO donors.
PVA, being a synthetic polymer, is nontoxic, water-soluble, noncarcinogenic and hydrophilic, making it ideal for producing films.11–14 This characteristic has led to its widespread use in various biomedical applications, such as wound dressings. PVA exhibits remarkable surface stabilization and chelation characteristics, which have led to its widespread application in various medical devices such as contact lenses, orthopaedic devices and drug delivery systems. Numerous scientific articles have detailed the integration of AgNPs into PVA films to create antimicrobial surfaces. 15
AgNPs synthesized using Calotropis gigantea leaf extract hold great potential for a wide range of biological applications, including antimicrobial therapy, anti-inflammatory treatment, cancer therapy, drug delivery, bio-sensing and bio-imaging.16–21 However, further research is needed to fully understand their biocompatibility, pharmacokinetics and long-term effects in biological systems.22–30
This study successfully created composite thin polymeric films of PVA/PEG with evenly distributed and monodispersed AgNPs. Calotropis gigantea plant leaf extract was used to biogenically synthesize AgNPs. The films were characterized using FTIR, UV-Vis, SEM, XRD and EDX, among other techniques. AgNPs-containing PVA/PEG films shown strong antibacterial activity against a range of bacterial species in addition to antioxidant and anticancer qualities. The synthesis, characterisation, antibacterial activity and cytotoxicity of PVA/PEG solid films containing biogenically synthesized AgNPs against cancer cell lines are described in this study to the best of our ability. These polymeric films, which are solid materials with antibacterial and antitumorigenic properties, may have extensive applications in biomedicine, including tissue engineering and wound healing.11,31,32
Materials and methods
Materials
Merck Limited provided the sodium hydroxide (NaOH), potassium bromide (KBr) and silver nitrate (AgNO3, 99.9%). Himedia, India supplied 1X PBS, while Sigma, USA, supplied Dulbecco’s Modified Eagle’s Medium (DMEM), Foetal Bovine Serum (FBS), Dimethyl Sulfoxide (DMSO), 5 mg/ml MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) and Doxorubicin. The 96-well tissue culture plates and wash beaker were provided by Tarson, India. Furthermore, Himedia Laboratories provided the polyvinyl alcohol (PVA) and polyvinyl glycol (PEG). Fresh leaves of the Apocynaceae species-Calotropis gigantea were gathered near Coimbatore, Tamil Nadu.
Preparation of leaf extract
We gathered fresh leaves of Calotropis gigantea, ensuring they were thoroughly washed, dried and finely powdered. The powder, approximately 10 mg, was subjected to boiling at 80°C in distilled water, resulting in the formation of a dark orange liquid (Figure 1(a)). Consequently, leaf extract served as a reducing agent in the synthesis of silver nanoparticles. 33

(a) Preparation of leaf extract. (b) Preparation of PVA/PEG thin film with AgNPs.
Preparation of silver nanoparticles
At room temperature, about 88 ml of a 1 mM silver nitrate solution was mixed with 12 ml of Calotropis gigantea as-prepared leaf extract, and the mixture was agitated for 15 min. It was confirmed that silver nanoparticles had formed when an orange to brown colour shift was seen. The analysis of the surface reactivity and structure of the silver nanoparticles made from the leaf extract was made easier by this reaction.
Preparation of thin film
PVA/PEG solid blended films with a 1:1 (w/w) PVA-to-PEG ratio were prepared by utilizing modified procedures from previous research. AgNPs were added to these blended films to improve their qualities. In order to create the films, 1 g of PVA was completely dissolved in 120 mL of deionized water at 80°C while being magnetically stirred. After adding 1 g of PEG to the PVA solution, the mixture was stirred magnetically at room temperature to homogenize it. Following the addition of 2.0 mL of an aqueous AgNPs suspension to the PVA/PEG solution, the mixture was further standardized at room temperature using magnetic stirrer (Figure 1(b)).[11]
Characterization of the prepared polymeric film
Several methods were used to evaluate the prepared polymeric film in order to ascertain its structure and characteristics. An Enraf Nonius CAD4-MV31 single crystal X-ray diffractometer was used for the X-ray diffraction (XRD) investigation. A Perkin Elmer Lambda spectrophotometer was used for UV-visible spectroscopy, and a BRUKER-ALPHA-Platinum-ATR-IR spectrometer was used for Fourier-transform infrared spectroscopy (FTIR). Using an FEI-Quanta FEG 200F, scanning electron microscopy (SEM) was used to analyse surface morphology. A microplate reader (Thermo Fisher Scientific, USA) was used to measure the amount of formazan product at a wavelength of 570 nm for the cytotoxicity analysis.
In vitro anticancer activity
Procedure
Cell culture
MCF-7 (Human Breast Cancer cells) were purchased from NCCS, Pune and were cultured in liquid medium (DMEM) supplemented with 10% Foetal Bovine Serum (FBS), 100 ug/ml penicillin and 100 µg/ml streptomycin and maintained under an atmosphere of 5% CO2 at 37°C.
MTT assay
The Ru-1 sample was tested for in vitro cytotoxicity, using MCF-7 cells by MTT assay. Briefly, the cultured MCF-7cells were harvested by trypsinization, pooled in a 15 ml tube. Then, the cells were plated at a density of 1 × 105 cells/ml cells/well (200 µL) into the 96-well tissue culture plate in a DMEM medium containing 10% FBS and 1% antibiotic solution for 24–48 hours at 37°C. The wells were washed with sterile PBS and treated with various concentrations of the Ru-1 sample in a serum-free DMEM medium. Each sample was replicated three times and the cells were incubated at 37°C in a humidified 5% CO2 incubator for 24 h. After the incubation period, MTT (20 µL of 5 mg/ml) was added to each well, and the cells were incubated for another 2–4 h until purple precipitates were clearly visible under an inverted microscope. Finally, the medium together with MTT (220 µL) was aspirated off the wells and washed with 1X PBS (200 µl). Furthermore, to dissolve formazan crystals, DMSO (100 µL) was added, and the plate was shaken for 5 min. The absorbance for each well was measured at 570 nm using a microplate reader (Thermo Fisher Scientific, USA), and the percentage cell viability and IC50 value were calculated using Graph Pad Prism 6.0 software (USA). 34
Antioxidant activity
Antioxidant evaluations of PVA/PEG Films Containing Biogenic AgNPs film against various free radical scavengers like DPPH, hydrogen peroxide, super oxide and nitric oxide were analysed with standard Ascorbic acid. 35
Antibacterial activity
The antibacterial activity of the synthesized PVA/PEG Films Containing Biogenic AgNPs film was tested using the agar well-diffusion method against pathogenic bacteria isolated from clinical samples: Klebsiella pneumoniae (Gramme-negative bacteria) and Pseudomonas aeruginosa (Gramme-negative bacteria).36,37
Result and discussion
Synthesis and characterization of PVA/PEG films containing biogenic AgNPs
The biogenic synthesis of silver nanoparticles (AgNPs) was achieved using an eco-friendly approach that employed Calotropis gigantea leaf extract as a natural reducing agent. This method exemplifies the principles of green chemistry, emphasizing sustainability and environmental safety. Unlike conventional techniques for synthesizing AgNPs – such as the chemical reduction of silver ions – this green approach avoids the use of hazardous chemicals that pose risks to biological systems and the environment. Traditional methods are often associated with high costs, resource-intensive processes and significant ecological concerns, making the need for alternative approaches critical.38–40
The use of C. gigantea leaf extract offers a cost-effective and sustainable solution, as the plant’s bioactive compounds effectively facilitate the reduction of silver ions to nanoparticles. This process not only reduces the ecological footprint of nanoparticle synthesis but also ensures the retention of high efficiency in nanoparticle production. Recent studies have further validated the potential of C. gigantea as a viable natural reducing agent. 41 Research findings have demonstrated the successful preparation of AgNPs using commercial extracts of this plant, highlighting its versatility and eco-friendly nature. The method underscores the importance of integrating green chemistry principles into nanoparticle synthesis to promote environmentally responsible scientific practices while maintaining the desired functional properties of the nanoparticles. 42
UV-Vis spectroscopy
The surface plasmon resonance (SPR) absorption band at about 409 nm was used to demonstrate the existence of silver nanoparticles (AgNPs) in PVA/PEG films (Figure 2). This phenomenon occurs when incoming light of a certain frequency causes the conduction electrons on the surface of the nanoparticle to vibrate coherently. The size, shape and surrounding dielectric environment of the AgNPs are some of the elements that affect the resonance situation. The homogeneous distribution of AgNPs throughout the PVA/PEG polymer matrix is strongly supported by the distinct and crisp SPR band seen at 409 nm. This feature not only shows that AgNPs have been successfully included, but it also shows how promising they are for applications that call for exact control over the interactions between nanoparticles and light. 43

Absorption spectrum of PVA/PEG films containing biogenic AgNPs.
A recent study reveals a similar SPR peak for AgNPs inserted in the polymeric matrix of the as-prepared thin film supports our findings. The size and uniformity of the dispersion of the nanoparticles are reflected in the band position and intensity. The study emphasized the critical role that nanoparticle production processes play in generating such well-defined optical characteristics, which is consistent with recent findings in the PVA/PEG system.
X-ray diffraction (XRD) spectroscopy
The XRD patterns of as synthesized nanocomposite thin film shows the diffraction peaks at of 32°, 37°, 46°, 55°, 57° and 76° correlate to the indexed planes (110), (200), (211), (220) and (311) of face-centred cubic (FCC). It also correlates with the JCPDS card no 04-0783. These peaks confirm that the AgNPs are crystalline within the polymer matrix (Figure 3). Since the AgNPs’ nanoscale diameters lead to smaller crystallites, the XRD peaks’ broadening is explained. These findings validate the effective integration of evenly distributed AgNPs into the amorphous PVA/PEG matrix. AgNPs produced in a PVA-based matrix also showed comparable diffraction patterns, according to a recent study. 44

X-ray diffraction patterns of the prepared PVA/PEG films containing biogenic AgNPs.
FT-IR spectroscopy analysis
The FTIR analysis aims to identify the functional groups present in the polyvinyl alcohol (PVA)/polyethylene glycol (PEG) films incorporated with biogenic silver nanoparticles (AgNPs). The result provides insights into the interaction between the polymer matrix and AgNPs, as well as any chemical changes in the film composition. From Figure 4 & Table 1, it was observed that Characteristic absorption bands in the spectra of pure PVA/PEG with the functional groups involved in hydrogen bonding and vibrations of the polymer backbone. The broad absorption peak at 3312 cm⁻¹ is a result of the -OH stretching vibration, which suggests that PVA and PEG have strong intermolecular hydrogen bonding. At 2965 cm⁻¹, the asymmetric stretching vibrations of -CH₂ groups are assigned. Bending vibrations of the -CH₂ and -CH₃ groups are represented by 1498 and 1431 cm⁻¹, respectively. Shift in 1067 cm⁻¹ indicates the stretching vibrations of the C-O-C bonds in the PEG element. The presence of these distinct bands indicates that the PVA and PEG mixture was properly blended. Hence, PVA/PEG/AgNPs Spectra reveals the spectral characteristics undergo noticeable alterations upon AgNPs incorporation: Changes in the shape of the -OH peak (3312 cm⁻¹ persists) suggest that the hydroxyl groups and AgNPs interact, perhaps as a result of weak bonding or coordination. 2993 cm⁻¹: A little change from 2965 cm⁻¹ indicates that the presence of nanoparticles has altered the -CH₂ environment. The structural rearrangements and possible physical adsorption or chemical interaction between the polymer matrix and AgNPs are indicated by the minor changes in the 1498, 1431 and 1067 cm⁻¹ peaks. Confirming the effective integration of AgNPs into the PVA/PEG matrix, the shifts and intensity variations provide a clue to potential coordination between Ag atoms and the oxygen-containing functional groups.

FTIR spectrum of PVA/PEG films containing biogenic AgNPs.
Assignments of FT-IR wavenumber of PVA/PEG films containing biogenic AgNPs.

Field emission scanning electron microscopy (FESEM) & energy-dispersive X-ray spectroscopy (EDS)
Field emission scanning electron microscopy (FESEM) was conducted to analyse the morphology of PVA/PEG films embedded with biogenic silver nanoparticles (AgNPs). The images revealed a distinct rod-like morphology with an approximate width of 15.8 nm, captured in view fields of 26.8 µm and 6.99 µm, as shown in Figure 6(a) and (b). This structural form suggests uniform dispersion and controlled growth of AgNPs within the polymer matrix.
Morphological study, energy-dispersive X-ray spectroscopy (EDS) (Table 2 & Figure 5(a) and (b)) analysis was performed to ascertain the elemental composition. The spectra indicated that the sample comprised 82.25% silver (Ag), 7.21% carbon (C), 5.82% oxygen (O), 3.41% nitrogen (N), 1.07% potassium (K) and 0.24% magnesium (Mg) (Figure 5(c)). The high silver content confirms the successful incorporation of AgNPs with minimal impurities, reflecting a high-purity nanocomposite film. These observations align well with recent findings reported, 32 who studied AgNPs-loaded PVA composites synthesized via a green method. Their FESEM images similarly showed rod-like silver nanoparticles with sizes in the nanometer range, while their EDS results confirmed predominant silver content (~80%) alongside minor elements originating from the polymer matrix and synthesis process. The consistency across these studies underscores the reproducibility of biogenic AgNPs synthesis and stabilization within polymer matrices.
Assignments of EDS on PVA/PEG films containing biogenic AgNPs.


(a–b) Scanning electron microscopy (SEM) with PVA/PEG films containing Biogenic AgNPs and (c) the spectrum of energy-dispersive x-rays (EDX).
Antioxidant activity
Antioxidant activity of PVA/PEG films containing Biogenic AgNPs and Ascorbic acid (standard medication) was tested using free radical tests (DPPH, SOD, NO and H2O2). Figure 6 (a)–(d) shows the percentage of inhibition of different free radicals by PVA/PEG films containing Biogenic AgNPs and Ascorbic acid. The study found that PVA/PEG Films containing Biogenic AgNPs have similar antioxidant activity compared to standard drugs against NO free radicals.

(a-d) In vitro antioxidant activities of PVA/PEG Film with AgNPs and ascorbic acid. (a) DPPH; (b) SOD; (c) NO and (d) H2O2
DPPH assay
In this experiment, a peak at 517 nm was seen due to the rich purple colour of the DPPH radical. With the addition of Pt complex, the peak intensity of the DPPH solution gradually dropped, and the colour of the DPPH vanished, showing that the synthesized PVA/PEG films containing Biogenic AgNPs has a strong radical scavenging ability. Figure 6(a) shows the percentage inhibition of PVA/PEG films containing Biogenic AgNPs by the DPPH radical.
SOD assay
The radicals known as (O2-) may bind to biomolecules (DNA/protein) and cause immediate damage to biological systems. The superoxide radical quenching activities of the PVA/PEG films containing Biogenic AgNPs increased as concentrations increased (Figure 6(b)).
NO assay
In addition to being an essential component of several biological systems, nitric oxide is a dynamic, pleiotropic mediator of a broad variety of physiological processes. The NO activity of a PVA/PEG films containing Biogenic AgNPs is depicted in Figure 6(c).
H2O2 assay
H2O2 can be switched to H2O by absorbing protons (H+) or electrons (e-). The Pt complex acts as an H2O2 radical scavenger in this experiment by donating azomethine protons to decrease the hydrogen peroxide to H2O2. Figure 6(d) depicts the H2O2 scavenging of a PVA/PEG films enclosing with biogenic AgNPs.
Antibacterial activities
The antibacterial efficacy of the synthesized PVA/PEG films with AgNPs was assessed. Two strains of Gram-negative bacteria, Klebsiella pneumoniae and Pseudomonas aeruginosa, were the subjects of an in vitro antibacterial activity test. By measuring the inhibition zones (in millimetre) and contrasting them with those of ampicillin, a positive control, the films’ efficacy was evaluated.
The films showed strong antibacterial activity against Klebsiella pneumoniae, with inhibition zones of 20 ± 1.0 mm for the plant extract and 22.12 ± 0.3 mm for the thin film. This was more effective than ampicillin, with inhibition zone of 15.5 ± 1.5 mm. Similarly, the films’ inhibition zones for Pseudomonas aeruginosa were 22 ± 1.0 mm for the plant extract and 24.3 ± 0.3 mm for the thin film, which were much larger than ampicillin’s 13 ± 1.0 mm inhibition zone. According to these findings, the films containing biogenic AgNPs have improved antibacterial qualities, which makes them attractive options for applications that call for antimicrobial materials (Figure 7).

Screening of antibacterial activity of PVA/PEG AgNPs film and histogram showing the zone of inhibition.
Anticancer activities
The anticancer properties of PVA-PEG AgNPs were evaluated through MTT assay by using Human Breast Cancer cell lines (MCF7) as illustrated in Figure 8. The PVA-PEG AgNPs demonstrated significant anticancer activity against MCF7 cells. Various Concentrations of 5, 12.5, 25, 50, 100 and 200 μg/mL nanoparticles was imparted with PVA-PEG AgNPs which effectively eliminated the cancer cells. These nanoparticles exhibit high permeability through cell membranes, making them potential carriers for antitumor agents.

Morphological assessment of PVA/PEG films containing biogenic AgNPs against MCF-7 cells lines at different concentrations.
The percentage cell viability and IC50 value were calculated using Graph Pad Prism 6.0 software (USA). The anticancer activity of percentage cell viability and IC50 value are 43.29 (μg/ml).
Morphological changes
Following a 24-hour treatment with PVA-PEG AgNPs at several concentrations to cause cytotoxicity in 3T3 cells, the cells were inspected under a microscope. The bulk of the cells perished of apoptosis, according to the cytological changes, with the exception of the 3T3 cells. A large number of the treated cells had early apoptotic morphological features, including different PVA-PEG AgNPs concentrations (Figure 9).

Histogram showing the cell viability percentage of PVA/PEG films containing biogenic AgNPs against MCF-7 cells with different concentrations.
Conclusion
In summary, PVA/PEG polymeric blended films embedded with AgNPs and Calotropis gigantea leaf extract were successfully developed, resulting in homogeneous solid films with well-dispersed nanoparticles. The structural and optical properties of these films were characterized using FTIR, XRD, SEM, EDS and UV-Visible spectroscopy. Antibacterial and antioxidant assays demonstrated that the inclusion of biogenic AgNPs enhanced the films’ antibacterial efficacy and radical scavenging activity. These findings suggest promising potential for PVA/PEG films containing AgNPs in biomedical applications, such as wound dressings for treating infected wounds. Preliminary results indicate localized cytotoxicity and antibacterial effects through NO and AgNPs release, providing an effective antibacterial barrier to prevent bacterial growth. However, future research must emphasis on in vivo toxicity studies and biocompatibility to ensure safety and efficacy before clinical implementation. In addition, exploring the integration of these films with other therapeutic agents could further enhance their biomedical utility.
Footnotes
Acknowledgements
The authors are thankful to SAIF, IIT Madras for the single crystal XRD, FTIR measurements. Authors are also thankful to Dr. K. Kadirvelu, DRDO, Bharathiyar University centre for extending facilities of UV studies.
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.

