Abstract
Novel metallic nanoparticles (NPs), such as biosynthesized gold nanoparticles (AuNPs), have been extensively studied because of their wide applications. Consequently, the present investigation aimed at the biosynthesis, characterization, and comparative assessment of biological activities of AuNPs. Biosynthesized AuNPs (ED-AuNPs) were synthesized using aqueous extract of Euphorbia dendroides (E. dendroides) aerial parts (EDAE), which functions as a reducing and stabilizing agent by its nature. Presence of tannins, saponins, flavonoids, alkaloids, glycosides/or carbohydrates, steroids, and terpenes was revealed by the qualitative phytochemical analysis of EDAE. Characterization of the biosynthesized AuNPs was attained by UV-Visible spectrophotometry, transmission electron microscopy (TEM), XRD, and Fourier transform infrared (FTIR). The cytotoxic effects of ED-AuNPs and EDAE were assessed in vitro against HepG2 and HCT-116 cancer cells. The MTT assay revealed a significant cytotoxic effect of ED-AuNPs and EDAE on HepG2 (IC50 = 41.72 ± 1.26 and 55.26 ± 2.25 μg mL−1) as well as HCT-116 (IC50 = 44.96 ± 3.23 and 69.83 ± 0.96 μg mL−1) cells, thus indicating their potential anticancer activity. Furthermore, ED-AuNPs exhibited potent anti-diabetic activity, with an IC50 value of 19.8 ± 1.97 µg mL−1, pointing to their plausible use as therapeutics in the treatment of diabetes, as well as a promising antimicrobial activity against H. pylori.
Introduction
Much attention has been paid to green synthesized nanoparticles (NPs) using plant extracts owing to its simplicity, prompt formation of NPs characterized by numerous and interesting morphologies, exclusion of detailed practices for the maintenance of cultured cells, and eco-friendliness.1–4 Hence, green nanotechnology used to synthesize NPs is considered an encouraging field of research and there has been a dramatic increase in the number of medicinal plants utilized for the biosynthesis of metallic NPs.5,6 Such medicinal plants are rich in phenolic compounds, alkaloids, diterpenoids, steroids, and other compounds which inhibit the development of various microorganisms.7,8 Furthermore, various phytochemicals in plant extracts have been extensively used in the biosynthesis of metal-based nanoparticles (MNPs), where they act as reducing and capping agents responsible for the reduction of metal ions to MNPs.9,10 Gold nanoparticles (AuNPs) in particular are among the various MNPs that possess special interest in various biological applications due to their unique properties, including easy preparation, large specific surface area (SSA), high surface plasmon resonance (SPR), adequate biocompatibility, and high antimicrobial activity.11,12 Gold nanoparticles find potential applications in, among others, treatment of cancer thermotherapy, electronics, catalysis, bio labeling, biosensors for the detection of viruses and bacteria, drug delivery, tissue/tumor imaging, and photothermal therapy (PTT).13–16 According to their interaction with cells, AuNPs were recently assessed for their cytotoxicity and biocompatibility. Interestingly, AuNPs exhibited size-dependent endocytosis, anti-proliferative effect on some kinds of cells as well as selective interaction with heparin-binding glycoproteins leading to the anti-proliferative and antiangiogenic activities reported against endothelial/fibroblast cells.17–19 Previous studies have pointed to the potent antibacterial activity exerted by AuNPs, with various shapes and sizes, on various Gram-negative and Gram-positive bacteria as well as multidrug-resistant (MDR) strains.20,21 As biocidal agents, AuNPs mainly target the plasma membrane and bacterial DNA.22–24
Alternative to chemical and physical synthesis methods,25–27 effective eco-friendly green synthesis approaches of MNPs have been adopted which utilize various biological materials, including microorganisms, enzymes, proteins besides plants or plant extracts.28–30 Compared to other biological processes, green synthesis of MNPs using plant derivatives has proven to be more valuable since it abolishes the time-consuming maintenance of microbial cultures and exhibits a high biological potential with various applications.4,9,31–33 The genus Euphorbia (Euphorbiaceae), the third largest genus of angiosperm plants, is estimated to include around 2000 described species and known to be widely distributed.34–36 In addition to the whole plant, there are different parts of the plants that are used, including roots, seeds, the more or less toxic milky latex, wood, barks, and leaves.34,37 Besides, the milky-white irritating latex is found in all Euphorbia plants and is known to play a crucial role as the first defense mechanism against insects, diseases, and herbivores. It is a rich source of phytochemicals, which have been studied extensively throughout time.38,39 Phytochemical analysis of various Euphorbia species indicated the presence of different phytochemicals, including sterols, diterpenes, triterpenes, carotenoids, flavonoids and alkaloids known to possess diverse biological activities.40–43 According to previous studies, over 5% of Euphorbia species are utilized in traditional medicine (TM) worldwide to treat various digestive, respiratory, and inflammatory diseases.44,45 Moreover, the anticancer and cytotoxic activities37,46,47 as well as antibacterial and antiviral activities 48–50 of some Euphorbia species have been previously reported.
Euphorbia dendroides L. (E. dendroides) is a tree-like semi-succulent spurge, known also as tree spurge. It grows in the Western Mediterranean area on rocky coastal places and cliffs, especially in Sardinia. Previous studies reported that the most abundant bioactive compounds in E. dendroides latex are diterpenes, particularly Jatrophane-type, which have been shown to exhibit antiviral activity against Chikungunya virus (CHIKV) and anticancer effect against multi-drug resistant cancers.51,52 Furthermore, E. dendroides are known to exhibit antiproliferative, antioxidant, and anti-inflammatory properties, mainly attributed to the high content of polyphenols particularly chlorogenic acid and rutin, the most representative compounds for phenolic acids and flavonoids, respectively.53,54 Recently, cytotoxic activity against various cancer cell lines of cycloartane triterpenes, including a newly identified one, isolated from the air-dried aerial parts of E. dendroides has been reported. 42 The qualitative and quantitative determinations of phenolic acid and flavonoid compounds of E. dendroides were determined using HPLC. The whole plant water extract of E. dendroides exhibited a variety of phytochemicals, including alkaloids, coumarins, flavonoids, glycosides/or carbohydrates, saponins, terpenes and tannins. 55 To our knowledge, there are no reports so far regarding the synthesis of AuNPs using E. dendroides aerial part extracts. In the present work, we report the biogenic production of AuNPs using hot water E. dendroides aerial part extracts as a modest, affordable, and reproducible method. In addition, we aimed to assess the anticancer, anti-diabetic and anti-Helicobacter pylori activities of the biosynthesized NPs in an attempt to unravel their potential bioactivities.
Materials and methods
Plant material
Euphorbia dendroides (synonym Tithymalus arboreus Tourn., E. divaricata Jacq. and E. laeta Aiton) aerial parts were collected from Mersa Matruh, on the northwest coast of Egypt, in March 2020. The plant’s taxonomic identity was established by comparing the voucher specimen acquired with a known identity found in the Department of Botany’s herbarium at Cairo University, Egypt. The aerial parts of the plant were air-dried, crushed into a fine powder, and stored at room temperature for subsequent analysis.
Preparation of E. dendroides extract for preliminary phytochemical screening
Fresh E. dendroides aerial parts were gathered, dried, and coarsely pulverized. Boiling deionized (DI) water was used to extract 100 g of coarse powder four times for 48 h each time. The extract was filtered and subjected to qualitative phytochemical screening. 56
Preparation of E. dendroides aqueous extract for synthesis of NPs
Aqueous extract of E. dendroides (1% w/v) was prepared via boiling of 1.0 g of E. dendroides dried aerial powder for 15 min, filtered using Whatman qualitative filter paper No.1, and completed up to 100 mL with DI water. Fresh plant extracts were prepared prior to each experiment.
Preparation of HAuCl4 stock solution
Hydrogen tetrachloroaurate (III) hydrate (HAuCl4. aq, ≥ 99.995% trace metals basis) was purchased from Sigma-Aldrich (Product # 254169). One gram of HAuCl4.aq was dissolved in 100 mL DI water to prepare a 1.0% w/v stock solution.
Biosynthesis of AuNPs using EDAE; ED-AuNPs
For the synthesis of ED-AuNPs, 0.05–2.0 mL of 1% w/v E. dendroides aerial part (EDAE) were added to 0.05 mL of 1.4 × 10−4 M HAuCl4.aq solution at room temperature and DI water was added to a final volume of 10 mL. The bioreduction of Au3+ ions to Au0 NPs was monitored by the change in the reaction mixture color from yellow to violet, dark pink or green based on the concentration of EDAE used. The pH of the solutions used to prepare ED-AuNPs was set to the appropriate values using 0.1 N H2SO4 and 0.1 N NaOH solutions. Sulfuric acid and sodium hydroxide used for pH monitoring were also purchased from Sigma-Aldrich.
Characterization of the biosynthesized ED-AuNPs
Ultraviolet-visible (UV/Vis) spectra of ED-AuNPs in the range of 300–1000 nm were recorded using a Shimadzu spectrophotometer (UV-2600, Shimadzu, Kyoto, Japan). The pattern of X-ray diffraction (XRD) was recorded using a Shimadzu XRD-6000 diffractometer with Cu-Kα radiation (λ = 1.54056 Å) to confirm the biosynthesis of NPs. Morphological and sizing analyses of ED-AuNPs were examined with transmission electron microscopy (TEM) and the images were recorded with JEOL-1200JEM. Fourier transform infrared (FTIR) spectra were obtained with a Nicolet 6700 FTIR spectrophotometer at room temperature. Thermogravimetric analysis (TGA) was performed with a 10°C min−1 rate of heating using a Shimadzu DT-50 thermal analyzer.
Cytotoxicity evaluation
Cell lines and cell culture
Human hepatocellular carcinoma (HepG2) and human colon carcinoma (HCT-116) cell lines were obtained from VACSERA Tissue Culture Unit (Cairo, Egypt). Cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM; Lonza, 12–604F) supplemented with 5% heat-inactivated fetal bovine serum (FBS), 100 IU/mL penicillin, and 100 µg/mL streptomycin (Lonza, 17-602E). Cultured cells were grown as monolayers at 37°C in a humidified atmosphere of 5% CO2 according to the provider’s instructions.
Determination of cell viability using MTT assay
In vitro anticancer activity of the biosynthesized ED-AuNPs was assessed against the standard anticancer drug doxorubicin (DOX) and EDAE using the tetrazolium salt (3-(4,5-Dimethylthiazol-2-yl)- 2,5-diphenyltetrazolium bromide, MTT).
57
HepG2 and HCT-116 cells at a density of 104 cells/well were seeded in 96-well plates and incubated for 24 h. The investigated ED-AuNPs, EDAE, and DOX were added at a final concentration (F.C.) ranging from 0 – 500 μg mL−1 to the seeded cells for another 48 h at 37°C and 5% CO2. Subsequently, treated cells were incubated with MTT at 5 mg/mL F.C. for 4 h at 37°C. Finally, 100 μL DMSO were added to dissolve the water insoluble purple formazan crystals resulting from the reduction of MTT by viable cells. Using microplate reader, the cell viability was measured by recording absorbance (Abs) at 540 nm after 15 min (Tecan Infinite 200 Pro, Austria). A negative control of 10 μL of the MTT stock solution added to 100 μL of medium alone was included. Cell viability was reported relative to control, untreated cells (viability of which was set at 100%) as follows:
Results were represented as mean values ± SD of three independent experiments.
The concentration required to inhibit 50% of cancer cell growth in μg mL−1 (IC50) was estimated using GraphPad Prism 6 software package. All experiments were performed in triplicate and data were represented as mean values ± SD.
In vitro determination of α-glucosidase inhibitory activity
The potential anti-diabetic efficacy of crude E. dendroides aqueous extract (EDAE) and phytosynthesized AuNPs (ED-AuNPs) was assessed in vitro via determination of α-glucosidase inhibitory activity.
58
The chromogenic substrate for alpha-D-glucosidase, p-nitrophenyl-α-D-glucopyranoside (pNPG), was used which was hydrolyzed to the yellow colored product, p-nitrophenol, that can be detected at 405 nm using a microplate reader (BioTek Instruments, Inc., Winooski, VT). Calculation of the percentage inhibition (% Inhibition) of α-glucosidase activity was carried out using the formula:
The 50% inhibitory concentration (IC50) was calculated using GraphPad Prism 6 software package. All experiments were performed in triplicate and data were represented as mean values ± SD.
Antibacterial assay against Helicobacter pylori
Helicobacter pylori strain
Helicobacter pylori (H. pylori) ATCC 43504 standard strain was supplied by American type culture collection (Manassas, VA) and obtained from the Regional Center for Mycology and Biotechnology (RCMP), Al-Azhar University.
Determination of the minimal inhibitory concentration
The in vitro antibacterial efficacy of EDAE and ED-AuNPs against H. pylori was determined by the microwell dilution method as previously described with few modifications.
59
For the determination of MIC in 96-well microtiter plate, serial two-fold dilutions of the examined samples in concentrations ranging from 125 to 0.24 µg mL−1 with bacterial inoculum adjusted to 106 CFU∕mL were used. As a reference, same concentrations of the standard antibiotic clarithromycin prepared in dimethyl sulfide (DMSO) were used. Each well of the 96- well microplate was filled with 40 µl of brain heart infusion (BHI) growth medium supplemented with 10% FBS, 10 µl of bacterial inoculum, and 50 µl of the two-fold serially diluted tested samples. The plates were incubated at 37°C for 72 h in 5% O2, 10% CO2, and 85% N2 atmosphere. Then, 40 μL of a freshly prepared (3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide, MTT) solution at 0.5 mg mL−1 final concentration was added to each well and incubated for a period of 30 min. Minimal inhibitory concentrations (MIC) of the tested samples where no change in color of MTT is observed was recorded using an automatic reader at 620 nm. The percentage of inhibition was calculated using the formula:
Statistical analysis
All the in vitro tests were performed in three independent experiments. Two-way analysis of variance (ANOVA) followed by post hoc Tukey’s HSD (Honestly Significant Difference) test was conducted to analyze the obtained data using by GraphPad Prism 6 software package. Results were represented as mean values ± standard deviation (SD) and considered significant at p ≤ .05.
Results and discussion
The phytochemical screening of EDAE
Various plant secondary metabolites, including among others phenolics, flavonoids, alkaloids, polysaccharides, and terpenoids, are used in the synthesis of metal-based NPs. Such metabolites are known to promote reduction of metal salt and serve as capping as well as stabilizing agents.60,61 In this regard, phytochemical screening of EDAE was carried out in an attempt to identify bioactive phytoconstituents with reducing properties. The preliminary screening detected the existence of flavonoids, carbohydrates, glycosides, coumarins, tannins, saponins as well as terpenoids. On the other hand, alkaloids and essential oils were absent. Similar results were previously reported. 62
Characterization of biosynthesized ED-AuNPs
Effect of E. dendroides aqueous extract
The optimum conditions for the biosynthesis of ED-AuNPs was determined by the nature of SPR peak obtained using UV/Vis spectroscopy. The SPR is a phenomenon which occurs in AuNPs, among other noble metallic NPs, which results in a strong absorption band in the visible region of the spectrum with maximum absorbance within 500–600 nm. Various parameters are known to be involved in the biosynthesis of NPs, which are modified and optimized for the purpose of the production of the desired size, shape and morphology of the biosynthesized NPs.63,64
Regarding the effect of EDAE quantity on the production of ED-AuNPs, the volume of plant extract was varied from 0.05 to 2.0 mL in 10 mL of 1.4 × 10−4 M HAuCl4. aq solution at room temperature (Figure 1(a) and (b)). By increasing the extract volume from 0.05 to1.0 mL, the absorbance was gradually increased with a shift in the λmax from 541 to 532 nm (Figure 1(a)). Increasing the volume of EDAE from1.2 to 2.0 mL results in the shift of λmax to 546 nm, this was accompanied by the development of the green colored ED-AuNPs solution and a slight decrease in absorbance (Figure 1(b)). In general, AuNPs are known to have surface SPR at around 530 nm.
65
Collectively, the dark pink color and the characteristic SPR band of Au NPs centered at about 532 nm recorded at 1.0 mL indicates that this is the optimum quantity of extract required for the production of ED-AuNPs. Similar sharp absorption peak of AuNPs biosynthesized from other plants has been formerly reported.66,67 The size of NPs and the maximum absorption peak are strongly correlated as has been previously reported, where an increase in the size of the particles is observed when the maximum absorption is red shifted.
68
Hence, it can be concluded that a reduction in the size of ED-AuNPs is achieved as the extract volume increased from 0.05 to 1.0 mL while the absorption spectra are blue shifted.69,70 UV/Vis spectra of ED-AuNPs using constant concentration of HAuCl4 (1.4 × 10−4 M) and different volumes of EDAE; (a) from 0.05 to 0.9 mL (b) from 1.0 to 2.0 mL.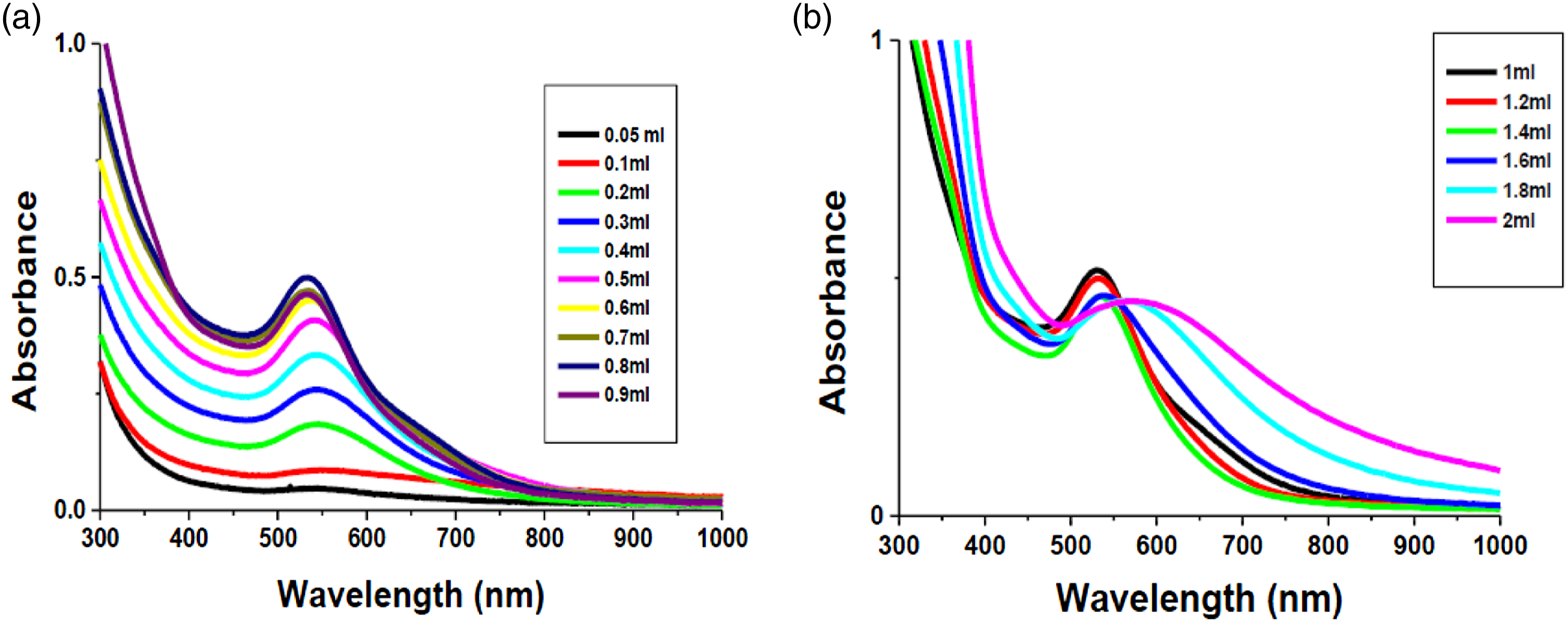
The morphology of the biosynthesized AuNPs, ED-AuNPs, was examined using the TEM analysis. The shape of the biosynthesized AuNPs is widely known to be dependent on the extract concentration. Although the low concentration of extract can reduce the Au3+ ions, it cannot prevent the aggregation of most quasi-spherical NPs due to the lack of biomolecules that serve as protective agents.
71
Hence, at low concentration of EDAE (0.1% w/v equivalent to 0.1 mL) ED-AuNPs exhibited anisotropic shapes, such as triangular and hexagonal nanoplatelets geometrical forms, with size range of 30–90 nm (Figure 2(a)). On the other hand, higher extract concentrations (1% w/v equivalent to 1.0 mL) results in the formation of almost spherical ED-AuNPs with an average size of 15 ± 3 nm, which replace the triangular and hexagonal geometrical structures (Figure 2(b)). Similarly, previous studies reported the synthesis of crystalline AuNPs of different shapes using different plant extracts.72–74 TEM analysis of ED-AuNPs synthesized from EDAE at (a) 0.1 mL (0.1% w/v) and (b) 1.0 mL (1% w/v) of extract concentration. The scale bar corresponds to 100 nm.
Effect of contact time
In addition to plant extract concentration, the reduction reaction (contact) time is among the factors influencing the shape, size, and stability of NPs to some extent.75,76 The effect of contact time between the Au3+ ions with the EDAE plant extract was studied at room temperature to follow the rate of reaction and the time necessary for the reduction process to be completed. Regarding the kinetics of AuNPs, synthesis of NPs was followed over different contact times (2–50 min) using UV/Vis spectroscopy. It was noted that AuNPs formation started 2 min after the interaction of EDAE with HAuCl4 and was completed within 30 min, where the characteristic absorbance peak of AuNPs was observed. Instead, a slight variation was observed after 30 min (Figure 3). This indicate that the time needed for the formation of gold NPs using E. dendriodes extract (ED-AuNPs) is 30 min and stay stable for 1 month. Similar findings were reported previously where the biosynthesis of AuNPs was attained after 30 min of incubation with various plant extracts.77,78 UV/Vis spectra as a function of contact time of ED-AuNPs synthesized using 1.4 × 10−4 M HAuCl4 and 1.0 mL EDAE at room temperature.
Effect of temperature
Temperature is one of the important factors that need to be considered during the synthesis of NPs where it controls the kinetics of the biosynthetic process as well as the size and shape of NPs.75,79 The effect of temperature on the biosynthesis of ED-AuNPs using EDAE is shown in Figure 4. The absorption spectra of ED-AuNPs increases with increasing the reaction temperature accompanied with a blue shift at 60°C compared to NPs synthesized at 30°C (Figure 4). Moreover, a decrease in the size of ED-AuNPs upon increasing the reaction temperature is observed as it is evident from the sharp and narrow SPR peaks. The obtained results are consistent with the previous findings reporting the increased reaction rate of NPs synthesis upon increasing the reaction temperature, indicating that it is tightly temperature-dependent.80–82 Most likely these findings are attributed to the effect of high reaction temperature that leads to the accelerated nucleation of metallic NPs implying the improved expenditure of most of the metal ions with the lowest extent of secondary reduction of the preformed nuclei.81,83 UV/Vis spectra as a function of temperature of ED-AuNPs synthesized using 1.4 × 10−4 M HAuCl4 and 1.0 mL EDAE.
Effect of pH
In addition to the previously mentioned factors, it has been reported that pH value of a plant extract can greatly affect the reduction process involved in the biosynthesis of NPs.75,79 Actually, the production of nucleation centers is affected by pH in a manner similar to temperature, where their number and the consequent production of metallic-based NPs increase as the pH increases. 84 Notably, the size and shape of NPs vary with the pH of the medium, where a change in pH leads to a change in charge of phytochemicals available within the plant extract. Hence, the ability to bind and reduce metal cations during production of NPs is affected.75,85,86
In the current study, the impact of pH on the biosynthesis of ED-AuNPs was investigated using UV/Visible spectrometry in the range from pH 2 to 12 (Figure 5). Initially, at pH values of 2 and 3, the absorption spectra showed two SPR absorption peaks. The peak at 554 nm was characteristic of the transverse mode of plasmon resonance of spherical AuNPs. On the other hand, that observed at around 820 nm was characteristic of the longitudinal mode of plasmon resonance of anisotropic AuNPs that are either nanorods, triangular or hexagonal shaped. This indicates the formation of larger and polydispersed AuNPs in the solution due to the agglomeration of the AuNPs. At pH values from 4 to 9, a blue shift occurs from 543 to 523 nm due to the decrease in size of AuNPs and the peak at longer wavelength disappeared, thus indicating the formation of uniform NPs that are smaller in size. By increasing the alkalinity of the surrounding media above pH 9, a change in the electron density on the surface of AuNPs is induced. Hence, it affects surface plasmon band and band intensity decreased, where a decrease in the absorption to about 50% and a slight shift in absorption band to 545 nm were observed at pH 12. Consequently, pH 9 of the plant extract was regarded to be the optimum pH whereas the smallest monodispersed ED-AuNPs (in the range of 5–10 nm) were obtained. The possible explanation of the obtained results might be based on the fact that in alkaline medium the rate of NPs formation is higher than the rate of their aggregation, while the aggregation of NPs exceeds the nucleation process in acidic medium.
87
Our results are in accordance with former studies which reported a decrease in the size of AuNPs by increasing pH.74,88,89 On the contrary, optimum acidic or neutral pH has been previously reported for the biosynthesis of AuNPs from other plant extracts.90–92 UV/Vis spectra as a function of pH of ED-AuNPs synthesized using 1.4 × 10−4 M HAuCl4 and 1.0 mL EDAE.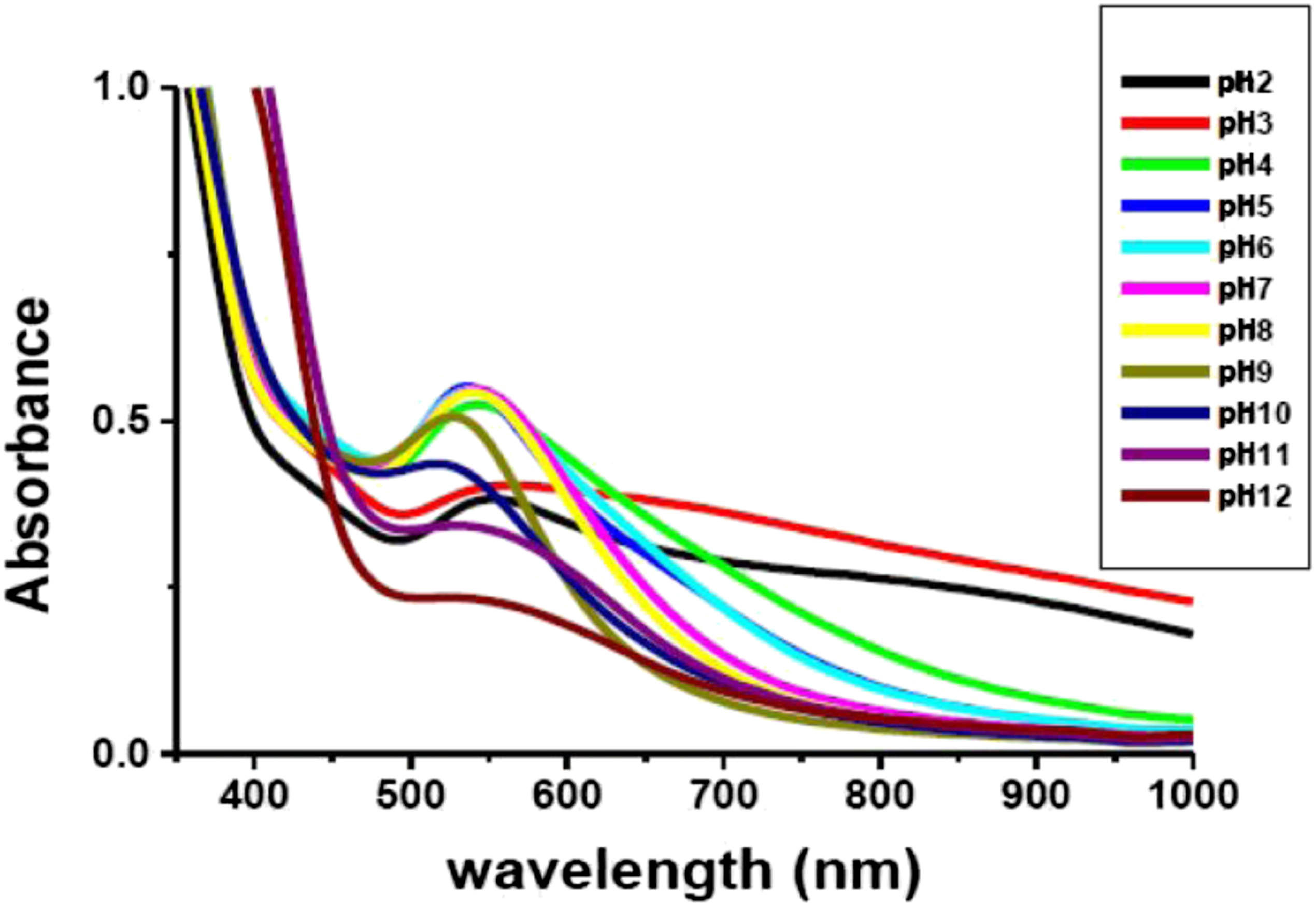
Indeed, it was concluded that formation of MNPs is highly dependent on the solution pH, as it controls the size and morphology of the biosynthesized NPs. Moreover, it affects the amount and stability of the produced NPs as well as influencing the rate of the reduction reaction.75,93 To that extent, the size of the biosynthesized ED-AuNPs was approved by TEM images taken at pH 3 and 9 (Figure 6). The obtained results revealed that the pH of the plant extract has a significant impact on the average particle size of the biosynthesized AuNPs, where the NPs prefer to aggregate at low pH values to form larger ones in preference to nucleate and form new NPs. The average size of the biosynthesized ED-AuNPs nanoplatelets at pH 3 (20-nm-sized particles) was larger than that at pH 9 (7-nm-sized particles) with different morphologies and regular spherical shapes, respectively. TEM micrographs of ED-AuNPs (a1) at pH 3 and (a2) at pH 9. The scale bar corresponds to 100 nm.
X-Ray diffraction analysis
Confirmation of the crystalline structure of the biosynthesized ED-AuNPs was achieved using X-Ray diffraction (XRD) analysis and the typical XRD patterns are shown in Figure 7 for ED-AuNPs synthesized using 1 mL extract at pH 9. The diffraction peaks observed at 2θ values = 38.17, 44.31, 64.44, and 77.34 could be assigned to the 111, 200, 220, and 311 crystallographic planes, respectively. These peaks are identical with those reported for the standard gold metal (Au0) (Joint Committee on Powder Diffraction Standards-JCPDS, USA). Hence, face center cubic (fcc) structure of the AuNPs synthesized using EDAE is confirmed. The Bragg’s peak broadening in the XRD pattern distinctly indicates the formation of small-sized NPs.94,95 The average crystallite size of AuNPs estimated using Scherrer equation and calculated using all four peaks was found to be 8.4 nm and is in good agreement with the average particle size of 7 nm obtained from the TEM image at 1 mL of extract concentration at pH 9. XRD pattern of ED-AuNPs synthesized using EDAE.
Fourier transform infrared spectroscopy
In the present study, an attempt was made to identify the plausible biomolecules implicated in the formation, capping, and stabilization of biosynthesized NPs via FTIR analysis. Fourier transform infrared spectrum of EDAE revealed peaks at 3426.6, 2959.1, 2853.3, 1739.4, and 1630 cm−1 (Figure 8(a)). The strong peak located at 3426.6 cm−1 is associated with O-H vibration in the biomolecules. The CH2 anti-symmetric and symmetric stretching vibrations were observed at 2959.1 and 2853.3 cm-1, respectively. The IR peak located at 1739.4 cm−1 is assigned to the carbonyl (C=O) group, while the one at 1630 cm−1 is assigned to amide I vibrations. In case of ED-AuNPs, FTIR spectra showed narrower peaks that are shifted relative to EDAE to 3395.5, 1727.7, and 1620.1 cm−1 (Figure 8(b)). Primary alcohols and carbonyl groups may be implied in the reduction as well as capping of the biosynthesized AuNPs.96–98 The presence of flavanones or terpenoids is indicated by the carbonyl, as they are adsorbed on the surface of metal nano-sized particles via interaction through the carbonyl groups. Carbonyl groups in proteins and amino acids have previously been shown to bind with MNPs or act as capping and stabilizing agents.99–101 Comparative FTIR spectra of (a) EDAE and (b) ED-AuNPs.
Thermogravimetric analysis of ED-capped AuNPs
Thermal stability of ED-AuNPs was assessed by TGA analysis. As shown in Figure 9, a steady loss of weight was observed up to 500°C. The recorded weight loss of ED-AuNPs was around 89.08% for up to 500°C (Figure 9). The perceived thermal behavior was most likely a consequence of the surface desorption of bio-organic compounds present in NP powder. Hence, it was anticipated that ED-AuNPs were formed of molecules involved in the reduction process of metal ions and that the EDAE is in charge of stabilizing the biosynthesized NPs. Comparable thermal behavior of biosynthesized AuNPs has been previously reported.102–104 Thermogravimetric analysis of capped AuNPs using EDAE.
Biological Activities of EDAE and the biosynthesized ED-AuNPs
Cytotoxic activity
Compared to conventional anticancer agents that lead to systemic toxicity and adverse side effects, nanotechnology-based promising anticancer agents could provide a less invasive alternative, increasing the patient’s life expectancy and quality of life due to their exceptional capabilities. 105 The noble metallic NPs, such as AuNPs, have been employed in a variety of biomedical applications, including anticancer. 13 Indeed, AuNPs are considered the most widely used NPs in nearly all aspects related to cancer management and research.106–108
In the current study, the in vitro anticancer effect of the biosynthesized NPs was assessed against the human hepatocellular carcinoma (HepG2) and the human colon carcinoma (HCT-116) cell lines using MTT assay. Comparative percentage of cell viability of DOX (+ ve control), ED-AuNPs, and EDAE is shown in Figure 10. Results obtained revealed that ED-AuNPs and EDAE exerted significant cytotoxic effect on the investigated cancer cells in a dose-dependent manner. A significant difference (p < .05) in the inhibitory effect on HepG2 as well as HCT-116 cancer cells between ED-AuNPs and EDAE was observed. The IC50 values, presented in Table 1, ranged from 0.38 ± 0.07 to 69.83 ± 0.96 µg mL−1. Similar results has been reported regarding the anticancer efficacy of biosynthesized AuNPs using different plant extracts against different cell lines.109–112 Comparative analysis of the cytotoxic effect of DOX, ED-AuNPs, and EDAE against (a) HepG2 and (b) HCT-116 cell lines. ns: not significant, **: p < .01, and ****: p < .0001. In-vitro cytotoxic activities of DOX, ED-AuNPs, and E. dendroides aqueous extract (EDAE) against HepG2 and HCT-116 cell lines. Determination of IC50 values (50% inhibition of cancer cell growth in μg mL−1) were performed in triplicate and expressed as mean values ± SD.

Anti-diabetic activity
Globally, diabetes mellitus (DM) is regarded as one of the most common metabolic disorders. 113 In spite of being available, most of the anti-diabetic therapeutic options are associated with multiple adverse side effects. Hence, attention has been paid toward herbal medicine, the use of which is associated with various concerns, including low degree of bioavailability. Consequently, the introduction of nanotechnology in the field of herbal medicine is considered an essential in order to eliminate the risks associated with conventional anti-diabetic medications. 114 The field of nanomedicine involves the management of diverse chronic non-communicable diseases including DM, using nanomaterials and nanostructures, which is considered an ultimate goal of all researchers due to their reduced side effects and better achievement of therapeutic efficacy. 115
In the current study, the anti-diabetic activity of ED-AuNPs compared to EDAE was evaluated. The biosynthesized NPs effectively inhibited intestinal α-glucosidase enzyme activity in a dose-dependent manner (Figure 11). The α-glucosidase inhibitory activity of ED-AuNPs exhibited the highest inhibitory activity (66.5% inhibition) when compared to EDAE (52.34% inhibition) with IC50 values of 19.8 ± 1.97 µg mL−1 and 214.67 ± 10.88 µg mL−1, respectively. The standard anti-diabetic drug acarbose has revealed the potent α-glucosidase inhibitory activity (90.1% enzyme inhibition) with an IC50 value of 20.66 ± 1.24 µg mL−1. Hence, the results obtained pointed to the promising antidiabetic potential of the biosynthesized ED-AuNPs. Biosynthesized AuNPs have previously been shown to exhibit similar in vitro antidiabetic effect in terms of α-glucosidase activity.67,116 Comparative analysis of the anti-diabetic effect of the anti-diabetic drug, acarbose, ED-AuNPs, and EDAE.
Anti-Helicobacter pylori activity
Helicobacter pylori (H. pylori) is a spiral Gram-negative bacterium known to be colonized in the stomach. It infects about half of the world population. Since its discovery, H. pylori infection has been linked to gastrointestinal disorders, including chronic gastritis, peptic ulcer, gastric cancer, and mucosa-associated lymphoid tissue (MALT) lymphoma.117–120 Moreover, various extra gastric diseases have been previously reported.121,122
The biosynthesized ED-AuNps and the aqueous extract of. E. dendroides (EDAE) were assessed for their anti-H. Pylori activity by the use of a micro-well dilution method and their MICs were compared to the standard drug, Clarithromycin, implied in anti-H. Pylori therapy (Figure 12). When compared to EDAE, ED-AuNPs exhibited a stronger bactericidal activity. The lower MICs besides the stronger antibacterial activities of ED-AuNPs (MIC = 15.63 μg mL−1) compared to EDAE (MIC = 125 μg mL−1) could be attributed to the smaller size and the presence of the miller index (111) in the cubic network of the former. Metal-based NPs provide a significantly large surface area surrounding the bacterial effluent, which is anticipated to improve the extent of bacterial elimination.123,124 Similarly, the antibacterial activity of the biosynthesized AuNPs against H. pylori has been previously reported.116,125 Comparative analysis of the anti-Helicobacter pylori effect of the antibiotic Clarithromycin, ED-AuNPs, and EDAE.
Conclusion
Owing to their unique physicochemical features, metal-based NPs have recently received considerable attention in biological applications. The purpose of the current study was to compare the potential pharmacological activities of E. dendroides aqueous extract (EDAE) and the gold nanoparticles synthesized from it (ED-AuNPs). To this extent, the presence of bioactive phytoconstituents was validated via established qualitative analyses. Moreover, FTIR spectra revealed the presence of reducing groups in EDAE that is utilized for AuNPs biosynthesis. Both the biosynthesized metal-based NPs showed obvious cytotoxic effects against HepG2 and HCT-116 cells. Moreover, ED-AuNPs were elucidated as antidiabetic agents as well as they exhibited a potent bactericidal activity against H. pylori. Our findings imply that the ED-AuNPs synthesized from aqueous extract of E. dendroides aerial parts (EDAE) may represent an encouraging source for further research as feasible agents for pharmaceutical and medicinal applications.
Footnotes
Acknowledgments
This work was supported by the Deanship of Scientific Research, Vice Presidency for Graduate Studies and Scientific Research, King Faisal University, Saudi Arabia [Project No. Grant 23].
Author contributions
All authors contributed to the design and implementation of the research, the formal analysis and investigation of the results, and to the writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.



