Abstract
A novel series of 2-[2-(7-chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-phenylacetamide derivatives is synthesized via substitution with 2-mercapto-4-methyl-5-thiazoleacetic acid at position 4 of 4,7-dichloroquinoline to obtain an intermediate acetic acid derivative. The chemical behavior of these reactants was investigated using different reaction conditions to optimize the nucleophilic substitution at position 4. The final compounds are prepared using a modified version of the Steglich esterification reaction between the acetic acid intermediate

Introduction
Malaria is a mosquito-transmitted (female Anopheles) infectious disease caused by Plasmodium parasites. Five parasite species can cause malaria in humans. In particular, Plasmodium falciparum and Plasmodium vivax are the most dangerous. Around 219 million cases of malaria were accounted for in 2017, in 87 countries, along with 435,000 deaths, mostly in sub-Saharan Africa.1–3
Vector control has been used as a preventive measure in malaria. However, mosquitos have become resistant to pesticides as well as there being an increase in parasite resistance, and consequently, the number of cases has not dropped. 4
Cancer has been defined as a severe disease caused by the uncontrolled growth of cells, tissue destruction, and death. It is the second principal cause of death worldwide. In 2018 alone, 17 million new cases were reported along with 9.6 million deaths. 5 Since cancer is a complex disease, treatment strategies that involve surgery, radiotherapy, and chemotherapy result in limited efficacy depending on the stage and type of cancer. Aggressive therapy is also associated with a significant number of side effects, and consequently, life quality is reduced. 6
Chloroquine (CQ), available since 1947, and its equivalents, have been successfully used for treatment of malaria infection.7–9 Despite widespread resistance to CQ in most of the malaria-endemic areas, compounds with the quinoline scaffold remain an important class of potential antimalarial and anticancer drugs that require more considerable research.10,11
A valuable approach to obtain novel antimalarial and anticancer drugs is represented by the assembly of fused molecules with a dual mechanism of action.12–17 A further approach refers to a superimposition in a single molecular scaffold of the structural features responsible for the activity of different antimalarial and anticancer agents.18–23
In our previous reports,24–27 different quinoline compounds were designed and synthesized. The in vitro and in vivo biological activities of these compounds showed promising potential as antimalarial and anticancer compounds.
We report herein the synthesis of a hybrid scaffold by incorporating 2-mercapto-4-methyl-5-thiazoleacetic acid (
Results and discussion
Chemistry
The synthetic route to compounds

Synthesis of 2-[2-(7-chloroquinolin-4-ylthio)-4-methylthiazol-5-yl)-N-phenylacetamide derivatives
Additional support for the assignment of the structure was based on 1H NMR data, which showed the presence of a singlet for the methyl protons at 2.32 ppm, a singlet at 3.87 ppm due to the methylene protons (2H,
The corresponding N-phenylacetamide derivatives
Data from 1H NMR and 13C NMR spectroscopy were used to identify the products. The 1H NMR spectra showed the presence of singlets for methyl protons (4-methylthiazol-5-yl) at 2.42–2.48 ppm, singlets at δ 3.78–3.92 ppm due to the methylene protons, the methoxy protons at 3.72–3.84 ppm, and methyl protons at 2.06–2.30 ppm indicating that the N-phenylacetamide had formed. All the structures reported were confirmed by 13C NMR spectroscopy, with the chemical shifts at 166.0–168.5 ppm confirming the existence of the
The IR spectra of compounds
Antimalarial activity
The novel synthesized compounds
The half maximal inhibitory concentrations (IC50) of quinoline derivatives for the formation of β-hematin (βHF) and the effect on Plasmodium berghei–infected mice (20 mg kg−1).

Com.: compounds
p < 0.001 compared to CiSS. n = 6.
Compounds
Structures
Cytotoxicity evaluation
All the synthesized compounds were assessed for cell death induction in vitro. The cell lines A549 (human lung tumor), MCF-7 (breast tumor), and normal human peripheral blood mononuclear cells (PBMCs), isolated from healthy donors, were used. The Bioethical Committee of the Institute of Immunology approved the study. Written consent was obtained from each donor.
Doxorubicin, CQ, and As2O3 were used as the reference drugs, and the results are summarized in terms of IC50 values (Table 2). Compounds
The activity of quinoline derivatives on cell viability.
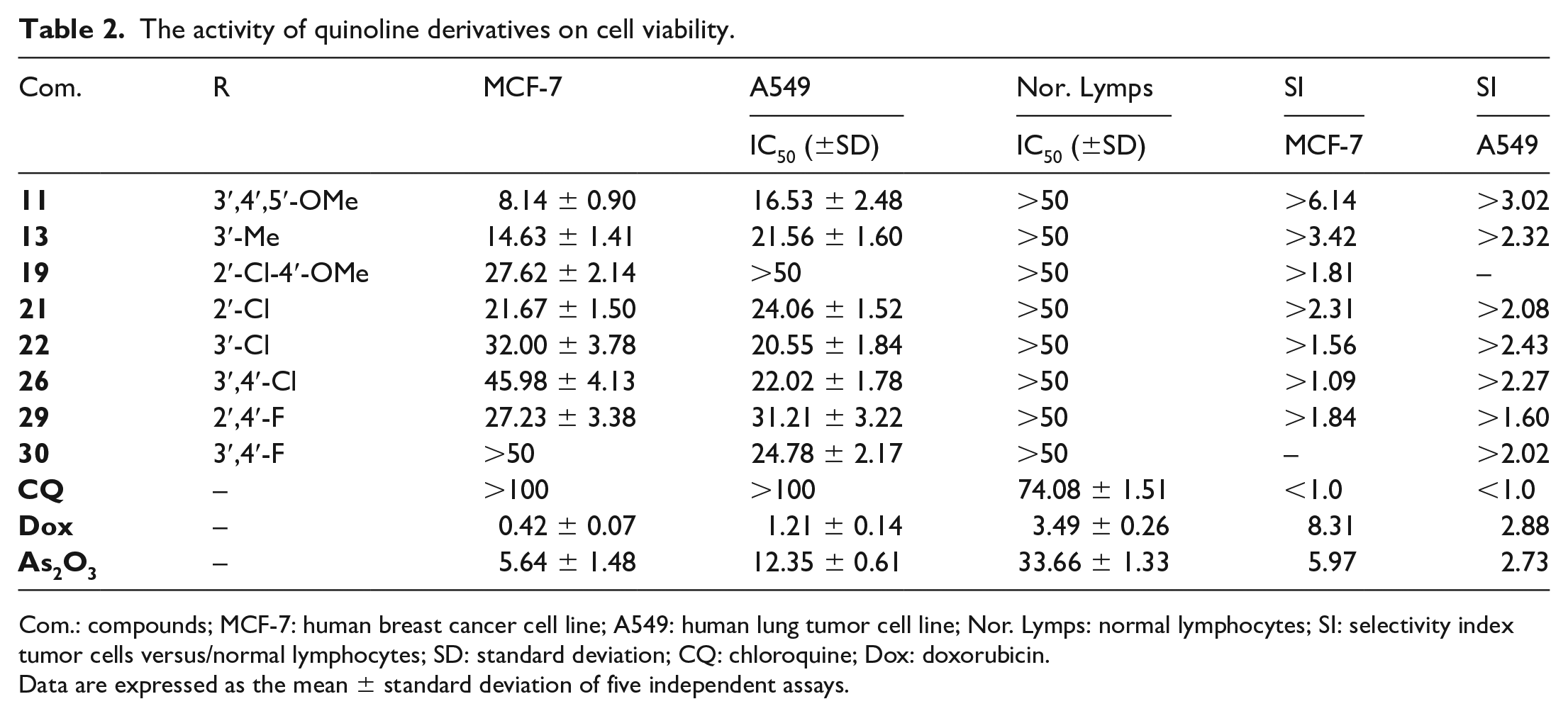
Com.: compounds; MCF-7: human breast cancer cell line; A549: human lung tumor cell line; Nor. Lymps: normal lymphocytes; SI: selectivity index tumor cells versus/normal lymphocytes; SD: standard deviation; CQ: chloroquine; Dox: doxorubicin.
Data are expressed as the mean ± standard deviation of five independent assays.
We can see that the type of substituent on the phenyl ring had a significant effect on the potential antimalarial and antiproliferative activities of the target compounds. When the phenyl group had three methoxy substituents at positions 3, 4, and 5, the corresponding compound exhibited excellent activity as an inhibitor of β-hematin formation, and as an antimalarial in vivo, and an antiproliferative in vitro. When the phenyl group was mono-substituted at position 3 with a methoxy group, the activity as a potential antimalarial compound was comparable to the trimethoxy-substituted compound. However, it did not exhibit antiproliferative activity against the two cancer cell lines. Compounds di- or mono-substituted with methoxy groups at other positions were not effective. It was found that when the phenyl group was substituted with Me, Cl, F, CF3, or Br, the corresponding compounds were not able to inhibit β-hematin formation in vitro, but exhibited moderate antiproliferative responses against the two tumor cell lines in vitro.
Our studies have demonstrated that compound
Conclusion
In summary, 29 novel compounds were obtained after substitution at position 4 of the 4,7-dichloroqunoline (
Experimental
NMR spectra were recorded on a JEOL EclipseTM 270 MHz for 1H NMR and at 67.9 MHz for 13C NMR using CDCl3 or dimethyl sulfoxide (DMSO)-d6, and are reported in ppm downfield from the residual CHCl3 or DMSO. Elemental analyses were obtained using a PerkinElmer 2400 CHN elemental analyzer; the results were within ±0.4% of the calculated values. A NicoletTM IS5 FTIR (ID3 Zn-Zr) spectrophotometer was used to determine the IR spectra. The melting points (m.p.) were determined by using a Thomas micro hot-stage device. The 4,7-dichloroquinoline and 2-mercapto-4-methyl-5-thiazoleacetic acid were purchased from Sigma-Aldrich Group, USA. Solvents were used directly or distilled and dried in the usual manner. Thin-layer chromatography was performed on Merck silica gel F254 (0.255-mm plates), and spots were analyzed by UV fluorescence at 254 nm.
Synthesis of 2-[2-(7-Chloroquinolin-4-ylthio)-4-methyl-thiazol-5-yl]acetic acid (
General technique for the synthesis of 2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-phenylacetamide derivatives 5-32
To a solution of 2-[2-(7-chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]acetic acid (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-phenylacetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2-methoxyphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3-methoxyphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(4-methoxyphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2,4-dimethoxyphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2,5-dimethoxyphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3,4,5-trimethoxyphenyl)-acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-o-tolylacetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-m-tolylacetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-p-tolylacetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2,4-dimethylphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2,5-dimethylphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3,4-dimethylphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3,5-dimethylphenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3-chloro-4-methoxyphenyl) acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(5-chloro-2-methoxyphenyl) acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2-chlorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3-chlorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(4-chlorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2,3-dichlorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2,4-dichlorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3,4-dichlorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3-fluorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(4-fluorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(2,4-difluorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(3,4-difluorophenyl)acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-[3-(trifluoromethyl)phenyl] acetamide (
2-[2-(7-Chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-(4-bromophenyl)acetamide (
Antimalarial activity
Inhibition of β-hematin formation
The assay was performed according to previously reported protocols.26,29,35 Hemin chloride (50 µL, 4 mM) in DMSO (5.2 mg mL−1) was pipetted into 96-well microplates. Different concentrations (5–100 mM) of the compounds in DMSO were added in triplicate (50 µL). Water (50 µL) or DMSO (50 µL) was used as control. Acetate buffer (100 µL, 0.2 M, pH 4.4) initiated the formation of β-hematin. Next, the plates were incubated at 37 °C for 48 h and subsequently centrifuged (4000 r min−1 × 15 min). The supernatent was washed twice with DMSO (200 µL) and resuspended in NaOH (200 µL, 0.2 N). The solubilized products were diluted (1:2) with NaOH (0.1 N), and the plates were read in an enzyme-linked immunosorbent assay (ELISA) reader at 405 nm (Microplate Reader, BIORAD-550). The results are expressed as %IβHF.
Parasite, the experimental host and strain maintenance
Male Balb-C mice, weight 18–22 g, were fed with a commercial diet at libitum under standard procedures of animal care following the aforementioned method approved by the Ethics Committee of the Institute of Immunology. The animals were infected with a rodent malaria strain of P. berghei ANKA. A million infected erythrocytes, in phosphate-buffered saline solution (PBS, 10 mM, pH 7.4, 0.1 mL), were inoculated intraperitoneally to infect the animal. The parasitemia was inspected by continuous microscopic examination of Giemsa-stained smears.26,29,36
Four-day suppressive test
One million P. berghei, injected in the caudal vein intravenously, were used to infect the mice (18–23 g, n = 6). After 2 h of inoculation, the compounds that were active in vitro were dissolved in DMSO (0.1 M) and subsequently diluted with Saline-Tween 20 solution (2%) and administered intraperitoneally for 4 days (10 mg kg−1). The parasite load was evaluated on day 4 by examining Giemsa-stained smears. The positive control was CQ (10 mg kg−1), and the negative control was saline solution. Non-treated mice were used as a baseline control for survival times.26,29,30 The percentage of parasitemia was used to express the results.
Antitumor activity
Cell lines
The human cell lines, obtained from the American Tissue Culture Collection (ATCC), MCF-7 (human breast cancer cell line) and A549 (human lung tumor cell line), were maintained in Roswell Park Memorial Institute (RPMI) 1640 media supplemented as recommended by the ATCC. The experimental analyses were performed by culturing 3 × 105 cells/well with the different compounds in 96 well microtiter plates. Five different assays in triplicate were performed per compound.26,29
Isolation of human total lymphocytes
Normal volunteers’ blood was used with written consent, as specified by the Ethical Committee at the Institute of Immunology, in order to obtain PBMCs by the standard Ficoll-Paque gradient (Histopaque 1077, Sigma, Poole, UK) as described previously.26,29 The cells, 85% T lymphocytes, 8% B lymphocytes, and 7% natural killer (NK) cells, were used as the primary culture for assessment of compound specificity. The selectivity index refers to the ratio of IC50 required to induce cell death in healthy cells divided by the IC50 required to induce cell death in tumor cell lines. Ideal compounds have high selectivity indices.
The 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide viability assay
The protocol used is a slight modification of Mossmann’s protocol
37
and was described previously.
38
Specifically, different concentrations of compounds
Data analysis
One-way analysis of variance (ANOVA) and t tests for specific group comparisons were used for data analysis. The program used was Graph Pad Prism 4.02. 39
Supplemental Material
Suplementary_matterial – Supplemental material for Synthesis and biological activity of 2-[2-(7-chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-phenylacetamide derivatives as antimalarial and cytotoxic agents
Supplemental material, Suplementary_matterial for Synthesis and biological activity of 2-[2-(7-chloroquinolin-4-ylthio)-4-methylthiazol-5-yl]-N-phenylacetamide derivatives as antimalarial and cytotoxic agents by Hegira Rámirez, Juan R Rodrigues, Michael R Mijares, Juan B De Sanctis and Jaime E Charris in Journal of Chemical Research
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Instituto de Investigaciones Farmacéuticas (IIF) (grant IIF.01-2014) and Consejo de Desarrollo Científico y Humanistico-Universidad Central de Venezuela (CDCH-UCV) (grant PG. 06-8627-2013/2).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
