Abstract
A cheap, reproducible, environmentally friendly, and biocompatible method for stabilizing and storing gold nanoparticles for prolonged time is presented. Adding a diluted pectin solution immediately after the synthesis of gold nanoparticles increases the stability of the aqueous nanoparticle dispersion, avoiding agglomeration and further precipitation, regardless of the initial size and shape of the nanoparticles. When the gold nanoparticles–pectin solution is freeze-dried, a sponge-like solid is obtained, improving significantly the pectin capacity to stabilize gold nanoparticles. Because the gold nanoparticles are immobilized into the freeze-dried pectin matrix, the size, size distribution, and shape remain unchanged, regardless of how long the solid is stored. The solid can be redispersed in water to obtain a solution that is able to maintain the stability of the gold nanoparticles, even for more than 20 weeks. This solution can be freeze-dried again, with no changes in the gold nanoparticles size. The proposed method is an alternative for stabilizing nanoparticles with no toxic agents, increasing significantly the storage time.
Introduction
In the last years, several studies have been focused on the effect of aging in the stability of nanoparticles. 1 –8 Agglomeration, precipitation, and even chemical–physical decomposition are commonly observed when aqueous suspension of nanoparticles is stored, 2 –4,9 forcing to use only freshly prepared nanoparticles, which limits their application in the real world. 5,10,11 Taking into account that the properties of nanoparticles depend on the size, it is highly recommended to use a stabilization method able to avoid agglomeration, even for long storage time. Several approaches proposed consist in the stabilization by adding surfactants. 12 –14 In this work, we present an alternative method based on pectin for stabilizing and storing gold nanoparticles (AuNPs). AuNPs have been extensively studied due to their wide application fields, 15 for example, drug delivery, 16 catalysis, 17 photonics, 18 and chemical sensing. 19 Pectin is a biopolymer with carboxyl and hydroxyl groups, which interact with the surface of metallic nanoparticles, avoiding agglomeration. 20,21 In the proposed method, a diluted pectin solution is added to a freshly prepared AuNPs, forming an aqueous solution able to maintain the initial size and shape of the AuNPs. The storage properties of the system are increased significantly when the solution is freeze-dried to obtain a solid-like sponge containing AuNPs. The advantages of the storage method based on freeze-dried pectin are readily evident: (i) It is able to maintain the size, size distribution, and shape regardless for how long it is stored; (ii) small storage volume is obtained even for high AuNPs concentration; and (iii) there is no need of special storage condition. When the freeze-dried solid is redispersed in water, AuNPs–pectin aqueous solution is obtained again, maintaining the size and shape of the AuNPs. Different cycles of freeze-dried and redispersion can be carried out, with no changes in the stability properties of the pectin. This is a cheap, fast, environmentally friendly, and reproducible method that is a good alternative to the use of surfactants to stabilize nanoparticles.
Materials and methods
Pectin (Aglupectin LA-S20) and a gold bar (99.99%) were purchased from Silvateam Co (supplied by Johe Productos Químicos S.A. de C.V., León, Guanajuato, México). and a local jewelry, respectively. Gold (III) chloride trihydrate (99.9%), nitric acid (90%), hydrochloric acid (37%), ascorbic acid (99%), and sodium citrate (99%) were purchased from Sigma-Aldrich (supplied by RG Laboratorios, Guadalajara, Jalisco, México).
Synthesis of gold chloride
Solution A1 was prepared by dissolving 1 g of a gold bar in a mixture of 2 mL of nitric acid and 9 mL of hydrochloric acid. Once the gold bar is completely dissolved, the solution is heated to evaporate the acids, obtaining a precipitate of HAuCl4·3H2O. The precipitate 1.48 g was dissolved in 20 mL of water. The solution was filtered with a 450 nm microfilter to separate the impurities. The final concentration of solution A1 was approximately 0.18 M. As a comparison, a solution of the commercial reagent gold (III) chloride trihydrated at the same concentration was prepared (solution A2).
Preparation of pectin stock solution
Eight grams of pectin was added slowly and under stirring to 200 mL of water previously heated at 55°C. Once the pectin is completely dispersed, the solution is maintained under stirring at 55°C for 15 min. Then, also under stirring, the solution was gradually cooled down to room temperature.
Synthesis of AuNPs with ascorbic acid
To synthesize the AuNPs, 1 mL of a solution 7.5 mM of ascorbic acid is added to 10 mL of water. Under stirring, 1 mL of solution A1 (or A2) 5 mM is added to the ascorbic acid solution by means of a peristaltic bomb (0.025 mL/s). The color of the solution rapidly changes to deep red, indicating the formation of the AuNPs. Four seconds after solution A1 (or A2) is completely incorporated, 2.5 mL of pectin stock solution is added. Finally, 5.5 mL of water is added to the previous solution to reach a final volume of 20 mL (solution B). As a control sample, a synthesis of AuNPs was carried out following the same procedure, without adding the pectin stock solution. The control samples were stored at 4°C.
Synthesis of AuNPs with sodium citrate
Four milliliters of solution A1 (or A2) 5 mM is added under stirring to 45 mL of water. The solution is heated to reach the boiling point. Then, 1 mL of sodium citrate solution 0.17 M is added, maintaining the stirring and heating for 10 min more, until the solution changes to a deep red color. The solution is cooled down at room temperature. Then, water is added to the solution to reach a volume of 50 mL. Finally, under stirring, 7.2 mL of pectin stock solution is added (solution C). In this case, control samples were also taken for the AuNPs dispersion prior the addition of the pectin solution. These samples were stored at 4°C.
Freeze-drying and redispersion in water
Twenty milliliters of solution B (or C) was freeze-dried in an ilShin apparatus, (TFD5503 model supplied by RG Laboratorios, Guadalajara, Jalisco, México), at −40°C and 4 Pa for 24 h. The final product is a sponge-like solid (solid A) formed by the mixture of pectin and AuNPs. When solid A is redispersed in water, solution B is obtained again. Five cycles “solution B–solid A–solution B, and so on” were performed with no evidence of nanoparticle agglomeration or precipitation.
UV–vis, zeta potential, TEM, and XRD
Ultraviolet-Visible spectroscopy (UV-vis) measurements were performed on an HACH DR3900 spectrometer (supplied by HACH México, Tlalneplantla de Baz, State of México, México). A Malvern ZetaSizer Nano ZS (supplied by Malvern Instruments México, Mexico City, México) was used to measure the Z potential. Samples were measured after a storage time of 1 week. Transmission electron microscopy (TEM) images were obtained in a microscope JEOL (JEM-1010 model, supplied by JEOL de México, México City, México) operating at 100 kV. X-ray diffraction (XRD) patterns were recorded with a PANalytical X-ray diffractometer (Empyrean model, provided by PANalytical México, San Pedro Garza García, Nuevo León, México) using a Cu-Kα radiation (λ = 0.15418 nm) at room temperature in the 2θ range 30–90°.
Results and discussion
The surface plasmon resonance (SPR) peaks measured by UV–vis spectroscopy can be related to the size and shape of metallic nanoparticles 22 ; thus, SPR peaks at the same wavelength indicate similar sizes. In Figure 1, the UV–vis absorption spectra for one sample prepared with ascorbic acid at different stages are shown: (a) before freeze-drying, (b) redispersed in water after freeze-drying, and (c) 20 weeks after redispersion in water. The SPR peaks are similar for the sample before and after freeze-drying, suggesting that this procedure does not affect the size and shape of AuNPs. The same behavior was observed for the AuNPs synthesized with solution A2 (data not shown), proving that the gold bar dissolution method is a good alternative to obtain a gold ions solution when it is not possible to purchase the commercial reagent. Samples stored for 20 weeks after redispersion in water show a small red shift (3 nm), indicating that agglomeration is negligible even when the pectin–AuNPs mixture is in solution. A representative spectrum of a control sample is also shown in Figure 1. This measurement was carried out after a storage time of 20 weeks. The SPR peak at 733 nm indicates the presence of larger particles than those stabilized by pectin. The broad signal can be attributed to polydispersity of the sample and coupling between the surface plasmons of the individual nanoparticles aggregates. 23 This observation is confirmed by TEM.
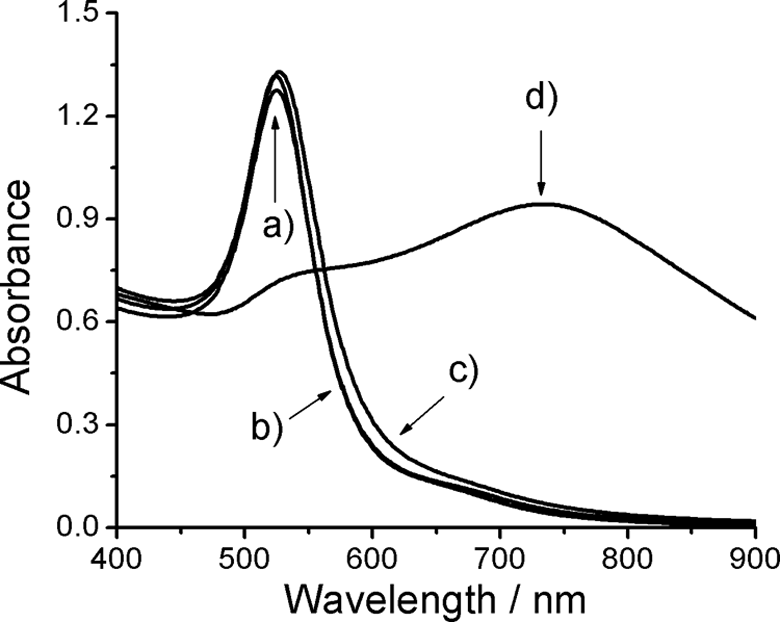
Absorbance spectra of the pectin–AuNPs mixture in solution: (a) before freeze-drying, (b) redispersed in water after freeze-drying, (c) 20 weeks after the redispersion, and (d) AuNPs without pectin after a storage time of 20 weeks. AuNPs: gold nanoparticles.
Representative TEM micrographs for samples prepared with ascorbic acid before (average size 34.1 nm) and after freeze-drying (average size 32.9 nm) are shown in Figure 2. In all cases, well-defined spherical nanoparticles were obtained. As it was observed in UV–vis spectra, the freeze-drying process has no significant effect on the AuNPs. The size distribution histograms are similar in both cases, proving that it is also possible to maintain the nanoparticle size distribution.
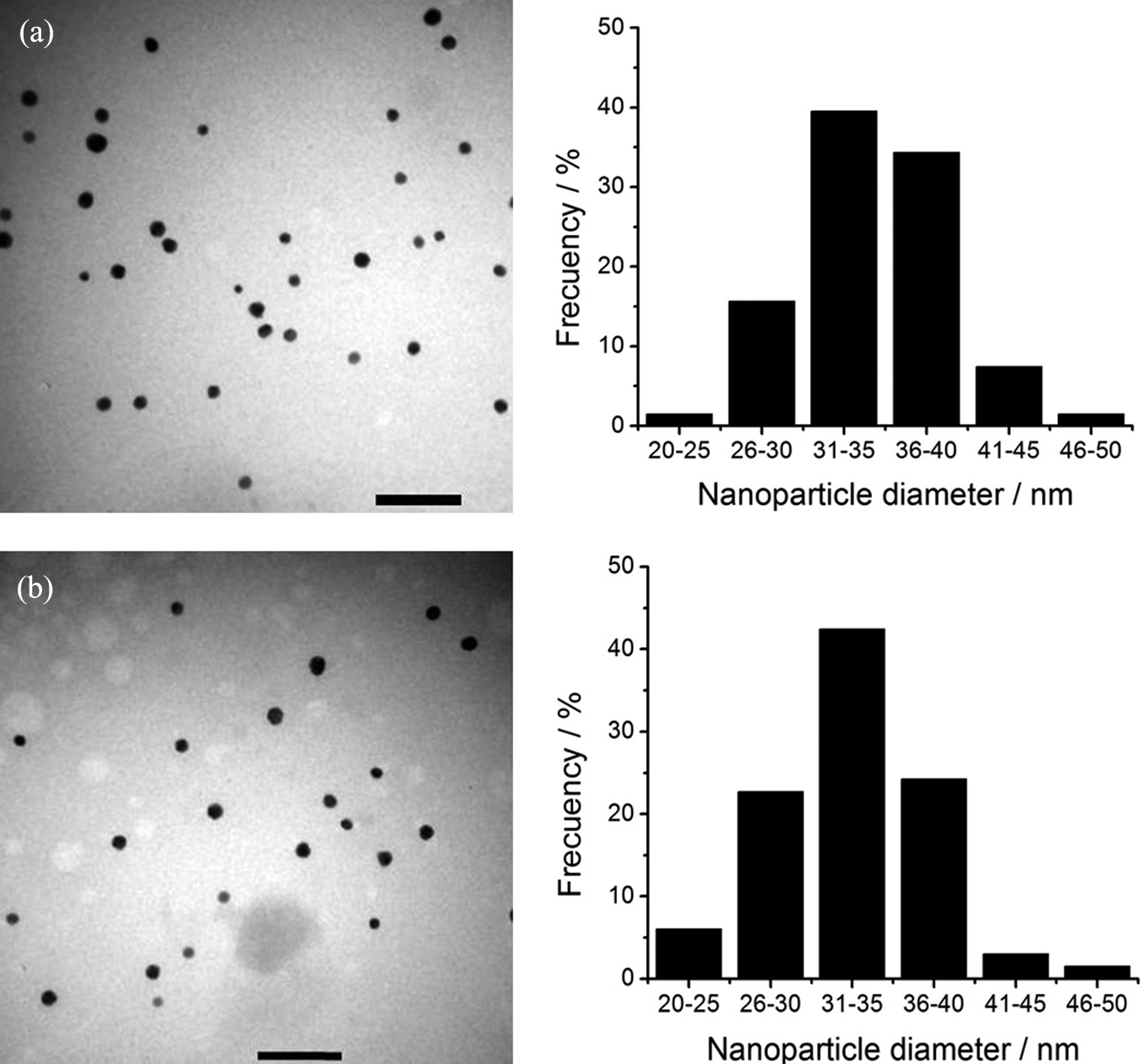
TEM micrographs and size distribution histograms of AuNPs reduced by ascorbic acid: (a) before freeze-drying and (b) redispersed in water after freeze-drying. Scale bars represent 200 nm. AuNPs: gold nanoparticles.
In order to determine if the proposed method is able to stabilize and store smaller nanoparticles, TEM micrographies of AuNPs prepared with sodium citrate were obtained, and they are shown in Figure 3. Not only the nanoparticles are smaller (average diameter 21.5 nm) than those synthesized using ascorbic acid but also they have a narrower size distribution. The presence of pectin agglomerates is evident; nevertheless, the AuNPs inside these agglomerates also maintain the same size than the rest of the nanoparticles. The size distribution histograms for samples before and after freeze-drying are also similar; thus, it can be concluded that pectin is able to avoid agglomeration when samples are freeze-dried and then redispersed in water, regardless of the initial size/size distribution of the AuNPs. This result is remarkable because it suggests that the capacity of pectin to stabilize nanoparticles does not depend on (i) the synthesis method; (ii) how long the AuNPs are maintained into the freeze-dried solid, and (iii) the amount of freeze-drying–redispersion cycles performed on the samples.

TEM micrographs and size distribution histograms of AuNPs reduced by sodium citrate: (a) before freeze-drying and (b) redispersed in water after freeze-drying. Scale bars represent 200 nm. AuNPs: gold nanoparticles.
Figure 4 shows the micrographs of control samples prepared with (a) ascorbic acid and (b) sodium citrate after a storage time of 1 week. Larger nanoparticles and agglomerates are observed. AuNPs synthesized with ascorbic acids are larger than those synthesized with sodium citrate, as it is expected taking into account that sodium citrate, besides being a good reducing agent, has stabilization properties. Control samples stored for longer time produce even larger nanoparticles. These results prove that adding pectin to fresh prepared AuNPs significantly increases the storage time, maintaining the nanostructure.

TEM micrographs of AuNPs without pectin after a storage time of 1 week. (a) AuNPs reduced by ascorbic acid and (b) AuNPs reduced by sodium citrate. Scale bars represent 200 nm. AuNPs: gold nanoparticles.
The interaction between AuNPs and pectin was followed by Z potential measurements. Absolute Z potential values above 30 mV provide good stability, because at these conditions, nanoparticles tend to repel each other. 24 This behavior is due to the adsorption of ions at the nanoparticle surface. The Z potential values for AuNPs synthesized with ascorbic acid and sodium citrate without pectin were −19.50 and −0.058 mV, respectively, far below the stability limit, which explain the growth and agglomeration observed in UV–vis and TEM. When pectin is added, the Z potential changes to −0.0151 and 0.078 mV, respectively, close to the point of zero charge. At this point, the electrostatic stabilization can be negligible due to the lack of repulsion forces between nanoparticles. These results, together with those obtained by UV–vis and TEM, suggest that the proposed systems are stabilized sterically. Large molecules, like pectin, tend to adsorb at nanoparticle surface through the carboxyl and hydroxyl groups, decreasing the Z potential, without affecting the stabilization properties. The capacity to maintain the size and shape of the AuNPs even after drastic temperature, pressure, and concentration changes, for example, freeze-drying and further redispersion in water, also supports that steric stabilization plays a major role in behavior of these systems.
XRD pattern of a freeze-dried sample is shown in Figure 4. The absence of diffraction peaks in the XRD pattern of the freeze-dried pectin indicates that the peaks obtained for the solid mixture are due only to the AuNPs. The major diffraction peaks can be indexed as the gold face-centered cubic phase based on the data of the Joint Committee Powder Diffraction Standard file no. 04-0784. The diffraction peaks of the AuNPs at 2θ of 38.4°, 44.6°, 64.7°, 77.7°, and 81.5° were indexed as (111), (200), (220), (311), and (222) crystalline plane diffraction peaks of gold, respectively (Figure 5).

XRD pattern for a freeze-dried mixture of AuNPs and pectin. As a comparison, XRD pattern for freeze-dried pectin is also included. AuNPs: gold nanoparticles.
Conclusions
Adding a diluted pectin solution to a dispersion of freshly prepared AuNPs in water avoids agglomeration, maintaining the original size, size distribution, and shape for a prolonged time regardless the initial nanoparticle size or synthesis method. This effect can be improved if the mixture AuNPs–pectin is freeze-dried. The obtained sponge-like solid containing AuNPs is a cheap and environmentally friendly method to store nanoparticles. By just adding water, the AuNPs can be redispersed for the required application. Several cycles of freeze-drying–redispersion can be performed on the pectin–AuNPs mixture, with no change in the nanoparticle size. This method can be easily extrapolated to stabilize and store nanoparticles of different metals.
Footnotes
Acknowledgment
The authors thank Silvateam Co. and its representatives in México Johe Productos Químicos for providing the pectin used in this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the University of Guadalajara, by means of the “Program for Financial Support to Improve the Production of SNI Members 2015” (PRO-SNI 2015).
