Abstract
Nitrocellulose/glycidyl azide polymer/2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane nanocomposites, in which 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane nanoparticles uniformly embedded in nitrocellulose/glycidyl azide polymer matrix, were synthesized using a sol–gel supercritical method. The micron morphology, crystal phase, molecular structure, specific surface area, and surface elements were characterized using scanning electron microscopy, X-ray diffractometry, infrared, Brunauer–Emmett–Teller, and X-ray photoelectron spectroscopy analyses, respectively. Thermal analyses were performed, and the kinetic and thermodynamic parameters, such as activation energy, per-exponent factor, rate constant, activation heat, activation free energy, and activation entropy, were calculated. The decomposition products of the nitrocellulose/glycidyl azide polymer matrix and nitrocellulose/glycidyl azide polymer/2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane were also investigated by differential scanning calorimetry–infrared analysis. The result indicated that the main decomposition product of nitrocellulose/glycidyl azide polymer is carbon dioxide and the –N3 group in glycidyl azide polymer decomposed to nitrogen without being detected by infrared spectrometer; the main decomposition products of nitrocellulose/glycidyl azide polymer/2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane are carbon dioxide, nitrous oxide, and water, and few carbon monoxide, methane, and nitrogen oxide are also detected. Energy performances of nitrocellulose/glycidyl azide polymer matrix and nitrocellulose/glycidyl azide polymer/2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane nanocomposites were evaluated, that is, the parameters such as standard specific impulse, characteristic speed, combustion chamber temperature, average molecular weight of combustion products, and explosion heat were calculated. The results illustrated that as the weight percentage of nitrocellulose increases, the values of standard specific impulse, characteristic speed, average molecular weight of combustion products, combustion chamber temperature, and explosion heat increase. This was ascribed to that the oxygen balance of glycidyl azide polymer is substantially lower than that of nitrocellulose, which results in that the chemical energy of glycidyl azide polymer does not release sufficiently. Additionally, as weight percentage of glycidyl azide polymer increases, the impact and friction sensitivity of the composites decrease obviously. This means that glycidyl azide polymer is much more insensitive than nitrocellulose.
Keywords
Introduction
The development of high-energy, low-sensitivity, low-cost explosives has long been the goal in the field of energetic materials. Update of energetic materials is an important indicator of update of weapons. 2,4,6,8,10,12-Hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane (CL-20) is the most outstanding nitroamine explosive with the highest density, the highest detonation velocity, the highest explosion heat, and high oxygen balance (OB). 1,2 It has cage-like molecular structure, which results in very high molecular tension. 3 Thus, the detonation performance of CL-20 is 10% higher than that of 1,3,5,7-tetranittro-1,3,5,7-tetrazocane (HMX). However, as a fatal defect, much high sensitivity substantially restricts the practical application of CL-20. Low safety is an inherent property of CL-20, and it is hard to change. Usually, there are two ways to alleviate this problem. One is the preparation of eutectic materials, that is, CL-20 co-crystallizes with a very insensitive explosive crystal to form an insensitive co-crystal. Ghosh and coworkers prepared CL-20/HMX co-crystal through solvent evaporation and proved that the performance of CL-20/HMX eutectic is more stable than CL-20 4 ; another discovery is that nano explosives have obviously lower sensitivities than the micron explosives. 5 So there are many researchers who try to fabricate nano-CL-20 by different methods. Bayat and Zeynali fabricated nano-CL-20 via precipitative crystallization. 6 Shang et al. prepared nano-CL-20 by the supercritical fluids method. 7 Guo et al. produced nano-CL-20 by mechanical milling method. 8 Similar studies were also reported by Wang et al. 9 and Bayat et al. 10 ; they prepared nano-CL-20 via ultrasonic spray-assisted electrostatic adsorption technology and micro emulsion method, respectively. Furthermore, to reduce the sensitivities of explosives, other nanoenergetics also were reported, such as nano-HMX, 11 –15 nano-RDX, 16 –18 nano-NTO, 19 nano-HNS, 20,21 and nano-TATB. 22,23
The energetic nanocomposites are another form of nanoenergetics. Although there are many preparation methods, the sol–gel method is the most commonly used one. It is because that the sol–gel method has many advantages, such as the convenience of low-temperature preparation, easy control over the stoichiometry and homogeneity of composites, and environment-friendly. 24,25 Moreover, the existence of the gel matrix effectively prevents the agglomeration of nanoparticles, controls the particle size at nanometer level, reduces the sensitivity of energetic materials, and enhances the energy and combustion performances. Previously, researchers used inert gel matrix such as ferric oxide, silicon dioxide, and Fluorine rubber (RF) to prepare nanocomposites. But this will bring a considerable penalty on energy performance. Thus, energetic gel matrix is a better choice to prepare nanocomposites.
Now, some nanocomposites using energetic materials as matrix have been fabricated, such as nitrocellulose (NC)/HMX,
26
glycidyl azide polymer (GAP)/HMX,
27
and RDX/GAP.
28
Because it is cheap and easily available, NC is the most widely used as the matrix. However, because NC has a high glass-transition temperature (
Experiment
Materials
NC (12.6% N, industrial grade) was purchased from Foshan Junyuan Chemical Co., Ltd (Foshan City, Guangdong Province, People’s Republic of China). CL-20 was purchased from Gansu Yinguang Chemical Co., Ltd (Baiyin City, Gansu Province, People’s Republic of China); GAP (number-average molecular weight = 4000, hydroxyl value of 0.49 mmol·g−1) was provided by the 42nd Institute of the Fourth Academy of China Aerospace Science and Technology Corporation. Ethyl acetate (purity of 99.5%) was purchased from Tianjin Fuyu Fine Chemical Co., Ltd (Tianjin city, People’s Republic of China). Toluene diisocyanate (TDI, purity of 98%) was purchased from Tianjin Guangfu Chemical Co., Ltd (Tianjin city, People’s Republic of China). Dibutyltin dilaurate (T-12, purity of 95%) was purchased from Tianjin Bodi Chemical Co., Ltd (Tianjin city, People’s Republic of China). Triethylenediamine (TED; purity of 98%) was purchased from Sinopharm Group Chemical Reagent Co., Ltd.
Synthesis of nanocomposites
NC, GAP, and CL-20 were dissolved completely in 20 mL of ethyl acetate at ambient temperature in various weight ratios (the ratios of NC:GAP:CL-20 were 0.67:0:0.33, 0.5:0.17:0.33, 0.33:0.34:0.33, 0.17:0.5:0.33, 0:0.67:0.33). Then, add the measured amount of TDI, the appropriate amount of TED solution (0.05 g of TED was dissolved into 1–2 mL of ethyl acetate), and T-12 (maintain the
The catalyst played an important role in the process. In general, there are two types of catalyst used in polyurethane production. Amine has been used in the early production of polyurethanes. TED is an amine catalyst commonly used. TED is an amine with weak alkalinity, but its steric hindrance is close to zero; so in practice, it exhibits excellent catalytic performance in promoting the polymerization reaction. The catalytic mechanism is shown in Figure 1(a). Tertiary amines easily undergo complex reaction with –NCO radicals to form complexes with high reactivity. This complex can quickly react with alcohol to produce carbamate and TED. Organotin catalysts such as stannous octoate, stannous oleate, and T-12 are commonly used for the production of polyurethanes and other products and are another catalyst in addition to amine catalysts. Organotin usually works at higher temperatures than amines. In theory, as shown in Figure 1(b), organotin can undergo coordination reaction with –NCO radicals to polarize it. After polarization, carbon atoms (positively charged in isocyanate molecules) exhibit high reactivity and are vulnerable to nucleophile attack. In the experiment, the use of these two catalysts alone did not achieve the desired effect. However, satisfactory results were achieved with both catalysts.
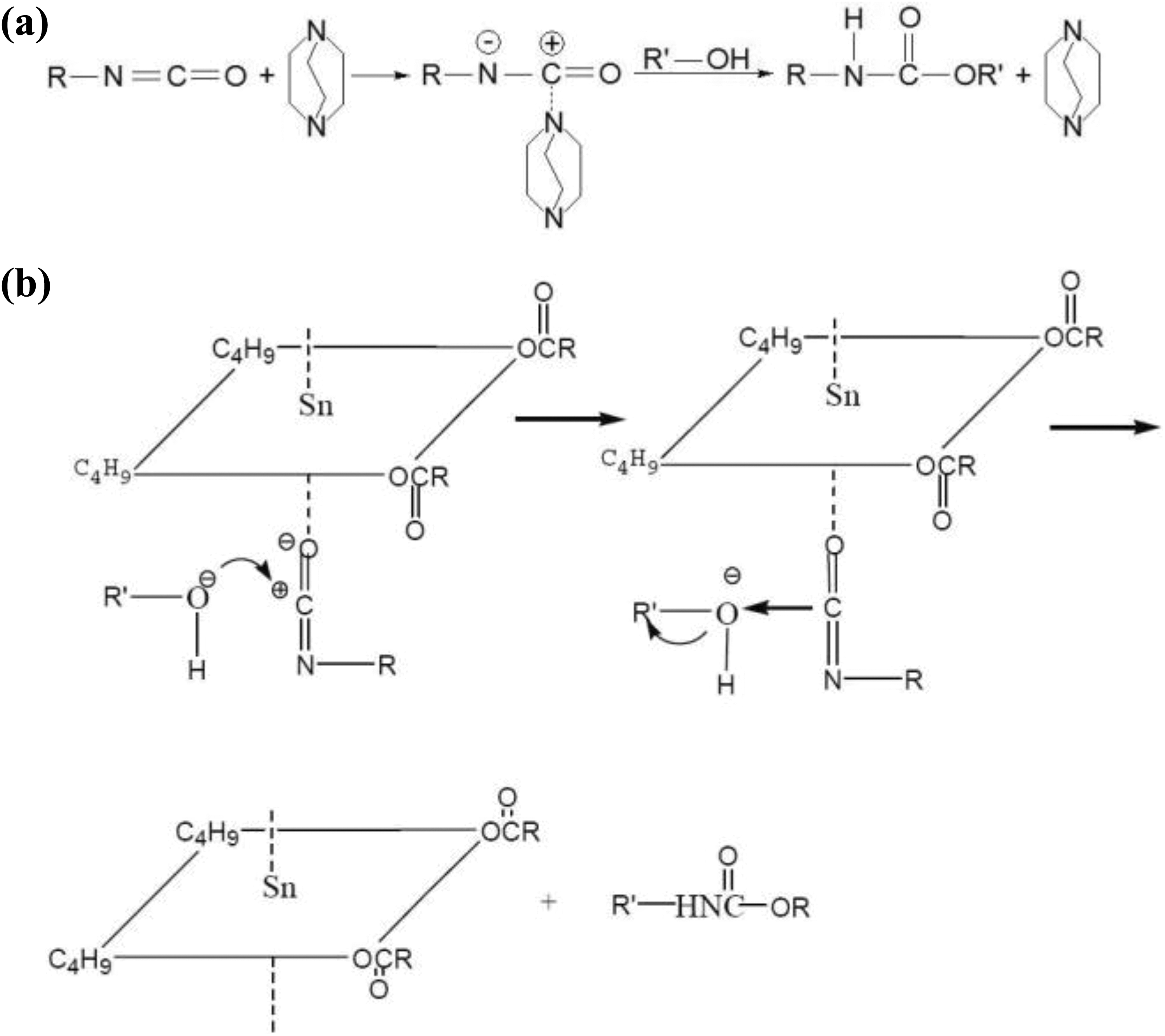
Catalysis mechanism of TED (a) and T-12 (b). TED: triethylenediamine; T-12: dibutyltin dilaurate.
Characterization and tests
The morphology was observed with a field-emission scanning electron microscope (SEM, JEOL JSM-7500, JEOL LTD.). The phases of the samples were investigated with an X-ray diffractometer (XRD, Bruker Advance D8, Bruker LTD.) using copper
The impact sensitivity of the samples was tested with an HGZ-1 impact instrument (HGZ-1 impact instrument). The special height (
Results and discussion
Morphology and structure
The morphology and structure are disclosed, and the SEM images are displayed in Figure 2. Figure 2(a) shows the image of the NC/GAP matrix. The NC/GAP matrix presents a cross-linked network structure, and the size is approximately 30 nm. There are abundant nanoscale voids formed among the network structures. Figure 2(b) depicts the morphology of NC/GAP/CL-20. It is clear that the morphology of nanocomposites is different from that of the matrix. The cross-linked network structures disappear because CL-20 particles embedded in the pores occupied the voids of the matrix. The size of the nanocomposite is about 100–200 nm and becomes lager than the size of the matrix.

SEM images of nanocomposites: (a) NC0.5/GAP0.5 and (b) NC0.33/GAP0.34/CL-200.33. SEM: scanning electron microscope; NC: nitrocellulose; GAP: glycidyl azide polymer; CL-20: 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane.
To identify the crystallinity of nanocomposites, XRD analysis is performed, and the diffraction pattern is shown in Figure 3(a). It is clear that there are obvious crystal peaks at 2
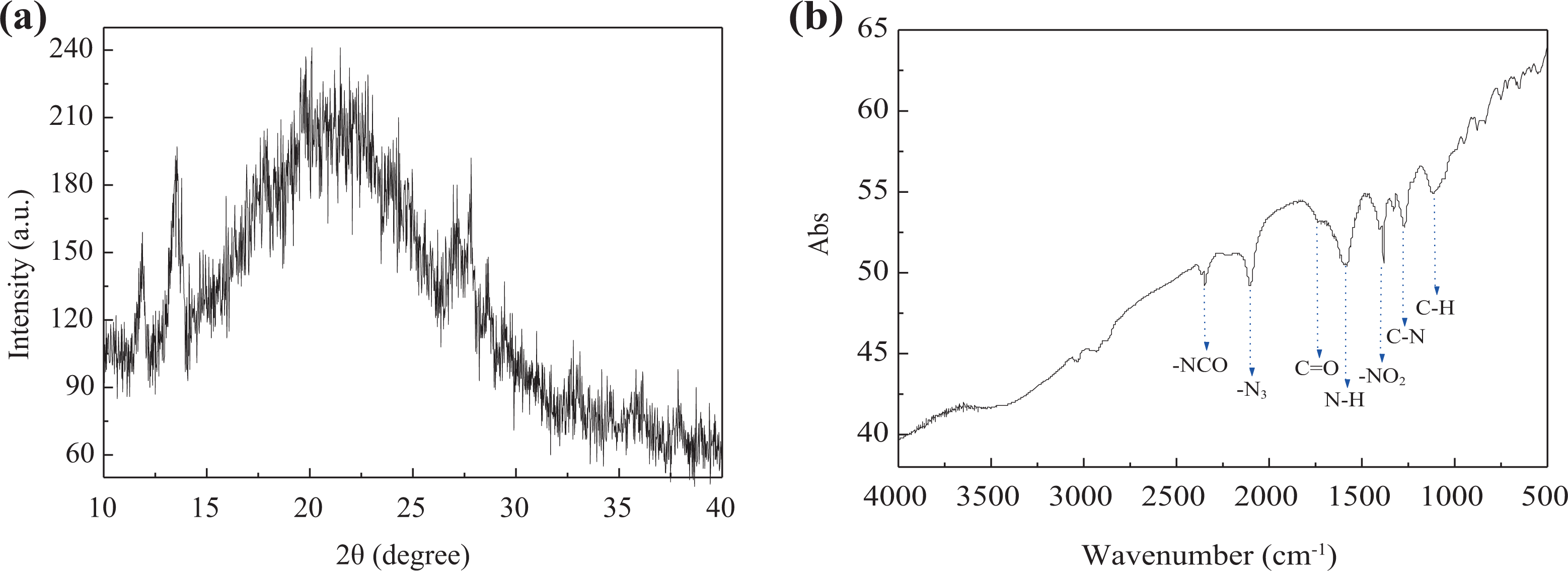
XRD pattern (a) and IR spectra (b) of NC0.33/GAP0.34/CL-200.33. XRD: X-ray diffractometer; IR: infrared; NC: nitrocellulose; GAP: glycidyl azide polymer; CL-20: 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane.
The specific surface area and the difference of porous properties of nanocomposites were investigated by nitrogen (N2) adsorption tests, and the results are exhibited in Figure 4. The curve of the low

BET data of samples: (a) isotherm linear plot and (b) Brunauer Emmett Teller (BET) surface area plot.
Pore structure parameters of samples.

NC: nitrocellulose; GAP: glycidyl azide polymer; CL-20: 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane.
The surface elements of nanocomposites were characterized by XPS, and the spectrum is shown in Figure 5. It can be seen that there are three obvious peaks corresponding to the electron excitation of the 1s orbital of O, N, and C atoms. The chemical bond information of O, N, and C elements was obtained from the narrow sweep of high resolution, and the results are exhibited in Figure 5(b) to (d). Electron excitation of the 1s orbital of O shows a peak, corresponding to oxygen atoms in the –NO2 group. Electron excitation of the 1s orbital of N shows three peaks, corresponding to nitrogen atoms in –NO2, –NH–, and –N3 groups. Electron excitation of the 1s orbital of C also shows three peaks, corresponding to carbon atoms in –CH2–, C=O, and –C– groups. These results are in accordance with the molecular structure of NC, GAP, and CL-20.

XPS spectra of NC0.33/GAP0.34/CL-200.33. XPS: X-ray photoelectron spectroscopy; NC: nitrocellulose; GAP: glycidyl azide polymer; CL-20: 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane.
Thermolysis properties
To study the thermolysis properties of NC/GAP/CL-20 nanocomposites, thermal analyses are performed, and the DSC traces are shown in Figure 6. Each curve displays two exothermic peaks corresponding to the decomposition of the nanocomposite. The peaks at about 166.9–170.8°C, that is, decomposition (i), should attribute to decomposition of the NC/GAP matrix; the peaks at about 238.1–246.6°C, that is, decomposition (ii), should attribute to decomposition of CL-20. The kinetic and thermodynamic parameters are calculated, and the results are listed in Table 2. The kinetic parameters, containing the activation energy (





where

(a) DSC traces of NC0.33/GAP0.34/CL-200.33 collected at different heating rates. (b) Kissinger plots of ln (
Thermodynamics and kinetics derived from DSC traces.
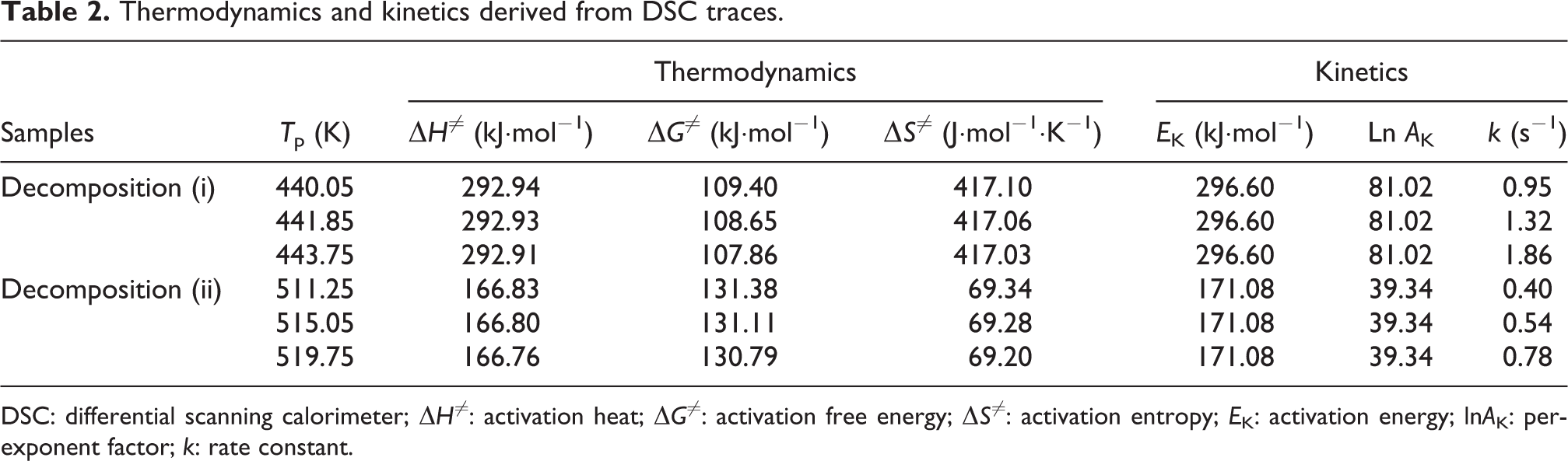
DSC: differential scanning calorimeter; Δ
For disclosing the decomposition mechanism of the nanocomposites, we investigate the gas products of thermal decomposition of NC/GAP and NC/GAP/CL-20 via DSC-IR analyses, and the results are shown in Figure 7. Firstly, for the NC/GAP matrix, Figure 7(a) indicates that its decomposition is not violent, and it decomposes gradually in the time range of about 623–2062 s. We extract the IR spectra at 623, 912, 1179, 1648, and 2062 s and illustrate them in Figure 7(b). The decomposition product of NC/GAP is very simple, that is, only carbon dioxide (CO2) is detected. Where is the N element? The –N3 groups in GAP molecules decompose to N2 which is a nonpolar molecule and cannot be detected by IR spectrometer. The –ONO2 groups in NC molecules split out and react with the C element, which accounts for the formation of CO2. The N element in –ONO2 groups are reduced to N2, and it evolves without being detected. Decomposition of NC/GAP/CL-20 is very different from that of NC/GAP. In Figure 7(c), it is very obvious that many gas products produce in the time range of about 600–1600 s, which attributes to the thermolysis of NC/GAP/CL-20. We extract the IR spectra at 704, 793, 865, 1110, and 1288 s and show them in Figure 7(d). Figure 7(d) indicates that the main decomposition products are CO2, nitrous oxide (N2O), and water (H2O); meanwhile, few carbon monoxide (CO), methane (CH4), and nitrogen oxide (NO) are also detected. Comparing these five IR spectra carefully, we can find that CO2 always appears in the whole decomposition process, but N2O appears only in the early stage of the decomposition. We have known that decomposition of NC/GAP does not produce N2O. Thus, as a product, N2O results from the reaction of –NO2 groups (splitting from homolysis of CL-20) with –C– and –N– fragments. Hence, CL-20 acts as an oxidizer in main decomposition of the nanocomposite. At 1110 s, no N2O is detected, but CO2 is still detected. Please note that at the end of the decomposition, ammonia (NH3) gas is released, and the signal of NH3 is very strong. It is much unexpected. No cation (just like ammonium), which can directly dissociate or to NH3, exists in the molecules of NC, GAP, or CL-20. Thus, we speculate that at elevated temperature, the remainder –N– and –CH– fragments (from homolysis of CL-20) take a molecular recombination to form NH2 radicals, and the NH2 radicals instantaneously combine each other to form hydrazine (H2N–NH2). And then the hydrazine decomposes to NH3, N2, and H2. In the IR spectrum at 1288 s in Figure 7(d), the formation of CH4, accompanying with the formation of NH3, confirms the molecular recombination. Additionally, the producing of NH3 and CH4 at the end of the decomposition reveals that the OB of NC0.33/GAP0.34/CL-200.33 is a negative value because of the lack of enough oxidizing groups to completely oxidize NH3 and CH4 to N2, CO2, and H2O.
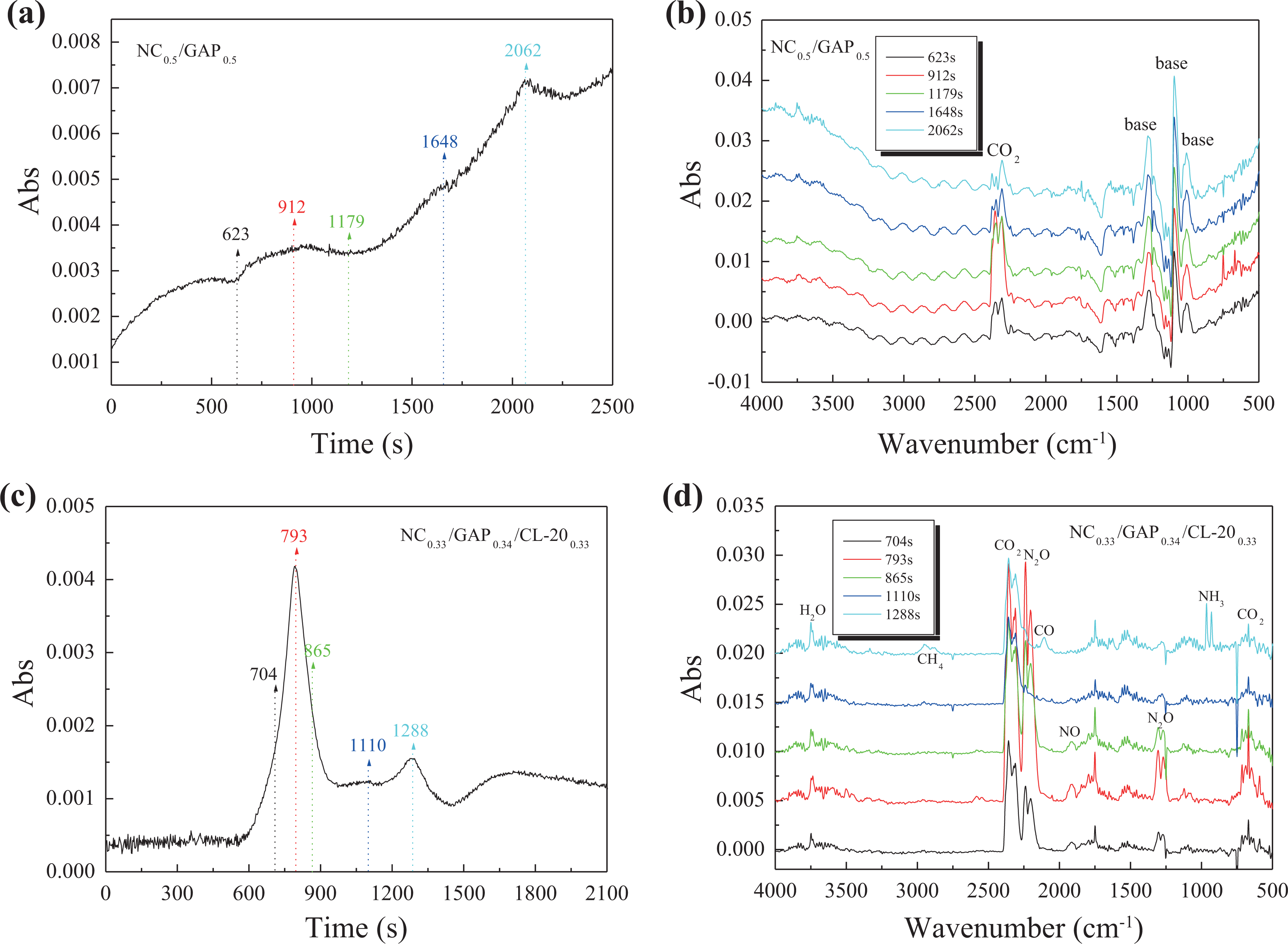
DSC-IR analysis for NC0.5/GAP0.5 (a) and (b) and NC0.33/GAP0.34/CL-200.33 (c) and (d) nanocomposites: (a) and (c) represent total absorbance of gas products; (b) and (d) represent IR spectra of gas products intercepted at different times (the time nodes of interception were illustrated in (a) and (c). DSC: differential scanning calorimeter; IR: infrared; NC: nitrocellulose; GAP: glycidyl azide polymer; CL-20: 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane.
Energy performance
For a further investigation on energetic properties of NC/GAP/CL-20, their energy performances are evaluated. Firstly, the evaluation on NC/GAP matrix is performed, and the results are listed in Table 3. It is clear that the standard specific impulse (
Energy performance of NC/GAP nanocomposites.a

NC: nitrocellulose; GAP: glycidyl azide polymer;
a All parameters were calculated by means of software ProPep 3.0 at condition of

Energy performance of NC/GAP nanocomposites as a function of weight percentage of NC. NC: nitrocellulose; GAP: glycidyl azide polymer.
The energy performances of NC/GAP/CL-20 nanocomposites are also evaluated, and the results are listed in Table 4. Figure 9 indicates that the
Energy performance of NC/GAP/CL-20 nanocomposites.a

NC: nitrocellulose; GAP: glycidyl azide polymer; CL-20: 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane;
a All parameters were calculated by means of software ProPep 3.0 at condition of

Energy performance of NC/GAP/CL-20 nanocomposites as a function of weight percentage of NC. NC: nitrocellulose; GAP: glycidyl azide polymer; CL-20: 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexaazaisowurtzitane.
Mechanical sensitivities
Mechanical sensitivities are other parameters that also characterize the energetic properties of the nanocomposites, that is, the safety of the nanocomposites. Figure 10(a) and (b) shows the special height (

Mechanical sensitivities of samples: (a) and (c) impact sensitivity; (b) and (d) friction sensitivity.
Conclusions
NC/GAP/CL-20 nanocomposites are prepared by sol–gel supercritical method, in which CL-20 nanoparticles are embedded in the NC/GAP matrix. XRD pattern indicates that the NC/GAP matrix is an amorphous material, while CL-20 is a crystal. IR spectrum shows that NC/GAP/CL-20 nanocomposite contains the molecular structure of NC, GAP, and CL-20. BET data of samples present that NC0.5/GAP0.5 is a kind of mesoporous material, which consists with the result in SEM images. In XPS spectrum, there are C, N, and O three elements. The results of thermal analysis indicate that two exothermic peaks corresponded to decomposition of NC/GAP and CL-20 from the low temperature peak (
Footnotes
Author contributions
Xiaolan Song and Yi Wang contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the Weapon Equipment Pre-Research Fund of China (grant no.: 6140656020201) and the National Natural Science Foundation of China (grant no.: 51206081).
