Abstract
In recent years, there has been growing interest in environmentally friendly methods for synthesizing semiconductor nanoparticles using naturally derived bio-based capping agents instead of conventional toxic solvents and reagents. In this study, an eco-friendly route for synthesizing cadmium sulfide nanoparticles is presented using bis(piperidinedithiocarbamato)cadmium(II) as a single-source precursor and anacardic acid, a bio-derived capping agent isolated from cashew nut shell liquid. The precursor complex was thermally decomposed in hot anacardic acid at temperatures of 140 °C, 160 °C and 180 °C. The reaction produced anacardic acid-capped cadmium sulfide nanoparticles with varying optical and structural properties depending on synthesis temperature. UV-Vis absorption spectra revealed blue-shifted excitonic peaks indicative of quantum confinement, while photoluminescence analysis revealed typical emission features associated with cadmium sulfide nanocrystals. Transmission electron microscopy analysis revealed a morphology evolution from quasi-spherical to irregular cuboidal particles with increasing temperature. X-ray diffraction patterns indicated a temperature-dependent phase transition from cubic to hexagonal crystal structures. This green synthesis approach demonstrates that naturally derived anacardic acid can effectively stabilize and tune cadmium sulfide nanocrystals, offering a sustainable alternative for nanoparticle synthesis with potential applications in optoelectronic devices and photocatalysis.
Keywords
Introduction
Cadmium sulfide (CdS) nanoparticles are type II–VI semiconductor materials used in photovoltaic,1,2 optoelectronics, catalysis3–5 and bioimaging6–8 due to their size dependent electronic and optical properties. Their outstanding properties are attributed to their narrow band gap, quantum confinement and large surface area supporting them as candidates for next generation technologies. 9 In a nanocrystal form, CdS appears as colloidal fluorescent quantum dots referred to as quantum dots (QDS) possessing outstanding optical and electrical properties which have led to their applications in diverse fields such as biosensing, nanomedicine, drug delivery and solar cells. 10
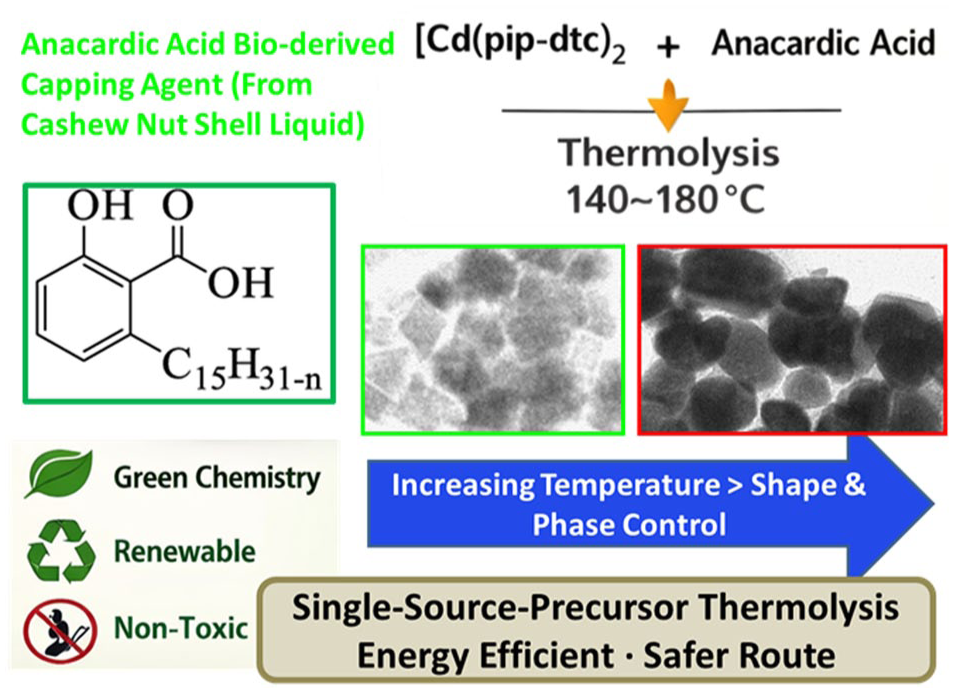
The conventional synthesis routes of CdS nanoparticles which rely on toxic reagents often generate hazardous by-products, posing environmental and health challenges.11–13 In light of increasing environmental concerns, green chemistry approaches have gained traction for nanoparticle synthesis seeking non-toxic reagents, renewable inputs and producing minimal waste. 14 Agro-waste products have recently been utilized as eco-friendly capping agents in the preparation of CdS nanoparticles.15,16 Among these, cashew nut shell liquid (CNSL) is of particular concern as it contains bioactive compounds such as anacardic acid, which possesses strong antioxidant and reducing properties. Anacardic acid shown in Figure 1 possesses carboxylate group and long hydrophobic alkyl chain 17 which structurally stabilizes nanoparticles by binding via carboxyl group while its alkyl tail ensures colloidal dispersion and hydrophobic compatibility. 18 Studies have demonstrated the ability of anacardic acid to serve as an effective capping and growth-directing agents in the preparation of metal chalcogenide nanocrystals. Our group has previously demonstrated the use of anacardic acid as a capping agent in the preparation of cadmium chalcogenide nanoparticles via a thermolysis of dual source. 19 In another study, we synthesized lead chalcogenide nanoparticles exhibiting controlled morphologies with PbS and PbSe forming spherical shapes and PbTe forming cubic structures.19,20 Compared with other bio-derived capping agents such as castor oil, banana peel extract and watermelon rind, anacardic acid offers unique advantages due to its carboxylate functionality for strong binding to nanoparticle surfaces and its long hydrophobic alkyl chain, which enhances colloidal stability and dispersion in nonpolar media, making it particularly effective for green synthesis of nanoparticles. 21

General structure of anacardic acid. 17
The choice of precursor strategy influences the properties and efficiency of CdS nanoparticle synthesis. The use of single-source precursor (SSP) containing cadmium and sulfur in a non-volatile, air-stable and non-pyrophoric form offers significant safety advantages during synthesis process.22–24 In addition to improved thermal and chemical control, the use of SSP further supports greener synthesis routes by minimizing the use of highly toxic cadmium salts traditionally employed in the synthesis of CdS.25–27 Using SSPs, CdS nanocrystals have been synthesized with narrow size distribution and quantum-confined, tunable optical properties through thermolysis under mild conditions in ambient air. 28 The method is notable for its simplicity and efficiency. The use of SSPs in CdS offers precise control over composition, phase and morphology by delivering both cadmium and sulfur in a pre-organized molecular structure. 29
Remarkable work on the use of bis(piperidinedithiocarbamato)cadmium(II) complex for the preparation of CdS nanoparticles employing tri-
Materials and methods
Chemicals
TOP 90% was supplied by Sigma-Aldrich, whereas acetone and methanol 99.5% were from Merck Chemicals. Chemicals were utilized as received, without additional purification. The organic salts of ligands and the corresponding cadmium complex were prepared following previously reported procedures.30,33,34 CNSL and anacardic acid were extracted and isolated following procedure outlined in prior work. 20
Preparation of CdS nanoparticles
The [Cd(pip-dtc)2] complex of the mass 0.5 g (1.2 mmol) was dissolved in TOP (6.0 mL). The solution was introduced into 6.0 g (32.3 mmol) of preheated anacardic acid in a three-necked flask at 140 °C/160 °C/180 °C under nitrogen gas flow. After maintaining the reaction time for 2 h, small portions of products were taken and methanol was added, resulting in the formation of a flocculent precipitate. The precipitate and solvent were separated by centrifugation, and then dispersed in acetone to yield yellow anacardic acid-capped CdS nanoparticles. A similar procedure was followed to check the influence of time where aliquot samples were taken after 5, 15 and 30 min for the reaction temperature of 160 °C. A schematic overview of the synthesis procedure is presented in Figure 2, showing the thermolytic decomposition of the cadmium SSP in hot anacardic acid, which serves as bio-derived capping agent.

Schematic illustration of the thermolytic synthesis of anacardic acid-capped CdS nanoparticles.
Characterization
Fourier transform infrared (FT-IR) was carried out using Perkin Elmer Paragon 1000 FT-IR Spectrometer and the spectra were obtained within the frequency range of 400–4000 cm−1. Spectra were recorded at room temperature and the sample was cast onto sample holder as liquid or solid. Optical absorption measurements were performed with a Varian Cary 50 UV-Vis spectrophotometer, using acetone as the reference solvent. A Perkin-Elmer LS 55 spectrofluorimeter was used to measure the photoluminescence (PL) of the CdS nanoparticles. The samples were placed in a quartz cuvette (1 cm path length) and all measurements were done at room temperature. The morphology was determined using a JEOL 1010 TEM and JEOL 2100 HRTEM operated at an accelerating voltages of 100 kV (equipped with a Megaview III camera, and Soft Imaging Systems iTEM software) and 200 kV (using Gatan camera and Gatan software), respectively. A Zeiss Ultra Plus FEG SEM was used for surface morphology analysis, equipped with an Oxford detector EDX at 20 kV which uses Aztec software for elemental analysis. X-ray diffraction (XRD) patterns were collected in the 2θ range of 20°–80° using a Bruker AXS D8 Advance diffractometer with Ni-filtered Cu Kα radiation (λ = 1.5406 Å) at 40 kV, 40 mA and room temperature.
Results and discussions
Optical properties
The thermal decomposition of bis(piperidinedithiocarbamato)cadmium(II) [Cd(S2CNC5H10)2] complex, dissolved in TOP and injected into a hot anacardic acid solution at 140 °C, 160 °C and 180 °C resulted in rapid decomposition of the complex and the formation of CdS nanoparticles capped with anacardic acid. IR spectroscopy was employed to analyse the surface characteristics of the anacardic acid-capped CdS nanoparticles capped with anacardic acid (Figure 3). The FT-IR spectrum of the CdS particles showed all necessary fingerprint peaks for the presence of anacardic acid in the sample. The spectrum of the anacardic acid-capped CdS display bands that are shifted to higher frequencies when compared to the free anacardic acid matrix. This shift may be due to coordination of the nanoparticles to the capping agent. Our previous investigations have also demonstrated similar findings,33,35 where the decrease in intensity of the carbonyl and hydroxyl peaks in the spectra of ricinoleic acid-capped CdS nanoparticles indicated that ricinoleic acid binds to the nanoparticle surfaces via both oxygen atoms of the carboxylic group.

Fourier transform infrared (FTIR) spectra of (a) anacardic acid and (b) anacardic acid-capped CdS nanoparticles.
The reaction was monitored over time using UV-Vis absorption and PL spectroscopy as shown in Figures 4 and 5 to investigate the optical properties of the prepared CdS nanoparticles. The effect of time showed minimal variation across the three temperature conditions. The initial investigation focused on the effect of reaction time at 160 °C as indicated in Figure 4. Samples were collected at 5, 10 and 30 min and analysed for their optical properties to pursue early nucleation processes, particle growth and phase evolution. All three samples exhibited broad and featureless absorption with minimal changes in the spectra over such time intervals, suggesting that the effect of time on particle growth and associated electronic properties may only become evident at longer time intervals.36,37

UV-Vis absorption spectra of anacardic acid-capped CdS nanoparticles prepared from Cd(pipdtc)2 complex at 160° C for (a) 5, (b) 15 and (c) 30 min.

(i) UV-Vis absorption spectrum of the as-synthesized anacardic acid-capped CdS nanoparticles prepared at (a) 140 °C, (b) 160 °C and (c) 180 °C obtained for 2 h. Inset shows a representative photoluminescence spectrum for sample (c) (excitation wavelength used was 420 nm). (ii) Tauc plot showing the estimated direct optical band gap of anacardic acid-CdS nanoparticles.
The reaction was further left to proceed for 2 h before being quenched and the resulting solution was cooled to approximately 70 °C. The resultant solution was washed thrice with methanol and then dispersed in acetone solvent for characterization. The influence of reaction temperature on the optical absorption of anacardic acid-capped CdS nanoparticles showed prominent excitonic absorption peaks around 445, 435 and 440 nm for samples prepared at 140 °C, 160 °C and 180 °C, respectively, as shown in Figure 5. The excitonic absorption bands at all temperatures are blue-shifted compared to those of bulk CdS, suggesting that the particles experience quantum confinement effects. 38 Bandgap values obtained by extrapolating the linear fit of the direct-allowed Tauc plot exhibit optical bandgaps in the range 2.6–2.7 eV (Figure 5(ii)). The representative PL spectrum of the prepared particles at 180 °C is shown as an inset of Figure 5. Anacardic acid-capped CdS particles obtained at this temperature shows usual bathochromically shifted emission peak located at 485 nm, the difference usually caused by shallow traps formed due to sulfur vacancies. 39 The relatively broader colour emission line width in the 8.5-nm range (~146 eV) is due to the broad size distribution of the as-synthesized anacardic acid CdS nanoparticles. The tunable optical properties, particularly the blue-shifted absorption and PL arising from quantum confinement, underscore the potential of these nanoparticles for photovoltaic and catalysis applications. 40
Morphological analysis
Effect of temperature during synthesis on the morphology of the prepared anacardic acid-capped CdS nanoparticles was also studied and the results are presented in Figure 6. It is observed that the morphology of the CdS nanoparticles evolves significantly with increasing synthesis temperature. It is noted that the morphology of the particles evolves significantly with increasing synthesis temperature. At 140 °C, broadly dispersed and quasi-spherical particles with sizes in the range of 5–60 nm are synthesized as indicated in Figure 6(a). The size distribution histogram shown in Figure 6(b) further confirms the broad size distribution of the particles synthesized at this temperature. Previous study show that the synthesis of anisotropic oblate to elongated shaped anacardic acid-capped CdS particles using dual source route at 140 °C with an average diameter of 14.02 ± 2.4 nm, the anacardic acid-capped CdS nanoparticles exhibited a well-ordered, mosaic-type arrangement, maintaining an interparticle spacing of roughly 3 nm. 19

Transmission electron microscopy (TEM) images of the prepared anacardic acid-capped CdS nanoparticles prepared at (a) 140 °C (and its corresponding histogram (b)), (c) 160 °C and (d) 180 °C (and its corresponding HRTEM image (e) and FFT (f)) using bis(piperidinedithiocarbamato)cadmium(II) complex as SSP obtained for 2 h.
At 160 °C, anisotropic growth is favoured, which produced irregular cuboidal particles with an average size of 19.28 ± 2.65 nm as indicated in Figure 6(c), corroborating the blue shift of the absorption band energy observed in Figure 5(b). Earlier study revealed that dual source route produced anisotropic shaped anacardic acid-capped CdS nanoparticles at this temperature giving particles with average width of 11.8 ± 2.4 nm and length 32.6 ± 4.2 nm. 41 A further increase in temperature to 180 °C produced spherical/cubic and irregularly shaped particles with sizes in the range of 20–39 nm as presented in Figure 6(d). This transition in morphology can be due to increased thermal energy which then overcomes the uniform stabilizing effect of anacardic acid. Higher temperatures usually promote accelerated surface atom mobility and facet-selective growth, thereby shifting the balance between anisotropic and isotropic growth modes. This behaviour suggests that the competition between ligand-controlled surface passivation and thermally driven atomic rearrangement governs the evolution of particle shape and size across the given temperature range. The representative high resolution TEM image (Figure 6(e)) of the anacardic acid-capped CdS nanoparticles synthesized at 180 °C shows presence of lattice fringes of the nanocrystals with the interplanar lattice spacing of 3.13 Å, which could be indexed to the (101) reflection of the hexagonal phase of CdS. Similarly, its fast Fourier transform (FFT) pattern (Figure 6(f)) displays distinct bright spots, indicating that the particles are highly crystalline. A summary of the sizes and morphologies observed for anacardic acid-capped CdS nanoparticles thermalized at different temperatures is presented in Table 1. Elemental analysis confirmed the presence of Cd and S in all samples (Electronic Supplementary Information Figure S1). SEM-EDX mapping showed consistent Cd and S signals across multiple probed areas confirming a uniform elemental distribution throughout the nanoparticles.
Summary of morphology, sizes calculated from TEM images and XRD data for anacardic acid-capped CdS nanoparticle.

Structural analysis
Thermolysis of the bis(piperidinedithiocarbamato)cadmium(II) complex in anacardic acid at 140 °C, 160 °C, and 180 °C was carried to observe the influence of temperature and capping agent on the crystallinity of the as-synthesized CdS nanoparticles. When the reaction was carried at 140 °C for 2 h as shown in Figure 7(a), cubic phase of anacardic acid-capped CdS was identified with broad diffraction peaks indexed to (111), (220), (311) mirror planes (Card No. 01-080-0019). The broad diffraction peaks observed in the XRD pattern of the 140 °C sample are consistent with the observed blue shift phenomenon in the UV-Vis absorption spectrum indicative of formation of small sized anacardic acid-capped CdS nanoparticles. XRD patterns of the CdS particles prepared at 160 °C as shown in Figure 7(b), matching the standard patterns of cubic structure with relatively narrow diffraction peaks, indicating the increase in crystalline sizes of the particles. However, thermolysis process carried at elevated temperature of 180 °C, led to the formation of highly crystalline hexagonal CdS nanoparticles as confirmed by the presence of (100), (002), (101), (102), (110), (103) and (112) reflections (Card No. 01-080-0006) as presented in Figure 7(c). No additional peak in XRD data was observed, indicative of the purity of the synthesized nanoparticles in all samples. Inspection of the full raw diffractograms, including low-angle regions (Electronic Supplementary Information Figure S2), showed no reflections attributable to CdO, CdSO₄, elemental sulfur or unreacted precursor, confirming phase-pure CdS. The particle sizes of anacardic acid-capped CdS nanoparticles obtained can be calculated using Debye-Scherrer equation (equation (1)) reported elsewhere, 42 and the results are tabulated in Table 1

where

XRD patterns of anacardic acid-capped CdS nanoparticles using bis(piperidinedithiocarbamato)cadmium(II) complex as SSP obtained for 2 h at (a) 140 °C, (b) 160 °C and (c) 180 °C.
Conclusion
This work highlights the successful thermolytic synthesis of CdS nanoparticles using an SSP in the presence of anacardic acid as an eco-friendly capping and stabilizing agent. The optical properties of the synthesized anacardic acid-capped CdS nanoparticles characterized by blue-shifted absorption confirm the quantum confinement effect. The study reveals that the synthesis temperature plays a critical role in determining particle morphology, size and crystalline phase. At lower temperatures of 140 °C, quasi-spherical particles with a broad size distribution were observed, while at elevated temperatures of 160 °C and 180 °C, anisotropic and irregular cuboidal shapes predominated. Notable transition from cubic to hexagonal crystalline phases as verified by XRD indicates the significant influence of thermal energy on phase stability. This green synthesis route offers an environmentally benign, cost-effective pathway for producing semiconducting nanoparticles with tunable properties for optoelectronic and catalytic applications. Although this study only focused to demonstrate the potential of anacardic acid to produce CdS nanoparticles at a laboratory scale, the simplicity and eco-friendly nature of the synthesis indicate likely potential for future scale-up, provided that nanoparticle uniformity can be carefully optimized.
Supplemental Material
sj-docx-1-chl-10.1177_17475198261425700 – Supplemental material for Eco-friendly thermolytic synthesis and characterization of anacardic acid-capped cadmium sulfide nanoparticles using a single-source precursor
Supplemental material, sj-docx-1-chl-10.1177_17475198261425700 for Eco-friendly thermolytic synthesis and characterization of anacardic acid-capped cadmium sulfide nanoparticles using a single-source precursor by Sixberth Mlowe in Journal of Chemical Research
Footnotes
Acknowledgements
The author acknowledges the University of Zululand, South Africa and University of Dar es Salaam for assisting with sample analyses. The author is also grateful to the Dar es Salaam University College of Education for facilitating the writing of this manuscript.
Ethical considerations
Not applicable.
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be made available on request from the corresponding author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
