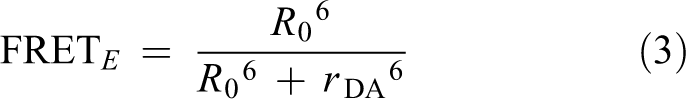Abstract
In recent years, palladium nanoparticles have been proved as energy acceptor candidates in fluorescence resonance energy transfer-based sensors for analytical and biological purposes. In this article, peptide-coated palladium nanoparticles were prepared using a simple one-step preparation method. The peptide Cys-Ala-Leu-Asn-Asn was used as a ligand, whereas hydrazine hydrate was used as a reductant to obtain water-soluble and stable peptide-coated palladium nanoparticles. Additionally, peptide-coated palladium nanoparticles were functionalized by adding the functional peptide CALNNGGARK(FITC) in combination with Cys-Ala-Leu-Asn-Asn during the preparation process. The prepared functionalized peptide-coated palladium nanoparticles were used for trypsin detection based on the fluorescence resonance energy transfer approach. Under optimized conditions, the proposed method can be used for the detection of trypsin concentrations in the range of approximately 0.2–8-μg/mL with a limit of detection of 0.18-μg/mL. The functionalized peptide-coated palladium nanoparticles were successfully applied for the detection of trypsin in urine samples. Our findings also indicated that peptide-coated palladium nanoparticles can highly quench fluorophores and are suitable for the manufacture of off–on state fluorescent sensors. We anticipated that the peptide-coated palladium nanoparticles proposed in this article will have great potential for the detection of trypsin in urine and other analytical, biological, and clinical applications.
Keywords
Introduction
Trypsin is one of the most specific proteases involved in digestive functions in a multitude of vertebrates and is able to activate the zymogens of chymotrypsin, carboxypeptidase, phospholipase, and other enzyme precursor. 1,2 The normal human urine samples are substantially free of trypsin, but the content of trypsin is high in the urine of patients with acute pancreatitis (average 84.4-μg/mL). 3 Therefore, the establishment of a method for determining trypsin in human urine is of great significance in clinical diagnosis. Nowadays, the detection methods of trypsin include colorimetry, electrophoresis, fluorescence, and so on. 4 –10 Among different methods, fluorescence resonance energy transfer (FRET)-based methods have attracted more and more attention. 11 –13 Especially a method with peptide-based probes containing fluorophore and quencher pairs has greater potential for monitoring the activity of various proteases by modifying the peptide. 12
In recent years, palladium nanoparticles (PdNPs), a new kind of functional materials, have been developed and have displayed excellent performance in catalysis applications. 14,15 Numerous preparation methods have been devised to meet the research requirements including peptide-templated preparations. 16 However, studies focused on the use of PdNPs for the analytical purposes are rare. Recently some studies have shown that PdNPs have the potential to exert a satisfactory quenching effect 17 and can be incorporated in FRET-based methods for the detection of DNA and cancer markers. 18,19 These studies demonstrate the potential of PdNPs as an energy acceptor in the establishment of FRET-based analysis methods. However, the PdNPs used in these reported FRET-based methods are unstable under detection conditions if they are not modified. Furthermore, the detections are based on the coordinated interactions between the heterocyclic structure of biomolecules and the palladium, which limits the selectivity of the detection methods and their further applications. To address these issues, a carboxyl group-modified PdNP was previously prepared and conjugated to DNA by post-synthetic processing for analytical purposes. 20 Based on this, a PdNPs-based FRET aptasensor was developed for the selective detection of aflatoxin M1 in milk. 21 But the post-synthetic processing complexifies the approach. Therefore, finding new caps and preparation methods that are more ideal for biological and analytical applications still represents an attractive area of research.
Previous works have focused on the preparation of water-soluble gold nanoparticle 22 and quantum dot, 23 which have good stability when they are coated with the pentapeptide Cys-Ala-Leu-Asn-Asn (CALNN) as a ligand. These extremely stable gold nanoparticles and quantum dots have been applied in various bioanalyses. 24 –26 Thus, we hypothesized that PdNPs, similar to the gold nanoparticles and quantum dots, can be also stabilized by CALNN because they are all metal materials that can form metal–thiolate bonds with the sulfydryl group of CALNN. In addition, the carboxyl group-modified PdNPs have been successfully prepared in aqueous solution 20 with 11-mercaptoundecanoic acid as a ligand, which provided great support for our assumption.
Inspired by the above, we aimed to prepare CALNN-coated PdNPs using the one-step synthesis method and to test their potential functionalization and application in the measurement of trypsin in human urine samples. The proposed synthesis method greatly simplified the process of modification and functionalization of PdNPs, and the peptide-coated PdNPs exhibited excellent stability. Furthermore, considering the high biocompatibility of peptide coating and the large libraries of the peptides into account, peptide-coated PdNPs will have great potential in biological and analytical applications such as FRET-based trypsin detection.
Experimental methods
Materials
Tris base, palladium chloride (PdCl2), sodium chloride (NaCl), sodium hydroxide, and hydrazine hydrate (N2H4·H2O) were purchased from Shanghai Macklin Biochemical Co., Ltd (China). Trypsin was purchased from Sigma-Aldrich Co., LLC (China-Mainland). Peptides (CALNN and CALNNGGARK(FITC)) were synthesized and purified by ChinaPeptides Co., Ltd (China). The Bicinchoninic Acid (BCA) Protein Assay Kit was obtained from Sangon Biotech (Shanghai) Co., Ltd (China).
Preparation of peptide-coated PdNPs
The disodium tetrachloropalladate solution (Na2PdCl4) was prepared by mixing 0.0876-g PdCl2 and 6-mL 0.2 mol/L of NaCl solution, followed by dissolution with distilled water to a volume of 250.00 mL.
Then, 0.0067-g CALNN was weighted in a three-necked round-bottomed flask, and 7 mL of distilled water was added to dissolve CALNN with gentle stirring. Thereafter, 0.07 mL of N2H4·H2O was added to the solution, and the pH was adjusted to about 12. Next, 7 mL of the Na2PdCl4 solution was quickly poured into the solution, followed by stirring for 30 min to ensure completion of the reaction. The prepared PdNPs were purified with 10K ultrafiltration tube (Amicon Ultra-4, Merck KGaA (Germany)) and rinsed with Tris-hydrochloride (HCl) buffer (0.05 M, 0.3-M NaCl, pH 8.0) for four times. Then, the resulting CALNN-coated PdNPs were re-dispersed in Tris-HCl buffer and sealed at 4°C. Besides, to prepare a specific peptide-functionalized PdNPs for trypsin (tPdNPs) detection, we simply mixed 0.0021 g of the functional peptide CALNNGGARK(FITC) with 0.0067-g CALNN during the synthesis process.
To further verify the coating of CALNN on PdNPs, we used the BCA method to detect CALNN qualitatively. The BCA method is widely used to determine proteins and peptides based on the reduction of Cu2+ by peptide bonds under alkaline condition. The resulting Cu+ is then reacted with a BCA reagent to form a purple coordination compound with a maximum absorption value at 562 nm. Therefore, we can verify the presence of CALNN by the change in absorption of solutions at 562 nm.
Characterization of the FRET efficiency
The characterization of the FRET of the nano-probe is calculated as previously described. 27 Förster distance (R 0) was calculated using equation (1):
where k 2 is set to 2/3 for fast isotropic rotation of donor, Φ D is a quantum yield of fluorescein isothiocyanate (FITC) donor in alkaline solution that is 0.9, 28 J is calculated overlap integral in the supplementary information, and n is the refractive index of water 1.333.
FRET efficiency (FRET E ) was calculated using the following equation
where I DA and I D represent the fluorescent intensity of FITC in the presence and absence of PdNPs, respectively.
Dynamic light scattering (DLS) and zeta potential analysis of peptide-coated PdNPs
Peptide-coated PdNPs for DLS and zeta potential (ZP) determinations were dispersed in disodium hydrogen phosphate–citric acid buffer (10-mM disodium hydrogen phosphate, 10-mM citric acid) at pH 4.0–8.0 and immediately filtered through a 0.25-µm membrane to remove any insoluble elements. The results of DLS were represented by the intensity of scattering light, and the Z-average sizes were recorded.
Detection of trypsin with tPdNPs
The concentration of tPdNPs was determined by Inductively coupled plasma atomic emission spectra (ICP-AES) and subsequently diluted to 80-ng/mL with a Tris-HCl buffer solution and mixed with a certain amount of trypsin. The mixture was incubated at 37°C for 100 min with a rotatory mixing. The fluorescence of the solution was determined by Tecan (Switzerland) Infinite M200 Pro. The detection parameters were set as follows: excitation at 490 nm and emission at 520 nm and 100 gains.
Collection of urine samples and detection of trypsin
The urine samples were collected from healthy individual volunteers. The study was approved by the ethics committee board of our city following the informed consent from subjects. About 20 mL of fresh urine samples was taken in a small beaker and was left for 0.5 h. The trypsin content was determined according to the experimental method described above, followed by the spiked recovery experiment.
Results and discussion
Preparation and characterization of PdNPs
Through the aqueous phase synthesis of nanomaterial, it is possible to obtain water-soluble nanoparticles, which are suitable for biological detections and analysis. Therefore, we chose water as a solvent in order to reach a satisfactory purpose. We hypothesized that the use of peptides instead of the usual sulfhydryl compounds as ligands in the formation process of PdNPs can result in the desired peptide-coated PdNPs. However, as reported in the literature, 20 the formation of Pd2+–S bonds can reduce the oxidizing ability of Pd2+. As a result, Pd2+ could not be reduced to form PdNPs and, for this reason, the Pd2+ solution must be poured into the mixture of the peptide ligand CALNN and the reducing agent N2H4·H2O during the preparation to avoid the formation of Pd2+–S bonds. Besides, as we know, the reducing capacity of N2H4·H2O is pH-dependent. Therefore, the pH of the ligand–N2H4·H2O mixture was adjusted to 12 prior to Pd2+ addition. By this operation, the preparation of CALNN-coated PdNPs was achieved in a single step. The transmission electron microscopic (TEM) image of prepared CALNN-coated PdNPs showed that the size of PdNPs was about 5 nm with a uniform spherical shape (Figure 1(a)). Furthermore, the lattice distances displayed in the high-resolution TEM image of the single particle were 0.229 and 0.202 nm (Figure 1(a), inset), which correspond to the {111} and {200} faces of the FCC structure of Pd0, respectively. These results indicated that PdNPs were successfully prepared. However, the peptide coated on the PdNPs could not be observed from the TEM image, since the major element of peptide displays low contrast in TEM.

(a) TEM image of CALNN-coated PdNPs. Inset: High-resolution image of single CALNN-coated PdNPs. (b) Detection of CALNN on the surface of PdNPs by the BCA method, showing the UV-vis absorption spectra of CALNN-coated PdNPs and CALNN solution as a positive control. (c) UV-vis spectrum of functional PdNPs specific for trypsin (tPdNPs) compared with CALNN-coated PdNPs and FITC. TEM: transmission electron microscopy; CALNN: Cys-Ala-Leu-Asn-Asn; PdNPs: palladium nanoparticles; BCA: bicinchoninic acid; UV-vis: ultraviolet–visible; FITC: fluorescein isothiocyanate.
Results shown in Figure 1(b) indicated that the ultraviolet–visible (UV-vis) absorption spectrum of CALNN-coated PdNPs treated with the BCA method exhibited an obvious absorption peak at 562 nm as that of the CALNN positive control. Comparatively, no significant absorption signal was obtained from the blank sample corresponding to PdNPs prepared without CALNN. These results indicated that the coating of CALNN on the surface of PdNPs was successful.
The advantage of this method is that the preparation and functionalization can be realized in a single step. In order to achieve specific functionalization of peptide-coated PdNPs, it is necessary to simply add a functional peptide cooperated with CALNN in the preparation. Therefore, the functional peptide CALNNGGARK(FITC) was designed to explore the functionalization of peptide-coated PdNPs. The functional peptide contained a ligand region of CALNN, a substrate region of Ala-Arg-Lys (ARK) recognized by trypsin, and a fluorescent signal reporter of FITC. The functional PdNPs specific for trypsin detection (tPdNPs) were prepared by simply making a peptide mixture composed of 90% CALNN and 10% CALNNGGARK(FITC) on a molar basis in the process. According to the UV-vis spectrum, the spectrum of tPdNPs, as well as FITC, displayed an obvious absorption peak at 490 nm compared with the CALNN-coated PdNPs (Figure 1(c)). This indicated that the functional peptides were successfully tagged to PdNPs to successfully obtain tPdNPs.
Stability of peptide-coated PdNPs
Since the stability of PdNPs is essential for a variety of applications, we aimed at investigating the stability of the prepared CALNN-coated PdNPs. The result showed that the prepared CALNN-coated PdNPs have excellent stability, and the results were similar to those previously reported for gold nanoparticles and quantum dots (QDs) coated with CALNN, 22,23 which maintain a good dispersion in aqueous solution in a wide pH range. As shown in Figure 2, the CALNN-coated PdNPs were well dispersed in pH 8.0 as measured by DLS, and the Z-average size was 30.6 nm. This size is different from that observed in the TEM image, because this is the hydrodynamic sizes of particle, which is calculated from the diffusion coefficient according to Stokes–Einstein equation. The ZP of the particles reveals the reason for the good dispersion of the particles. The ionization of terminal carboxyl groups of the peptides under alkaline conditions results in a negatively charged surface of the particles, making them repulsive to each other. With the decrease of pH, the ZP is reduced correspondingly to the protonation of carboxyl groups, thus resulting in a slight aggregation of particle and increase of the particle size. However, the particle size was finally maintained at about 70 nm, even at the pH of 4.0, which is lower than the pKa of the carboxyl group. This is because the outwardly hydrophilic asparagine (N) residues of the peptide ligands induce the electronic repulsion. When the pH is 4.0, the protonation of the N residues reverses the charge of particle surface to positive, thereby providing a little repulsive force responsible for the stability of the particles. As reported in the literature, 22,23 the high stability of the particle resulted from a high-density coating of the peptides, which was estimated around 2.5 peptides/nm2 on the surface of tPdNPs based on the determination of the concentrations of FITC and tPdNPs (see Online Supplementary Material, Figures S1 and S2).

Size distributions of CALNN-coated PdNPs at different pH conditions were measured by DLS. Relative ZP and Z-average size of CALNN-coated PdNPs are displayed in each panel. ZP: zeta potential; CALNN: Cys-Ala-Leu-Asn-Asn; PdNPs: palladium nanoparticles.
Design and verification of FRET-based trypsin probe
On the basis of the stable tPdNPs, a FRET-based trypsin probe was designed as indicated in Figure 3(a) to explore the application of peptide-coated PdNPs. Basically, it was constituted of a PdNPs quencher and FITC, which are connected by a peptide substrate of trypsin. Due to the high quenching effect of PdNPs on fluorescent signals, as claimed in the literature, 18 FITC fixed on PdNPs via substrate peptides could not emit fluorescent signals due to the FRET to PdNPs. Following specific cleavage of the CONH bond between arginine (R) and lysine (K) by trypsin, the fluorescent dye becomes free and gets away from PdNPs, resulting in the restoration of fluorescence intensity (Figure 3(a)). On this basis, a FRET-based sensor for trypsin quantitative detection was established by measuring the change of fluorescence intensity.
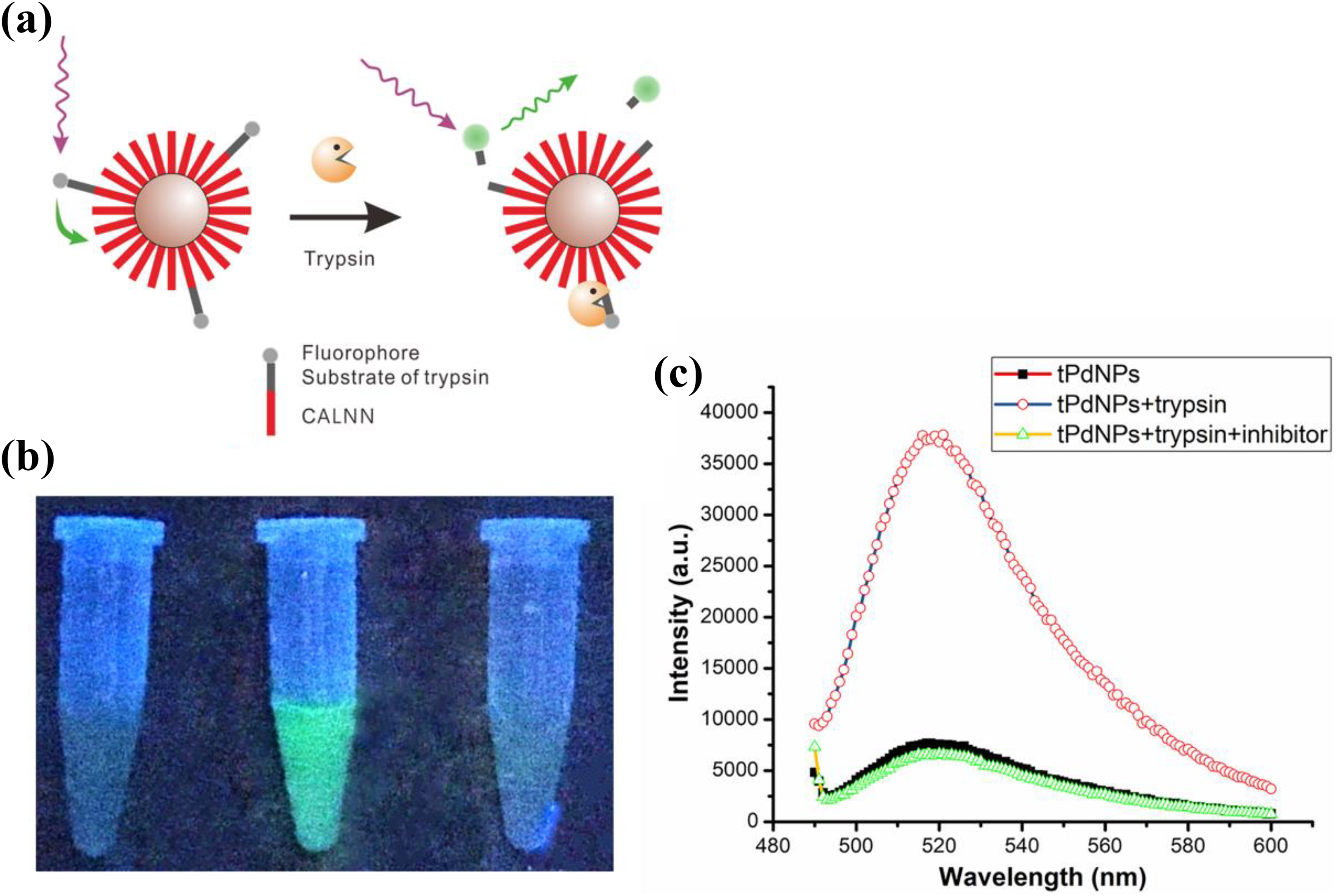
(a) Scheme of a nano-fluorescence probe based on the FRET technology for the detection of trypsin. (b) Photograph of tPdNPs, tPdNPs in the presence of trypsin, and tPdNPs in the presence of trypsin and trypsin inhibitor, respectively. Photographs were taken under radiation of 360-nm UV light in dark. (c) Relative fluorescence spectra of solutions showing the actual fluorescence intensity change of the solutions. For photographing, 8-μg/mL tPdNPs, 100-μg/mL trypsin, and 200-μg/mL trypsin inhibitor were added in the reaction, and the fluorescence spectra of solutions were measured with 470-nm excitation. FRET: fluorescence resonance energy transfer; UV: ultraviolet; PdNPs: palladium nanoparticles.
The feasibility of the design was validated, and the results are shown in Figure 3(b) and (c). Due to the high quenching effect of PdNPs, the prepared tPdNPs showed a weak background signal under the test conditions. In the presence of trypsin, an obvious green bright fluorescence was observed (Figure 3(b)). The quantitative analysis of fluorescence intensity (Figure 3(c)) showed that the fluorescence was enhanced by more than five times in the presence of trypsin compared with the original intensity, indicating that the FITC tagged on the tPdNPs was quenched over 84% through FRET. This fluorescence enhancement is probably due to the trypsin-specific cleavage of the substrate because the addition of trypsin inhibitor, which totally inactivates the trypsin, resulted in the absence of fluorescence in the solution.
To better understand the FRET between FITC and PdNPs, we tried to calculate the Förster distance (R 0) between the selected FRET pairs. Surprisingly, using the spectral data of PdNPs and FITC (see Online Supplementary Material, Figure S3), the calculated R 0 obtained from the wide absorption and high molar extinction coefficient of PdNPs at 400–700 nm (over 1 × 106 M−1·cm−1) was approximately 12 nm. As a consequence, the distance between donor and acceptor (r DA) lesser than 12 nm (R 0) will result in high FRET E (over 50%), and r DA lesser than 6 nm (0.5 R 0) will even exhibit 100% FRET E , 27 which is significant for the off–on state fluorescent probe. Therefore, the high fluorescent quenching of FITC in nano-probe over 84% (Figure 3(c)) was reasonable. Additionally, since PdNPs are very suitable for FRET-based sensors, our data were then analyzed using equation (3), which relates the FRET E to R 0, to determine the FITC donor-PdNPs acceptor center-to-center separation distance (r DA)
This analysis resulted in a calculated center-to-center distance between FITC and PdNPs of around 9 nm. The radius of PdNPs was around 2.5 nm, whereas the length of the stretched peptide CALNNGGARK(FITC) measured using Chem3D was approximately 5 nm. Thus, the calculated distance is approximately equal to the sum of both the radius and the peptide length. This indicated that the calculation of R 0 and r DA was precise.
Moreover, tPdNPs were able to be stored and maintain their excellent performance for a long run. The tPdNPs in the aqueous solution remained well dispersed at 4°C for more than 8 months. Importantly, there was no change in the fluorescence background and the fluorescence intensity of stored tPdNPs treated with trypsin compared with those freshly prepared (Figure 4), which means that peptide ligands coated on the surface of tPdNPs remain tightly coated after a long-term storage. Therefore, the tPdNPs were still well water-soluble, and the functional substrate regions remained active against trypsin as well. Additionally, as reported in the literature, CALNN-coated nanoparticles can be freeze-dried for long-term storage. 22 Thus, if CALNN-coated PdNPs are freeze-dried and stored at −20°C, long-time preservation can be realized, which is an important prerequisite for the clinical applications of tPdNPs.
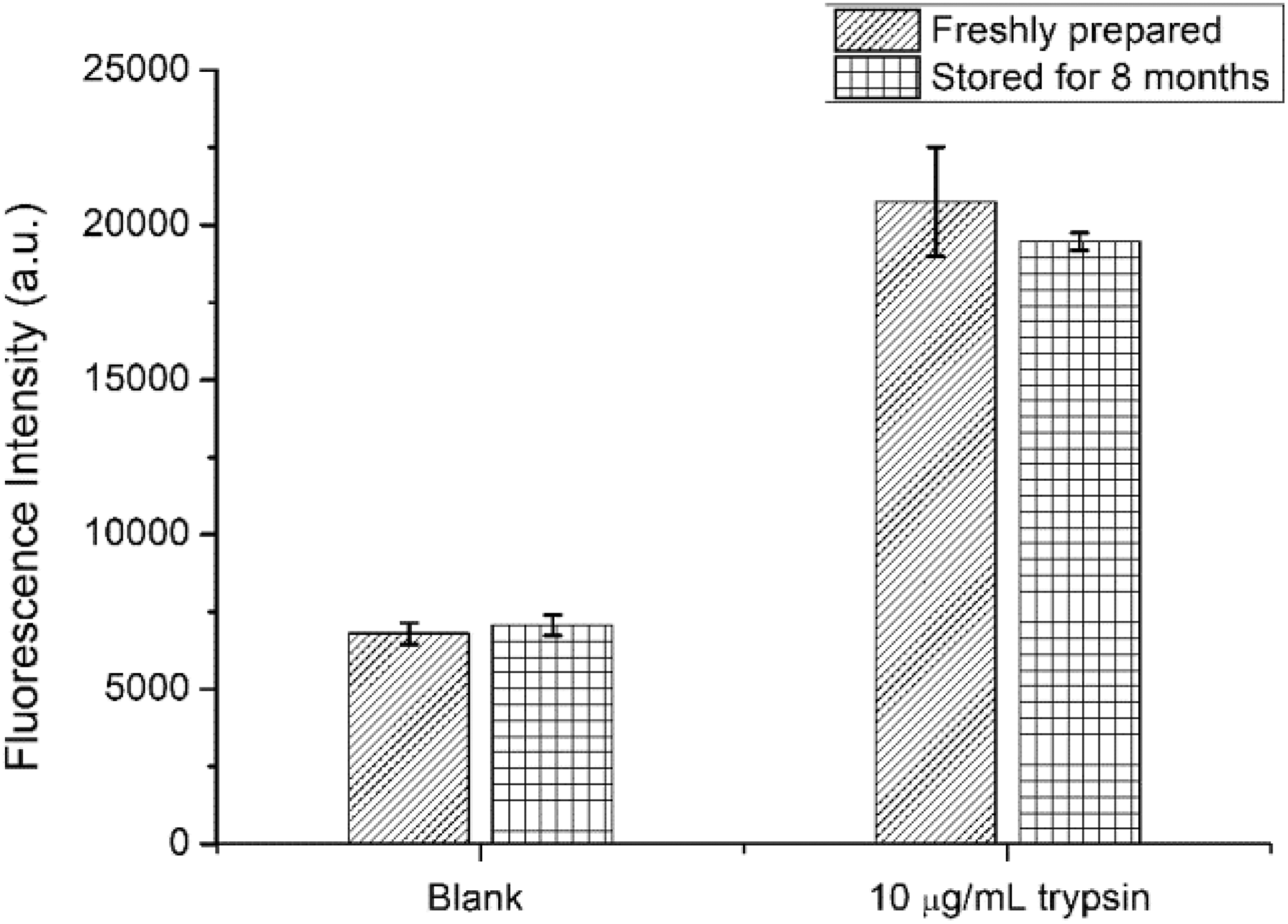
Fluorescence intensity of freshly prepared and stored tPdNPs without or with 10-μg/mL trypsin.
The detection of trypsin
Prior to detection, optimization of the conditions was performed. The concentration of tPdNPs was considered according to the upper detection limit of equipment, while the pH and temperature were 8.0 and 37°C, respectively. Thus, we only optimized the incubation time. As shown in Figure 5(a), the fluorescent intensity increased with time, linearly at the beginning, and reached the plateau within 100 min. Therefore, 100-min incubation should be performed prior to fluorescence detection to obtain a high and stable fluorescent signal. The relationship between fluorescence intensity and trypsin was tested under optimal conditions. As shown in Figure 5(b), there was a good linear correlation between the fluorescence intensity and the logarithm of concentration in the range of approximately 0.2–8-μg/mL. The calculated limit of detection (LOD) was 0.18-μg/mL, which was sufficient for the quantitative analysis of trypsin in human biological fluids. 3,29 The LOD was calculated using the formula: LOD = (3σ/s), wherein σ represents the standard deviation of the blank signal and s represents the slope of the calibration curve. 13 Compared with other reported fluorescent methods, this tPdNPs-based probe displayed a comparable linear range and LOD for trypsin determination (Table 1). Compared with the previous studies, 11 the method we proposed has a similar linear range but is more sensitive. Furthermore, the one-step preparation method of tPdNPs has significant advantages compared with other reported fluorescent methods.

(a) Curve of fluorescence intensity versus time of trypsin detection with the nano-fluorescent probe. Fluorescence was recorded by Tecan Infinite M200 Pro with the settings of 37°C, automatic cycling fluorescence intensity detection with 490-nm excitation, 520-nm emission, and 5-min time interval. The concentration of trypsin was 10-μg/mL. (b) The linear curve of the fluorescence intensity changes with the logarithm of the concentration of trypsin. ΔF = F − F 0, where F is the fluorescence intensity in the presence of trypsin, and F 0 is the blank control signal of nano-probe.
Comparison of different fluorescent methods for trypsin detection.

PdNPs: palladium nanoparticles; AuNC: gold nanocluster; GQD: Graphene quantum dot; FRET: fluorescence resonance energy transfer; LOD: limit of detection; AuNP: gold nanoparticle; BSA: bovine serum albumin; CdTe: cadmium telluride; QD: quantum dot; CRET: chemiluminescence resonance energy transfer.
To evaluate the applicability of the proposed method, the concentration of trypsin in urine samples was determined by a standard method. Human urine samples were diluted 10-fold with Tris-HCl buffer (pH = 8.0) for analysis, and the results were listed in Table 2. It can be seen that trypsin was not detected in urine samples collected from healthy individuals, which was consistent with the results of the literature report. 3 In addition, the recoveries of different concentrations were in the range of 95.5–99.2%, and the relative standard deviation was lesser than 4.87%, indicating that the proposed nano-probe for trypsin has potential application value in clinical diagnosis. The accurate detection of trypsin in real samples reveals the great selectivity of the proposed method because these samples consist of complex components.
Results of the determination of trypsin in human urine samples.
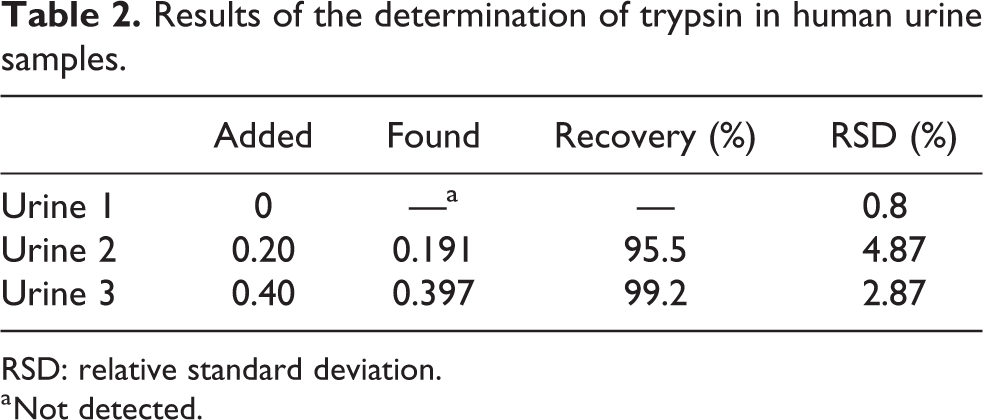
RSD: relative standard deviation.
a Not detected.
Conclusions
In summary, water-soluble peptide-coated PdNPs with excellent stability were synthesized using a one-step preparation method with the peptide CALNN as a stabilizing ligand. With this method, peptide-coated PdNPs can be functionalized by simply adding a functional peptide (such as CALNNGGARK(FITC)) complexed with CALNN during the preparation. Based on the prepared functional tPdNPs, a FRET-based detection for trypsin was achieved. Under optimized conditions, the proposed method can be used for the determination of trypsin in the range of approximately 0.2–8-μg/mL, and capable to detect active trypsin in a human biological fluid with good accuracy and selectivity. Additionally, the data show that PdNPs are very suited for FRET-based sensors. It is inferable that different functional PdNPs can be easily obtained by varying functional peptides. Therefore, considering the high quenching efficiency of PdNPs for varying fluorescent labels, the good biocompatibility of CALNN-coated and the large libraries of peptides, the functional peptide-coated PdNPs prepared by the simple and fast one-step method proposed in this article are expected to have a great potential for analytical, biological, and clinical applications.
Supplemental material
Supplemental Material, ESI - Peptide-coated palladium nanoparticle for highly sensitive bioanalysis of trypsin in human urine samples
Supplemental Material, ESI for Peptide-coated palladium nanoparticle for highly sensitive bioanalysis of trypsin in human urine samples by Guohua Zhou, Huimin Jiang, Yanfang Zhou, Peilian Liu, Yongmei Jia, and Cui Ye in Nanomaterials and Nanotechnology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (21605074, 21705071), the Natural Science Foundation of Guangdong Province (2014A030310274, 2016A030310362, 2017A030310604), the Research Group of Rare Earth Resource Exploiting and Luminescent Materials (2017KCXTD022), and the Natural Science Foundation of Lingnan Normal University (ZL1404).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.