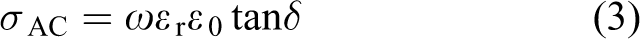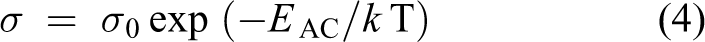Abstract
Magnesium Oxide/Praseodymium Oxide (MgO/Pr6O11) nanocomposite was prepared by sol–gel method. X-ray diffraction analysis confirmed the cubic structure. The morphology was studied using high-resolution scanning electron microscopy picture. The AC/DC conductivity and dielectric properties were investigated in the frequency range of 1–4 KHz and in the temperature range of 303–573 K. The results showed that the dielectric constant and the tangent loss were frequency and temperature dependent. The AC/DC conductivity at different temperatures indicated that the conductivity was thermally activated. The activation energy for AC/DC conductivity was calculated using Arrhenius plot.
Introduction
In recent years, nanocomposites have been the subject of renewed attention, due to their enhanced physical, chemical and biological properties and a wide range of applications. It has been found that the properties of nanomaterial differ from those of their individual atoms and molecules or bulk matter. The added advantage of nanocomposite is that the unstable bulk material can be rendered stable in the composite form. Ferrite–Magnesium Oxide (Fe–MgO), 1 Nickel–Titanium di-oxide (Ni–TiO2), 2 Aluminium–Aluminium Oxide (Al/Al2O3), 3 Aluminium/Silicon carbide (Al/SiC) 4 and Copper/Neobium (Cu/Nb) 5 are some of the nanocomposites that were used in various automotive and general industrial applications, such as aerospace, electronic and military, 6 battery cathodes, 7,8 microelectronics, 9 non-linear optics, 10 sensors 11 and so on. Rare earth-doped metal oxides are potentially attractive because such materials exhibit unique physical and chemical properties used for various optical and electronic applications. In this work, we synthesized the MgO/Pr6O11 nanocomposite through a sol–gel method. ‘Sol–gel’ route has proved to be a viable technique involving hydrolysis and polycondensation of a molecular precursor, such as metal alkoxides, to yield hydroxides or oxides under different conditions. Other advantages of sol–gel synthesis are that the final products prepared are homogeneous and products are relatively higher in purity.
In the present work, the AC conductivity, dielectric constant and dielectric loss measurements have been performed for MgO/Pr6O11 nanocomposites. The temperature and frequency dependence of the electrical conductivity and the dielectric constant for MgO/Pr6O11 nanocomposite in the frequency range of 1–4 KHz and in the temperature range of 303–573 K have been investigated.
Experimental details
For the synthesis of MgO/Pr6O11 nanocomposite, magnesium acetate tetrahydrate (MgAc) and praseodymium acetate (PrAc) are taken in the ratio 1:0.5 (1 MgAc:0.5 PrAc). Both precursors were dissolved separately with 50 ml double distilled water. The Pr acetate and Mg acetate solutions were mixed and stirred well to get homogeneous solutions. Then, 0.5 M of acetic acid was added dropwise to the aforementioned mixture and maintained under the temperature range of 50–60°C for 4 h. A gel that formed was allowed to dry at room temperature. The white powder product obtained was dried at a temperature of 90°C for 6 h in a hot air oven. The dried powder sample is further annealed at a temperature of 500°C for 2 h.
Results and discussions
Phase analysis
Figure 1 shows the X-ray diffraction pattern of MgO/Pr6O11 nanocomposites. From Figure 1, the samples that exhibit major peaks at 28.6447° (111)*, 32.0867° (200)*, 47.36° (220)* and 56.08 (311)* are assigned to cubic Pr6O11 (Joint Committee on Powder Diffraction Standards [JCPDS] 42-1121). The other peaks at 43.274° (200) and 62.242° (220) are assigned to cubic MgO (JCPDS 04-0829). The asterisk (*) marked in Figure 1 denotes the Pr6O11 phases. The crystallite size of the nanocomposite is calculated using the Scherrer formula: 12,13

XRD pattern of MgO/Pr6O11 nanocomposite. XRD: X-ray diffraction; The asterisk (*) marked is to denotes the Pr6O11 phases. It distinguishes MgO peak and Pr6O11 peaks.
where t is the crystallite size, λ is the wavelength of copper Kα radiation (λ = 1.54056 Å), β is the full width at half maxima and θ is the angle of diffraction. The crystallite size was found to be 12.4 nm.
High-resolution scanning electron microscopy and X-ray spectra analysis
The surface morphology of MgO/Pr6O11 nanocomposite studied using high-resolution scanning electron microscopey (HR-SEM) is shown in Figure 2(a) and (b). From the micrographs, it can be seen that the nanocomposite is flake shaped with small agglomeration. The energy dispersive X-ray spectra are shown in Figure 2(c). The inset shows the measured atomic concentrations of the elements with respect to the dopant ratio. The atomic percentage of Mg (36.14%) is more than Pr (9.73%).

(a) and (b) HR-SEM pictures of MgO/Pr6O11 nanocomposite. (c) Energy Dispersive Analysis of X-rays (EDAX) spectra of MgO/Pr6O11 nanocomposite. HR-SEM: high-resolution scanning electron microscopy.
Dielectric studies
The temperature dependence of dielectric constant (∊ r) and loss tangent (tanδ) of MgO/Pr6O11 nanocomposite in the temperature range of 303–573 K for four selected frequencies (1 KHz, 2 KHz, 3 KHz and 4 KHz) is shown in Figures 3 and 4. The relative dielectric constant (∊ r) of the samples can be obtained using the following formula:
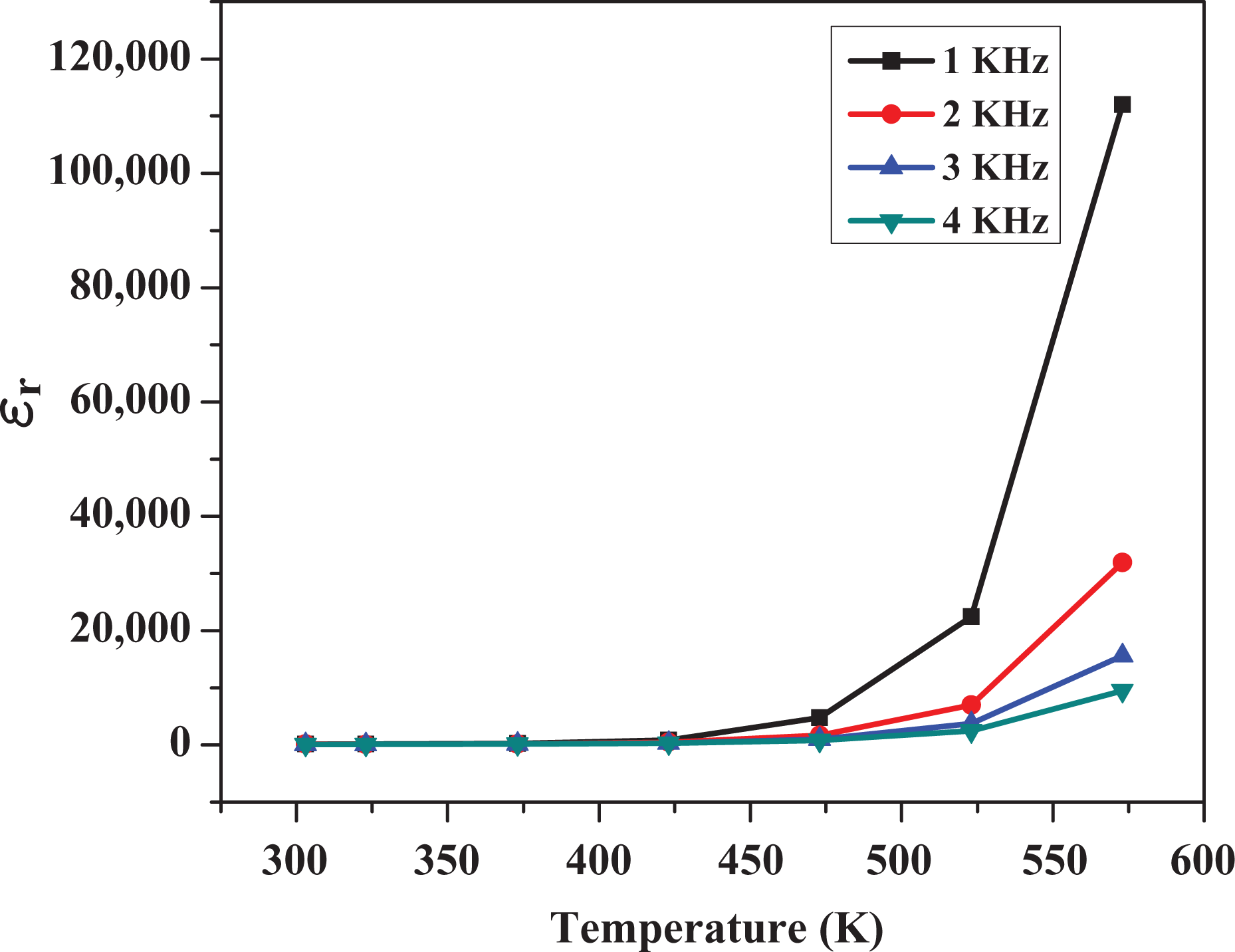
Variation of dielectric constant with temperature.

Variation of tangent loss with temperature.
where C, d, ∊ 0 and S are the measured capacitance, the thickness of the sample, the dielectric constant in vacuum (∊ 0 = 8.854 × 10−12 F/m) and the area of the sample, respectively. The dielectric constant decreases significantly with an increase in the frequency. This phenomenon can be attributed to the decreasing dipole polarization at higher frequencies. From Figure 3, it can be concluded that the dielectric constant increases with the increasing temperature. This is because the orientation polarization is related to thermal motion of the molecules and dipoles are hard to orient spontaneously at low temperature. Therefore, as the temperature increases, the orientation of dipoles becomes easier and thus the orientation polarization increases; this consequently increases the dielectric constant. At low temperature, high relaxation time exists and hence a low dielectric constant has been observed. At higher temperature, the thermal energy provided to the sample increases. Hence, the dielectric constant increases. The peak dielectric constant is higher for the sample at 573 K and is 112,251 at 1 KHz with a loss tangent of about 17.8. The tangent loss also decreases with the increasing frequency. The dielectric loss is rather high at low frequency but falls quickly with the rising frequency. Similar to the dependence of dielectric constant on temperature, the dielectric loss increases with the increase temperature. This indicates the thermally activated nature of the dielectric relaxation of the system. This type of thermally activated dielectric materials is useful for Complementary metal-oxide-semiconductor (CMOS) technology such as gate dielectrics, transistors and so on.
Figure 5 shows the dependence of AC conductivity, σ AC, on frequency (1–4 KHz) at different temperatures in the range of 303–473 K. The AC electrical conductivity (σ AC) of the dielectric material can be calculated using the following equation: 14

Variation of AC conductivity with log f.
where ω (=2πf) is the angular frequency and f is the applied frequency. It is clear from the figure that σ AC increases with the increasing frequency in the temperature range of 303–373 K. At higher temperature, a higher AC conductivity value has been observed but it decreases with increasing frequency. This is due to the short range translational hopping of charge carriers between the localized states. Figure 6 shows the AC conductivity log (σ AC) as a function of the reciprocal temperature (1000/T) in the investigated temperature range at different frequencies for the MgO/Pr6O11 nanocomposites. From the figure, it is seen that σ AC increases linearly with the increasing temperature. This may indicate that the AC conductivity is a thermally activated process, and it can be analysed according to the well-known Arrhenius equation: 15

Variation of AC conductivity with temperature.
where σ 0 is the constant, E AC is the activation energy for conduction, T is the absolute temperature, k is the Boltzmann’s constant and σ 0 is the pre-exponential factor including the charge carrier mobility and density of states. The obtained values of the AC activation energy (E AC) for frequencies 1 KHz, 2 KHz, 3 KHz and 4 KHz are 0.2284, 0.2279, 0.2266 and 0.2253 eV, respectively.
Figure 7 shows the variation of DC conductivity (σ DC) with frequency of range 303–473 K. The DC conductivity (σ DC) of MgO/Pr6O11 nanocomposite at various temperatures is calculated using the following relation: 16

Variation of DC conductivity with log f.
where L is the sample thickness, R is the resistance and S is the electrode area on the sample surface. From Figure 7, it is observed that the DC conductivity of the sample remains low and it increases linearly with increase of operating frequency. The temperature dependence of the DC electrical conductivity of MgO/Pr6O11 nanocomposite is shown in Figure 8. The DC activation energy (E DC) is calculated from the Arrhenius equation (using equation (4)). 15 The value of E DC was found to be 0.2139, 0.1999, 0.1913 and 0.1849 eV for 1 KHz, 2 KHz, 3 KHz and 4 KHz, respectively.

Variation of DC conductivity with temperature.
Conclusion
Nanocrystalline MgO/Pr6O11 composite is prepared by a simple sol–gel method. X-ray analysis confirmed the cubic structure. Electrical parameters such as dielectric constant, tangent loss, AC/DC conductivity as a function of frequency range 1 KHz–4 KHz and temperature range of 303–473 K were studied through impedance spectra. The frequency-dependent AC and DC conductivity at different temperatures indicated that the conductivity process was thermally activated. The temperature dependence of AC and DC conductivity showed a linear increase with increasing temperature. The calculated activation energy was found to decrease with the increasing frequency for AC and DC conductivity. The dielectric constant and the tangent loss were found to decrease with the increasing frequency and increase with the increasing temperature. This high dielectric MgO/Pr6O11 nanocomposite is potentially useful for microelectronic devices.
Footnotes
Acknowledgements
The authors wish to express their thanks to Sophisticated Analytical Instrumnent facility (SAIF), IIT Madras, India, for providing the facilities of HR-SEM and Energy Dispersive Analysis of X-rays (EDAX).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.