Abstract
Background:
Methotrexate (MTX) is the first-line treatment for rheumatoid arthritis (RA), and folic acid is routinely co-prescribed to prevent folate deficiency. However, no consensus exists regarding the optimal dose and frequency of folic acid supplementation.
Objectives:
This study aimed to compare the incidence of major adverse cardiovascular events (MACE) in RA patients receiving high versus low doses of folic acid alongside MTX.
Design:
A population-based retrospective cohort study.
Methods:
RA patients using MTX without baseline MACE were recruited retrospectively from a citywide database in Hong Kong between 2006 and 2015 and followed until 2018. The primary outcome was the first occurrence of MACE. Cox regression analyses with time-varying covariates were used to assess the association between folic acid dosage and incident MACE, adjusting for demographics, traditional cardiovascular risk factors, markers of inflammation and anti-rheumatic drug use.
Results:
A total of 8405 RA patients on MTX were identified. Of these, 6854 (78.5%) were female and the mean age was 56.0 ± 13.5 years. Among the cohort, 2967 patients (35.3%) received ⩾5 mg of folic acid daily, while 5438 (64.7%) received 0–<5 mg. After a median follow-up of 9 years (interquartile range: 5 years), 504 patients (6.0%) developed MACE. Multivariable analyses showed that the use of folic acid ⩾5 mg daily was associated with a significantly higher risk of MACE (adjusted hazard ratios were 1.34 (95% confidence interval (CI) 1.09–1.64) in the erythrocyte sedimentation rate model and 1.39 (95% CI 1.14–1.71) in the C-reactive protein model) compared to lower doses. The association remained significant after inverse probability treatment weighting and machine-learning gradient boosted regression modelling.
Conclusion:
High-dose folic acid supplementation may be associated with an increased risk of MACE in RA patients taking MTX.
Plain language summary
Rheumatoid arthritis (RA) causes joint pain and swelling. Patients often take methotrexate to ease symptoms and folic acid to reduce its side effects. This study looked at RA patients in Hong Kong from 2006 to 2018 to see if high doses of folic acid (5 mg or more daily) affect cardiovascular disease compared to lower doses. Among 8,405 patients, those taking high-dose folic acid had a higher risk of cardiovascular disease than those on lower doses. After adjusting for age, sex, and inflammation markers, the risk was about 34-39% higher with high doses. Since RA patients already face higher cardiovascular risks, doctors should carefully consider folic acid doses and discuss risks with patients. More research is needed to confirm these results and guide safer treatment.
Introduction
Methotrexate (MTX) is the first-line treatment for rheumatoid arthritis (RA). 1 Its inhibition of dihydrofolate reductase (DHFR) may lead to folate deficiency. 2 Thus, it is a routine practice to prescribe folic acid in combination with MTX. However, there has not been a consensus on the frequency and dose of folic acid supplements with MTX. The few randomized trials of folic acid supplements in doses from 1 to 5 mg daily have shown a significant reduction in the incidence of side effects such as mouth ulcers or deranged liver function from MTX.3–5 Globally, clinical practices for folic acid dosing vary widely. In a recent survey among dermatologists and rheumatologists in Ireland, folic acid supplementation ranges from 0.4 mg 6 days per week to 5 mg daily, with 23 practitioners (44%) prescribing 5 mg per week and 22 (40.4%) opting for 30–35 mg per week. 6 In Asian countries, doses exceeding 5 mg/week are more commonly used. 7
There have been recent concerns about the safety of high-dose folic acid supplements. Folic acid supplements and higher serum folate were associated with prostate cancer risk. 8 An analysis of over 14,000 participants in the National Health and Nutrition Examination Survey in the US found a J-shaped association that modest intake of folate was associated with lower risk of cardiovascular disease mortality and all-cause mortality, whereas excess folic acid supplementation was deleterious. 9 A pooled analysis of two randomized trials of B vitamins, including folic acid, found a negative interaction between folic acid and aspirin in cognitive outcomes in older people with mild cognitive impairment. 10 One potential mechanism of some of these adverse effects is the propensity of excessive unmetabolized folic acid and oxidized metabolites of folate to compete with active folates for folate receptors in cells and folate transporters.11,12 The accumulation of unmetabolized homocysteine in blood as a result of impaired active folate function could lead to severe oxidative stress. 13 Another possible mechanism is the effect of folic acid on gut microbiota, which have been implicated as a risk factor for cardiovascular events and mortality.14,15 A recent in vitro study on faecal samples of healthy young adults in China showed significant changes in the composition of gut microbiome and production of short-chain fatty acids upon inoculation of folic acid. 16
Patients with RA are at high risk of cardiovascular disease. 17 However, the effect of different doses of folic acid supplement on cardiovascular outcomes in RA patients has not been studied. It is postulated that high-dose folic acid supplements (5 mg daily or more) might lead to excessive cardiovascular risk. We therefore compared the incidence of major adverse cardiovascular events (MACE) in a cohort of RA patients on MTX on high versus low doses of folic acid.
Methods
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 18
Data source and subjects
This population-based retrospective cohort study utilized the Hospital Authority Data Collaboration Laboratory database in Hong Kong, which encompasses all clinical data from patients managed in 43 public hospitals and 49 specialist outpatient clinics. Mortality data were obtained from death certificates issued by the Hong Kong Immigration Department. The platform has been used in territory-wide epidemiological studies.19,20 Patients diagnosed with RA were identified based on physician diagnoses recorded using the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD9-CM) coding. The study included patients with the RA diagnostic code (ICD9-CM: 714) from January 2006 to December 2015, with follow-up extending until December 2018. Inclusion criteria were: (1) age 18 years or older, (2) documented prescription of MTX and (3) follow-up periods lasted at least 2 years and RA diagnostic codes lasted ⩾6 months. Patients with MACE prior to the RA diagnosis date were excluded. The study was approved by the Joint Chinese University of Hong Kong-New Territories East Cluster Clinical Research Ethics Committee (Ref. No.: 2021.024).
Outcome and covariates
The primary outcome of interest was the first occurrence of MACE, defined in this study as a composite of myocardial infarction, unstable angina, ischaemic or haemorrhagic stroke, transient ischaemic attack and cardiovascular death. Relevant ICD9-CM codes are detailed in Table S1. The exposure period was defined as the duration from study inclusion to either the first occurrence of MACE or the end of the study.
Demographics (age, sex and disease duration) and traditional cardiovascular risk factors (hypertension (HT), hyperlipidaemia (HL), diabetes mellitus (DM)) at baseline or ever were collected. The definitions of HT, HL and DM were based on ICD9-CM codes and/or dispensed prescription records for these conditions (Table S1). Medications were monitored utilizing British National Formulary codes, as detailed in Table S2. At each visit, markers of inflammation were collected, including serial measurements of erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) levels, along with medication prescription. The medications tracked included glucocorticoids, MTX, sulphasalazine (SLZ), leflunomide, hydroxychloroquine (HCQ), biological disease-modifying antirheumatic drugs (bDMARDs) and non-steroidal anti-inflammatory drugs (NSAIDs). The daily dose of folic acid was calculated using data extracted from prescription records, including drug strength (e.g. 5 mg), administration frequency (e.g. once daily), dosage form (e.g. 1 tablet), dispensed quantity (e.g. 14 tablets) and duration.
Statistical analysis
Descriptive statistics were reported as counts with percentages or as means ± standard deviations, with comparisons conducted using the χ2 test for categorical variables and the t test for continuous variables. Covariates significantly associated with MACE development (p < 0.05) in univariate analysis were included in the multivariable analysis. Time-dependent Cox proportional hazards regression models were utilized to examine the association between covariates, including age, sex, disease duration of RA, HT, HL, DM, ESR, CRP, and the use of NSAIDs, DMARDs, glucocorticoids and folic acid, and the occurrence of MACE. ESR and CRP were treated as time-varying covariates, updated annually, with yearly levels calculated as the mean of all measurements taken during that year. Medications dispensed throughout the follow-up period were likewise updated annually and analysed as time-varying exposures. Therefore, both ESR and CRP levels, as well as medication exposure, were expressed as yearly intervals in the Cox regression models. Folic acid usage was categorized into maximum daily dose of 0–5 and ⩾5 mg (i.e. ever prescribed folic acid 5 mg or more per day in 1 year). Missing ESR/CRP values were addressed using multiple imputations based on a fully conditional specification model, generating five imputed datasets via chained equations. The missing data represented less than 10% of the total records, and the imputation model included the outcome, all covariates and the treatments. Two sensitivity analyses were conducted for additional confounder control: (1) an inverse probability treatment weighting (IPTW) modelling with stabilized weights to reduce baseline variability; (2) a machine-learning algorithm to fit a gradient boosted regression model (GBM) using a flexible estimation method for adjusting a large number of pretreatment covariates. 21 A standardized mean difference (SMD) of <0.1 was regarded as the cutoff for achieving adequate balance. Covariate-adjusted survival curves were generated using Kaplan–Meier methods with IPTW applied to compare MACE-free survival between the groups receiving a maximum daily dose of folic acid of 0–<5 mg and those receiving ⩾5 mg. All analyses were performed in R V.4.0 (https://www.r-project.org/) using ‘dylpr’, ‘MICE’, ‘twang’, ‘survival’, ‘survminer’, ‘survey’, ‘mitools’ and ‘ggplot2’ packages.
Results
A total of 8405 patients with RA who had ever used MTX during the follow-up period were included in this study for analysis, encompassing 75,679 person-years of follow-up. The median follow-up time for the cohort was 9 years (interquartile range = 6–11 years). Their baseline characteristics are summarized in Table 1. The mean age was 56.0 ± 13.5 years, and 81.5% of them were female. At baseline, 31.4%, 7.7% and 2.9% of patients had HT, HL and DM, respectively. MTX was the most prescribed conventional synthetic DMARD (csDMARD), accounting for 80.1% of the cohort, followed by HCQ (32.5%), while only a small minority (3.7%) were on bDMARDs initially.
Baseline characteristics of rheumatoid arthritis patients in the entire cohort.

bDMARD, biological disease-modifying anti-rheumatic drug; COX2, cyclooxygenase 2; CRP, C-reactive protein; csDMARD, conventional synthetic disease-modifying anti-rheumatic drug; ESR, erythrocyte sedimentation rate; HDL, high-density lipoprotein; LDL, low-density lipoprotein; NSAID, non-steroidal anti-inflammatory drug; RA, rheumatoid arthritis; TNF, tumour necrosis factor.
The analysis included a total of 59,210 patient-years in the 0–<5 mg daily folic acid group and 16,469 patient-years in the ⩾5 mg folic acid group. The details of the folic acid prescription practice are shown in Figures S1 and S2. Among the 38,784 patient years with <5 mg daily folic acid prescribed, 20,780 prescriptions (53.6%) were 10 mg/week and 9509 (24.5%) were 5 mg/week. For prescriptions of ⩾5 mg daily, the majority were 5 mg daily (10,620/16,469; 64.8%) and 10 mg daily (4217/16,469; 25.6%). Table 2 compares characteristics during follow-up between the groups receiving 0–<5 mg folic acid daily and those receiving ⩾5 mg daily. Patients receiving a higher dose of folic acid were slightly older and more likely to be female. A higher proportion of them had HT and were prescribed NSAIDs or glucocorticoids. They also had higher ESR and CRP levels. After IPTW adjustment, all SMDs improved and most were <0.1 (Table 2). The Love plot is shown in Figure S3.
Before and after applying the IPTW in patients who used folic acid (0–<5 vs ⩾5 mg per day), considering the demographic variables as fixed over time and other characteristics as time-dependent predictors.

SMD: a standardized mean difference of less than 0.1 has been suggested as a cutoff for adequate balance.
Adjusted for age, sex, disease duration, CRP, traditional cardiovascular risk factors (ever hypertension, ever diabetes, ever dyslipidaemia) and medications (anti-TNF, non-anti-TNF, COX2 inhibitors, non-COX2 inhibitors, MTX, SLZ, HCQ, systemic glucocorticoid) during follow-up.
Adjusted for age, sex, disease duration, ESR, traditional cardiovascular risk factors (ever hypertension, ever diabetes, ever dyslipidaemia) and medications (anti-TNF, non-anti-TNF, COX2 inhibitors, non-COX2 inhibitors, MTX, SLZ, HCQ, systemic glucocorticoid) during follow-up.
bDMARD, biological disease-modifying anti-rheumatic drug; COX2, cyclooxygenase 2; CRP, C-reactive protein; csDMARD, conventional synthetic disease-modifying anti-rheumatic drug; ESR, erythrocyte sedimentation rate; HCQ, hydroxychloroquine; IPTW, inverse propensity of treatment weighting; MTX, methotrexate; SLZ, sulphasalazine; SMD, standardized mean difference; TNF, tumour necrosis factor.
A total of 504 patients (6.0%) developed their first MACE, yielding a crude incidence rate of 6.7 events per 1000 patient-years. Results from the time-varying univariate analysis for unadjusted hazard ratios (HRs) associated with MACE risk factors are presented in Table S3. All conventional cardiovascular risk factors – such as age, male sex, HT, HL and DM – along with markers of inflammatory burden (disease duration, ESR and CRP) were significantly associated with MACE. When comparing the two folic acid dosage groups, the use of ⩾5 mg daily was associated with a higher MACE incidence (HR 1.34; 95% confidence interval (CI) 1.10–1.63) in the univariate analysis. Most anti-rheumatic medications, including MTX, HCQ, anti-TNF bDMARDs and NSAIDs, were associated with a lower risk of MACE, while glucocorticoids and SLZ were detrimental.
In the multivariable analysis, daily folic acid use of ⩾5 mg was associated with increased adjusted HRs compared to 0–<5 mg. The HRs were 1.34 (95% CI 1.09–1.64) and 1.39 (95% CI 1.14–1.71) in the time-dependent event-specific risk models for ESR and CRP, respectively, after adjusting for traditional cardiovascular risk factors and other medications (age, sex, disease duration, CRP, ever HT, ever DM, ever HL, anti-TNF, non-anti-TNF, NSAIDs, MTX, SLZ, HCQ and systemic glucocorticoids) as shown in Table 3). Other independent predictors of MACE included older age, male sex and elevated ESR/CRP levels, while exposure to NSAIDs and MTX was linked to a lower risk of MACE. Following the application of IPTW, these findings remained consistent with the primary results from the regression models. When comparing folic acid use of ⩾5 to 0–<5 mg daily, the adjusted HRs were 1.77 (95% CI 1.36–2.31) and 1.66 (95% CI 1.40–2.12) in the ESR and CRP models utilizing stabilized weighting, respectively. Using GBM, daily folic acid doses of ⩾5 mg significantly increased the incidence of MACE compared to 0–<5 mg, with HRs of 1.95 (95% CI 1.52–2.49) in the ESR model and 1.89 (95% CI 1.47–2.42) in the CRP model. Figure 1 presents the adjusted MACE-free Kaplan–Meier survival curves for patients on high and low folic acid doses. Individuals exposed to folic acid ⩾5 mg daily had significantly reduced MACE-free survival compared to those receiving 0–<5 mg daily.
Multivariable Cox proportional hazards regressions after IPTW using the demographic variables as time fixed and the other features as time-dependent predictors (being updated at each visit) in the rheumatoid arthritis cohort.
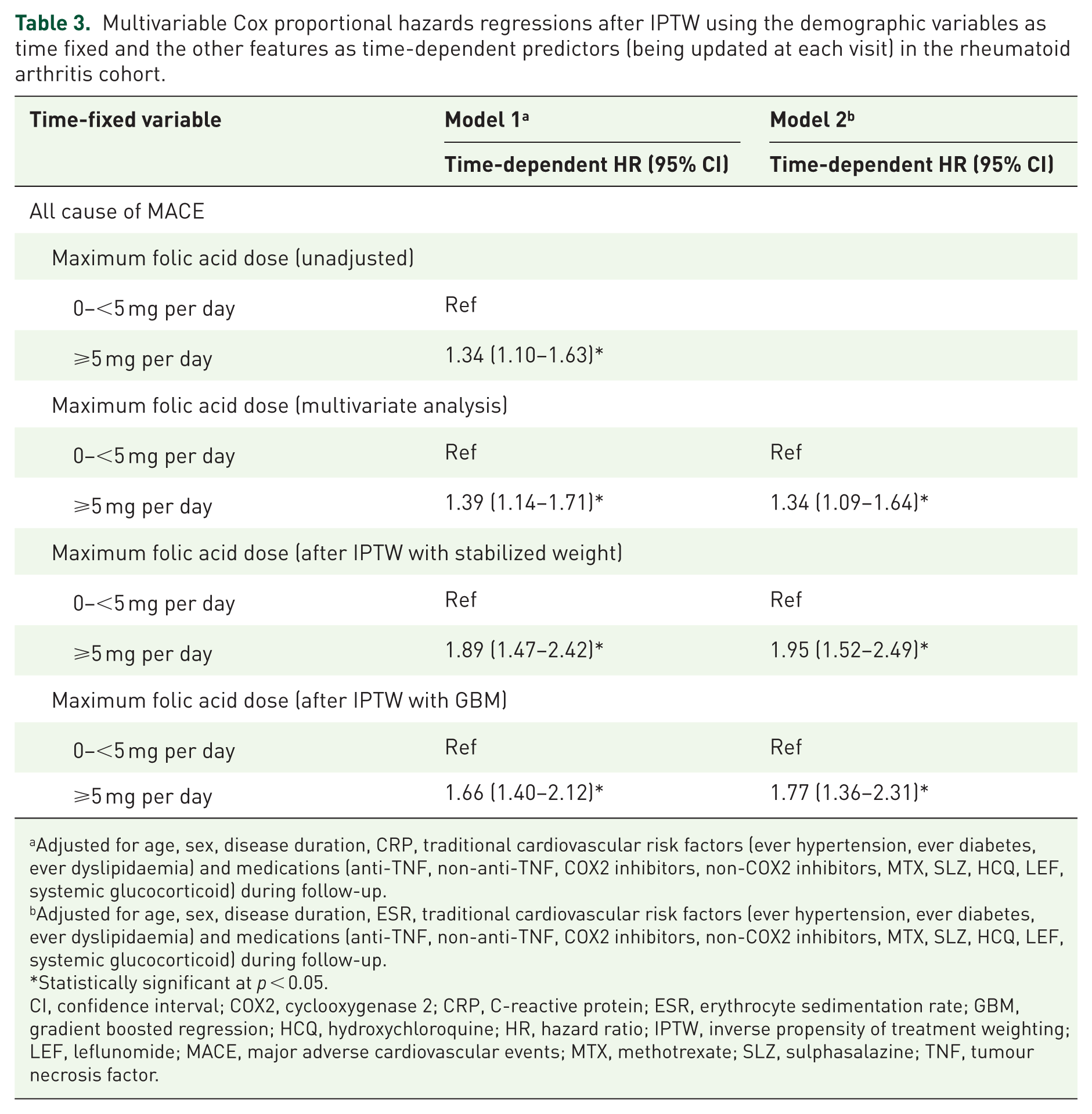
Adjusted for age, sex, disease duration, CRP, traditional cardiovascular risk factors (ever hypertension, ever diabetes, ever dyslipidaemia) and medications (anti-TNF, non-anti-TNF, COX2 inhibitors, non-COX2 inhibitors, MTX, SLZ, HCQ, LEF, systemic glucocorticoid) during follow-up.
Adjusted for age, sex, disease duration, ESR, traditional cardiovascular risk factors (ever hypertension, ever diabetes, ever dyslipidaemia) and medications (anti-TNF, non-anti-TNF, COX2 inhibitors, non-COX2 inhibitors, MTX, SLZ, HCQ, LEF, systemic glucocorticoid) during follow-up.
Statistically significant at p < 0.05.
CI, confidence interval; COX2, cyclooxygenase 2; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; GBM, gradient boosted regression; HCQ, hydroxychloroquine; HR, hazard ratio; IPTW, inverse propensity of treatment weighting; LEF, leflunomide; MACE, major adverse cardiovascular events; MTX, methotrexate; SLZ, sulphasalazine; TNF, tumour necrosis factor.

Adjusted survival curves for overall MACE survival by treatment group (0–<5 vs ⩾5 mg group), weighted by the inverse probability of treatment using the demographic variables as time fixed and the other features as time-dependent predictors (being updated at each visit) in the rheumatoid arthritis cohort.
In subgroup analyses examining cardiac and cerebrovascular outcomes separately, daily folic acid use of ⩾5 mg compared to 0–<5 mg was also associated with significantly increased risks. For cardiovascular diseases, the adjusted HRs were 1.90 (95% CI 1.33–2.72) and 1.81 (95% CI 1.27–2.58) in the ESR and CRP models after IPTW with stabilized weights, and 1.70 (95% CI 1.17–2.47) and 1.62 (95% CI 1.13–2.32) after IPTW with GBM. Additionally, HRs for stroke outcomes were 2.06 (95% CI 1.46–2.92) and 2.01 (95% CI 1.42–2.84) in the ESR and CRP models after IPTW with stabilized weights, and 1.89 (95% CI 1.30–2.76) and 1.79 (95% CI 1.26–2.55) after IPTW with GBM (Table S4).
Discussion
In this population-based retrospective cohort study with relatively large sample size, we report a 1.3-fold increased risk of cardiovascular outcomes associated with high doses (⩾5 mg daily) of folic acid compared to doses of less than 5 mg daily in RA patients on MTX, after controlling for multiple contributing factors. We also presented the prescribing patterns of folic acid in real-world clinical practice, capturing commonly used dosages across multiple centres over an extended observation period. In our study, 96.2% (6476/6731) of RA patients receiving MTX were prescribed folic acid at baseline. Despite co-prescription of folic acid in patients receiving MTX is universally recommended, a previous study examining a nationwide cohort of 2467 new users of oral MTX within the Veterans Health Administration revealed that only 73.0% of patients were given folic acid within 30 days of starting MTX. 22 Notably, patients not seen by rheumatologists were 23% less likely to receive folic acid.
The primary adverse events linked to MTX use include gastrointestinal issues and hepatic dysfunction. A meta-analysis involving 7 studies with 709 patients found that for RA patients treated with MTX, those supplied with folate were less likely to have gastrointestinal side-effects (odds ratio (OR) 0.71; 95% CI 0.51–0.99) and elevated transaminase (OR 0.15; 95% CI 0.10–0.23). 23 Folate also appeared to reduce MTX discontinuation compared to placebo (OR 0.29; 95% CI 0.21–0.42), though there was no statistical difference for mouth sores (OR 0.83; 95% CI 0.57–1.22). Several reviews and guidelines recommended a folic acid dosage of at least 5 mg per week with MTX therapy.24–27 However, only three double-blind RCTs assessed the effect of high versus low doses of folic acid supplementation in both MTX toxicity and efficacy. Morgan et al. aimed to evaluate the effects of two different weekly doses of folic acid (5 and 27.5 mg) compared to placebo in 94 patients. They found that folic acid did not affect the efficacy of MTX, as measured by joint tenderness, swelling scores and the Health Assessment Questionnaire at either dose. However, patients receiving folic acid had lower toxicity scores compared to those given a placebo (p < 0.001), with no significant differences observed between the two folic acid regimens. 28 The FOLVARI Study recruited 100 RA patients to be started on MTX and randomized them to receive folic acid 10 or 30 mg per week. 29 At 24 weeks, there was no difference in the co-primary endpoints of incidence of toxicity and change in disease activity between the two groups. In another trial, Stamp et al. 30 compared two doses of folic acid (5 and 0.8 mg per week) in 40 patients with active RA. At the 6-month follow-up, there was no significant difference in the change in Disease Activity Score (DAS) between the two regimens. Furthermore, there was no significant difference in MTX-related adverse effects between the two groups. All in all, current evidence was insufficient to determine the optimal dose of folic acid supplementation. The updated European Alliance of Association of Rheumatology (EULAR) recommendations for managing RA no longer specify the dosages for folic acid supplements in MTX users. 1 Similarly, the American College of Rheumatology guidelines recommended the use of folic acid to reduce the toxicity associated with MTX without mentioning the exact dosing. 31 Our study results showed that most patients used folic acid at a dosage of 10 mg/week (27.5%), followed by 5 mg daily (14.3%) and 5 mg per week (12.6%). This suggests that, in real-world practice, physicians tended to prescribe dosages higher than the minimal recommended dose of 5 mg per week, although overall the high-dose regime (5 or 10 mg/day) was less popular compared to 5 or 10 mg weekly dose (14,837 vs 30,289 patient years). This may be related to the need for larger doses of MTX for effective disease control, and the notion that higher doses of folic acid could better mitigate the adverse side effects associated with MTX.
Folic acid supplementation is known to lower plasma homocysteine levels. Several observational studies have demonstrated that high plasma homocysteine levels are predictors of cardiovascular diseases.32,33 However, it remains unclear whether lowering homocysteine can reduce the occurrence of cardiovascular diseases. A meta-analysis in 2006 found no benefits of folic acid supplementation for patients with a prior history of vascular disease, 34 while another comprehensive review in 2011 reported similar findings but did not stratify participants based on their cardiovascular disease history. 35 Additionally, a third analysis indicated that while folic acid showed no effect on primary cardiovascular disease or stroke endpoints, there might be potential harm for participants with high baseline homocysteine levels. 36 Conversely, a more recent meta-analysis found that folic acid supplementation reduced the risk of stroke by 10%. This effect remained stable regardless of baseline folate levels, folic acid dosage, baseline vitamin B12 levels, vitamin B12 dosage, homocysteine reduction, intervention duration or whether folic acid was taken alone or in combination (all p-interactions >0.05). 37 Of note, more than 80% of subjects in the trial intervention groups were on folic acid supplementation ⩽0.8 mg/day, and the folic acid efficacy was greater in areas without fortified grain. Another comprehensive review involving a total of 884 RCTs evaluating 27 types of micronutrients found that folic acid supplementation (median dose of 3 mg/day; range of 0.5–15 mg/day) reduced the risk of stroke (Risk Ratio (RR): 0.84; 95% CI 0.72–0.97), though no benefits on myocardial infarction and cardiovascular disease mortality could be demonstrated. 38 Specifically, the evidence regarding folic acid supplementation at a dosage of 5 mg/day remains inconsistent across different populations. Two randomized trials have reported potential cardiovascular benefits associated with this regimen in high altitude inhabitants and dialysis patients, respectively. Two other trials on post-myocardial infarction and end-stage renal failure patients have demonstrated neutral or non-significant associations.39,40 The vast heterogeneity of populations may have led to the discrepancies in cardiovascular outcomes observed in these studies. More importantly, these studies did not directly compare the effects of different doses of folic acid. Therefore, further dose–response meta-analyses or real-world studies with large sample sizes are essential to identify the optimal folic acid dosage that protects against the adverse effects of MTX without jeopardizing cardiovascular health.
Currently, most large-scale studies on the effects of folic acid on lowering homocysteine and its impact on MACE involve combining it with other B vitamins.41–44 Our study explored the sole impact of high-dose folic acid supplementation on MACE in RA patients with MTX. An adverse effect of high-dose folic acid supplementation on atherothrombotic risk has hypothetic biological plausibility. 45 First, folic acid promotes cell proliferation through its role in thymidine synthesis, which may also occur in atherosclerotic. 46 Second, hyperhomocysteinaemia, potentially linked to impaired active folate metabolism due to excessive folic acid intake, might be associated with altered methylation potential in vascular cells, possibly relating to changes in cell phenotype that could be observed in plaque development.45,47,48 Additionally, folic acid exposure has been suggested to influence the methylation of L-arginine to asymmetric dimethylarginine, an endogenous inhibitor of nitric oxide synthase, which might theoretically affect vascular function. 49 Lastly, despite the disappointing results of the CIRT (Cardiovascular Inflammation Reduction Trial), 50 observational studies repeatedly showed that patients with RA who received MTX had fewer cardiovascular events than patients who received other therapies or placebo.51,52 This speculative cardiovascular protective effect of MTX might be offset by the co-administration of high doses of folic acid, resulting in an apparent elevated risk of adverse events. A deeper understanding of these mechanisms is essential for clarifying the complex and potential dose-dependent relationship between folic acid supplementation, blood homocysteine levels and vascular health, especially in RA patients receiving MTX. In the meantime, it is advisable to review the indications of high-dose folic acid supplementation and keep good control of all modifiable cardiovascular risk factors, including arthritis disease activity.
This study has several limitations. First, causality cannot be definitively established and temporality between folic acid exposure and MACE cannot be fully confirmed. Despite adjustment with multivariable models, IPTW and GBM, residual and unmeasured confounding cannot be entirely excluded. Data on some potential confounders – such as smoking status, obesity, homocysteine levels and genetic background (including the methylene tetrahydrofolate reductase A1298C polymorphism) – were unavailable in the database. Additionally, MTX and glucocorticoid dosages were not adjusted for, which could have dose-dependent effects on cardiovascular outcomes. These factors, together with the dynamic interplay among inflammation, anti-rheumatic therapy and conventional cardiovascular risks, may have introduced additional sources of bias. Second, selection bias may arise from the non-random allocation of folic acid doses, as prescribing decisions might reflect patient characteristics (e.g. older age, female sex or comorbidities like hypertension) not fully captured in our data. Third, patients with shorter follow-up or early events may be underrepresented, introducing potential survival bias. Fourth, disease activity scores were not directly captured. However, we utilized markers of inflammation, such as ESR and CRP, over time as surrogates for disease activity, which can partially reflect the status of disease control. However, the influence of other causes of elevated inflammatory markers cannot be eliminated. Fifth, the study relied on prescription records to estimate folic acid exposure. It was not possible to assess adherence and to account for over-the-counter or dietary consumption of folic acid. Therefore, the actual intake of folic acid remains uncertain. In addition, information on over-the-counter supplements or dietary folate intake was not available, which may have led to exposure misclassification. The binary cutoff of folic acid dose also could not capture the potential non-linear effects or dose–response relationships which warrant further studies. Additionally, co-administration of folinic acid with MTX is not a common practice locally; otherwise, it would be intriguing to perform a subgroup analysis as folinic acid does not require metabolism by DHFR and thus will not lead to circulating unmetabolized folic acid. However, due to its potential dose-dependent attenuation of therapeutic effect of MTX and higher cost, folinic acid is primarily reserved as a rescue treatment for MTX toxicity.53,54 Moreover, outcome data were extracted from the electronic health record (EHR), which may be subject to coding errors. Nevertheless, prior validation studies of the territory-wide EHR system have demonstrated high coding accuracy.55,56 Finally, as the study population was derived from a single healthcare region in Hong Kong, the findings may not be generalizable to populations with different genetic backgrounds, prescribing practices or healthcare systems. Future prospective and mechanistic studies with more complete data capture and longer follow-up are warranted to validate these associations and clarify potential biological mechanisms.
Conclusion
The study found that high-dose folic acid supplementation may be associated with a higher risk of MACE compared with lower doses among patients with RA receiving MTX. Further prospective and mechanistic studies are required to evaluate the cardiovascular safety of high-dose folic acid.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X261423837 – Supplemental material for High-dose folic acid supplement is associated with increased cardiovascular risk in rheumatoid arthritis patients on methotrexate
Supplemental material, sj-docx-1-tab-10.1177_1759720X261423837 for High-dose folic acid supplement is associated with increased cardiovascular risk in rheumatoid arthritis patients on methotrexate by Huan Meng, Ho So, Xianfeng Yan, Yingzhao Jin, Steven H. Lam, Timothy Kwok and Lai-Shan Tam in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X261423837 – Supplemental material for High-dose folic acid supplement is associated with increased cardiovascular risk in rheumatoid arthritis patients on methotrexate
Supplemental material, sj-docx-2-tab-10.1177_1759720X261423837 for High-dose folic acid supplement is associated with increased cardiovascular risk in rheumatoid arthritis patients on methotrexate by Huan Meng, Ho So, Xianfeng Yan, Yingzhao Jin, Steven H. Lam, Timothy Kwok and Lai-Shan Tam in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors are grateful to all the staff of the Hospital Authority Data Collaboration Laboratory (HADCL) for their assistance. We would also like to acknowledge the S. H. Ho Foundation for its support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
