Abstract
Background:
Rheumatoid arthritis (RA) is complicated by a high risk of cardiovascular disease and requires the initiation of biological or targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) for persistently active disease despite first-line therapies. The influence of b/tsDMARDs, especially tsDMARDs, on cardiovascular risk in Taiwanese patients with RA remains unclear.
Objectives:
To compare the risk of major cardiovascular adverse events (MACEs) or venous thromboembolism (VTE) amongst RA patients initiating approved b/tsDMARDs for up to 5 years.
Design:
A nationwide, population-based, retrospective cohort study.
Methods:
Using Taiwan National Health Insurance (NHI) Research Database, we identified patients with RA initiating NHI-reimbursed b/tsDMARDs indicated for RA between 2001 and 2020. Study outcomes were newly developed MACEs or VTE within 5 years of the first b/tsDMARD initiation. Time-dependent Cox regression analysis was performed to determine the association between b/tsDMARDs and MACEs or VTE and independently associated or protective factors. Subgroup analyses by age at b/tsDMARD initiation and cardiovascular risk levels, as well as sensitivity analyses of b/tsDMARD initiation after 2012, were performed.
Results:
We enrolled 12,332 adults with RA initiating the first b/tsDMARD during pre-determined period. The incidence rates of MACE and VTE were 894 and 283 per 100,000 person-years, respectively. After adjustment, other b/tsDMARDs were not associated with a higher risk of MACEs or VTE than tumour necrosis factor inhibitors (TNFis) up to 5 years after initiation. Subgroup analyses by age at b/tsDMARD initiation and cardiovascular risk levels revealed consistent findings. Factors associated with or protective against MACEs or VTE were identified.
Conclusion:
No non-TNFi b/tsDMARD had a higher risk of MACEs or VTE than TNFis up to 5 years after initiation amongst patients with RA, and this remained consistent for those initiating their b/tsDMARD at age 65 years and older or with high cardiovascular risk.
Keywords
Introduction
Rheumatoid arthritis (RA) is a systemic chronic inflammatory disease characterised by additive and symmetric polyarticular synovitis, impacting approximately 0.5%–1.0% of the global population. 1 According to an updated nationwide longitudinal study, the estimated prevalence of RA in Taiwan was 97.5 per 100,000 population, and the incidence rate was 15.8 per 100,000 person-years (PYs) from 2002 to 2007. 2 In Taiwan, patients are diagnosed with RA if they fulfil the 1987 American Rheumatism Association revised criteria 3 or the 2010 RA classification criteria. 4 Clinical practice, including management of RA comorbidities, in Taiwan has adhered to updated management recommendations.5,6 Most medication prescriptions can be reimbursed by the Taiwan National Health Insurance (NHI) system under its co-payment guidelines. 7 Biological or targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) can be approved under NHI reimbursement if patients have two records of high disease activity at least 4 weeks apart, defined as Disease Activity Score 28 > 5.1, despite having received the following treatments: (i) ⩾2 conventional synthetic DMARDs (csDMARDs) for ⩾6 months, with full target doses for ⩾2 months, respectively, at the same medical institution unless intolerance occurs; or (ii) concomitant use of ⩾2 csDMARDs for ⩾3 months, with full target doses for ⩾2 months, respectively, and prednisolone at a dose of ⩾15 mg per day for ⩾3 months unless intolerance occurs. NHI-reimbursed rituximab is indicated only for those patients with an inadequate response to NHI-reimbursed tumour necrosis factor inhibitors (TNFis). 8
RA is associated with multiple extra-articular manifestations and comorbidities at diagnosis and throughout disease evolution owing to persistent systemic inflammation.9,10 Increased risks of incident cardiovascular events 11 and venous thromboembolism (VTE) 12 have been documented in patients with RA. These, along with the common manifestations of tendon and joint damage, further complicate the disease, lead to functional disability, impaired quality of life, 13 and increased premature mortality. 9 Comparative studies have explored the influence of various b/tsDMARDs on cardiovascular safety.14,15 After the ORAL Surveillance study reported a higher risk of major adverse cardiovascular events (MACEs) in cardiovascular risk-enriched patients (e.g. those aged 65 years and older, current smoking, and relevant history) using tofacitinib than TNFis, 16 several investigations prioritised the cardiovascular safety of tsDMARDs. However, many recent cohort studies, predominantly from Europe, have reported conflicting results,17–21 even amongst patients with cardiovascular risks.22,23 Currently, there is relatively limited data from large-scale comparative analyses of cardiovascular risk amongst patients with RA using different b/tsDMARDs in Taiwan. Despite rising global concerns, studies addressing safety issues amongst tsDMARDs users with RA in Taiwan are lacking. Thus, further research is required to optimise the management of patients with RA, which can be explored using population-based database research in Taiwan. 24 In this study, we aimed to compare the risk of MACEs or VTE amongst RA patients initiating NHI-reimbursed b/tsDMARDs up to 5 years after initiation and to identify potential associated factors.
Materials and methods
Data source
This was a population-based retrospective cohort study. Claims data for each patient requested in this study were obtained from the Taiwan National Health Insurance Research Database (NHIRD), with which data from the Taiwan Catastrophic Illness Registry could be interlinked from 1 January 2000 to 31 December 2020. The Taiwan NHI system, first implemented on 1 March 1995, is an exclusive, obligatory, single-payer scheme covering approximately 99.9% of the Taiwanese population. 24 The NHIRD is a repository of NHI claims data used in numerous population-based longitudinal epidemiological studies. The collected information includes claims data concerning registration, demographics, orders and dates of ambulatory and inpatient services utilisation, diagnoses and procedures with corresponding International Classification of Diseases-Ninth and Tenth Revision-Clinical Modification (ICD-9/10-CM) codes, medication prescriptions with corresponding Anatomical Therapeutic Chemical classification codes, and medical expenditures reimbursed by NHI. Nevertheless, it lacks information such as weight, height, substance use (including tobacco), prescriptions without NHI reimbursement, laboratory measures, disease activity, physical injury and functional measures.
Enrolment of study patients
Newly diagnosed biologic-naïve adult patients with RA were identified via the NHIRD using ICD codes (Supplemental Table 1) 25 and catastrophic illness certification for RA diagnosis from 1 January 2001 to 31 December 2020. Patients without b/tsDMARD use, with first b/tsDMARD use that did not meet the NHI co-payment guidelines for RA treatment, with first b/tsDMARD use before the first date of RA diagnosis or with missing data on urbanisation and insured amounts were excluded to ensure all selected patients were newly prescribed the NHI-reimbursed b/tsDMARDs approved for active RA. The patients were categorised into four mutually exclusive b/tsDMARD groups according to the mechanism of action of their first b/tsDMARD. Rituximab was not considered for primary analysis owing to its role as a second-line bDMARD under current NHI co-payment guidelines. Included b/tsDMARDs were those approved for first-line treatment of persistently active RA in Taiwan between 2001 and 2020, with details presented in Supplemental Table 2. Each selected patient was assigned an index date, defined as the date of initiation of the first NHI-reimbursed b/tsDMARD, which could help censor the occurrence of potential confounders within 1 year before b/tsDMARD initiation capable of influencing the risk of outcomes after initiation. The selection process for patients included in the final analysis is shown in Figure 1.

Flow diagram of inclusion and selection for the study population and categorisation into four b/tsDMARD groups based on first NHI-reimbursed b/tsDMARDs. Those initiated with rituximab as their first NHI-reimbursed b/tsDMARDs were not included.
Study outcomes
The investigated outcomes were newly developed MACEs or VTE within 5 years after initiation of the first NHI-reimbursed b/tsDMARDs, identified by their ICD-9/10-CM codes (Supplemental Table 1). MACE was defined as the composite of myocardial infarction necessitating ⩾3 days of admission unless death, ischaemic stroke, cardiovascular mortality, coronary artery bypass graft or procedures of coronary revascularisation, 26 and VTE as a composite of deep venous thromboembolism or pulmonary embolism. The positive predictive value of claims data in defining myocardial infarction, ischaemic stroke and VTE has been validated previously.27–29 The follow-up period for each patient was the interval between the index date and any of the following censoring events: 31 December 2020, occurrence of any outcome event, date of discontinuation as 90 days after the last prescription of the first b/tsDMARDs, date of direct switching of the first b/tsDMARDs, date of withdrawal from NHI system for any reason, death or reaching the maximum duration of 5 years after b/tsDMARD initiation, whichever occurred first.
Potential confounders of outcomes
Gender, age at index date, disease duration before the first b/tsDMARD initiation, socioeconomic status such as urbanisation levels, and insured monthly incomes were included as covariates. Urbanisation level was determined collectively by population density and physician number per 100,000 residents, as well as proportions of older individuals, agricultural workers and residents with a college-level education or above in the residential area of each patient. Selected comorbidities were recognised as one inpatient visit or ⩾3 ambulatory visits with corresponding ICD-9/10-CM codes within 1 year before the index date. Composite events, such as MACEs, cancers, mental illness, sepsis and VTE, were included to determine their association with cardiovascular outcomes. The use of concomitant medications within 1 year before the index date was determined by the corresponding Anatomical Therapeutic Chemical classification codes; these included immunosuppressants and systemic corticosteroids, with prednisolone equivalent daily dosages, antiplatelet drugs and anticoagulants. The included diseases, medications and their corresponding administrative codes are summarised in Supplemental Table 1.
Methods to enhance diagnostic accuracy and sensitivity analyses
Given that catastrophic illness certification of RA was issued after the diagnosis was certified by ⩾2 additional qualified rheumatologists, we included patients with corresponding diagnostic codes and the certification, which could be obtained from Taiwan Catastrophic Illness Registry, to increase diagnostic accuracy. Comorbidities were defined as one inpatient visit or ⩾3 ambulatory visits using administrative codes. B/tsDMARDs under NHI reimbursement can only be accessed after review and approval by qualified rheumatologists to ascertain the appropriate indications.
For data processing validity, we excluded patients with missing data on urbanisation and insurance amounts in NHIRD. In the case of withdrawal from the study, we only used available data before patients withdrew from NHI.
To further stratify the impact of comorbidities, we defined patients with high cardiovascular risk as those with initiation at age 50 years and older plus a recorded history of hypertension, diabetes mellitus, or hyperlipidaemia within 1 year before the index date or a recorded history of coronary heart disease before the index date, which could support an external comparison. Additionally, we analysed the interactive effects of hypertension and diabetes mellitus on the risk of outcomes using regression analyses.
We also conducted subgroup analyses amongst our crude incidence rate analyses and time-dependent Cox regression analyses according to age at index date with a cut-off point of 65 years (those aged 65 years or older were deemed older individuals in Taiwanese administrative regulations) and high cardiovascular risk.
To minimise potential bias resulting from advancing changes across the follow-up period and due to patients with initiation in different periods, sensitivity analyses focusing on the period after 1 January 2012, when golimumab was approved for RA treatment under NHI reimbursement in Taiwan, were performed. Furthermore, we performed propensity score matching amongst the TNFi, tocilizumab and abatacept groups by gender, year of birth and year of the index date, with the following incidence rate ratios (IRR) of outcomes and time-dependent Cox regression analysis presented.
Statistical analysis
Categorical variables are presented as the number (percentage) of patients, while continuous variables are presented as mean ± standard deviation. Amongst the b/tsDMARD groups, we compared categorical variables using the chi-square or Fisher’s exact test and continuous variables using the independent t-test. The crude incidence rates and IRR of the outcomes amongst study patients were analysed. Kaplan–Meier survival curves of the study outcomes were plotted. The risk of outcomes following b/tsDMARD initiation was determined using time-dependent Cox proportional hazards models to adjust for covariates after examining the proportional hazard assumption with Schoenfeld residuals and presented as adjusted hazard ratios (aHR) with 95% confidence intervals (CI). Cramer’s V correlation tables for comorbidities within 1 year before the index date were provided. A probability (p) value of <0.05 was considered statistically significant. Statistical analyses were performed using SAS version 9.3 (SAS Institute Inc., Cary, NC, USA).
Reporting standards
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 30
Results
Baseline characteristics of study patients
In total, we enrolled 12,332 newly diagnosed patients with RA who were initiated on b/tsDMARDs between 2001 and 2020. Amongst them, 8902 patients were using TNFis, 974 were taking tocilizumab, 994 were taking abatacept and 1462 were using tsDMARDs (Figure 1).
The baseline patient characteristics are shown in Table 1. The study patients were mainly female (76.3%), and the mean age at first NHI-reimbursed b/tsDMARD initiation was 54.3 ± 13.1 years. Upon comparison by treatment groups, we found that patients initiating b/tsDMARDs other than TNFis started their therapies at an older age, had a longer disease duration before initiation and had a lower proportion of lower monthly income. The abatacept group had an older age at initiation (58.0 ± 13.0 years). In TNFis group, a lower proportion of patients had metabolic comorbidities, such as diabetes mellitus (9.3%) and hyperlipidaemia (10.0%), before initiation. In tsDMARD group, a relatively higher proportion of patients had hyperlipidaemia (13.1%), but a lower proportion had MACEs before initiation (0.5%). The majority of study patients were given concomitant nonsteroidal anti-inflammatory drugs (NSAIDs) (98.7%) and methotrexate (91.0%). A higher proportion of patients in TNFis group were prescribed leflunomide (32.7%), methotrexate (91.4%), sulfasalazine (70.3%), cyclosporine (15.8%), azathioprine (3.8%), NSAIDs (98.8%) and a higher daily dose of corticosteroids; the tsDMARD group exhibited the opposite results. Baseline characteristics of the excluded patients with missing data are presented in Supplemental Table 3.
Baseline characteristics of study patients.

MACE was defined as the composite of myocardial infarction, ischaemic stroke, cardiovascular mortality, coronary artery bypass graft or procedures of coronary revascularisation. VTE was defined as the composite of deep venous thromboembolism or pulmonary embolism. High cardiovascular risk patients were those with initiation at age ⩾ 50 years plus recorded HTN, DM or hyperlipidaemia within 1 year before the index date, or recorded coronary heart disease before index date. A p-value of <0.05 is considered statistically significant.
Age at initiation of first b/tsDMARDs.
Insured monthly incomes lower than median income (⩽22,800 New Taiwan dollars/month).
bDMARD, biological disease-modifying antirheumatic drug; DM, diabetes mellitus; HTN, hypertension; MACEs, major adverse cardiovascular events; TNFis, tumour necrosis factor inhibitors; tsDMARD, targeted synthetic disease-modifying antirheumatic drug; VTE, venous thromboembolism.
Crude incidence rates of MACEs or VTE amongst study patients
Analyses of crude incidence rates and IRRs of the outcomes are shown in Table 2 and Supplemental Table 4. After a total of 45,300 PYs of follow-up, 2.42% of the study patients developed incidental MACEs, while 0.77% developed incidental VTE up to 5 years after initiation, with an incidence rate of 894 and 283 per 100,000 PYs, respectively. Considering MACEs, only abatacept had higher IRR versus TNFis (IRR, 1.45; 95% CI, 1.00–2.10), although no significant difference in risk was detected amongst b/tsDMARD groups (Log-rank test p-value = 0.232). No b/tsDMARD had a higher IRR, nor a difference in risk, in subgroup analyses of patients initiated at <65 years, at ⩾65 years and patients without and with high cardiovascular risk. All 29 patients who developed MACE in the tsDMARDs group were initiated with tofacitinib but reached a non-significantly higher IRR (1.27; 95% CI, 0.84–1.92; Supplemental Table 4). Regarding VTE, no b/tsDMARD had a higher IRR, and there was no difference in risk amongst the groups (log-rank test p-value = 0.554). Owing to the limited number of VTE events, analyses based on the respective b/tsDMARDs and subgroup analyses were not performed. Figure 2 presents the Kaplan–Meier curves.
Crude incidence rates of outcomes up to 5 years after b/tsDMARDs initiation.

MACE was defined as the composite of myocardial infarction, ischaemic stroke, cardiovascular mortality, coronary artery bypass graft or procedures of coronary revascularisation. VTE was defined as the composite of deep venous thromboembolism or pulmonary embolism. High cardiovascular risk patients were those with initiation at age ⩾ 50 years plus recorded hypertension, diabetes mellitus or hyperlipidaemia within 1 year before index date, or recorded coronary heart disease before index date. A p-value <0.05 is considered statistically significant.
bDMARD, biological disease-modifying antirheumatic drug; CI, confidence interval; IRR, incidence rate ratio; MACEs, major adverse cardiovascular events; TNFis, tumour necrosis factor inhibitors; tsDMARD, targeted synthetic disease-modifying antirheumatic drug; VTE, venous thromboembolism.

Kaplan–Meier curves of MACE (a, b) or VTE (c, d) up to 5 years after NHI-reimbursed b/tsDMARDs initiation.
Factors associated with MACEs or VTE amongst study patients
Tables 3 and 4 show results of time-dependent multivariable Cox regression analyses of factors for MACEs up to 5 years following b/tsDMARDs initiation, along with subgroup analyses. No b/tsDMARD was associated with higher risk than TNFis after adjustment. Factors associated with MACEs occurrence within 1 year before initiation were male gender, age at initiation, hypertension and prescription of corticosteroids >5 mg/day across all subgroups. For younger initiators (<65 years), associated factors also included disease duration before initiation (Multivariable Analysis B; aHR, 1.05; 95% CI, 1.01–1.09), valvular heart disease (aHR, 3.46; 95% CI, 1.61–7.44) and coronary heart disease (aHR, 2.29; 95% CI, 1.35–3.88). For older initiators (⩾65 years), associated factors also included cerebrovascular disease (aHR, 2.06; 95% CI, 1.24–3.42) and history of MACEs/VTE (aHR, 2.21; 95% CI, 1.14–4.28). Several comorbidities were identified as associated factors amongst patients with high cardiovascular risk, while valvular heart disease was identified as a factor amongst those without high cardiovascular risk (aHR, 3.70; 95% CI, 1.09–12.59). Factors associated with a lower risk included methotrexate and sulfasalazine prescription amongst younger initiators or patients without high cardiovascular risk and NSAID prescription amongst older initiators or patients with high cardiovascular risk. Hyperlipidaemia was associated with a lower risk for older initiators (aHR, 0.51; 95% CI, 0.31–0.84). Considering VTE, as shown in Table 5, no b/tsDMARD was associated with higher risk than TNFis after adjustment. Factors associated with VTE occurrence within 1 year before initiation included age at initiation (aHR, 1.03; 95% CI, 1.01–1.05), hypertension (aHR, 1.69; 95% CI, 1.05–2.71), chronic obstructive pulmonary disease (aHR, 2.03; 95% CI, 1.11–3.71), thyroid diseases (aHR, 3.45; 95% CI, 1.46–8.14), sepsis (aHR, 3.24; 95% CI, 1.09–9.60), history of MACEs/VTE (aHR, 25.51; 95% CI, 13.41–48.51), and prescription of corticosteroids >5 mg/day (aHR, 3.97; 95% CI, 2.18–7.22). Methotrexate, sulfasalazine and NSAID prescriptions were associated with a lower risk. Finally, Cramer’s V correlations amongst comorbidities within 1 year before the index date revealed low correlations between one another (Supplemental Table 5).
Associated factors associated with MACEs up to 5 years after b/tsDMARDs initiation, with subgroup analyses regarding age at initiation.
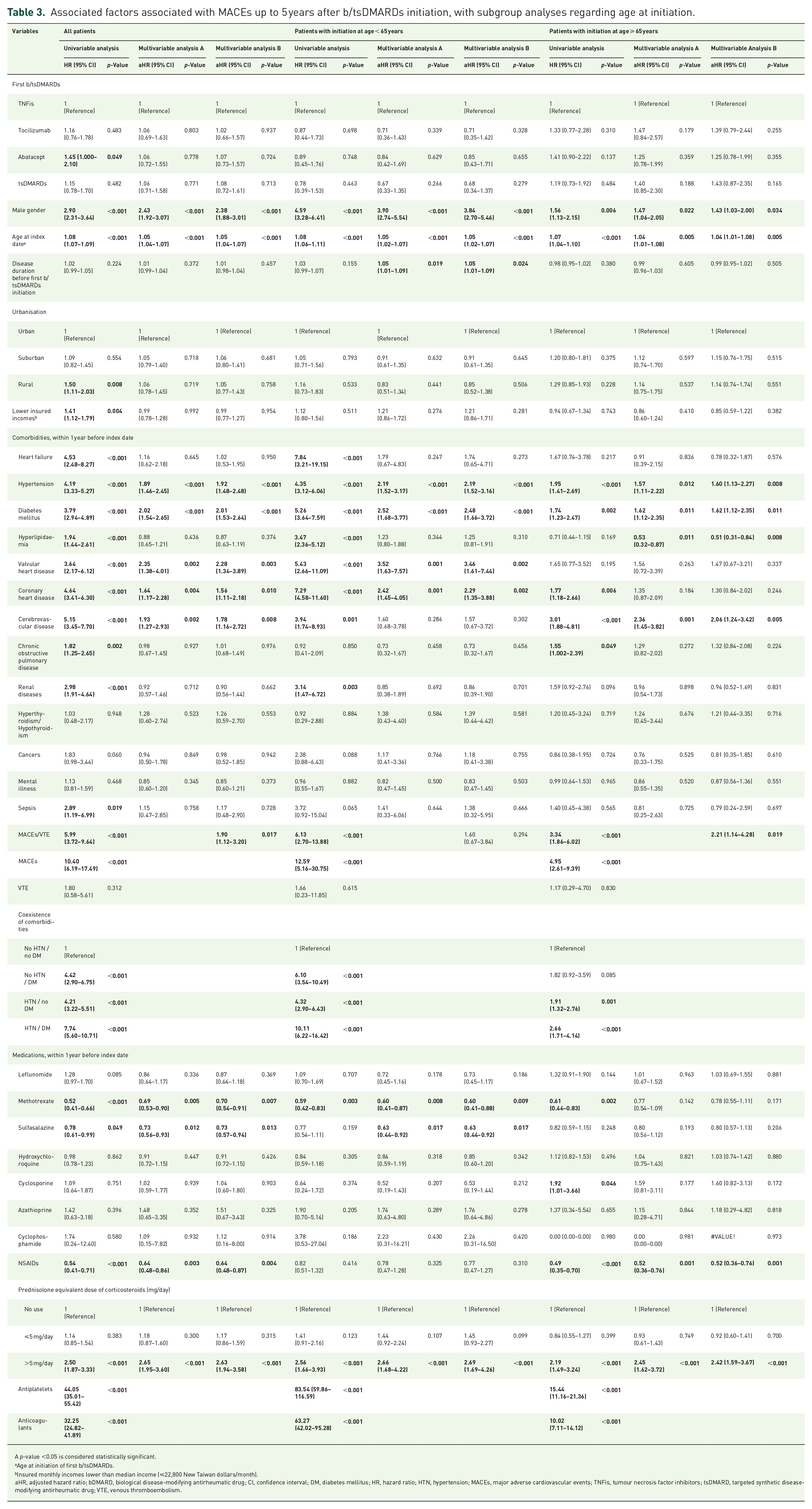
A p-value <0.05 is considered statistically significant.
Age at initiation of first b/tsDMARDs.
Insured monthly incomes lower than median income (⩽22,800 New Taiwan dollars/month).
aHR, adjusted hazard ratio; bDMARD, biological disease-modifying antirheumatic drug; CI, confidence interval; DM, diabetes mellitus; HR, hazard ratio; HTN, hypertension; MACEs, major adverse cardiovascular events; TNFis, tumour necrosis factor inhibitors; tsDMARD, targeted synthetic disease-modifying antirheumatic drug; VTE, venous thromboembolism.
Associated factors associated with MACEs up to 5 years after b/tsDMARDs initiation, with subgroup analyses regarding high cardiovascular risk.

A p-value <0.05 is considered statistically significant.
Age at initiation of first b/tsDMARDs.
Insured monthly incomes lower than median income (⩽22,800 New Taiwan dollars/month).
aHR, adjusted hazard ratio; bDMARD, biological disease-modifying antirheumatic drug; CI, confidence interval; DM, diabetes mellitus; HR, hazard ratio; HTN, hypertension; MACEs, major adverse cardiovascular events; TNFis, tumour necrosis factor inhibitors; tsDMARD, targeted synthetic disease-modifying antirheumatic drug; VTE, venous thromboembolism.
Associated factors associated with VTE up to 5 years after b/tsDMARDs initiation, with sensitivity analyses regarding initiation after 2012.

A p-value <0.05 is considered statistically significant.
Age at initiation of first b/tsDMARDs.
Insured monthly incomes lower than median income (⩽22,800 New Taiwan dollars/month).
aHR, adjusted hazard ratio; bDMARD, biological disease-modifying antirheumatic drug; CI, confidence interval; DM, diabetes mellitus; HR, hazard ratio; HTN, hypertension. MACEs, major adverse cardiovascular events; TNFis, tumour necrosis factor inhibitors; tsDMARD, targeted synthetic disease-modifying antirheumatic drug. VTE, venous thromboembolism.
Sensitivity analyses in the period after 2012
For MACEs, sensitivity analyses of patients initiated after 2012 showed that the associations were almost consistent (Supplemental Table 6). The results of propensity score matching amongst patients initiating TNFi, tocilizumab and abatacept after 2012, as well as the corresponding analyses, are presented in Supplemental Tables 7–10 and Figure S1. With 474 matched study patients for each bDMARD, no bDMARD had a higher IRR or a higher MACE risk than TNFis after adjustment, while profiles of associated factors were moderately altered. For VTE, sensitivity analyses revealed that the associations were almost consistent, whereas tocilizumab initiators were associated with a lower risk (Table 5).
Discussion
In this cohort study analysing 12,332 patients with RA initiated on b/tsDMARDs, our primary finding was that risk of MACEs or VTE was comparable amongst initiators of all first-line b/tsDMARDs in Taiwan over a follow-up period of up to 5 years, which remained consistent amongst patients initiating at the age of 65 years and older or with high cardiovascular risk. Moreover, associated factors and protective factors stratified by subgroup analyses based on age at b/tsDMARDs initiation and high cardiovascular risk were identified.
Given that the ORAL Surveillance Study raised the issue of cardiovascular safety of tsDMARDs, we showed that crude incidence rates of MACEs were 971 per 100,000 PYs for tsDMARDs and 1008 per 100,000 PYs for tofacitinib and that of VTE was 233 per 100,000 PYs for tsDMARDs. According to prior studies using NHIRD, a higher risk of premature cardiovascular events was detected amongst patients with RA than in the general population (crude incidence rate: 0.76 and 0.36 per 100 PYs; aHR, 1.21; 95% CI, 1.13–1.28), 31 and the crude incidence of VTE was 15.9 per 100,000 PYs in the Taiwanese population. 32 Under the Taiwanese NHI system, the maximum approved dose for RA treatment is 5 mg twice daily for tofacitinib and 4 mg once daily for baricitinib; however, prescriptions of daily dosages above these are possible. We also included patients with a history of MACEs or VTE to verify their potential role as associated factors. These factors should be considered when interpreting the crude incidence rates of the study outcomes. Currently, incidence rates reported by available large-scale research have varied considerably, mainly reflecting differences in study design, follow-up duration, definition of outcomes and ethnic or regional prevalence of cardiovascular diseases.17,18,23,33 Our results were similar to those of recent studies on MACEs in Western countries16,22; however, our VTE values were lower than those reported in a study conducted in France. 19 From an Eastern Asian perspective, we revealed a lower crude incidence rate of both MACEs and VTE amongst tsDMARD initiators than in Korean studies.21,34
Herein, we found no association between MACEs or VTE and tsDMARDs versus TNFis after adjustment, and this finding remained valid amongst those with a high cardiovascular risk, which coincided with most of the currently available large-scale studies. However, the limited number of VTE events restricted the subgroup and association analyses between VTE and tofacitinib. Regarding non-TNFi bDMARDs, only abatacept exhibited a higher crude IRR of MACEs, which was not observed in either subgroup analysis. This could be explained by the confounding effects of older age at initiation; higher proportions of patients with hypertension, chronic obstructive pulmonary disease, renal diseases, mental illness, history of MACEs and corticosteroid use; and a lower proportion of methotrexate users amongst abatacept initiators. After adjusting for covariates, no association was observed between abatacept use and MACEs. Likewise, no non-TNFi bDMARDs were associated with VTE versus TNFi after adjustment, consistent with most cohort studies. 35 The tendency of tocilizumab initiators to have a lower VTE risk may be influenced by the limited number of events.
Regarding factors associated with MACEs occurrence within 1 year before initiation, male gender, initiation at older ages, hypertension and prescription of corticosteroids >5 mg/day 36 could impact patients across all ages or cardiovascular risk levels. Later initiation of indicated b/tsDMARDs and concomitant valvular 37 and coronary heart disease were associated factors specific to younger initiators, whereas cerebrovascular disease and a history of MACEs/VTE 38 were factors specific to older initiators. Notably, factors such as male gender, hypertension, valvular heart disease and prescription of corticosteroids >5 mg/day conferred a higher risk to younger initiators or patients without high cardiovascular risk. We observed an adverse impact of later b/tsDMARD initiation on necessary control in younger patients through factors such as age at initiation and disease duration before initiation, which was consistent with that reported previously 39 and may reflect suboptimal awareness of inadequate RA treatment amongst younger Taiwanese patients. This indicates that strategies aimed at timely and intensive treatment and a reduction in misperception of RA progression are mandatory. Considering protective factors, the cardioprotective effects of methotrexate and sulfasalazine were consistent with those reported in previous studies, which were mainly attributed to their modulation of lipid profile, anti-inflammatory and anti-atherogenic effects. 40 Both csDMARDs benefited younger initiators or patients without high cardiovascular risk, suggesting their role in early disease stages and primary prevention. NSAID prescriptions benefited older initiators or patients with high cardiovascular risk, 41 highlighting the importance of anti-inflammation for high-risk patients. We identified hyperlipidaemia as a protective factor for older initiators, despite adjustment and a low correlation with other comorbidities. As it is a prevalent noncommunicable disease and lipid-lowering agents are commonly administered to older Taiwanese individuals, this may be biased by the effect of drug control.
Considering factors associated with VTE occurrence within 1 year before initiation, initiation at older ages, concomitant hypertension, chronic obstructive pulmonary disease, thyroid diseases, sepsis, history of MACEs/VTE, and prescription of corticosteroids >5 mg/day were associated factors, which were known risk factors for endothelial dysfunction and hypercoagulability. Methotrexate, sulfasalazine and NSAID prescriptions were identified as protective factors. Although this may be attributed to immunomodulation of inflammation or endothelial dysfunction, our findings contrast those of several prior studies. Given that we focused on b/tsDMARD initiators amongst Taiwanese patients with RA, our study probably reflected different study designs, ethnicities, patients of interest, or dosages.42,43 Notably, our results regarding factors associated with VTE may be skewed owing to the limited number of events.
This study had some limitations. First, database research is associated with several limitations. Although validated in previous studies, the diagnostic accuracy of covariates and outcomes in administrative databases has limited reliability, particularly for VTE. 29 The database did not contain data on body mass index, laboratory recordings, disease activity, 44 history of smoking or substance use, family history, or stressful events, which are potential confounders. Use of alternative medications, prescription dosage, prescription compliance and the actual date of the first b/tsDMARD discontinuation or switching could not be accurately determined. However, we made efforts to enhance the diagnostic accuracy and adjusted for associated factors as covariates for study outcomes as much as possible, using the accessible resources of the administrative database. Disease activity can be partially reflected and adjusted by using a daily equivalent dose of corticosteroids. Second, the retrospective use of claims data may limit causal relationships; therefore, we merely claimed associations and associated factors. Third, we set the time scale to within 1 year before the index date to examine continuous exposure to potential confounders while maintaining a substantial number of study patients for large-scale analysis. The influence of potential confounders occurring beyond 1 year before the index date could not be determined. Fourth, owing to selection bias, our results could not be extrapolated to patients with RA without NHI-reimbursed b/tsDMARDs. The global generalisability of our results is also limited, owing to the nature of regional research.
Despite the above-mentioned limitations, this was a population-based study using the NHIRD, which provides reliable data and minimises selection and recall bias. Compared with published studies from East Asia, we further conducted subgroup analyses of study outcomes based on age at b/tsDMARD initiation and cardiovascular risk levels. Though the treatment experience of RA with tsDMARDs is relatively limited, we still provide real-world evidence showing no preference for b/tsDMARD selection in terms of 5-year cardiovascular safety, regardless of the unanalysed factors. However, those initiated at younger ages, who are expected to be prescribed b/tsDMARDs for longer durations, still require stringent risk stratification before b/tsDMARD selection. As RA is a major cardiovascular risk factor, adequate monitoring and management of cardiovascular comorbidities are mandatory for patients using b/tsDMARDs. 6 Patients with associated factors should receive appropriate instructions and individualised preventive measures. This study provides reference data for global epidemiological analyses and Taiwanese health policymakers for the pharmacovigilance of b/tsDMARDs, as well as a basis for future Taiwanese research on immunologic mechanisms and translational medicine. Future studies supporting more definite causal relationships, accounting for smoking status and RA disease activity, and allowing censoring events to occur within a longer period are recommended.
Conclusion
This population-based study found no non-TNFi b/tsDMARD was associated with a higher risk of MACEs or VTE than TNFis up to 5 years after their initiation amongst Taiwanese RA patients, and this remained consistent for those initiating their first b/tsDMARD at age 65 years and older or with high cardiovascular risk.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251321917 – Supplemental material for Major adverse cardiovascular events or venous thromboembolism in patients with rheumatoid arthritis initiating biological or targeted synthetic disease-modifying antirheumatic drugs: a nationwide, population-based cohort study
Supplemental material, sj-docx-1-tab-10.1177_1759720X251321917 for Major adverse cardiovascular events or venous thromboembolism in patients with rheumatoid arthritis initiating biological or targeted synthetic disease-modifying antirheumatic drugs: a nationwide, population-based cohort study by Chung-Mao Kao, Yen-Ju Chen, Yi-Ming Chen, Der-Yuan Chen and Hsin-Hua Chen in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
We would like to thank Ms. Chia-Hui Yu and the Biostatistics Task Force of Taichung Veterans General Hospital for their assistance in performing the statistical analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
