Abstract
Background:
Evidence on the safety of intravenous neridronate (IV NER) in children with conditions other than osteogenesis imperfecta (OI) remains limited. This retrospective study describes adverse events, including acute phase reactions (APR) and hypocalcemia, over a median follow-up of 20 months in a monocentric pediatric cohort with rheumatological conditions.
Methods:
Thirty-seven children with OI and other bone diseases undergoing infusion of IV NER were included. Clinical, demographic, and serologic data were recorded.
Results:
The most common side effect was APR (62%), which occurred less frequently in patients on chronic oral cholecalciferol supplementation: 43% of the 14 patients who did not experience APR were supplemented, while none of the 23 subjects who developed APR received supplementation (p = 0.001). Conversely, pre-infusion 25-dihydroxycholecalciferol and calcium levels did not differ between patients who experienced APR and patients without APR (24.23 ± 12.96 vs 27.56 ± 11.81 ng/mL, respectively; p = 0.496). Premedication with prednisone does not seem to be effective in the prevention of APR. Hypocalcemia was rare and asymptomatic in all cases.
Conclusion:
NER appears safe across different pediatric conditions, with predictable first-infusion reactions. The role of vitamin D and premedication in children candidate to NER infusion should be further confirmed in larger cohorts.
Introduction
Bisphosphonates (BPs) are stable derivatives of inorganic pyrophosphate in which two phosphate groups are covalently linked to a carbon group (P-C-P structure) with two side chains (R1 and R2) attached at the central carbon. Similar to pyrophosphate, BPs bind to hydroxyapatite crystals, resulting in the inhibition of mineral growth and dissolution. 1 BPs also interact directly with cells as osteoclasts and monocytes, interfering with specific intracellular biochemical cascades. 1 Based on the presence of nitrogen or amino groups in the R2 position, BPs can be classified as non-nitrogen-containing or nitrogen-containing aminobiphosphonates (N-BPs). In particular, N-BPs exert their pharmacological action by blocking the farnesyl pyrophosphate synthase (FPPS), an enzyme in the mevalonate pathway catalyzing the biosynthesis of essential isoprenoids like carotenoids, sterols, and ubiquinones. FPPS is essential for the isoprenylation and geranylation of proteins: blocking this enzyme leads to an accumulation of unprenylated proteins in monocytes and osteoclasts, resulting in osteoclast loss of function 2 (Figure 1).
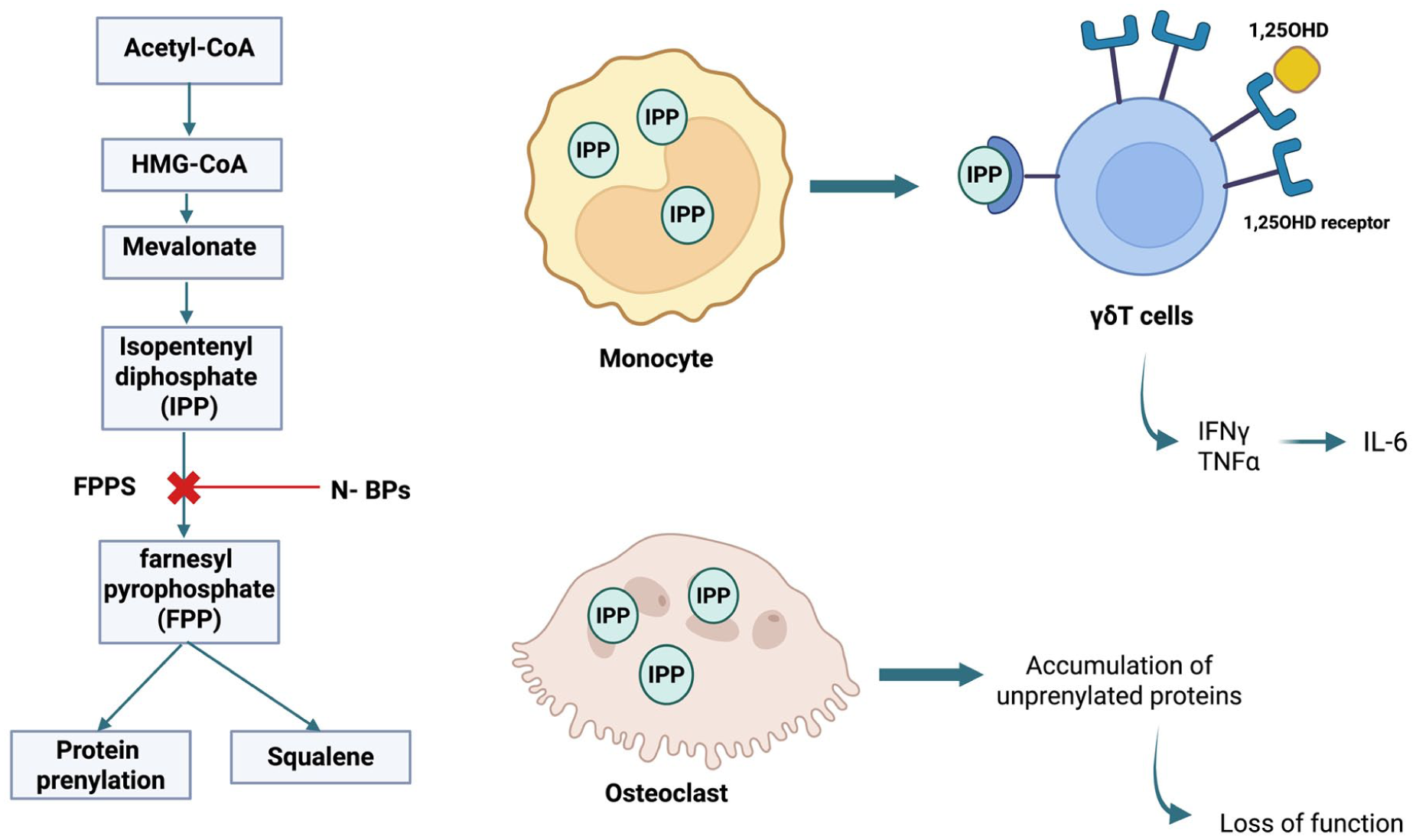
Visual representation of the pathogenic paradigm of acute phase reaction resulting from N-BPs. N-BPs inhibit FPPS, a key enzyme in the mevalonate pathway, leading to the accumulation of the upstream metabolite IPP in monocytes and osteoclasts. Monocytes interact with adjacent γδT cells with a consequent release of IFNγ and TNFα, promoting the release of other inflammatory cytokines. The upregulation of 1,25(OH)D receptors in γδT lymphocytes may suggest a role of cholecalciferol in immune modulation.
The main indication for BPs in pediatric age is osteogenesis imperfecta (OI), a setting where intravenous (IV) pamidronate is the most prescribed agent. Not surprisingly, the strongest evidence about the safety of BPs in pediatric patients can be extrapolated from studies on patients with OI mainly receiving pamidronate. 3 Similar to adult populations, acute phase reaction (APR) emerged as the most common adverse effect of IV BPs. APR consists of a transient inflammatory state, characterized by reversible flu-like symptoms such as fever, headache, arthralgia, myalgia, and chills. 4 APR usually occurs 24–36 h after the first IV BPs administration and typically does not recur at subsequent infusions. Although the pathogenesis of APR has not yet been fully elucidated, some studies demonstrated that the accumulation in monocytes of isopentenyl diphosphate and dimethylallyl diphosphate, secondary to FPPS blockade, may result in activation of adjacent γδT cells with the release of interferon-γ, tumor necrosis factor, and other inflammatory cytokines5,6 (Figure 1). Other side effects commonly reported in the pediatric population are asymptomatic hypocalcemia and the radiological finding of transversal metaphyseal linear bands of increased density (“zebra lines” sign) that result from alternate periods of increased and normal bone mineralization. 3 Atypical femoral fractures and medication-related osteonecrosis of the jaw have been reported anecdotally in the pediatric population. 3
Neridronate (NER) is another N-BP with a chemical structure and potency close to that of pamidronate, but with a less binding infusion schedule. In pediatric age, NER is currently licensed for OI, while indications in adults comprise Complex Regional Pain Syndrome (CRPS) and Paget’s disease. Data on the safety of NER in children are still scarce and limited to OI, even though interest in this molecule is rising in other pediatric settings. 7 Therefore, the present study aims to describe the safety of NER therapy in pediatric patients with rheumatological conditions.
Methods
Pediatric patients who received NER infusion at the Pediatric Rheumatology Unit, ASST G. Pini-CTO in Milan, Italy, were retrospectively included in the NAPOLEON registry. Approval by the local ethical committee was obtained (Approval number 5600_12.03.2025_P_bis, March 12, 2025, Comitato Etico Territoriale Lombardia 3). Clinical, demographic, and laboratory data were collected from patients’ medical records. Biochemical parameters, including serum levels of 25-OH vitamin D (25(OH)D), calcium, phosphorus, parathyroid hormone (PTH), and creatinine, assessed within 1 week before the first NER infusion and 4 weeks after the last IV NER, were recorded. Reference ranges of serum calcium, PTH, and phosphorus were established according to the patient’s age. 25(OH)D and PTH serum determinations were all assayed in the biochemical laboratory of Gaetano Pini Hospital, to avoid methodological issues. 25(OH)D cutoffs were defined according to the Italian Pediatric Society and the Italian Society of Preventive and Social Pediatrics, 8 the Endocrine Society, 9 and the Society for Adolescent Health and Medicine 10 as follows: deficiency <20 ng/mL; insufficiency 20–29 ng/mL; sufficiency ⩾30 ng/mL.
Chronic vitamin D supplementation consisted of a daily cholecalciferol intake of 2.000–3.000 IU lasting at least 4 weeks at the time of the first NER infusion. All subjects received NER infusion as outpatients.
Side effects of IV NER were monitored from the day of the first infusion to the latest follow-up. APR was defined as flu-like symptoms (pyrexia, arthralgia, myalgia, headache, nausea, malaise, and chills) occurring within 48 h after NER infusion. Patients self-recorded APR symptoms in standardized diaries for 48 h after each infusion. As for clinical practice, all patients were advised to take paracetamol (10–15 mg/kg every 8 h) in case of APR occurrence until symptom resolution.
Descriptive statistics were used to summarize data. Categorical variables were expressed as percentages. The normality of variable distribution was tested by Shapiro–Wilk test. Continuous variables with non-parametric distribution were expressed as median values (interquartile range, IQR), while continuous variables with parametric distribution were expressed as mean (standard deviation, SD). The Mann–Whitney test assessed differences in non-parametric continuous variables between groups, while associations between categorical predictors and outcomes were evaluated by Chi-square or Fisher’s exact test, as appropriate. Univariate and multivariable logistic regression models were used to measure the strength of association (odds ratio (OR) and related 95% confidence interval (95% CI)), adjusted for potential confounders. p-values ⩽0.05 were considered as statistically significant. Analyses were performed with R commander software (R Foundation for Statistical Computing, Vienna, Austria).
The study was conducted in agreement with the Declaration of Helsinki. All patients and parents provided written informed consent. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material).
Results
Thirty-seven children received NER infusion
Between 2010 and 2024, 37 pediatric patients (mean age 11.54 ± SD 3.27 years) received IV NER at our Institution, for a total of 138 infusions. Demographic and clinical features are reported in Table 1. Treatment with IV NER was most commonly administered because of bone marrow edema (BME) syndromes (48%): 7 patients (39%) had post-traumatic BME, while 10 patients (55%) had an orthopedic predisposing condition (osteochondritis (n = 4); skeletal dysplasia (n = 2); Blount disease (n = 1); flatfoot (n = 2); valgus knee (n = 1)). Eight patients received IV NER because of chronic recurrent multifocal osteomyelitis (CRMO, 22%); all had vertebral involvement with or without vertebral fractures. Additional indications included CRPS and OI. Patients with OI were treated with a single infusion of NER at a dose of 2 mg/kg every 3 months, according to the schedule authorized by the Italian Drug Agency.3,11 In case of conditions other than OI, the protocol consisted of four consecutive infusions 3 days apart according to the authorized schedule in adults with CRPS, 12 adopting the same dosing used in OI (2 mg/kg). Patients receiving NER after 01/2023 were also administered oral prednisone premedication 0.5 mg/kg 30 min before and 24 h after NER infusion.
Demographical and clinical features of the 37 patients who underwent neridronate infusion.
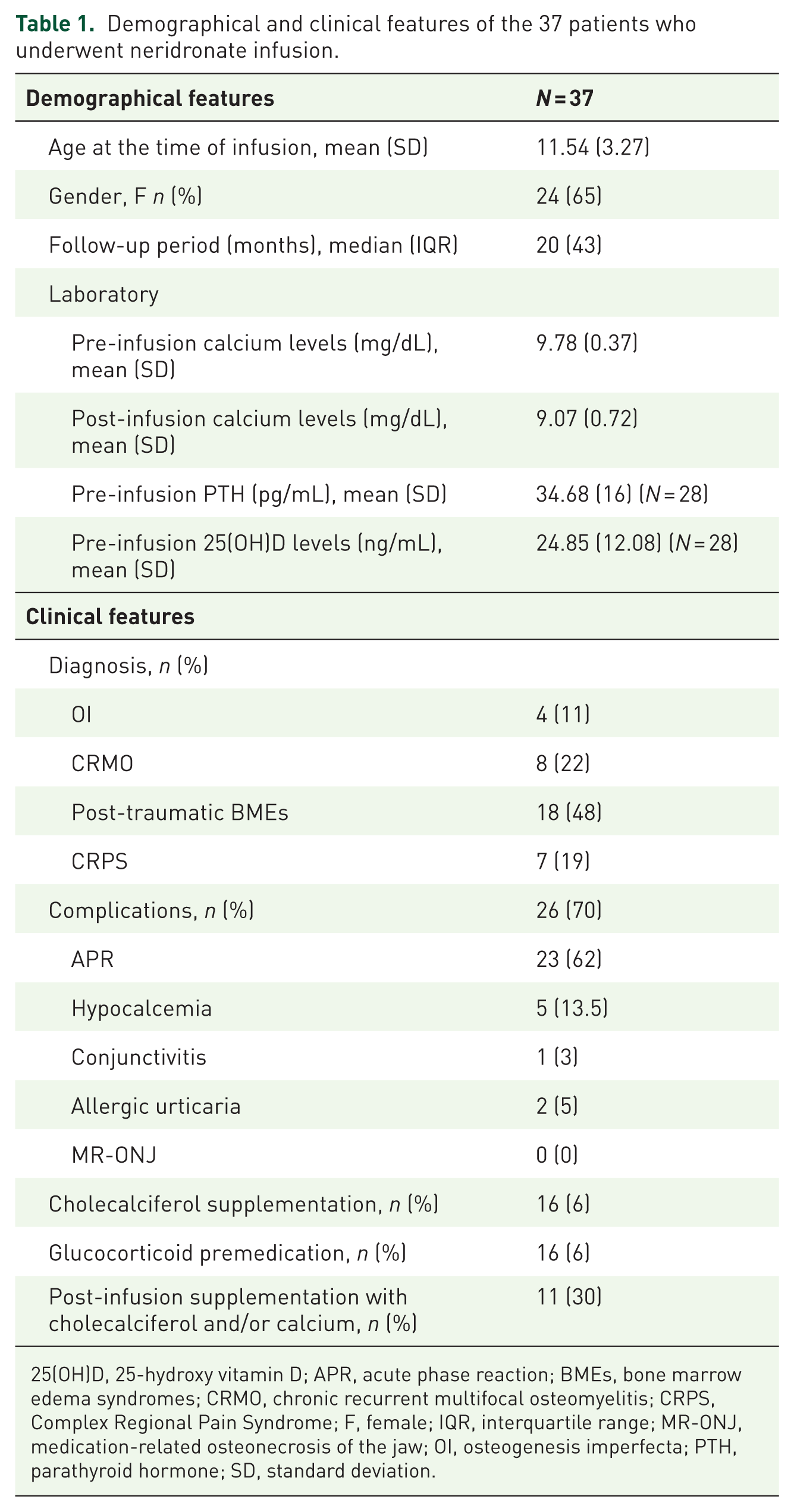
25(OH)D, 25-hydroxy vitamin D; APR, acute phase reaction; BMEs, bone marrow edema syndromes; CRMO, chronic recurrent multifocal osteomyelitis; CRPS, Complex Regional Pain Syndrome; F, female; IQR, interquartile range; MR-ONJ, medication-related osteonecrosis of the jaw; OI, osteogenesis imperfecta; PTH, parathyroid hormone; SD, standard deviation.
Occurrence of APR
Over a median follow-up time of 20 months (IQR 43), complications occurred in 26 patients (70%), consisting mainly of APR (n = 23/26; 88%). All complications manifested only after the first infusion of NER and did not recur with subsequent infusions. The occurrence of side effects did not differ upon the adopted therapeutic regimens (a single NER infusion every 3 months vs four consecutive NER infusions 3 days apart, p = 0.41).
Arthromyalgias and fever were responsive to paracetamol in 15 of 23 patients (65%) and to paracetamol plus non-steroidal anti-inflammatory drugs combo in 3 additional subjects (13%). The remaining five patients (22%) did not require analgesic or antipyretic therapy since symptoms were mild. The efficacy of NER therapy on pain reduction was not impaired by APR occurrence (p = 1).
Logistic regression did not disclose any association between age or gender and APR onset; premedication with prednisone did not affect the rate of APR occurrence (p = 0.27).
Vitamin D and APR
As shown in Table 2, ongoing oral cholecalciferol supplementation exerted a protective role against the development of APR: 43% of the 14 patients who did not experience APR were supplemented before NER infusion, while none of the 23 subjects who developed APR received supplementation (p = 0.001). The incidence of APR did not differ based on the adopted cholecalciferol dosage regimen (p = 0.09).
Differences between patients with and without APR.

25(OH)D, 25-hydroxy vitamin D; APR, acute phase reaction; IQR, interquartile range; SD, standard deviation.
No patient receiving cholecalciferol supplementation before IV NER presented flu-like symptoms suggestive of APR (Figure 2).

Incidence of acute phase reaction in patients taking ongoing cholecalciferol supplementation before neridronate infusion (a) and in patients with 25(OH)D deficiency (b).
The pre-infusion 25(OH)D levels were similar between patients who experienced APR and those who did not (24.23 ± 12.96 vs 27.56 ± 11.81 ng/mL, respectively; p = 0.496), even though data were available for 28 patients only. Logistic regression did not disclose any association between 25(OH)D levels and APR onset. In 8/28 patients (30%), pre-infusion 25(OH)D levels were below 20 ng/mL (vitamin D deficiency). In this subgroup of patients, the prevalence of APR was 88%, as compared to 40% in patients with higher 25(OH)D levels (⩾20 ng/mL, OR: 8.67; 95% CI: 0.8–453.6; p = 0.044; Figure 2). There were no statistically significant differences in the rate of APR between patients with vitamin D insufficiency (20–29 mg/mL) and those with sufficient levels (>30 mg/mL; p = 1). In addition, male and female subjects had similar serum 25(OH)D (p = 0.344).
Hypocalcemia
Only 5 out of 37 patients (13.5%) experienced post-infusion hypocalcemia, presenting exclusively after the first infusion. All patients who experienced hypocalcemia were asymptomatic, and none of them required specific IV rescue therapy. Hypocalcemia was resolved in all cases by prescribing a supplementation with calcium and cholecalciferol for a maximum period of 30 days. None of the patients who developed hypocalcemia was receiving cholecalciferol or calcium before the infusion. The lowest recorded blood calcium value was 7.5 mg/dL, with post-infusion mean calcium values of 9.07 mg/dL (±SD 0.72). Pre- and post-infusion serum mean calcium levels were similar (9.78 ± 0.37 vs 9.07 ± 0.72; 95% CI: 0.49–1.10; p = 0.001). No statistically significant association emerged between post-infusion hypocalcemia and prior calcium supplementation (p = 0.63) or cholecalciferol supplementation (p = 0.39); the incidence of hypocalcemia did not differ in relation to the cholecalciferol dosage regimen adopted (p = 0.08). No association was found between APR and calcium supplementation preceding the infusion (p = 0.43) or pre-infusion serum calcium levels (p = 0.19).
At a multivariable regression analysis adjusted for cholecalciferol supplementation, pre-infusion calcium, and 25(OH)D levels, lower age at first infusion approached statistical significance in predicting post-infusion hypocalcemia (OR: 2.29; 95% CI: 0.94–5.58; p = 0.068).
Other adverse events
Post-infusion conjunctivitis manifested in one subject, not requiring specific therapy; two patients developed allergic urticaria, treated with antihistamines and oral prednisone with complete resolution. No case of osteonecrosis of the jaw, phlebitis, or atypical femoral fracture manifested in our cohort.
Discussion
To our knowledge, the present study provides the first evaluation of the safety profile of IV NER in children and adolescents with conditions other than OI. IV NER may be useful in a large spectrum of pediatric disorders, but solid data on safety in children still need to be collected. Our study confirms that even in the pediatric population, APR is the most common side effect of IV NER, presenting in 62% of patients. Such an incidence is higher than what is reported in the literature. In two previous studies on pediatric patients with OI treated with IV NER, APR occurred in 47% 13 and 24% 11 of cases, respectively. In a recent description of five pediatric patients with pediatric type I CRPS, APR presented in one case. 7 Although it is a transient, benign reaction, APR can affect adherence to therapy. In the NAPOLEON cohort, APR occurred invariably after the first infusion only. Interestingly, a protective role emerged for chronic oral cholecalciferol supplementation before infusion, a finding that should be corroborated in larger studies due to our limited sample size. Despite the protection ensured by supplementation, 25(OH)D levels were higher in the population that did not experience APR, but without obtaining statistical significance, possibly due to the small sample size. Nevertheless, APR was more common in patients with 25(OH)D deficiency as compared to patients with values equal to or greater than 20 ng/mL (88% vs 44%, Figure 2). These findings suggested a potential protective role of 25(OH)D levels in preventing APR, as emerged in adult literature.14–16 Such protection might be mediated by the immunoregulatory effects of vitamin D 17 : in vitro, 1,25(OH)D negatively modulates the function of γδ T lymphocytes via the vitamin D receptor, which is upregulated upon cell activation (Figure 1). 18 However, despite seasonal fluctuation with higher cell rates in winter compared to summer, the number of circulating γδ T cells was not shown to directly correlate with serum levels of 25(OH)D. 14 The present study could not confirm the negative association between serum calcium and APR that had been described in adults receiving IV BPs,15,16,19–21 a dissimilarity that can be mediated by the impact of age on metabolism and pharmacological clearance.
In the adult population, several premedication regimens with corticosteroids (e.g., a 3-day course of dexamethasone 4 mg 22 or dexamethasone 4 mg daily for at least 3 days plus paracetamol 500 mg 4 times daily23,24) have demonstrated efficacy in preventing APR after zoledronic acid infusion. Following the emerging evidence in adults, we have recently adopted a premedication regimen. In our cohort, prednisone 0.5 mg/kg 30 min before IV NER and 0.5 mg/kg the following day did not affect the occurrence of APR, a finding that needs to be further confirmed in additional and larger studies in pediatric populations. The poor protective role exerted by premedication with oral steroids in the pediatric population can be explained by the short therapeutic schedule (two administrations only, to minimize side effects in pediatric subjects) compared to the 3-day scheme adopted on adult patients.
In the NAPOLEON cohort, hypocalcemia occurred rarely (13.5%), with an incidence rate comparable to literature data. 25 In a recent study on OI patients, Maines et al. reported a positive correlation between baseline 25(OH)D levels and post-infusion calcium values, identifying low 25(OH)D values as a candidate risk factor for post-infusion hypocalcemia. In our patients, prior calcium or cholecalciferol supplementation or normal serum 25(OH)D or calcium values were not associated with a lower risk of post-infusion hypocalcemia. Furthermore, the incidence of hypocalcemia did not differ with the IV NER dosage regimen. Lower age at first infusion is associated with post-infusion hypocalcemia, even after adjusting for cholecalciferol supplementation, pre-infusion calcium, and 25(OH)D levels. To date, there are no recommendations about calcium supplementation after infusion of BPs; however, it can be helpful to check dietary calcium intake and eventually prescribe a brief post-infusion supplementation with oral calcium carbonate in selected patients in order to avoid secondary hypocalcemia.
This study has several limitations. The retrospective design impinges on the quality of data; pre- and post-infusion biochemical determinations were not available for all patients. The limited sample size, with heterogeneity across clinical diagnoses and NER dosing regimens, led to an insufficient statistical power of subgroup analyses and prevented the generalization of our results. Despite these limitations, the overall safety profile of NER appears favorable.
Conclusion
Our data suggest that IV NER is a safe therapy in pediatric subjects with OI, CRMO, post-traumatic BMEs, and CRPS. APR represents the most frequent side effect of IV NER in children too, and may affect compliance; however, it is usually a mild and transient reaction. In all cases, NER-related complications emerged only after the first infusion, and pre-infusion cholecalciferol supplementation might prevent this frequent complication, while premedication with prednisone appeared not to be effective. The observations on strategies to prevent APR should be cautiously interpreted due to the limited statistical power in a small cohort and the potential influence of confounding factors. Studies on a larger cohort of pediatric patients are needed to explore and confirm these findings.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X261419235 – Supplemental material for Safety of intravenous neridronate in pediatric patients: insights from the monocentric retrospective NAPOLEON cohort
Supplemental material, sj-docx-1-tab-10.1177_1759720X261419235 for Safety of intravenous neridronate in pediatric patients: insights from the monocentric retrospective NAPOLEON cohort by Stefania Costi, Andrea Amati, Raffaele Di Taranto, Francesco Baldo, Chiara Crotti, Michela Ricci, Teresa Giani, Maurizio Virgilio Gattinara, Massimo Varenna, Roberto Caporali, Cecilia Beatrice Chighizola and Achille Marino in Therapeutic Advances in Musculoskeletal Disease
