Abstract
Background:
Up to one-third of people living with psoriasis develop psoriatic arthritis (PsA), and the majority have active psoriasis prior to the development of arthritis. Clinical risk factors, such as nail involvement, in conjunction with novel blood biomarkers, could improve PsA risk monitoring and early diagnosis.
Objectives:
The aim of the HIPPOCRATES Prospective Observational Study (HPOS—www.hpos.study) is to follow a cohort living with psoriasis and identify risk factors for the development of PsA.
Design:
HPOS is a patient-driven online prospective European observational cohort.
Methods:
Adult participants with psoriasis but with no prior diagnosis of PsA are eligible. Participants are invited to provide consent and join the study online. They complete a semi-structured questionnaire to collect data on demographics, psoriasis, comorbidities, risk factors for PsA, and the Psoriasis Epidemiology Screening Tool screening questionnaire. Follow-up is conducted through a questionnaire every 6 months. The primary outcome is the new onset of PsA confirmed by a diagnosis from their doctor. The study will also collect peripheral blood samples from a subset of participants for biomarker identification.
Ethics:
This study follows the principles of the Declaration of Helsinki. To date, ethical approval has been granted by independent ethical committees in 10 countries.
Discussion:
Studying a cohort of individuals with psoriasis will allow us to identify risk factors for arthritis development and to develop a risk calculator. This can support focused efforts on screening, patient education, and even studies looking to delay or prevent the onset of arthritis. This study, run via remote online data collection, provides an efficient way to recruit a large cohort (25,000) across multiple countries. However, challenges have had to be addressed with some key changes in study design, ethical review, and recruitment strategies required for each individual country.
Trial registration:
HPOS, Clinicaltrials.gov ID: NCT05858528, IRAS number 325080; https://clinicaltrials.gov/study/NCT05858528?locStr=United%20Kingdom&country=United%20Kingdom&cond=Psoriasis&term=HPOS&aggFilters=status%3Anot%20rec&rank=1.
Plain language summary
The HPOS Study, part of the HIPPOCRATES project, aims to find out what signs or factors can show which people with psoriasis might later develop Psoriatic Arthritis (PsA). PsA is a type of inflammatory arthritis that is related to the skin condition psoriasis. It occurs in about 1–2% of the general population but can develop in up to 30% of people who already have skin or nail psoriasis. Diagnosing PsA early can be difficult because symptoms can be vague or inconsistent, which means treatment often starts only after joint damage has already happened. By learning more about how psoriasis develops into PsA, researchers hope to find new ways to treat the disease earlier—or even prevent or delay it. The HPOS Study is an observational study that uses online questionnaires. Adults (aged 18 or older) who have psoriasis but not PsA can take part. Participants fill out a questionnaire every six months for three years. These questionnaires collect information about age, psoriasis details, lifestyle and health factors, early joint symptoms (using the PEST questionnaire), daily function, treatment satisfaction, disease impact, fatigue, and mental health. If early signs of PsA appear, participants are advised to contact a doctor for assessment. The study plans to recruit 25,000 people across 14 European countries (including the UK, Ireland, France, Germany, and others) and expects that around 675 participants will develop PsA each year. A smaller group of 3,000 participants will also provide a small finger-prick blood sample, which will help researchers look for blood markers that might predict PsA development. HPOS is the first large-scale European study to track how psoriasis progresses to PsA. The findings could lead to a “risk calculator” that helps doctors identify people at high risk of developing PsA earlier.
Introduction
Psoriatic arthritis (PsA) is a chronic immune-mediated inflammatory disease that affects peripheral joints, entheses, and axial sites and which typically develops alongside skin and/or nail psoriasis in up to 30% of cases. 1 It is estimated that 1%–2% of the general population have PsA, which represents 5–10 million people in the EU.2,3 It is increasingly recognized that PsA is associated with comorbidities, particularly those which promote the development of accelerated atherosclerosis and contribute to cardiovascular morbidity and mortality.4,5 While PsA can be classified using a mixture of clinical, laboratory, and radiographic features such as in ClASsification criteria for Psoriatic Arthritis (CASPAR), 6 evidence suggests that the disease is developing for several years before sufficient classifiable clinical features emerge. 7 The evolution from psoriasis to the point at which the patient meets CASPAR may occur in several stages. These stages include (1) patients with skin and/or nail psoriasis only but with risk factors for subsequent development of PsA; (2) an immune activation phase when there is evidence of cytokine (e.g., IL-23/IL-17 and/or TNF) over-production at a cellular or tissue level; (3) a stage where there is asymptomatic evidence of synovio-entheseal inflammation on imaging by MRI or ultrasound; (4) a “prodromal stage” where psoriasis patients may have musculoskeletal symptoms such as arthralgia and/or stiffness but without sufficient signs to make a diagnosis of PsA; and (5) PsA meeting CASPAR. 8
There is an important possibility that at some of these stages, the transition to PsA may be reversible. 9 At present, treatment is focused on those patients who receive a PsA diagnosis (stage 5 above), have ongoing inflammatory disease, and often already have evidence of radiographic damage. Future treatment intervention needs to focus on earlier stages of disease to possibly prevent the development of PsA and limit irreversible long-term outcomes. Improving our knowledge of the molecular basis of these early stages and the transitions between them will enable us to have a deeper understanding of the progression of psoriasis to PsA. Our underlying hypothesis is that a greater understanding of the early stages of transition from psoriasis to PsA will open the possibility of treatment strategies aiming to prevent PsA.
Therefore, the aim of the HIPPOCRATES Prospective Observational Study (HPOS), a flagship study conducted as part of the HIPPOCRATES consortium, is to identify demographic, clinical, and biological predictors for the development of PsA, thus facilitating the development of an individual risk model for individuals with psoriasis at future risk of developing PsA.
Objectives
Our primary objective is to identify demographic, clinical, and biological predictors for the development of PsA using data from all assessments prior to the development of PsA. We will also test the performance of the Psoriasis Epidemiology Screening Tool (PEST) 10 in identifying PsA.
Our secondary objectives will explore the impact of psoriasis and psoriasis treatments. Impact of psoriasis will be measured using patient-reported outcomes (PROs), including PsA impact of disease (PsAID), 11 Multi-Dimensional Health Assessment Questionnaire (MD-HAQ), 12 Patient Health Questionnaire (PHQ-9), 13 FACIT-fatigue, 14 and the itch numerical rating scale. 15 Impact of treatment burden will be measured using the treatment satisfaction questionnaire for medication (TSQM). 16
Study design
This study is a prospective observational cohort study using a web-based platform where participants will be enrolled and followed up on a 6 monthly basis (6 registrations/visits over a 3-year study period). Participation will involve the completion of web-based questionnaires and for 3000 participants who consent, the option of patient-centric sampling to collect a small sample of whole blood for biomarker discovery.
Participants will be recruited across Europe from the following countries: UK, Ireland, France, Germany, Belgium, Netherlands, Sweden, Portugal, Greece, Norway, Switzerland, Czech Republic, Poland, and Romania, including HIPPOCRATES consortium partners and additional interested dermatology/rheumatology teams. The University of Oxford is the sponsor for the study across all countries, but local regulations will be followed, and local ethical approval will be sought for each individual country. Within the Participant Information Sheet (PIS) and consent form (Supplemental Documents 1 and 2), we have asked for participants to give consent for the study team to keep their contact details to see if they would be interested in taking part in future studies. This provides flexibility for future substudies to be developed but these will not be implemented without further ethical review.
Selection of population
Adults with psoriasis but without a pre-existing diagnosis of PsA are eligible to join the study. Inclusion criteria include adult ⩾18 years, self-reported diagnosis of skin psoriasis (any form), and participant willingness and ability to give informed consent for participation in the study and complete data in one of the HPOS languages. Participants may not enter the study if they have a pre-existing diagnosis of PsA. Screening will be carried out via the website, and eligibility will be checked automatically during the online consent process. The website contains:
(a) The information about the study, including the study adverts, website registration instruction leaflet, the patient information sheet, consent forms, as well as more specific and general information about the research project using documents and videos.
(b) An e-mail address for potential participants to ask questions about the study (
(c) A secure registration form requiring first name, surname, and e-mail address to establish a website account.
Since participation in the study is fully online and there is no contact with a clinician or study personnel for evaluation, there can be a degree of uncertainty in terms of eligibility. If participants, who are not eligible due to a preceding PsA diagnosis joined the study, these individuals are notified, and their data will be removed from the study. Remote recruitment online supports rapid recruitment across multiple countries, and the recruitment of a large cohort, but does restrict participation to people with some degree of digital literacy.
Interventions, patient follow-up, visits, and trial procedures
There are no hospital or other healthcare visits required for HPOS. All data are self-reported by the participants and do not require travel to any study site. The primary outcome is a diagnosis of PsA, and this will initially be self-reported by participants. If participants report a diagnosis of PsA, they will be asked to confirm this diagnosis by uploading a clinic letter from their rheumatologist or primary care practitioner (Figure 1). Questionnaires, including screening for arthritis and assessment of the impact of psoriasis on each individual, are collected at baseline and at each follow-up assessment. Data will be requested from participants every 6 months for 3 years, and they will be prompted to upload their data by email. The reminder email includes a link to the study website so that the participant can easily complete the required questionnaires. If they do not respond, a total of three reminder emails will be sent.
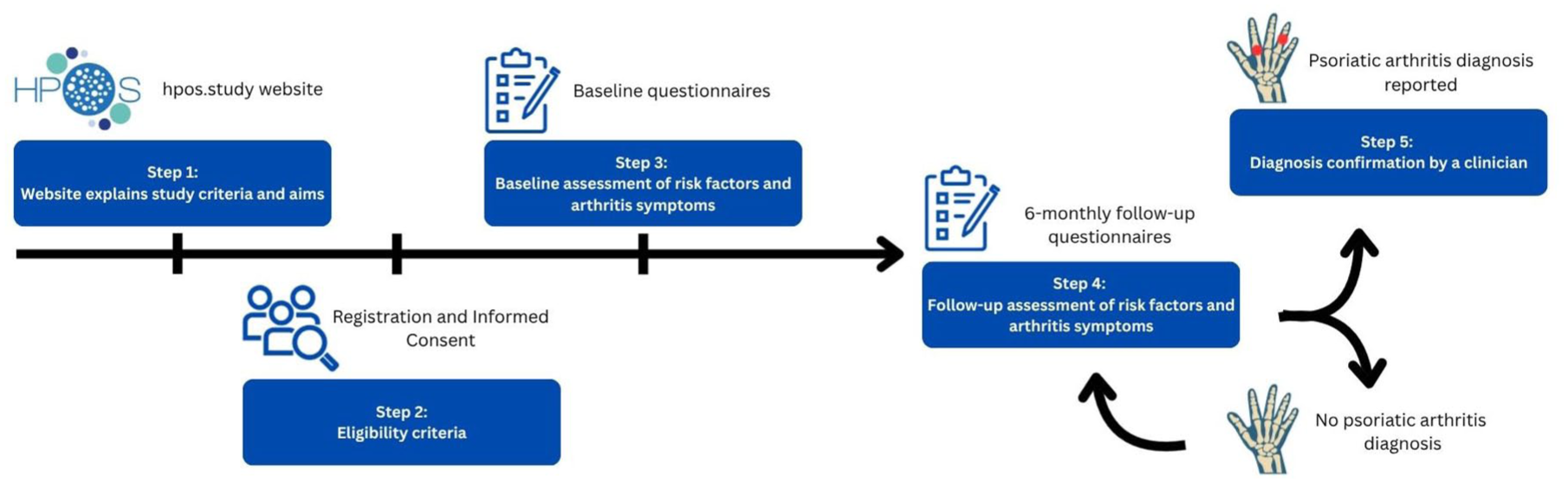
Flowchart of participation with eligibility and data checkpoints.
Data on demographics, psoriasis, co-morbidities, and PsA risk factors are collected. All participants then complete a screening questionnaire for PsA and a number of PROs assessing the impact of disease (Table 1).
Data collected in HPOS study.

HPOS, HIPPOCRATES Prospective Observational Study; MD-HAQ, multi-dimensional health assessment questionnaire; PEST, Psoriasis Epidemiology Screening Tool; PHQ-9, patient health questionnaire-9; PsA, psoriatic arthritis; PsAID, PsA impact of disease; TSQM, treatment satisfaction questionnaire for medication.
Patient research partner participation in study design
PROs were selected in co-design sessions with HPOS patient research partners (PRPs) and in additional focus groups with people with lived experience of psoriatic disease (patient representatives from the Psoriasis Association and the Psoriasis and Psoriatic Arthritis Alliance). The key PRO selected was to measure patient impact using the PsAID questionnaire. 11 This PRO was chosen as PsAID was co-designed with PRPs and reflects many different facets of the impact of PsA. If the PEST questionnaire is positive and a participant may have PsA, an additional questionnaire is used to measure functional impairment. After review of a selection of measures, PRPs chose the multi-dimensional health assessment questionnaire (MD-HAQ) as this includes more difficult tasks (e.g., walking 3 km, participate in recreational activities) and is shorter than many older alternatives. 17
While the focus of the study is on development of PsA, the majority of participants will not develop PsA. Discussions with PRPs and academic dermatologists in HIPPOCRATES suggested that cardiovascular risk, treatment satisfaction, and mental health impact would be key areas of interest amongst those participants with only skin disease. Therefore, it was decided that if the PEST questionnaire is negative and the participant have indicated that they are using some form of treatment for their psoriasis, they will be asked to complete the TSQM to assess the impact of their psoriasis treatment. 16
The HPOS PRPs were keen to collect additional information about mental health but concerned about overburdening participants. Thus, if patients score high on the PsAID questionnaire, they will be invited to complete some additional specific questionnaires, but these will be optional. If depression is scored ⩾5, the participant will be invited to complete the PHQ-9. 13 If fatigue is scored ⩾5, participant will be invited to complete the FACIT-fatigue questionnaire. 14
It was important for the PRPs that participants could see and interpret their own results. A dashboard (Figure 2) was designed so that after the participant has completed the questionnaire, the scores will be calculated and displayed. To assist participants with the interpretation of the scores, a brief explanation of the score and any necessary associated advice is provided.

The dashboards indicating the PEST Score and PsAID Score, and associated messages that are received with the scores. Green areas show a low risk and good outcomes, while orange to red areas show a high risk and suggest to take action by consulting with a health professional.
Alongside the data collected within the main study website, additional data may be collected via routinely collected datasets such as NHS and social care records with appropriate permissions. Consent for this is included in the consent form, where this is permitted according to individual country’s data protection rules.
Understanding who is likely to develop PsA is a priority for people living with psoriasis. This was reflected in the recent PsA James Lind Priority Setting Partnership where the question “Can tests be developed to predict whether a person has or will develop psoriatic arthritis?” was ranked third in the top 10 unmet needs. 18 PRPs living with psoriasis/PsA are part of the HIPPOCRATES work package steering committee overseeing the HPOS cohort and will help with dissemination of future results.
Sample size
Given the exploratory nature of the analysis, we aim to recruit 25,000 participants by year 3 and anticipate that 2.7% equivalent to 675 participants per annum will develop PsA. 19 Follow-up will ensure recruitment of a significant number of individuals who develop PsA during the study. Using risk stratification models developed from the other studies within the HIPPOCRATES consortium, participants were planned to be classified as high or low risk.
Blood sample collection
A subset of the HPOS population (n = 3000) were planned to be selected via an algorithm for remote finger-prick blood sampling. This algorithm was planned to be prepared via statistical modeling of routine data sources across Europe. Unfortunately, this separate HIPPOCRATES workshop did not develop an efficient, externally validated model. Therefore, a decision was made to use proven, published risk factors to identify those at high or low risk. 20 The participants who consented to take part in the blood sampling study were grouped into three risk groups: (i) a low risk group with no risk factors and a low PEST Score (<3), (ii) a high risk group with ⩾2 risk factors but no musculoskeletal symptoms (PEST score <3), and (iii) a high risk group with positive PEST Score (>3 points) and at least two other risk factors. The risk factors included were severe psoriasis, nail psoriasis, BMI >30 kg/m2, uveitis, or family history of PsA. The participants will be evenly selected across the European countries participating in HPOS.
The finger-prick blood samples will be collected with the Mitra® kit from Neoteryx by Trajan, which will be shipped to the selected participants. A logistics firm will be hired to coordinate the kit shipment across countries. Samples will be returned to the University College Dublin, Ireland, to coordinate long-term storage and for proteomic, metabolomic, lipidomic, and genomic biomarker analysis. The aim of this substudy is to identify blood biomarkers that are early indicators for PsA development. These biomarkers, in conjunction with the risk profile evaluation, could be used as a predictive or diagnostic tool.
Outcomes
The primary outcome is a new diagnosis of PsA. During the study, if a diagnosis of PsA is reported by a participant, participants will then be asked to upload a scan or photo of a hospital clinic letter confirming their diagnosis. As this is a pragmatic study, this will usually be a local rheumatologist, and so the expertise of the physician may vary.
Secondary outcome measures include PsAID, 11 MD-HAQ, 12 PHQ-9, 13 FACIT-fatigue, 14 itch numerical rating scale, 15 and the TSQM. 16 Data are collected directly via the HPOS website and held on secure servers at the University of Oxford.
Ethics
The HPOS trial is being conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. Informed consent was given in writing via an online questionnaire, which was translated into the participating country or region-specific languages (e.g., German, Italian, and French for Switzerland).
The consent questionnaire requested mandatory agreement: (i) that the information about the study was understood, (ii) the participation was voluntary and participation could be withdrawn at any point, (iii) that collected data will be looked at routinely by researchers from the University of Oxford and University College Dublin, as well as regulatory authorities, (iv) that data would be pseudo-anonymized, and these pseudo-anonymized data could be shared with industry partners to complete HPOS objectives. Additionally, optional consent was requested to: (i) participate in finger-prick blood sampling, (ii) agree to be contacted again for future studies, (iii) agree that genetic risk factors will be studied with the collected blood samples, (iv) agree that blood samples can be used in future studies that will have received ethical approval, (v) agree that HPOS will communicate with participants via email, letter, or telephone to send reminders for completing questionnaires, (vi) agree that HPOS will share study updates on the progress of the research (Supplemental Document 2).
Collection of personal data is minimized within the study, with identifiable data being held securely to maintain confidentiality before, during, and after the study. The study is sponsored by the University of Oxford in all countries, but ethical approval must be gained in each country. This has meant that the core protocol has had to be adapted on a country-specific basis to adjust for specific research rules and requirements.
In three planned countries (Italy, Spain, and Denmark), it was not possible for the study to gain ethical approval. Although the study is primarily observational, results of the screening questionnaires are fed back to participants with advice about seeing a rheumatologist if they have symptoms of PsA. Thus, the study is considered interventional by some ethics committees. In these countries, ethical committees (and in some cases, national law) required participants to be consented face-to-face with a member of the research team (e.g., in Italy and Spain). As recruitment is completed online, including online consent, this was not feasible, and the decision was taken not to open the study in these countries. In France, there are ongoing discussions to nest the study within an already ethically approved online platform: Community of Patients for Research (ComPaRe). 21
Due to a number of initially planned countries (Spain, Italy, Denmark) not participating in the HPOS recruitment, additional countries (Poland, Romania, Czech Republic) were invited to participate to reach the target of 25,000 participants across Europe. This also provides further diversity in European countries participating in the project.
Statistical analysis plan
As the diagnosis of PsA will be observed over time, time-to-event data will also be included in the primary outcome analysis to identify demographic, clinical, and biological predictors of the development of PsA. A Cox proportional hazards model will be employed to estimate hazard ratios for developing PsA, associated with various risk factors. This model will account for the time to diagnosis and appropriately handle censored data due to participants’ dropouts or due to study completion without a PsA diagnosis. In relation to the treatment of censored cases, all data in the HPOS study are right-censored, because individuals who were diagnosed with PsA before the study registration (i.e., left-censored) are removed from the study due to ineligibility. Censored data will be characterized into either independent, random, or informative censoring, and censoring time points will be documented thoroughly.
Potential factors to be investigated include:
Demographics—age, height, weight, ethnicity, skin tone, waist circumference, smoking, alcohol and postcode (to estimate social deprivation), family history of psoriasis, and PsA.
Diagnostic and descriptive data related to their psoriasis—duration, subtype, extent, severity (allowing calculation of self-assessed psoriasis area and severity index), nail involvement, current and previous treatment.
The PEST questionnaire, a validated screening questionnaire to identify any musculoskeletal symptoms and questions on back pain potentially indicating peripheral or axial PsA including a manikin to mark areas of musculoskeletal pain.
Additional risk factors for PsA—recent trauma/injury, recent infection.
Co-morbidities—uveitis, inflammatory bowel disease, depression, anxiety, metabolic syndrome, cardiovascular disease.
Patient impact—PsAID, MD-HAQ.
Additional optional questionnaires on patient impact.
Additional advanced modeling will be utilized to attempt to develop an individualized predictive model using data from this study and other studies in the HIPPOCRATES consortium. In general, machine learning models will be trained and tested on separate subsets of the data to avoid overfitting. Models will be subject to cross-validation workflows including comparison using datasets between different countries. The models could be validated using data from one country, and testing could be performed on data from other countries.
Missing data will be analyzed with multiple imputations via, for example, a random forest model. Data will be analyzed with and without imputations. We will optimize the models based on standard measures such as accuracy, precision, and recall. Variable selection will be based on each variable’s respective contribution to the classification performance as measured, for example, by Shapley values. Additionally, we aim to perform a time-to-event analysis. Where temporal data are available at sufficient resolution, the goal is to predict the time until a PsA diagnosis.
Monitoring
The study is managed by a trial management committee including the chief investigator (CI), co-investigators from University College Dublin (UCD), and study staff. As part of the HIPPOCRATES consortium, additional oversight and input are provided by the wider HIPPOCRATES team, including the HIPPOCRATES Executive Team.
The SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) guidelines 22 and the CASP (Critical Appraisal Skills Programme for Cohort Studies) checklist 23 were completed to follow appropriate reporting guidelines (Supplemental Documents 3 and 4).
Preparations and study launch
All materials were originally developed in English with PRP input, and this allowed review and input into a draft website. The PROs chosen were available in multiple languages in most cases, but some additional translation was required, particularly for Eastern European countries. All of the study documentation (patient information sheet, consent form) and website text also need translation for each country. This was initially done using DeepL software (DeepL, Cologne, Germany), then the translations were reviewed, checked, and corrected by native speakers in each country.
The HPOS Study is launched in each country after ethical approval has been obtained. To date, HPOS has been launched in the UK, Ireland, Greece, Portugal, Poland, Czech Republic, Romania, the Netherlands, and Norway (Table 2). Additional countries are currently awaiting ethical approval. The study recruitment is being coordinated in each country by local PIs who are consultant rheumatologists or dermatologists.
Ethics submission and proposed HPOS study launch times.
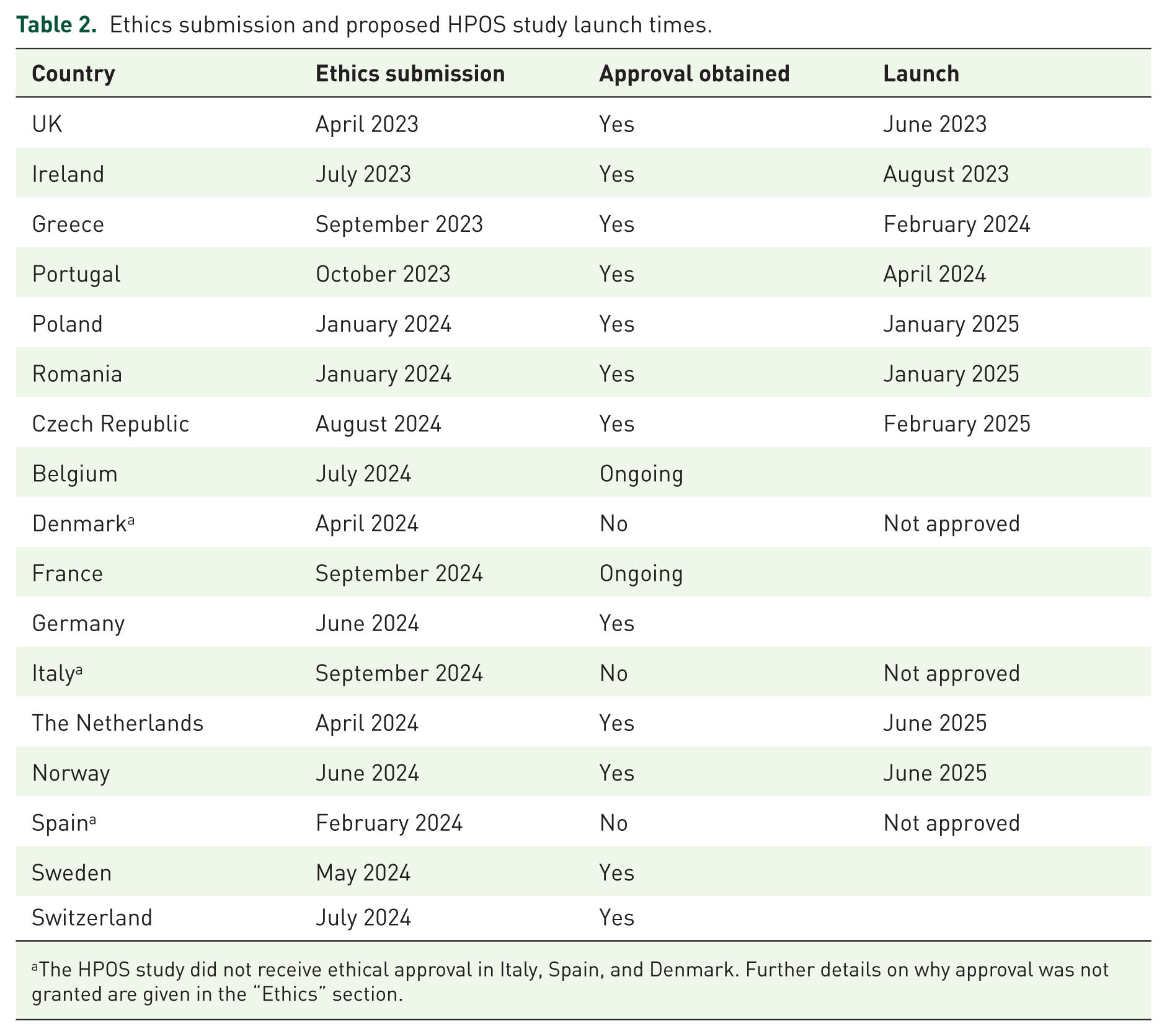
The HPOS study did not receive ethical approval in Italy, Spain, and Denmark. Further details on why approval was not granted are given in the “Ethics” section.
Recruitment strategies
Launch meeting
Prior to each country’s launch, a launch meeting is held with the HPOS study team, country PI and other investigators, patient organizations, and any additional member of the team. For example, in Ireland, this included key stakeholders including patients, University College Dublin communications staff, local pharmaceutical company representatives, dermatologists, rheumatologists, CEOs of both the Irish Skin Foundation and Arthritis Ireland, and the HPOS team. This helps to support a local strategy and allows the team to consider different key recruitment opportunities as outlined below.
Networking
Recruitment of 25,000 participants with psoriasis across Europe is no small endeavor. Therefore, it was clear from the outset that an extensive network with clinicians and patient organizations would be required to raise awareness about the HPOS Study in each participating country. The study teams are hence encouraged to contact clinical umbrella organizations to help connect with dermatologists (in private and public dermatology clinics and hospitals), and general practitioners (GPs) across the country. For example, in Ireland, the Irish Association of Dermatologists and the Irish College of General Practitioners were contacted to share information about the HPOS Study with all their members. Additionally, dermatologists from across the country were contacted with a letter and a pack of study flyers, to request help with recruitment of psoriasis patients directly in the dermatology clinics and hospitals. In the UK, in addition to dermatologists and GPs, other medical clinics such as dentists were contacted to display a study poster in their patient waiting area or on the clinic screens (Supplemental Figure 1).
Patient organizations
Another route to directly reach patients is via patient organizations. Connections were made with national (e.g., Psoriasis Association UK, and Irish Skin Foundation) and international patient organizations (e.g., The European Umbrella Organisation for Psoriasis Associations (EUROPSO), European Alliance of Associations for Rheumatology, and The International Federation of Psoriasis Associations (IFPA)). The national patient organizations support HPOS recruitment by sharing HPOS information in the form of email newsletters and study flyers with their subscribers, while the international patient organizations supported HPOS by providing contacts between the HPOS country teams and national patient organizations or individual PRPs. Additionally, both national and international patient organizations supported HPOS via sharing posts and news on social media.
In the modern era of social media, there are many psoriasis patients who are active on social media reporting about living with psoriasis, PsA, or other comorbidities. Connecting with social media influencers can provide an additional effective avenue for reaching out to many psoriasis patients at once.
Study flyers
Study flyers have been prepared by media designers from EURICE, a partner of the HIPPOCRATES consortium, and by university communications teams from the University of Oxford, to support the study launch. These flyers have been translated to all the required languages. Flyers have been distributed to clinics, hospitals, community centers, and churches. Digital versions of the flyers have been displayed on screens in clinics, hospitals, and on university campuses, or have been used for social media posts (Supplemental Figure 1).
Health care system
Some health care systems have registries of psoriasis patients who are interested in participating in medical research. This was utilized in the UK, with the NIHR Be Part of Research mailing list (https://bepartofresearch.nihr.ac.uk/).
Traditional media
As potential study participants are recruited and followed online, it is crucial that information is shared as widely as possible to support recruitment. A coordinated press launch has been developed with communications teams from universities (University of Oxford and University College Dublin), and in the case of Ireland with a Public Relations (PR) media consultant, to ensure maximum awareness of the study. The PR consultant in Ireland prepared newspaper article drafts and letters to the editor in preparation for a 6-week PR campaign. This traditional media campaign was launched on February 18, 2024 by contacting national and regional newspapers, radio stations, and medical journals, with the prepared articles. In total, 43 articles were published, and 21 radio interviews with national and regional media were held. Furthermore, a TV interview with a dermatologist and a psoriasis patient and influencer was arranged. In Ireland, this was by far the most effective method of driving recruitment, whereas in the UK, most patients were referred from GPs.
Social media
In conjunction with traditional media, HPOS was actively promoted on social media (Facebook, Instagram, LinkedIn, and X). The benefits of social media posting are that this is a free method to share information about the study to a wider community. Furthermore, the specific psoriasis awareness events on social media can help raise awareness about the study (such as Psoriasis Awareness Month in August, or World Psoriasis Day in October).
Nonetheless, without a crowd of followers, social media posts may show limited success. To overcome this issue, social media influencers and patient organizations were contacted to help raise awareness of the HPOS study. Major newspaper articles, radio interviews, and the TV interview were shared on social media platforms. Additionally, national and international patient organizations were linked with each social media post. With the help of influencers and patient organizations, a wider community was reached. Social media posts can also be boosted using paid advertising.
Monitoring recruitment success
As part of the HPOS registration questionnaire, participants are being asked to report where or how they learned about the study. This helped the study coordinators to evaluate the effectiveness of different recruitment approaches. Overall, the recruitment methods listed above either led to steady, slow rise in recruitment numbers, or to sudden rapid increases in recruited participants. Thus far, each country has implemented a different strategy for recruitment and has observed a country-specific impact on recruitment success (Table 3 and Figure 3).
Recruitment method reported by participants in individual country.

Snapshot of study recruitment from June 24, 2025. Radio includes radio shows and podcasts. Newspaper/articles include print and online newspapers, as well as articles in online journals. Medical visits include GP, Dermatologists, Rheumatologists (in private practice or hospitals), and in the UK specifically also any other medical office that displayed a poster (e.g., Dentists). Social media includes Facebook, Instagram, TikTok, Twitter/X, LinkedIn. Other/unknown includes blank answers and unknown answers, but also the “Be part of research” email list of psoriasis patients in the UK (n = 464), and the UCD University in Ireland (n = 33), assisting with advertising.
GP, general practitioners; UCD, University College Dublin.

Recruitment monitoring over time with key campaigns highlighted.
For example, recruitment through GPs and dermatologists or flyers in clinics was most successful in the UK, with more than 300 participants being recruited this way. The UK was also able to recruit psoriasis patients interested in participating in research via the NIHR emailing list, which led to more than 370 recruited participants.
The PR campaign in Ireland was conducted from February to late March 2024 and led to the recruitment of 1400 participants in this timespan. Unsurprisingly, this was the most successful recruitment approach thus far, however, a significant amount (~€15,000) of budget was required to hire the PR consultant and to run the PR campaign.
Social media posting was particularly successful in Portugal, with the lead PI having a large followership in addition to obtaining support from EUROPSO and the Portuguese Psoriasis patient organization, where more than 400 participants were thus far recruited through social media posting. In contrast, patient partners in Greece advised the team that advertising was not likely to encourage recruitment for Greece and that people would only be likely to take part if they were approached by their treating physician. Recruitment in Greece has therefore focused on psoriasis clinics.
Of course, advertising and recruiting budget for studies are rarely considered in grant proposals, and additional funding support from industry partners already involved in the HIPPOCRATES consortium was negotiated and generously forthcoming. The recruitment of study participants is never easy, and reaching sufficient study numbers in multiple countries in a timely manner can only be achieved with a supportive network and funds to cover printing and advertising costs.
Dissemination
As an ongoing cohort, data will be analyzed at key timepoints based on recruitment and follow-up. Results will be shared via conference presentations and publications supported by the HIPPOCRATES Communication and Dissemination work package. Working with HPOS and wider HIPPOCRATES patient partners, we will also disseminate results via patient organizations, patient conferences, and lay summaries. A newsletter is sent to HPOS participants twice a year to share key updates, including the initial sharing of results with all participants.
Discussion
HPOS is an ambitious study, designed to investigate the progression from psoriasis to PsA in a population of 25,000 individuals. Due to the large study population number, the study is conducted Europe-wide via online questionnaires. The 6-monthly frequency of follow-up questionnaires ensures the close monitoring of study participants from non-symptomatic to PsA development. The risk evaluation strategy employed in the HPOS Study enables early detection of PsA symptoms and alerts study participants to seek medical advice. This will lead to timely diagnosis and treatment of PsA. Previous studies that investigated PsA progression often recruited individuals with symptoms or imaging findings, or performed retrospective data analysis on electronic healthcare records rather than performing prospective targeted studies, and might therefore have been less suited for understanding the early warning signs of PsA development. Furthermore, the involvement of PRPs in the study planning ensured that the HPOS Study reflects the needs and interests of the patients.
Limitations
There are some limitations in the design and recruitment strategies of the HPOS study, which are addressed here. The HPOS study may suffer from selection bias due to its digital nature of recruitment and the completion of questionnaires via an online platform. The study team are aware that this might exclude individuals with limited digital skills, older patients, or possibly those who are less engaged online. However, due to the large sample size (25,000 participants), recruitment in multiple European countries, and limited funding, study recruitment in person was not possible. To address potential under-representation of specific population groups and ensure broad participation, we have been promoting the study through multiple well-established channels. HPOS was advertised through psoriasis organizations and diverse traditional media, including television, radio, newspapers, newsletters, hospitals, dermatology clinics, referrals from family and friends, and patient support groups, and through social media.
Given the remote nature of the study, and the participants’ interaction with their own regional medical professionals, there is no possibility of standardizing diagnosis within and across countries. Therefore, both psoriasis and PsA diagnosis are initially based on a participant’s self-report. The authors cannot guarantee that participants don’t misdiagnose eczema for psoriasis, however, the number of individuals who do so is expected to be very low. There are many initiatives from patient organizations globally which help distinguish between the skin conditions. Also, the HPOS questionnaire about psoriasis severity uses images that help the participant to evaluate the body location, the body surface area affected, the redness, scaliness, and thickness of the psoriasis plaques. For a diagnosis of PsA, the participants are requested to upload a confirmation document, such as a template letter for their next clinic visit, provided by the HPOS team, for their GP, or rheumatologist to sign. If the documentation is missing, it is being followed up, and once uploaded, the document is verified by the HPOS clinical team to ensure a diagnosis of PsA was confirmed by a clinician. Of course, there will be some variation in expertise of the GPs or rheumatologists making this diagnosis, nonetheless, a local rheumatology diagnosis is a reasonable gold standard. Furthermore, the data analysis will include a sensitivity analysis to compare patient-reported diagnosis with those diagnoses confirmed by medical professionals.
Finally, retention of study participants in an online study can be challenging, and volunteer numbers are expected to drop after the completion of the baseline questionnaires. The HPOS team attempts to engage participants who are already enrolled in the study but have not completed all questionnaires with up to three reminders per timepoint. In addition, study newsletters are being sent to participants to engage them and encourage participation.
Conclusion
The HPOS study, run via remote online data collection, provides an efficient way to recruit a large cohort (25,000) across multiple countries. However, with this novel, remote study, challenges have had to be addressed with some key changes in study design, ethical review, and recruitment strategies required for each individual country. Studying the HPOS cohort of individuals living with psoriasis will allow us to identify risk factors for the onset of arthritis, aiming to develop a risk calculator that could be applied to individual people with psoriasis. This will support focused efforts on screening, patient education, and even studies looking to delay or prevent the onset of arthritis.
Protocol amendments
The HPOS study currently uses V1.5 of the protocol. Significant changes prior to this include
V1.1—Inclusion of questions on key comorbidities to assess frailty.
V1.2—Addition of “Be Part in Research” for recruitment.
V1.3—AE reporting removed from protocol as not required, clarification on registration process.
V1.4—Addition of new recruitment posters/flyers and redesign of study landing site.
V1.5—Clarification of blood sampling process and statistical analysis plans.
Role of study sponsor and funder
The study is sponsored by the Research Governance, Ethics and Assurance Team, University of Oxford (
The study sponsor and the funder had no involvement in study design, data collection, management, analysis and interpretation of data, writing of the report, and the decision to submit the report for publication. However, the sponsor approved to conduct this study.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251412835 – Supplemental material for The protocol for a patient-driven online prospective European observational cohort aiming to determine risk factors for the development of psoriatic arthritis among people living with psoriasis: the HIPPOCRATES Prospective Observational Study
Supplemental material, sj-docx-1-tab-10.1177_1759720X251412835 for The protocol for a patient-driven online prospective European observational cohort aiming to determine risk factors for the development of psoriatic arthritis among people living with psoriasis: the HIPPOCRATES Prospective Observational Study by Teresa Grohmann, Timeo Mtenga, David Kerr, Joe Barrett, Nathanael Gray, Heidi Bertheussen, Juan D. Cañete, Kurt de Vlam, Maarten de Wit, Theodoros Dimitroulas, Sam Groothuizen, Glenn Haugeberg, Mari Hoff, Rudolf Horváth, Jana Hurnakova, Michaela Koehm, Przemyslaw Kotyla, Jean-Guillaume Letarouilly, Rik Lories, Mari Løset, Christine Möser, Burkhard Möller, Maria Mytilinaiou, Mikkel Østergaard, Ana Rodrigues, Caitriona Ryan, Carlo Selmi, Liv Eidsmo, Axel Svedbom, Tiago Torres, Florentin Ananu Vreju, Marleen G. H. van de Sande, Lars Werner, Stephen R. Pennington, Oliver FitzGerald and Laura C. Coates in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
We want to thank all our HPOS participants for taking part in the study. We want to thank the HIPPOCRATES consortium for the continued support throughout from planning to execution of the study. We also want to acknowledge the support of all the patient organizations who have supported the design and launch of the HPOS study including HIPPOCRATES patient partners who are not named authors, Psoriasis Association, Psoriasis and Psoriatic Arthritis Alliance, EuroPsO (Jan Koren, David Trigos, Russ Cowper), Dutch Psoriasis Patient Organization, SPAE (Association of Patients with Psoriasis and Atopic Dermatitis), the Irish Skin Foundation, and IFPA. We want to thank our EURICE partners, Nina Weiler and her team, for assisting with poster and website design. For continued support in promoting HPOS on social media, we want to thank Zoe Ryan (itchingtotellyou), Helen Hanrahan (theflakyfashionista), Ciara Kelly (ciarakellydoc), Laura Dowling (fabulouspharmacist), Christina Psomadakis (dr.somaskin), and Alan O’Neill. We also want to acknowledge the recruitment support and rollout of HPOS, in countries that were not initially part of HIPPOCRATES, within the iPROLEPSIS consortium.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
