Abstract
Background:
The neutrophil-to-lymphocyte ratio (NLR) is a potential surrogate marker for disease activity in many rheumatic diseases.
Objective:
To study the correlation between NLR and disease activity of systemic lupus erythematosus (SLE) using the SLE disease activity score (SLE-DAS).
Design:
Retrospective, cross-sectional.
Methods:
Consecutive adult patients who fulfilled the American College of Rheumatology (ACR) or Systemic Lupus International Collaboration Clinic (SLICC) criteria for SLE were recruited between March 2023 and February 2024. SLE activity was assessed by physicians’ global assessment (PGA), SLE-DAS, and SLE disease activity index-2000 (SLEDAI-2K). The calculated NLR was correlated with disease activity indices and serological parameters (anti-dsDNA, C3/4) by Spearman’s rank correlation. Patients were stratified into SLE-DAS remission, mild, and moderate/severe disease activity, and a comparison of the NLR was performed among these subgroups by one-way ANOVA.
Results:
A total of 420 SLE patients were studied (93.1% women, age 31.6 ± 13.1 years, SLE duration 15.7 ± 8.1 years). Moderate/severe, mild disease activity and SLE-DAS remission were present in 70 (16.7%), 65 (15.5%), and 285 (67.9%) patients, respectively. SLE-DAS correlated significantly with SLEDAI-2K (rho 0.90; p < 0.001) and PGA (rho 0.60; p < 0.001). The mean NLR of all patients was 3.54 ± 4.0 and NLR correlated significantly with SLE-DAS (rho 0.17; p < 0.001). The NLR was significantly higher in patients with active disease in the SLE-DAS renal domain than those in remission (5.17 ± 7.11 vs 3.22 ± 3.19; p = 0.03). The NLR in patients with moderate/severe SLE-DAS activity (5.25 ± 6.89) was significantly higher than those with mild activity (3.12 ± 2.26; p < 0.01) or remission (3.22 ± 3.19; p < 0.01). ROC analysis showed that an NLR cut-off of 3.11 showed a sensitivity of 55.6% and specificity of 68.7% in detecting moderate/severe SLE-DAS activity (area under the curve 0.67 (0.6–0.7); p < 0.001).
Conclusion:
The NLR is a convenient marker that correlates significantly with disease activity in SLE.
Introduction
Systemic lupus erythematosus (SLE) is a multisystem autoimmune disease that mainly affects women of childbearing age. 1 It is more prevalent in certain ethnic groups, such as Asians and African Americans. 2 Despite the improvement in survival of SLE in the past few decades, survival rates plateaued after the mid-1990s. 3 Mortality and organ damage of SLE are mainly due to uncontrolled disease activity and treatment-related toxicities. As a result, assessment of disease activity is important during routine clinical practice. Moreover, serial measurement of disease activity is also crucial for the evaluation of the efficacy of a novel therapeutic agent or strategy.
Conventional serological markers of SLE activity include the levels of anti-dsDNA antibodies and complement C3 and C4. Although there have been a myriad of studies on novel biomarkers of SLE, none have been thoroughly validated in longitudinal studies for clinical use in different ethnic groups. 4 Moreover, these biomarkers are costly and may not be readily available in many localities. In recent years, the neutrophil-to-lymphocyte ratio (NLR) has been explored as a marker of disease activity or prognosis for many medical illnesses, including SLE, adult-onset Still’s disease, rheumatoid arthritis, cardiovascular disease, metabolic syndrome, sepsis, and traumatic brain injury.5 –13 Although NLR is not a disease-specific marker, it has great potential to be used as a surrogate for disease activity in autoimmune diseases such as SLE.
SLE-DAS is a new disease activity score that shows better performance than the SLEDAI-2K in discriminating improvement or worsening disease activity defined by a change of PGA by 0.3 points or more. 14 SLE-DAS also involves scoring some variables, such as proteinuria, thrombocytopenia, and leukopenia, as continuous instead of dichotomous variables to reflect the severity of these manifestations. Moreover, it also includes activity assessment in the cardiopulmonary and gastrointestinal systems. 14 SLE-DAS has been validated in SLE patients of different ethnicities.14 –17
A number of studies have shown a positive correlation between NLR and disease activity in SLE.18 –22 However, there have not been any data regarding the relationship between NLR with disease activity in our local Chinese patients with SLE as assessed by the SLE-DAS. This prompts the current study, which aims to investigate the role of the NLR as a surrogate marker for disease activity in SLE. We hypothesize that there is a positive correlation between NLR and SLE-DAS.
Patients and methods
Study population
This is a cross-sectional study on the relationship between NLR and disease activity of SLE. We recruited consecutive SLE patients from our rheumatology outpatient clinics and hospital admissions between March 2023 and February 2024. Clinical data were retrieved by review of the electronic medical records from the hospital system. These included demographic information, disease manifestations, lymphocyte count, neutrophil count, urine total protein to creatinine ratio (uP/Cr), renal biopsy (if any), use of immunosuppressive drugs, and glucocorticoid dosages. Disease activity was assessed by the SLE disease activity index (SLEDAI)-2K, 23 SLE-DAS, 14 and physicians’ global assessment (PGA) 24 by the attending rheumatologists. The peripheral blood NLR (within 14 days prior to clinical visits) was calculated and correlated with the disease activity indices by statistical analyses.
The current study was approved by the Central Institutional Review Board of the Hospital Authority, Hong Kong (CIRB-2024-256-4). Patients were de-identified during data analyses.
Inclusion and exclusion criteria
The inclusion criteria of this study were: (1) adult patients ⩾18 years old; (2) patients fulfilling the 2012 Systemic Lupus International Collaboration Clinic (SLICC) 25 or the 1997 American College of Rheumatology (ACR) classification criteria 26 for SLE. Exclusion criteria were: (1) active infection requiring antibiotic use or hospitalization within 30 days of recruitment; (2) active malignancy receiving therapies; (3) hematological diseases affecting the neutrophil or lymphocyte counts, or those who had a history of splenectomy; (4) liver cirrhosis; (5) patients who were hospitalized within 30 days of clinic visits due to acute coronary syndrome or stroke; and (6) patients who were treated with high-dose prednisolone >40 mg/day or intravenous cyclophosphamide within 30 days of recruitment.
Disease activity assessment
Patients recruited were assessed for disease activity using the SLE-DAS, SLEDAI-2K, and PGA. SLE-DAS is an index that consists of 17 items, including four continuous variables (arthritis, proteinuria, thrombocytopenia, and leukopenia) and 13 dichotomous variables that enable the calculation of a composite score from an online formula. 14 Index-based remission, mild activity, and moderate/severe activity were defined as a score of ⩽2.08, >2.08 to 7.64, and >7.64, respectively.
SLEDAI-2K is an updated version of SLEDAI, a global disease activity index that scores clinical features of nine organ systems on a binary scale. 23 PGA is a simple visual analog scale (0–3) that captures the overall disease activity according to the clinical judgment of attending physicians. 24 It does not describe individual SLE manifestations or discriminate activity between organ systems.
Statistical analysis
Values of this study were expressed as mean ± standard deviation (SD) unless otherwise specified. The relationship between NLR and disease activity (SLE-DAS, PGA, SLEDAI, C3/4, dsDNA titer) was studied by Spearman’s rank correlation test. Comparison of continuous variables among three groups was performed by one-way ANOVA, and post hoc comparison of the groups was performed by Tukey’s test for unequal samples.
The sensitivity and specificity of NLR cut-offs to predict moderate/severe disease activity (as defined by SLE-DAS) were studied by receiver operating characteristic (ROC) analysis. The area under the curve (AUC) and the 95% confidence interval (CI) were calculated for each model. A p value of <0.05, two-tailed, was considered statistically significant. All statistical analyses were performed by the SPSS program (IBM; version 29.0).
Results
Study population
A total of 517 SLE patients were identified, with 97 subjects excluded for the following reasons: hematological disease affecting cell counts (n = 72), recent infection (n = 4), splenectomy (n = 12), liver cirrhosis (n = 2), active malignancy (n = 1), cyclophosphamide use in the preceding month (n = 5), and prednisolone 40 mg daily or equivalent use in the preceding month (n = 1). Analyses were performed on the remaining 420 patients. All were ethnic Chinese. There were 391 (93.1%) women, and the mean age was 31.6 ± 13.1 years. The mean SLE duration was 15.7 ± 8.1 years.
Table 1 shows the cumulative clinical manifestations of the 420 SLE patients studied. The most common manifestations were arthritis (65%), renal disease (57.1%), and facial rash (48.3%). Positive anti-dsDNA was present in 73.1% of patients, and the most common immunosuppressive medications ever received by patients were prednisolone (84.8%), hydroxychloroquine (75.7%), and azathioprine (50.5%).
Cumulative manifestations of SLE in all the patients studied (N = 420).

aCL, anticardiolipin IgG (moderate/high titer); SLE, systemic lupus erythematosus.
The mean SLE-DAS score of all the patients studied was 3.41 ± 4.19. SLE-DAS remission, mild and moderate/severe activity was present in 285 (67.9%), 65 (15.5%), and 70 (16.7%) patients, respectively. The mean SLEDAI-2K score was 2.95 ± 2.76, and the mean PGA was 0.52 ± 0.64. A total of 294 (69.8%) patients had a PGA < 0.5; 60 (14.2%) patients had a PGA of 0.5 to 1.0; 54 (12.8%) patients had a PGA of >1.0 to 2.0; and 12 (2.8%) patients had a PGA score of >2.0 to 3.0.
The mean daily prednisolone dose was 3.0 ± 4.7mg (IQR 0.0–5.0) for all 420 SLE patients studied. The number of patients using prednisolone, mycophenolate mofetil, azathioprine, calcineurin inhibitors, and hydroxychloroquine in those 135 patients with active SLE was 90 (66.2%), 73 (53.7%), 16 (11.8%), 23 (16.9%), and 91 (66.9%), respectively. The mean daily prednisolone dose used in these patients was 5.0 ± 7.0 mg (IQR 0.0–5.0) before treatment augmentation.
Correlation between NLR and disease activity
The mean NLR of all the 420 patients studied was 3.54 ± 4.0 (IQR 1.72–4.00). NLR correlated positively and significantly with SLEDAI-2K (rho 0.14, p < 0.05) and SLE-DAS (rho 0.17, p < 0.01). Figure 1 shows a scatter plot of SLE-DAS with NLR for the patients studied. However, NLR did not correlate significantly with anti-dsDNA titer, C3/4 level, or the histological class of LN (data not shown). SLE-DAS correlated significantly with SLEDAI-2K (rho 0.90; p < 0.001) and PGA (rho 0.60; p < 0.001).
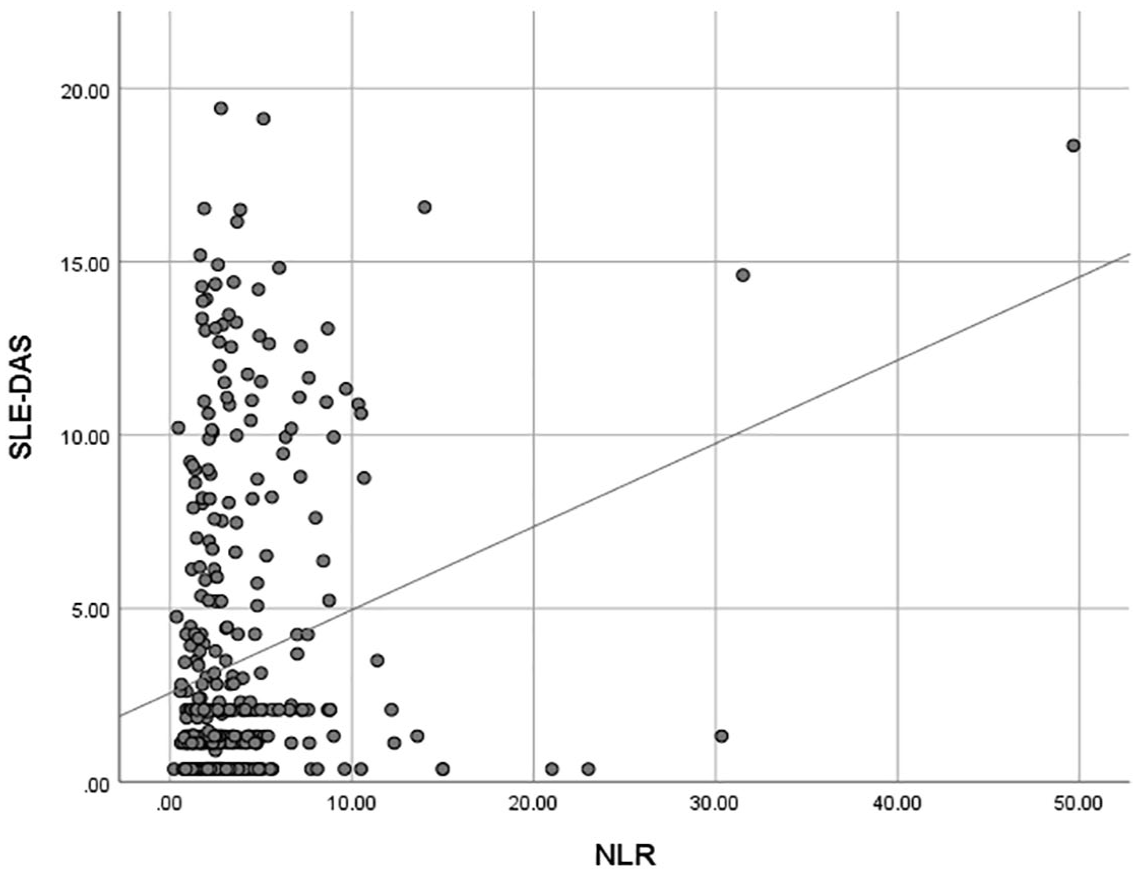
Scatter plot diagram of NLR versus SLE-DAS.
A significantly higher NLR was observed in SLE patients with moderate/severe activity than in mild activity (5.25 ± 6.89 vs 3.12 ± 2.26; p < 0.01) or remission (5.25 ± 6.89 vs 3.22 ± 3.19; p < 0.01). However, the NLR in patients with SLE-DAS mild activity was not significantly different from those in remission (3.12 ± 2.26 vs 3.22 ± 3.19, p = 0.72). Figure 2 shows a box plot of NLR in different SLE-DAS activity status.

Box plot showing NLR in different SLE-DAS activity categories.
There were 135 clinically active SLE patients who scored in the SLE-DAS domains. The major active manifestations were renal disease (n = 63), mucocutaneous disease (n = 42), arthritis (n = 7), and thrombocytopenia (n = 7; Table 2). Among patients with active lupus nephritis, 46 had renal biopsy performed: class I LN (n = 1), class II LN (n = 5), class III ± V LN (n = 19), class IV ± V LN (n = 15), and pure class V LN (n = 6). Table 2 also shows the mean NLR of the individual SLE-DAS domains in the 135 clinically active SLE in comparison with those with SLE-DAS remission (mean NLR 3.22 ± 3.19). Only active renal disease showed a statistically significant higher NLR compared to SLE-DAS remission (5.17 ± 7.12 vs 3.22 ± 3.19, p = 0.03), while the other manifestations such as mucocutaneous (4.74 ± 7.50, p = 0.20) and thrombocytopenia (8.50 ± 11.0, p = 0.25) only showed a numerically higher NLR than SLE-DAS remission.
NLR in patients with active SLE scored in different SLE-DAS domains versus SLE-DAS index-based remission.

Comparison of NLR between active manifestations with SLE-DAS remission.
Mucocutaneous manifestation was defined as lupus rash, alopecia, or oral ulceration, scored in the SLE-DAS.
N/A, not available; NLR, neutrophil-to-lymphocyte ratio; SD, standard deviation; SLE, systemic lupus erythematosus; SLE-DAS, systemic lupus erythematosus disease activity score.
A ROC analysis on the optimal cut-off of NLR in detecting moderate/severe SLE-DAS activity was performed (Figure 3). An NLR cut-off of 3.1 showed a sensitivity of 55.6% and specificity of 68.7% in the prediction of moderate/severe SLE-DAS (AUC 0.66 (0.56–0.73); p < 0.001). No optimal cut-off of NLR could be found to predict mild SLE-DAS activity (data not shown).
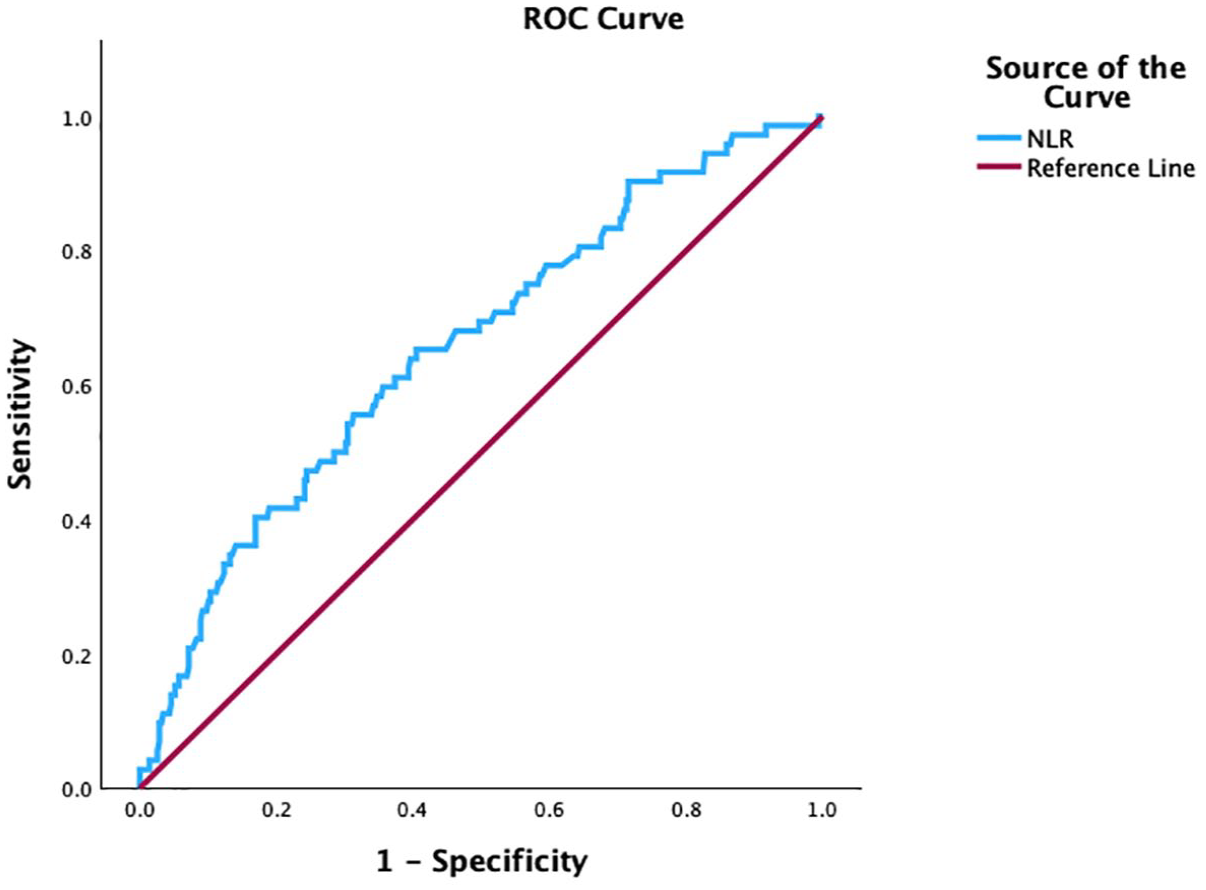
ROC curve analysis of NLR to moderate to severe SLE disease activity (as defined as SLE-DAS ⩾7.64). AUC = 0.66, p ⩽ 0.001, 95% CI = 0.59–0.73. An NLR value of 3.11 shows a sensitivity of 55.6% and a specificity of 68.7% in detecting moderate to severe SLE disease.
Discussion
In this cross-sectional study, we investigated the relationship between NLR and disease activity in our local Chinese SLE patients. We demonstrated that NLR correlated with SLE disease activity as assessed by both the new SLE activity index, SLE-DAS, and the conventional SLEDAI-2K. The NLR was significantly higher in patients with active renal disease or moderate to severe SLE-DAS disease activity compared to those with SLE-DAS remission, respectively.
Lymphopenia is a characteristic hematological feature of SLE, with a reported prevalence of 15%–82%. 27 Lymphocyte count in SLE correlates inversely with disease activity, but the exact mechanisms remain uncertain.28,29 Lymphopenia in SLE has been associated with the presence of anti-lymphocyte antibodies 30 and increased lymphocyte apoptosis.31 –33 Different classification systems of SLE have included lymphopenia as a criterion. In the 1997 ACR criteria, leukopenia (white cell count <4.0 × 109/L) or lymphopenia (lymphocyte count <1.5 × 109/L) for at least two occasions is one criterion under hematological disorder. 26 In the 2012 SLICC criteria, leukopenia of <4.0 × 109/L or lymphopenia <1.0 × 109/L also counts as an immunological criterion. 25 However, in the more recent 2019 European Alliance of Associations for Rheumatology (EULAR)/ACR classification criteria for SLE, a total white cell count of <4.0 × 109/L instead of lymphopenia is selected as a criterion. 34 Although lymphopenia reflects SLE activity, it is not incorporated into the conventional disease activity indices such as the SLE disease activity index (SLEDAI)-2K in which the total leukocyte count of <3.0 × 109/L is regarded as a parameter instead. 23 The newer disease activity index, SLE-DAS, 14 also adopts the total white cell count instead of the lymphocyte count as a marker for activity, although it is computed as a continuous variable in the formula, which may contribute to the improved sensitivity to change in this scale.
On the other hand, neutrophils also play an important role in the pathogenesis of SLE. Neutrophils are responsible for defense against pathogens by a number of mechanisms that include phagocytosis, degranulation, generation of reactive oxygen species (ROS), production of pro-inflammatory cytokines, and the formation of neutrophil extracellular traps (NETs). 35 A subset of pro-inflammatory neutrophils, known as low-density granulocytes (LDGs), is increased in patients with SLE and correlated with disease activity. 36 LDGs in SLE patients have increased capacity for NETosis in the absence of added stimulation, phagocytosis, mitochondrial ROS production, and production of cytokines and have a prominent type I interferon signature. 37 The increased NETosis in SLE patients promotes in vitro activation of plasmacytoid dendritic cells (pDCs) and the production of type I interferons.37 –39 Therefore, the neutrophil count may indirectly reflect the degree of inflammation in SLE.
Previous studies of NLR in SLE patients with different ethnic groups showed a significant positive relationship between disease activity score and NLR values.18 –22 This is consistent with our findings that the NLR of our patients correlated with both the SLEDAI-2K and SLE-DAS. However, previous works have exclusively adopted the global index SLEDAI for disease activity assessment. Our study is unique in that we have adopted the SLE-DAS, a new disease activity index scale that uses the total white cell count as a continuous variable in the formula. Moreover, the SLE-DAS categorizes disease activity into remission, mild, and moderate/severe subgroups, and we were able to demonstrate a higher NLR in the moderate/severe group as compared to those with mild activity or in remission. In our study, two-thirds of patients in the moderate/severe SLE-DAS activity group had renal disease, while active mucocutaneous manifestations and leukopenia accounted for 80% of those with SLE-DAS mild activity scores. NLR did not differ significantly between patients with mild disease and remission. While this could possibly be related to the relatively small number of patients with mild SLE activity, our results illustrated that the NLR could be a better surrogate marker for renal activity of SLE.
The best cut-off value of NLR in determining the level of disease activity in SLE has not been established in the literature. In the study by Wu et al., 19 the best NLR cut-off value for predicting severe SLE disease activity (SLEDAI > 9) was 2.26 (sensitivity 75%; specificity 50%). Similarly, a cut-off NLR value of 2.2 was reported to best correlate with active SLE (SLEDAI > 4) in Soliman’s study. 18 In our analysis, a cut-off of NLR or 3.1 best predicted moderate/severe SLE activity by the SLE-DAS. Further studies using the same definitions of the level of SLE disease activity are needed to validate an optimal NLR cut-off for clinical applications.
In the current study, we demonstrated that the NLR was significantly higher in patients with SLE-DAS moderate/severe activity than index-based remission status. Similarly, the NLR was significantly higher in patients with clinically active renal than remitted SLE. These observations were driven by the high proportion of patients in the SLE-DAS moderate/severe activity group having renal activity. Our findings corroborate those reported by Soliman et al. 18 and Han et al., 21 who also showed that the NLR was significantly higher in patients with active LN compared to those without. Previous studies have reported a positive relationship between NLR and histological features of LN. In the study by Han et al., 21 higher NLR was observed in those with higher activity score and active cellular crescents, whereas NLR was associated with a positive anti-dsDNA and histologic class IV or V disease in the study by Soliman et al. 18 A higher NLR has also been associated with heavier proteinuria, lower eGFR and a worse renal prognosis in patients with LN in terms of progression to end-stage kidney disease or death. 20 However, we were unable to demonstrate in our study that the NLR values correlated with the histologic classes of LN or serological activity of SLE. While we correlated the NLR with the severity of disease activity by SLE-DAS, our study was not originally designed to look at the relationship between NLR and histological activity or chronicity of LN.
There are several limitations of our study. First, most patients were recruited from the outpatient clinics. The proportion of patients with more severe SLE activity was relatively small. The large proportion of patients with lower SLE-DAS scores may limit the power of the ROC analysis to calculate the best NLR cut-off for predicting mild disease activity. Second, although we excluded patients with recent cyclophosphamide use, the use of other immunosuppressive drugs such as prednisolone, azathioprine, and mycophenolate mofetil might have an effect on the NLR. Third, as the study was cross-sectional, we did not have longitudinal data regarding the correlation between the change in NLR and disease activity over time, as well as the effects of NLR on damage accrual and other outcomes such as cerebrovascular, cardiovascular disease, and infective complications. Finally, although the correlation between NLR and SLE-DAS was highly significant statistically, the correlation coefficient was low. A similar magnitude of the correlation coefficients was also reported in several previous studies using SLEDAI for assessing SLE activity.19,21,22 This indicates a weak correlation between the two parameters, and multiple other factors may be associated with clinical disease activity.
Conclusion
In this study, we demonstrate that the NLR is a potential marker for SLE disease activity, particularly renal activity, in our local Chinese patients. It is a convenient and inexpensive marker for SLE disease activity that has pathogenetic significance. Thus, it may have the potential to stratify patients into different levels of disease activity to guide personalized therapies. However, the relationship between NLR and SLE-DAS should be interpreted with caution because of the weak correlation shown. Similar to other conventional SLE markers such as anti-dsDNA and complements, NLR has to be considered together with clinical settings to make an accurate judgment of disease activity. Further validation in longitudinal cohorts regarding the relationship between NLR and specific SLE organ manifestations, in particular lupus nephritis, and accrual of organ damage and other complications, is needed to clarify the role of NLR in the routine monitoring of patients with SLE.
Supplemental Material
sj-doc-1-tab-10.1177_1759720X251347272 – Supplemental material for The neutrophil-to-lymphocyte ratio as a surrogate marker for disease activity in systemic lupus erythematosus: a correlation study using the SLE-DAS
Supplemental material, sj-doc-1-tab-10.1177_1759720X251347272 for The neutrophil-to-lymphocyte ratio as a surrogate marker for disease activity in systemic lupus erythematosus: a correlation study using the SLE-DAS by Mandy Ming Yan Wong, Fung Lam and Chi Chiu Mok in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
