Abstract
Background:
Ankylosing spondylitis (AS) is a progressive chronic disease that primarily affects the axial skeleton, leading to significant musculoskeletal impairment. This condition may extend its inflammatory impact to peripheral muscles, potentially resulting in muscle wasting.
Objectives:
The study aimed to investigate the prevalence of sarcopenia among AS patients, to determine the specific cutoff values for muscle status, and to identify risk factors that may impact muscle status in AS patients.
Design:
A cross-sectional study was conducted involving consecutive AS patients attending the Department of Rheumatology and Immunology at the First Medical Center of the Chinese PLA General Hospital. Clinical characteristics and body composition were evaluated.
Methods:
Based on the Asian Working Group for Sarcopenia criteria, the prevalence of sarcopenia among AS patients were calculated. Additionally, specific cutoff values for muscle mass and muscle strength in AS patients were analyzed. Univariable and multivariable logistic regression analyses were employed to scrutinize risk factors impacting muscle status in AS patients, including appendicular skeletal muscle mass index (ASMI) and grip strength.
Results:
Measurements of body composition, including ASM at 22.79 kg and ASMI at 7.69 kg/m², were significantly lower compared to the healthy control group (p < 0.05). The prevalence of sarcopenia, as identified by the sex-specific fifth percentile, is 8.66% in AS patients. In our study, the ASMI cutoffs were set at 7.48 kg/m² for males and 5.63 kg/m² for females. Logistic regression analysis revealed that AS patients who had a longer disease duration, lower level of education, elevated inflammatory markers, and a higher Bath Ankylosing Spondylitis Metrology Index (BASMI) were more likely to experience a decrease in ASMI. Regarding the muscle strength, lower education level and higher Bath Ankylosing Spondylitis Functional Index were correlated with a reduced grip strength in individuals with AS.
Conclusion:
Our study indicates that AS patients may have a diminished muscle status. Although the incidence of sarcopenia among AS patients is relatively low, a notable subset does suffer from muscle atrophy. Variables such as disease duration, education level, inflammatory markers, and BASMI are likely to influence muscle status in AS patients, warranting consideration in clinical management and intervention strategies.
Introduction
Sarcopenia is a systemic disease that leads to muscle atrophy and a degradation of physical capabilities, heightening the risk of falls, fractures, physical disability, and a reduction in life expectancy. 1 Although primary sarcopenia is a natural accompaniment to the aging process, it may also arise as a secondary affliction within the spectrum of chronic inflammatory diseases. These conditions are characterized by their impact on the compromised functionality of skeletal muscles, affecting their contractile abilities, metabolic efficiency, and hormonal equilibrium.2,3
Ankylosing spondylitis (AS) is a chronic inflammatory condition that predominantly impacts the axial skeleton, including the spine and sacroiliac joints, potentially culminating in diminished mobility and disability. 4 The disease’s persistent inflammation is driven by immune cell dysregulation and the production of inflammatory cytokines such as C-reactive protein, tumor necrosis factor (TNF)-α, and interleukin-6. 5 Evidence suggests that these cytokines may contribute to muscle wasting. With the progression of AS, patients often experience significant changes in their body composition, including fluctuations in muscle and fat mass, as well as alterations in body mass index (BMI). The pain associated with AS can limit physical activity, leading to the compounding difficulties of muscle wasting and fat gain. These physiological changes can substantially influence the efficacy of treatments and the disease’s progression, deeply affecting the patients’ overall quality of life. 6
The clinical implications of body composition changes in AS are noteworthy, yet the existing evidence within this patient group is scarce and fails to uniformly demonstrate a reduction in muscle mass.7,8 Such discordant results could stem from differences in the populations studied and the analytical techniques for body composition assessment. Therefore, a comprehensive nutritional assessment of AS patients is imperative for gaining a deeper understanding of the nuances of sarcopenia and its impact on the disease course. The study aimed to ascertain the prevalence of sarcopenia in the AS patients, to establish specific thresholds for muscle mass, and to uncover potential risk factors that could affect muscle health in individuals with AS. By addressing these objectives, the study seeks to enhance the clinical management of AS by providing targeted insights into the muscular aspects.
Methods
Study population
This study collected data from patients with AS at the Department of Rheumatology and Immunology of Chinese PLA General Hospital. 9 These patients were recruited between October 2023 and June 2024, strictly adhering to predefined inclusion criteria.
Inclusion criteria
(i) Patients who met the revised 1984 New York criteria. 10
(ii) Patients aged 18 years or older.
(iii) Patients capable of undergoing assessments for body composition, grip strength, and gait speed.
(iv) Patients with complete clinical records and who had completed the pertinent questionnaires.
Exclusion criteria
(i) Patients with concurrent rheumatic diseases such as rheumatoid arthritis (RA), osteoarthritis, etc.
(ii) Patients with chronic debilitating diseases such as nephrotic syndrome, inflammatory bowel disease, etc.
(iii) Patients with concomitant severe health conditions such as malignancies, severe congestive heart failure, etc.
(iv) Patients with implanted cardiac pacemakers, internal metallic or electronic devices, or those who have undergone spinal surgery.
(v) Patients unable to walk or stand independently and cooperate with the assessments.
(vi) Other medical or personal conditions deemed incompatible with study requirements.
Additionally, a control group comprising 127 healthy subjects, matched for age and sex, was assembled from the Department of Nutrition at the First Medical Center of the Chinese PLA General Hospital. These individuals were selected in accordance with the established exclusion criteria, ensuring a robust comparison group for the study. During our sample size calculation, anticipating a 34.3% 7 prevalence, we determined that approximately 243 participants would be required. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 11
Measurements of body composition
Human body composition analysis was carried out utilizing the H-Key 350 body composition analyzer, employing a multifrequency bioelectrical impedance analysis (BIA) method. To prepare for the assessment, participants were instructed to remove all metallic accessories, stand barefoot on the foot electrodes, and hold the hand electrodes with their arms hanging naturally and away from their body, thereby initiating the measurement process. The brief yet comprehensive test, lasting approximately 30 s, facilitated the simultaneous acquisition of various body composition metrics. In the context of this study, key parameters such as appendicular skeletal muscle mass (ASM) and trunk skeletal muscle mass (TRSM) were examined. To normalize these metrics and account for the influence of body size, height measurements were integrated. The formula of appendicular skeletal muscle mass index (ASMI) is as follows: ASMI = ASM/(Height in meters). 2 Likewise, the formula of trunk skeletal muscle mass index (TRSMI) follows a comparable approach.
Measurements of grip strength and gait speed
Grip strength was ascertained using the EH101 electronic hand dynamometer. For the procedure, patients were asked to stand upright with feet shoulder-width apart, grasp the dynamometer firmly with their dominant hand, and apply force gradually until a stable reading appeared on the dynamometer’s screen. After a 30-s respite, this process was repeated a total of three times, and the highest value obtained was taken for further analysis to ensure accuracy. Gait speed was assessed through a 6-m walk test, a standard measure of physical function. A straight, obstacle-free, and quiet path section was selected, with the 6-m distance clearly marked at the starting and ending points. Participants were instructed to walk the marked distance at their quickest safe pace, maintaining a consistent speed without acceleration or deceleration. The test was conducted twice, and the average of the two speeds was calculated for record-keeping purposes. It was imperative that if participants exhibited any adverse symptoms, such as pallor, chest tightness, or excessive sweating during the test, the procedure would be halted immediately, and appropriate medical intervention would be initiated without delay. This precaution ensured the safety and well-being of all individuals involved in the study.
Diagnosis of sarcopenia
Sarcopenia was diagnosed in accordance with the Asian Working Group for Sarcopenia (AWGS) criteria, 12 defining low muscle mass, low grip strength, and low gait speed by referencing the sex-specific lowest quintile as a benchmark. Our analysis identified four groups of individuals based on these three variables and calculated the prevalence for each: sarcopenia, which includes reduced muscle mass coupled with decreased muscle strength and/or physical function; presarcopenia, marked solely by reduced muscle mass; possible sarcopenia, indicated by a decline in muscle strength and/or physical function; and severe sarcopenia, where all three—reduced muscle mass, strength, and physical function—are concurrently affected.
Other covariates
Demographic information collected in this study included sex (male/female), age (years), disease duration (years), education level (below college/college or above), and smoking (yes/no). Additionally, the presence of human leukocyte antigen (HLA)-B27 status (positive/negative) was noted, as was the inflammatory marker status (normal/elevated), with elevated erythrocyte sedimentation rate (>20 mm/h) or C-reactive protein (>5 mg/L) used as indicators of inflammation. The usage of TNF-α inhibitors was also recorded (used/unused). Given the widespread and low-dose use of nonsteroidal anti-inflammatory drugs, along with a stable dosage regimen sustained for a minimum of 4 weeks, these medications are regarded as a consistent background factor in our study. The clinical indicators assembled in this study included the Bath Ankylosing Spondylitis Metrology Index (BASMI), the Bath Ankylosing Spondylitis Functional Index (BASFI), and the Ankylosing Spondylitis Disease Activity Score (ASDAS). These composite indicators are instrumental in evaluating the disease activity, functionality, and overall health status of AS patients. 13 Missing data were imputed using multiple imputation method.
Statistical analysis
Descriptive statistics were employed to encapsulate the demographic and clinical traits of our study cohort. To determine the data distribution, normality tests were meticulously applied. Continuous variables, adhering to a normal distribution, were presented as mean (standard deviation, SD), while those not conforming to normality were depicted as median (interquartile range). Categorical variables were articulated in terms of counts and percentages (number of observations, n; percentage, %). In the realm of quantitative data exhibiting normal distribution, independent samples t tests were utilized. In contrast, for non-normally distributed data, nonparametric Mann–Whitney U tests were strategically deployed. For categorical data comparison across groups, the Fisher exact test was selected for scenarios with expected frequencies less than 5, and for all other cases, the Chi-squared test was the preferred analytical tool.
To scrutinize the risk factors associated with muscle status in AS patients, univariable and multivariable logistic regression analyses were applied. Variables with p-values below 0.2, deemed to be of significance, were incorporated into the multivariable model. Collinearity diagnostics were conducted to examine the potential for multicollinearity among the variables. All statistical analyses were performed using R 4.2.2 (The R Foundation for Statistical Computing, Vienna, Austria). A threshold of p < 0.05 was considered statistically significant.
Results
Baseline characteristics of study patients
Compared to the sex- and age-matched healthy control group, AS patients exhibited a notable decrease in ASM and ASMI (ASM: 22.79 kg vs 23.83 kg; ASMI: 7.69 kg/m2 vs 7.95 kg/m2). However, there were no statistically significant differences in TRSM or TRSMI. Furthermore, AS patients showed lower levels of various body composition indicators, such as inorganic salt, total body water, and basal metabolic rate, compared to the healthy controls (p < 0.05), as detailed in Table 1. Interestingly, there was no significant difference in protein and body fat content between AS patients and the healthy controls; indeed, the percent body fat (PBF) in AS patients was marginally higher than in the controls.
Body composition of AS patients and healthy control group.

AS, ankylosing spondylitis; ASM, appendicular skeletal muscle mass; ASMI, appendicular skeletal muscle mass index; TRSM, trunk skeletal muscle mass; TRSMI, trunk skeletal muscle mass index.
Regarding the demographic profile and body composition of AS patients in our study, the mean age and disease duration for the AS cohort were 36.11 and 10.94 years, respectively. Notably, within these groups, male patients were observed to be younger and better educated (p < 0.05), with no significant variation in disease activity between them. In relation to body composition, female AS patients exhibited lower measurements across several parameters, including PBF. However, when assessing gait speed, no significant difference emerged between them (p = 0.731). Additional details were provided in Table S1.
Characteristics of sarcopenia-related features
We conducted a comprehensive survey to determine the prevalence of sarcopenia in AS patients. In our study of 254 AS patients, we discovered that sarcopenia affected 8.66% of the participants; presarcopenia was present in 11.42%; possible sarcopenia accounted for 36.61%; and severe sarcopenia was identified in 1.18% of the cases. When compared to the control group, the sarcopenia group was characterized by a longer disease duration, lower education level, and higher inflammatory marker levels. The presarcopenia group was marked by lower education levels and elevated BASMI scores. The possible sarcopenia group showed extended disease durations and reduced education level. Notably, all three groups with muscle wasting conditions had significantly lower grip strength than the control group (p < 0.05), as shown in Table 2. In the sensitivity analysis, we evaluated the classification of sarcopenia using the diagnostic criteria defined by the AWGS, further details are outlined in Table S2.
Baseline demographics and clinical features of AS patients with different sarcopenia-related trait.

The prevalence is calculated according to the diagnostic criteria set by AWGS, utilizing the sex-specific lowest quintile as the benchmark. Patients in the control group were those with AS without sarcopenia, presarcopenia, or possible sarcopenia.
p < 0.05. **p < 0.01. ***p < 0.001.
AS, ankylosing spondylitis; ASDAS, Ankylosing Spondylitis Disease Activity Score; ASM, appendicular skeletal muscle mass; AWGS, Asian Working Group for Sarcopenia; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; BMI, body mass index; HLA, human leukocyte antigen; PBF, body fat percent; TNF-α, tumor necrosis factor alpha; TRSM, trunk skeletal muscle mass.
According to the criteria recommended by AWGS, we categorized ASMI based on the lowest quintile cutoffs by sex: low-ASMI group (below the sex-specific fifth percentile) and the control group, which was shown in Table 3. Specifically, the cutoff values of ASMI were 7.48 kg/m2 for males and 5.63 kg/m2 for females. Meanwhile, the cutoff values of TRSMI were 7.79 kg/m2 for males and 6.21 kg/m2 for females. The mean age for the low-ASMI and control groups was 35.69 and 36.21 years, respectively, with no statistically significant difference (p = 0.134). The disease duration for these groups was recorded at 13.31 and 10.34 years, respectively. A higher prevalence of elevated inflammatory markers was observed in the low-ASMI group, indicating a significant difference (p = 0.001). When assessing disease activity, the BASMI scores revealed a statistically significant disparity (p = 0.003). In evaluating body composition, the low-ASMI group consistently exhibited lower values across nearly all metrics compared to the control group (p < 0.05), aligning with anticipated outcomes. Grip strength was notably higher in the group with high ASMI scores (p < 0.05), but no significant variation was found between the two groups in terms of gait speed (p = 0.205).
Baseline demographics and clinical features of AS patients with low-ASMI and control groups.
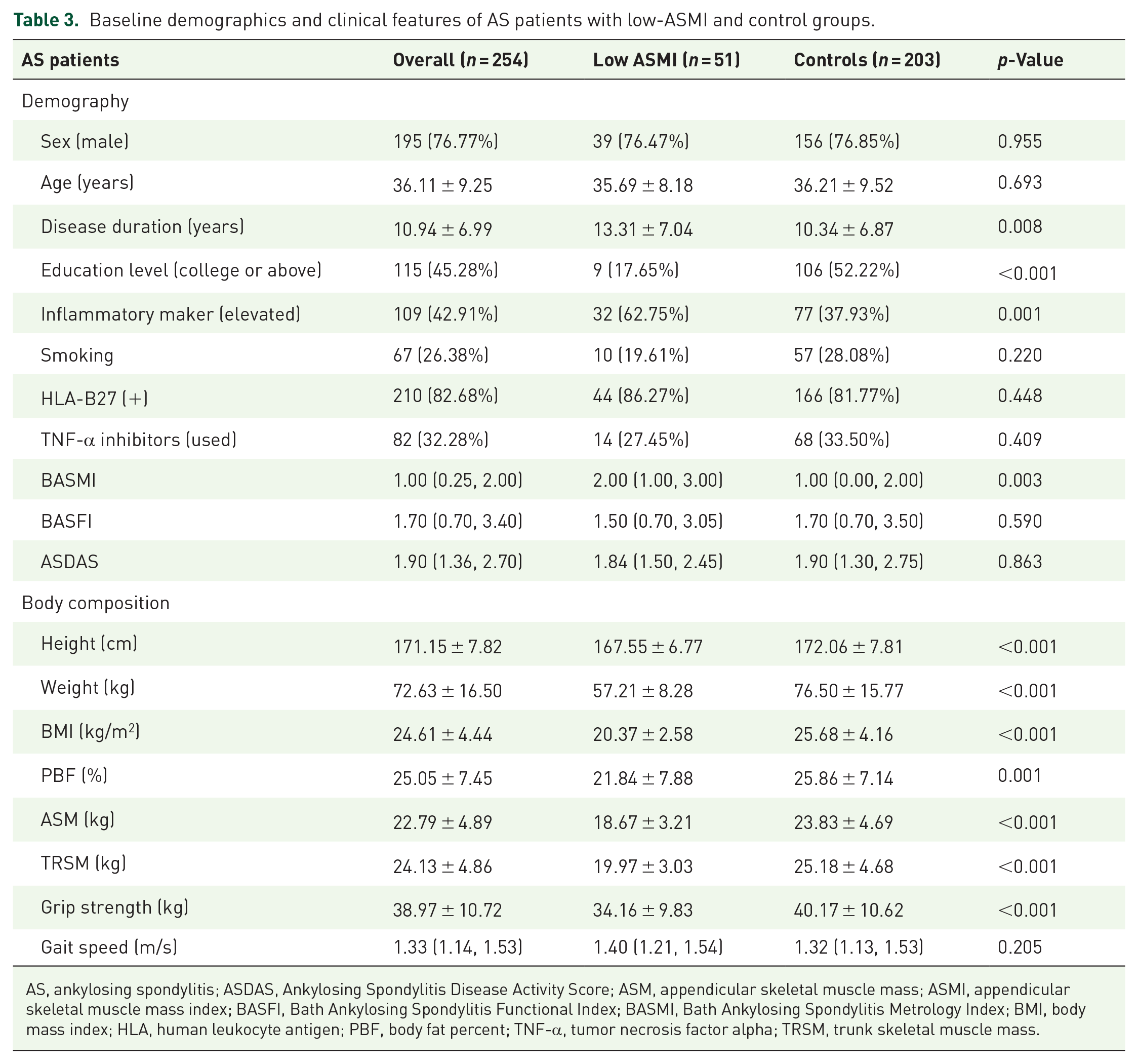
AS, ankylosing spondylitis; ASDAS, Ankylosing Spondylitis Disease Activity Score; ASM, appendicular skeletal muscle mass; ASMI, appendicular skeletal muscle mass index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; BMI, body mass index; HLA, human leukocyte antigen; PBF, body fat percent; TNF-α, tumor necrosis factor alpha; TRSM, trunk skeletal muscle mass.
Similarly, we applied quintile-based cutoffs for grip strength and gait speed, with the thresholds set at 37.4 kg for males and 21.8 kg for females for grip strength, and 1.09 m/s for them in terms of gait speed. In the examination of the grip strength, BASFI was identified as the only demographic differentiator among the patient groups, which was shown in Table 4. The distinctions in TRSM and gait speed for individuals with AS were detailed in Tables S3 and S4.
Baseline demographics and clinical features of AS patients with low grip strength and control group.

AS, ankylosing spondylitis; ASDAS, Ankylosing Spondylitis Disease Activity Score; ASM, appendicular skeletal muscle mass; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; BMI, body mass index; HLA, human leukocyte antigen; PBF, body fat percent; TNF-α, tumor necrosis factor alpha; TRSM, trunk skeletal muscle mass.
Factors associated with skeletal muscle atrophy in AS patients
Upon performing multivariate analyses on 254 AS patients, the following variables were identified with significant associations with low ASMI, as detailed in Table 5: disease duration (odds ratio (OR): 1.086, 95% confidence interval (CI): 1.025–1.150), education level (OR: 0.140, 95% CI: 0.059–0.332), inflammatory marker (OR: 2.950, 95% CI: 1.290–6.745), and BASMI (OR: 1.322, 95% CI: 1.029–1.700). An analysis was also conducted to examine the risk factors associated with low TRSMI, with the outcomes presented in Table S5. Additionally, we had conducted a further analysis of the factors influencing grip strength. Our findings indicated that education level and BASFI were significant factors affecting grip strength, as presented in Table 6. Regrettably, we did not identify any factors influencing gait speed, which was detailed in Table S6.
Univariate and multivariate analyses of influencing factors of ASMI (logistic regression).

ASDAS, Ankylosing Spondylitis Disease Activity Score; ASMI, appendicular skeletal muscle mass index; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; CI, confidence interval; HLA, human leukocyte antigen; OR, odds ratio; TNF-α, tumor necrosis factor alpha.
Univariate and multivariate analyses of influencing factors of grip strength (logistic regression).

ASDAS, Ankylosing Spondylitis Disease Activity Score; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; CI, confidence interval; HLA, human leukocyte antigen; OR, odds ratio; TNF-α, tumor necrosis factor alpha.
Discussion
This study collected a detailed dataset on body composition and sarcopenia-related traits among AS patients. When compared with a healthy control group, a significant reduction in ASM and ASMI was observed in AS patients. Further analysis indicated that patients with low ASMI exhibited diminished values across a spectrum of body composition parameters relative to the control group. Influential factors such as disease duration, education level, and inflammatory markers, and BASMI were identified to potentially affect the appendicular muscle status of AS patients. Regarding grip strength, education level and BASFI were correlated with a reduced muscle strength in individuals with AS.
The incidence of AS has been on an upward trajectory in recent years. A review reported the mean prevalence of AS per 10,000 individuals as 23.8 in Europe, 16.7 in Asia, 31.9 in North America, 10.2 in Latin America, and 7.4 in Africa. 14 Although AS is primarily characterized by chronic pain in the axial skeleton and predominantly impacts young males who are not typically at high risk for muscle atrophy, emerging research suggests that sarcopenia can affect younger demographics as well. 15 The perpetual state of chronic inflammation in AS patients, compounded by factors such as diminished physical activity, escalates the risk of muscle atrophy and ensuing disability, profoundly affecting their daily life and work capacity. 16 Consequently, understanding the nuances of body composition and sarcopenia-related traits in AS patients or initiating early preventative measures is essential for enhancing their overall quality of life.
In our study, the AS patient was predominantly male, with a median age of 35.00 years old and an average disease duration of 10.91 years. This demographic profile diverges from the typical demographic affected by sarcopenia, which is generally observed in older populations. 17 The discrepancy in our findings may be attributed to the protracted chronic inflammation associated with the extended disease course in AS patients. Given that AS predominantly affects young men, this population may experience an accelerated onset of muscle wasting. Our results are somewhat aligned with another study on AS, where 74% of the patients were male, with a mean (SD) age and disease duration of 42.6 (12.2) and 8.3 (8.5) years, respectively. 18
A case-control study utilizing ultrasound did not detect any significant differences in ASM and fat mass between AS patients and the control group. 8 In contrast, a different study employing dual-energy X-ray absorptiometry observed statistically significant reductions in ASM in male AS patients when compared to age-matched healthy controls. 7 The inconsistencies in these outcomes are likely due to the use of different methodologies for measuring skeletal muscle mass, which can introduce variability in the results. Our study employed BIA, a method that allows for the rapid and efficient assessment of AS patients in outpatient settings. The study revealed ASM and TRSM values of 22.79 kg and 24.13 kg, respectively. These findings offer a reference point for BIA examinations specifically tailored for the assessment of AS patients.
Although AS predominantly affects a younger demographic, sarcopenia can manifest in these patients as well. The reported prevalence of sarcopenia in AS patients exhibits variability across studies, with estimates ranging from 10% to 34.3%.7,18,19 Strikingly, our study indicated a notably lower prevalence rate of 8.66%, which deviates from the findings of prior research. Existing criteria for sarcopenia are generally formulated for the broader population and may not account for the unique impact of chronic diseases. Consequently, the AWGS suggests adopting one-fifth as the cutoff point. 20 In our study, the cutoff point established for ASMI was 7.48 kg/m2 for males and 5.63 kg/m2 for females, which indeed differ from the AWGS criteria. We have also established cutoff values for low muscle strength and low physical function in patients with AS. For low muscle strength, the cutoff values were 37.4 kg for males and 21.8 kg for females. For low physical function, the specific cutoff value was 1.09 m/s. A systematic literature review has unveiled that presarcopenia or possible sarcopenia are more frequently observed conditions in AS patients compared to overt sarcopenia. 21 Our research findings corroborate this pattern, highlighting the presence of muscle atrophy in the AS patient population, underscoring the critical necessity for the prompt identification and implementation of intervention strategies.
In individuals with RA, muscles are perpetual state of chronic inflammation in AS patients, compounded by factors such as diminished physical activity, escalates the risk of muscle atrophy and ensuing disability, profoundly affecting their daily life and affected early in the disease process, and indicators of muscle atrophy persist even during periods of clinical remission. 21 Similarly, in AS, the chronic inflammation that causes pain and limits mobility may also take a toll on muscle health. 21 Our logistic regression analysis pinpointed factors that contribute to muscle wasting in AS patients. We observed that individuals with higher levels of education tend to have a reduced risk of muscle atrophy, possibly due to the influence of education on lifestyle choices. 22 With advanced education, people are more likely to gain access to exercise and nutritional knowledge, elevating their cognitive awareness and motivation for physical activity. Consequently, this may lead to more effective self-management of their health conditions and enhanced muscle health overall. Other studies have suggested that factors such as older age, lower BMI, and higher BASFI scores are more significantly associated with sarcopenia than the activity level of the disease. 18 The extent of spinal structural damage in AS patients is not uniform, BASFI and BASMI provide valuable insights into physical impairment. 23 However, while BASFI is based on patient self-reports, BASMI derives its values from clinical examinations, offering a more accurate reflection of the structural status of AS patients. Our research has identified that higher scores on BASMI should prompt healthcare professionals to vigilantly track the muscle condition of AS patients.
Longitudinal research on grip strength indicates that both males and females face an age-related decline in strength, with females typically reaching peak strength earlier and experiencing an earlier onset of decline. 24 While aging and exercise deficiency can partially account for the age-associated weakening of strength, they do not explain the entirety of the phenomenon. In the case of AS, there is a dearth of research examining the disease’s impact on grip strength, which complicates the determination of whether such changes are attributable to the disease itself, to inactivity, or to the normal aging process. For RA patients, the hands are notably affected, often resulting in pain, deformity, and impaired functionality. 25 Grip strength assessments can provide a sensitive reflection of the localized impact of RA on the hands, a dimension that cannot be wholly captured by measures of disease activity alone. 26 In our study, we observed that individuals in the low-ASM and TRSM groups exhibited significantly diminished grip strength compared to the control group, suggesting a general reduction in muscle strength. Furthermore, our study identified low education levels and high BASFI scores as risk factors for decreased grip strength, highlighting the need to be vigilant about the detrimental impact of the disease on grip strength. Our study also assessed the physical functioning of AS patients and found that the majority maintained normal gait speeds, indicating that few had progressed to a stage of muscle atrophy severe enough to impact their ambulatory pace.
The strength of our study is its meticulous assessment of body composition through BIA, effectively addressing a significant gap in the current literature. Our research also provides an in-depth comparative analysis between AS patients with different muscle status and their respective control groups, yielding a thorough understanding of the body composition characteristics in AS patients. However, the primary limitation of our study lies in the selection of diagnostic criteria for sarcopenia in AS. In the absence of universally agreed-upon criteria, we followed the guidelines of AWGS and established our own data-derived lowest quintile as the cutoff value. Additionally, our study is limited by the absence of follow-up data that would track changes in body composition post-intervention. To advance the field, we are setting our sights on a future research agenda that will focus on formulating diagnostic criteria for sarcopenia within the context of AS. We aim to develop and execute a suite of targeted interventions, encompassing tailored exercise regimens and nutritional support. Additionally, we are committed to initiating longitudinal studies aimed at exploring and addressing the factors leading to muscle atrophy, thereby enhancing the overall musculoskeletal health of patients with AS.
Conclusion
AS patients demonstrated a diminished muscle status compared to healthy individuals. Although sarcopenia is not prevalent in AS patients, a subset does indeed suffer from muscle atrophy as the disease progresses. Several factors were identified as potentially influencing appendicular skeletal muscle status in AS patients, including disease duration, education level, inflammatory markers, and BASMI. Education level and BASFI could potentially influence muscle strength. These findings illuminate the intricate aspects of muscle health among patients with AS, providing significant insights and practical intervention strategies for the better management of muscle health in this population.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X251335591 – Supplemental material for Muscle health in ankylosing spondylitis: effects of disease duration, inflammation, education level, and spinal mobility on skeletal muscle atrophy
Supplemental material, sj-docx-1-tab-10.1177_1759720X251335591 for Muscle health in ankylosing spondylitis: effects of disease duration, inflammation, education level, and spinal mobility on skeletal muscle atrophy by Jiawen Hu, Yiwen Wang, Zhimeng Yu, Yinan Zhang, Xiaojian Ji, Lulu Zeng, Jiaxin Zhang, Shiwei Yang, Kunpeng Li, Jian Zhu, Jianglin Zhang, Yinghua Liu and Feng Huang in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X251335591 – Supplemental material for Muscle health in ankylosing spondylitis: effects of disease duration, inflammation, education level, and spinal mobility on skeletal muscle atrophy
Supplemental material, sj-docx-2-tab-10.1177_1759720X251335591 for Muscle health in ankylosing spondylitis: effects of disease duration, inflammation, education level, and spinal mobility on skeletal muscle atrophy by Jiawen Hu, Yiwen Wang, Zhimeng Yu, Yinan Zhang, Xiaojian Ji, Lulu Zeng, Jiaxin Zhang, Shiwei Yang, Kunpeng Li, Jian Zhu, Jianglin Zhang, Yinghua Liu and Feng Huang in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
