Abstract
Background:
Switching between therapies is a recommended strategy for rheumatoid arthritis (RA) and psoriatic arthritis (PsA) patients who experience treatment failure; moreover, data on switching due to primary non-response and subsequent failures are limited.
Objectives:
To obtain information from clinical practice regarding failures due to primary non-response in patients on biologic and target synthetic disease-modifying antirheumatic drugs (ts/bDMARDs), assessing the incidence rate (IR) of switching due to primary non-response and risk of subsequent treatment failure by cycling compared to swapping.
Design:
A longitudinal retrospective study, spanning from 2007 to 2022, was conducted on patients with RA or PsA treated with ts/bDMARDs at an outpatient rheumatology clinic.
Methods:
The main outcomes were as follows: (1) ts/bDMARD failure due to primary non-response and (2) subsequent discontinuation of prescribed ts/bDMARD due to lack of efficacy. The independent variable was switching between classes compared to switching within class. As covariates, clinical, sociodemographic, clinical, and treatments were considered. To estimate ts/bDMARDs switching rates, survival techniques were used, expressing the IR per 100 patients * year with their 95% confidence interval. Cox multivariate regression analyses were run to assess the role of switching between/within class in the subsequent treatment failure.
Results:
In total, 327 patients were included. Of these, 50 patients in 77 treatment courses developed primary non-response with an IR of 4.25 (3.4–5.3). The IR was quite similar between RA and PsA, higher in women, and in those who started ts/bDMARDs after 2018. In those with primary non-response, there were 42 subsequent treatment failures with an IR of 26.38 (19.49–35.69). The multivariate model showed that cycling increased the risk of subsequent failure compared to swapping (hazard ratio: 2 (1.1–3.77),
Conclusion:
This study provides support to consider swapping a better alternative rather than cycling after primary non-response.
Plain language summary
Introduction:
The studied drugs are biological therapies that are arthritis-modifying drugs designed early in the last decade to prevent or reduce inflammation caused by the disease. This study focuses on initial lack of efficacy, in patients with rheumatoid arthritis or psoriatic arthritis who are treated with biologic therapies. We wanted to know how often these treatments fail to work right from the start and how it impacts future treatment choices in real-life practice.
Methods:
We included patients from 2007 to 2022 in which their consultant rheumatologist had decided to commence them on biologic therapy. We studied the changes due to initial lack of efficacy, we also included sociodemographic, clinical and treatments information.
Results:
The results showed that, of the 327 patients, 50 experienced primary initial lack of efficacy. This means about 4 out of every 100 patients had this treatment failure due, with a higher rate seen in women and those starting treatment after 2018. Among those who didn’t respond to their initial treatment, there were 42 further treatment failures, with changes within the same drug class (cycling) more often than changes between classes (swapping).
Conclusion:
This study provides support to consider swapping (changes between drug classes) a better alternative rather than cycling (changes within the same drug class) after initial lack of efficacy. We consider these finding useful for the management of patients. Implementation of more effective strategies may change the disease course.
Keywords
Introduction
Rheumatoid arthritis (RA) and psoriatic arthritis (PsA) are chronic inflammatory joint diseases that can impair patients’ ability to work and perform daily activities, reducing quality of life.1,2
Advances in early diagnosis, targeted treatment strategies, and the development of biologic and synthetic therapies (target synthetic disease-modifying antirheumatic drugs, ts/bDMARDs) have significantly improved their prognosis.3,4
Biologic agents and small molecules target specific pathways involved in disease pathogenesis, aiming to achieve remission or low disease activity.5–12
However, 30%–40% of patients still experience poor outcomes, adverse events, or lack of response, leading to treatment withdrawal.8,13–16 This variability reflects differences in study designs, populations, healthcare practices, and systems. For these patients, successive biologic switching—whether within the same class (cycling strategy) or to a different mechanism of action (swapping strategy)—is advocated by all major international treatment guidelines.8,11,12,17 Nevertheless, the evidence for directing switching from one agent to another is limited with most switches being based on clinical criteria and guidelines algorithms are not clear enough and can differ from one another.
Focusing on inefficacy, treatment response to ts/bDMARDs in RA or PsA can be classified as primary or secondary non-response, based on evidence of an initial response. Primary non-response is considered if the drug is ineffective, with no clinical response and secondary non-response would be considered if, after an initial response, the effectiveness is lost over time.18,19 While primary non-response may be due to a mechanistic failure, secondary non-response may be driven by immunogenicity. 20 We believe that studying the type of non-response separately is a key requirement to improve the current knowledge on patient approach and management. Focusing on primary non-response, it seems reasonable to consider a different mode of action for the next treatment rather than switching within class, but studies addressing the best possible strategy in rheumatic diseases are controversial,14,16,21–27 and except for a few, mainly are related to tumor necrosis factor-alpha inhibitors (TNFi).28,29
A recent study of 12 European registries found similar retention rates for first, second, and third TNFi treatments in ankylosing spondylitis, though rates were lower for second TNFi in cases of primary non-response compared to secondary non-response. Both second and third TNFi treatments were deemed potentially beneficial. 21 Another real-world study in PsA showed low 3-year persistence with bDMARDs as second-line therapy after TNFi, with higher persistence rates for IL-17 and IL-12/23 inhibitors compared to TNFi, though reasons for discontinuation were not analyzed. 30
In RA, both cycling and swapping strategies for b/tsDMARDs are supported by clinical trials and real-world data, with no clear preference in international recommendations. A 2012 meta-analysis by Schoels et al. 31 included one trial on cycling (GO-AFTER) and three on swapping (ATTAIN, REFLEX, and RADIATE), showing both strategies to be more effective than placebo. A 2021 meta-analysis suggested swapping might be superior to cycling after first-line TNFi failure, though it often did not distinguish between primary and secondary failures.26,32 Trials focusing on primary non-responder support cycling as effective compared to placebo.14,33,34 Recently, two head-to-head trials comparing TNFi to non-TNFi suggest swapping may be more beneficial than cycling to another TNFi for primary non-responders.35,36
In all these studies, it is important to consider the heterogeneity in terms of design, study duration, inclusion criteria, treatment regimens, comparators, covariables, and definition of main outcomes (many of them do not specifically focus on primary non-responders but on inefficacy or even persistence as a whole). This makes comparing studies and concluding difficult. Most studies emphasize the failure of the first biologic treatment, especially TNFi, yet in real-world practice, patients often experience multiple therapeutic failures. Thus, it is crucial to examine switches across the follow-up period for all types of ts/bDMARDs. This study aims to analyze long-term data on treatment failures due to primary non-response in PsA and RA patients, comparing the risk of subsequent failure between cycling and swapping strategies.
Materials and methods
Setting, study design, and patients
The study was conducted at a public reference tertiary hospital, HCSC, in Madrid, Spain. It was an observational, retrospective longitudinal study involving patients with chronic inflammatory arthritis who attended the HCSC rheumatology outpatient clinic from January 2007 to April 2021. The study included all patients whose data were recorded in the hospital’s electronic health records and followed them until they were either lost to follow-up or until the study concluded on December 30, 2022.
In the present study, we selected patients with the following criteria: (a) 16 years of age or older; (b) had at least two medical claims with an International Classification of Diseases, Ninth or Tenth Revision, Clinical Modification diagnosis of PsA or RA, and fulfilled the PsA and RA diagnosis according to The CASPAR criteria, 37 ACR/EULAR 2010 classification criteria, 38 respectively; (c) PsA patients were included from January 2007 to April 2021 and followed up until December 2022. RA patients were included from January 2007 to December 2015 and followed up until December 2022; (d) had to be on ts/bDMARDs in the inclusion period.
Exclusion criteria: (a) Patients with a concomitant diagnosis of inflammatory bowel disease and (b) missing data related to the study’s primary outcomes.
The study was conducted in accordance with the Declaration of Helsinki and the principles of Good Clinical Practice and ethics review board approval was obtained. This research is executed in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 39
Data sources
Data were obtained from our EHR, which is used in all outpatient interactions between health professionals and patients.
Variables
Dependent variable
Two main outcomes were established: (a) switching between ts/bDMARDs due to primary non-response defined if the duration of the ts/bDMARDs course was <6 months. When the duration was longer (6–12 months), researchers (D.F-N., M.R-L., and L.A.) reviewed health records and classified patients as primary non-response according to their information and medical criteria. In the case of patients on rituximab, they were reviewed individually even though their treatment duration was longer than 12 months. (b) In those patients who achieved switching between ts/bDMARDs due to primary non-response, the main outcome was subsequent discontinuation of prescribed ts/bDMARD due to lack of efficacy.
The independent variable was switching between classes compared to switching within class. As a class, we included the following exposures: (a) Tumor necrosis factor-alpha inhibitors (TNFi); (b) Janus kinase inhibitors (JAKi); and (c) other biologics: Anti-IL6, Anti-CD20, CTLA4-Ig, Anti-IL17, Anti-IL12/23, and Anti-IL23.
The following covariates were considered including (1) demographic; (2) disease-related; (3) baseline comorbidities; (4) laboratory parameters at baseline: erythrocyte sedimentation rate (ESR), C-reactive protein, rheumatoid factor (RF), and HLAB27; and (5) treatment-related: current use of glucocorticoids, nonsteroidal anti-inflammatory drugs (NSAIDs), and concomitant conventional synthetic DMARDs (csDMARDs) during the whole follow-up of the study. (6) Calendar time: Dividing the starting time of each ts/bDMARDs into year intervals based on treatment strategies and the commercialization of anti-IL17 or JAKi (cutoff in January 2015 and January 2019).
Statistical analysis
A descriptive analysis was conducted. Frequency distribution was used for qualitative variables, mean and standard deviation, or median and percentiles for quantitative variables. Bivariate associations were studied using the Student’s
Switching rates between ts/bDMARDs due to primary nonresponse were estimated by including all PsA or RA patients, measuring from the baseline visit to loss of follow-up, the main outcome, or the end of the study. Discontinuation due to lack of efficacy was estimated for patients with primary nonresponse, measuring from the date of primary nonresponse to loss of follow-up, the main outcome, or the end of the study. Incidence rates (IR) of switching due to primary nonresponse and subsequent failure due to lack of efficacy were estimated by survival techniques (allowing for multiple failures per patient), expressing the IR per 100 patient-years with their respective 95% confidence interval (CI).
Cox bivariate analyses were done to assess the differences between demographic, clinical covariates, and the risk of subsequent failure. Cox multivariate regression analyses were run to assess the role of switching between/within class in the subsequent treatment failure. We included age, sex, calendar time, other known related factors, and all variables with a
The results of the regression models were expressed by hazard ratio (HR) and 95% CI. We considered the influence of covariates if the prevalence was >10%, and if the frequency was lower (in comorbidities or several treatments), categories were grouped based on affinity.
40
The number of variables in the multivariate model followed the rule of Freeman.
41
Missing values of more than 10% were not used in the multivariate analysis. The proportional hazard assumption was tested using Schoenfeld residuals and the scaled Schoenfeld residuals. All statistical analyses were performed using STATA software. A two-tailed
Results
A total of 327 patients were included (141 PsA and 186 RA patients), with a total follow-up of 1807.9 patients-year, a median of 5.1 (2.3–8.9) years, and a maximum follow-up of 16 years. Table 1 includes a detailed description of the baseline characteristics of the patients. As expected, individuals with RA were older and had a higher prevalence of females compared to PsA. The median lag time from diagnosis to first ts/bDMARD was 2.2 (0.45–5.26) years, being higher for RA compared to PsA.
Description of baseline (at first ts/bDMARD) demographic and clinical characteristics of patients.

ACPA, anti-citrullinated peptide antibodies; Anti-CD20, anti-CD20 monoclonal antibody; Anti-IL12/23, anti-interleukin 12/23; Anti-IL17, anti-interleukin 17; Anti-IL6, anti-interleukin 6; CI, confidence interval; COPD, chronic obstructive pulmonary disease; CRP, C-reactive protein; csDMARDs, conventional synthetic disease modifying anti-rheumatic drugs; CV, cardiovascular; ESR, erythrocyte sedimentation rate; JAKi, Janus kinase inhibitors; NSAIDs, non-steroidal anti-inflammatory drugs; PsA, psoriatic arthritis; RA, rheumatoid arthritis; RF, rheumatoid factor; SD, standard deviation; TNFi, TNF-alpha inhibitors; ts/bDMARDs, targeted synthetic and biologic disease-modifying anti-rheumatic drugs.
Focusing on therapy at the beginning of the study, 63.9% and 42.2% of the patients were on glucocorticoids and NSAIDs, respectively, with differences between RA and PsA. The most frequent drugs used as first ts/bDMARDs were TNFi in both groups (79.5%), followed by rituximab in RA and by Anti-IL17 in PsA. Concomitantly, 76.45% of the patients used csDMARDs, and the most frequent was methotrexate in both groups.
As shown in Figure 1, 327 patients received 609 courses of ts/bDMARDs therapy during the study period (405 TNFi, 50 RTX, 42 JAKi, 43 Anti-IL17, 33 Abatacept, 25 Anti-IL6, and 11 Anti-IL23), of which 190, 66, 37, 13, and 11 patients received one, two, three, four, and five courses ts/bDMARDs, respectively. Of them, 126 patients (38.5%) presented 263 switches between ts/bDMARDs over time (70 patients (37.6%) and 142 events in RA; 56 patients (39.7%) and 121 events in PsA). Regarding inefficacy (191 events), 50 of all patients developed 77 switches (12.6%) due to primary non-response (25 patients and 37 events in RA; 25 patients and 40 events in PsA).

Therapy flow chart during the study period.
The estimated IR due to primary non-response was 4.25 (3.4–5.3) per 100 patients-year (Table 2). It was quite similar between RA and PsA and among different patient ages. However, it seemed to be lower in men compared to females and in those with negative RF. The calendar time effect seems to be important as well, it was quite low in the first two periods, increasing after 2018. Regarding the ts/bDMARDs, anti-TNF was one of the drugs with the smallest IR, together with rituximab, JAKi, or Anti-IL6. In relation to other therapeutic co-variables, IR was lower in those on concomitant csDMARDs or in those using NSAIDs, but higher in those on glucocorticoids.
IR of switching due to primary non-response.
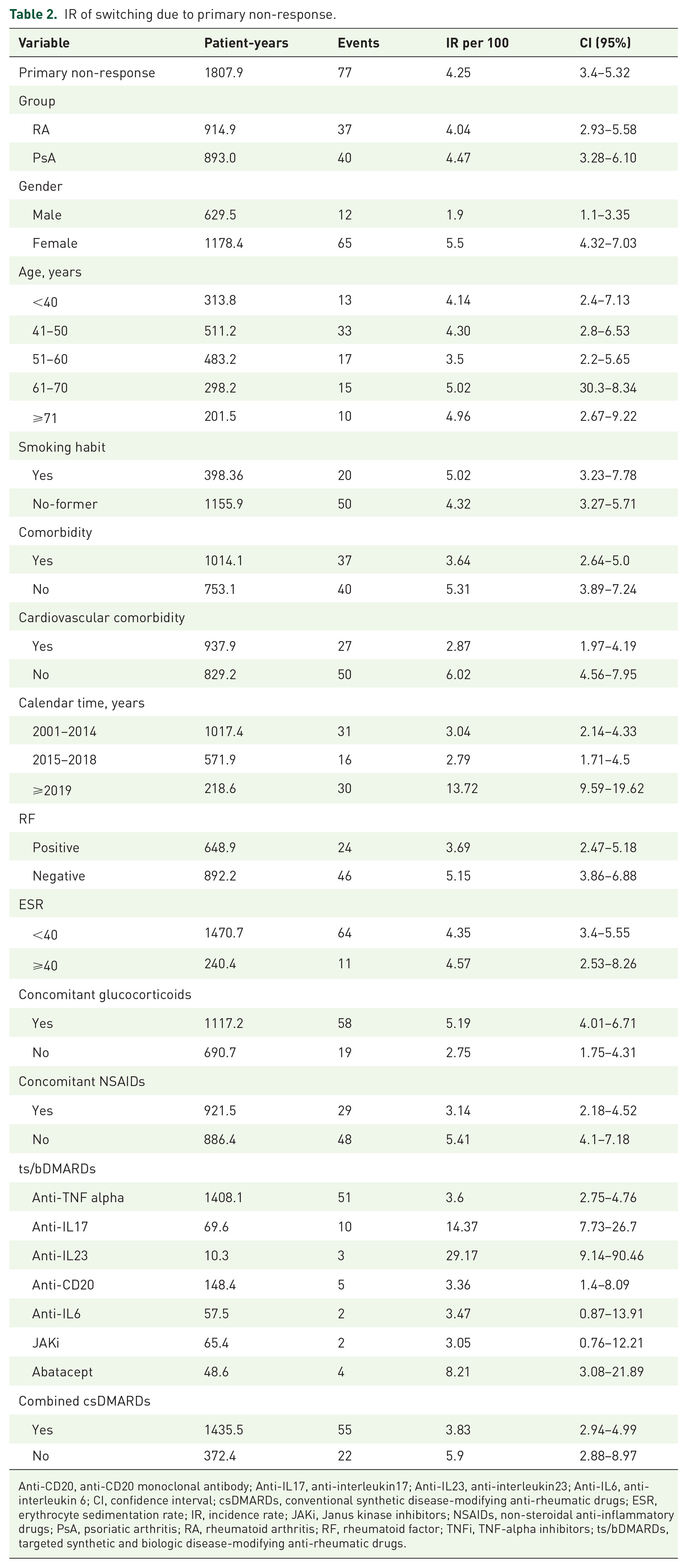
Anti-CD20, anti-CD20 monoclonal antibody; Anti-IL17, anti-interleukin17; Anti-IL23, anti-interleukin23; Anti-IL6, anti-interleukin 6; CI, confidence interval; csDMARDs, conventional synthetic disease-modifying anti-rheumatic drugs; ESR, erythrocyte sedimentation rate; IR, incidence rate; JAKi, Janus kinase inhibitors; NSAIDs, non-steroidal anti-inflammatory drugs; PsA, psoriatic arthritis; RA, rheumatoid arthritis; RF, rheumatoid factor; TNFi, TNF-alpha inhibitors; ts/bDMARDs, targeted synthetic and biologic disease-modifying anti-rheumatic drugs.
In those patients with primary non-response (
Incidence rate of subsequent ts/bDMARD discontinuation due to lack of efficacy in patients with primary non-response (

CI, confidence interval; csDMARDs, conventional synthetic disease-modifying anti-rheumatic drugs; ESR, erythrocyte sedimentation rate; NSAIDs, non-steroidal anti-inflammatory drugs; PsA, psoriatic arthritis; RA, rheumatoid arthritis; ts/bDMARDs, targeted synthetic and biologic disease-modifying anti-rheumatic drugs.
To evaluate differences between demographic, clinical covariates, and the risk of subsequent failure among patients who experienced primary non-response, we initially conducted bivariate analyses (Table 4). These analyses revealed no statistically significant differences between the principal variable and swapping versus cycling, nor other covariates. However, after adjusting for age, gender, disease group (RA vs PsA), and the year of ts/bDMARD initiation, cycling was associated with an increased risk of subsequent failure compared to swapping (HR: 2 (1.1–3.77),
Bivariate analysis: risk of subsequent failure in patients who have experienced primary non-response.

CI, confidence interval; csDMARDs, conventional synthetic disease-modifying anti-rheumatic drugs; HR, hazard ratio; NSAIDs, non-steroidal anti-inflammatory drugs; PsA, psoriatic arthritis; RA, rheumatoid arthritis; ts/bDMARDs, targeted synthetic and biologic disease-modifying anti-rheumatic drugs.
Multivariate analysis: risk of subsequent failure in patients who have experienced primary non-response.

CI, confidence interval; HR, hazard ratio; ts/bDMARDs, targeted synthetic and biologic disease-modifying anti-rheumatic drugs.
Discussion
This longitudinal real-world study examines primary non-response in chronic inflammatory joint diseases, enabling a direct comparison of switching strategies. The findings support swapping as a more effective approach than cycling after primary non-response, regardless of the ts/bDMARD type, treatment course, or other factors. In addition, the study provides descriptive insights, including the incidence of ts/bDMARD failures due to primary non-response in these patients. This study included patients with PsA and RA treated with all available ts/bDMARDs in a single center. Sociodemographic and clinical data were comparable to other national PsA and RA populations studied.42,43
It is reasonable to consider that the underlying mechanisms for treatment failure may differ between primary or secondary non-response. But nonetheless, there are limited scientific publications distinguishing between them. 16 In this study, primary non-response was defined as no improvement observed within 6 months, prompting a switch in ts/bDMARD therapy. 19 To accurately identify all cases of switching due to primary non-response, and given the real-world nature of the data, treatments lasting longer than 6 months were individually reviewed, particularly for patients on rituximab and infliximab. Considering these factors, it is essential to account not only for the 6-month cutoff but also for the specific type of b/tsDMARD and the context from which the data were derived.
In accordance with previously published results,14,16 around 40% of our patients required switching between ts/bDMARDs. This percentage was similar between RA and PsA. As in other studies, the main reason for switching was drug inefficacy, being somewhat less marked in RA compared to PsA, at the expense of more adverse events in RA, maybe because of sex or age differences, more comorbidities rather than the disease per se.
In relation to the total number of switches due to inefficacy, 40% were due to primary non-response, with an estimated IR of 4.25 per 100 patient-years, similar between disease groups or age, but higher in females, and differed between ts/bDMARDs. The role of sex has been reported previously in both diseases and other rheumatic diseases.44,45 In this sense, physiological differences, but also differences in subjective assessment of patient-reported outcomes, could influence the response to pharmacotherapy in men and women.46,47 Regarding the ts/bDMARDs, in other studies, the efficacy shown was similar between drugs,22,48,49 but in our study, TNFi together with rituximab, JAKi, or Anti-IL6 had the lowest incidence of primary failure. However, the data are crude and linked to bias. It must be taken into consideration that this study has included patients with ts/bDMARD since 2007 when there were fewer therapeutic alternatives, conditioning the small patients-year in those more recent ts/bDMARDs, unable to provide robustness to these results.
In our study, the trend to switch between different mechanisms of action became notably more pronounced in 2018. This difference is shown in the crude incidences of primary non-response and subsequent failures, and it has been confirmed in the multivariate analysis. This change is also found in other publications27,50 and can be attributed to the increased availability of treatment options and the full implementation of a Treat-to-Target approach in clinical practice.
Interestingly, in those patients with primary non-response, their subsequent failures due to lack of inefficacy increased by sixfold reflecting the challenges in managing treatment failures in chronic inflammatory arthritis.19,51 Although the incidence of lack of inefficacy was higher in the PsA group compared to RA, in those with high ESR, or in those without concomitant csDMARDs, after adjustment in the multivariate analysis none of these variables achieved statistical significance.
The IR was quite similar between switching within and between classes; nevertheless, after the adjustment in the multivariate analysis, the cycling strategy increased independently the risk of subsequent failure compared to the swapping strategy. There has been scientific literature in RA14,16,22,23,26,29 and PsA 27 with controversial results. In more recent observational studies, data supporting the evidence of swapping strategy are increasing, and just two observational studies did not find differences in both strategies.23,29 This fact is also demonstrated in two head-to-head clinical trials.35,36
Bearing in mind our results, prolonged treatment with the same mechanism of action might not be recommended for primary non-responders. It might be associated with disease progression, clinical complications, and a reduced quality of life. In addition, it may incur unnecessary costs and expose patients to potentially harmful side effects or the development of blocking auto-antibodies. 32
Nowadays, switching between ts/bDMARDs and mainly TNFi due to primary non-response, albeit controversial, still occurs because of confidence and experience with these drugs. In addition, it is not yet clearly established in the guidelines,5,11,12 although the trend seems to be downward. Besides, it cannot be forgotten that certain individual characteristics of the patients such as their preferences, their comorbidities, and/or extra-articular manifestations.11,14,52
It is important to note that all TNFi target the same molecular and inflammatory pathways; thus, it is not surprising that most patients who are primary non-responders to their initial TNFi fail to achieve their treatment targets when cycled through alternative TNFi. The same reasoning may apply to other families such as Anti-IL17 or Anti-IL6. On the other hand, available JAKi differ in terms of affinity for the receptor-associated tyrosine kinases of the JAK family. Maybe in the case of JAKi, cycling to other JAKi after primary failure could be considered an alternative as shown in a recent study by Pombo-Suarez et al. 29 ; however, our study was not able to differentiate by types of JAKi due to the small number of courses. Even so, this group was too small to influence the overall result of this study.
The main limitations of this study are those that affect any retrospective observational study. On one hand, prescription bias cannot be completely ruled out. On the other, there was missing data; however, those included in the analysis have less than 10% missing data. Besides, given the extended follow-up, clinical practice has changed over time as more therapeutic options have become available. This reflects the small sample of the most recent ts/bDMARDs but also has conditioned different therapeutic management, prevailing the switch between TNFi mostly in the first years of the study. To alleviate this imbalance, we have included the calendar time variable in the analysis. Third, we did not report direct parameters of disease activity, but we used ESR as a subrogate variable. Finally, the number of patients and patient-years for the final model is somewhat modest, which might reduce the strength of the analysis. With this in mind, we still consider that these results add to the current knowledge on the real-world management of patients with chronic inflammatory arthritis.
The main strength of this study lies in its longitudinal design with a median follow-up of 5 years and a maximum follow-up of 16 years; the inclusion of a broad non-selected real-world patient spectrum and a wide variety of treatment options. Moreover, all the data were available for analysis, allowing adjustment for confounders to elude possible bias.
In summary, this study provides valuable data on the course of treatment of patients with PsA and RA, as well as on the switch pattern of ts/bDMARDs in the long term. This study can directly compare swapping versus cycling of ts/bDMARDs. We consider that our results add to current knowledge on the real-world management of chronic inflammatory arthritis including the treatment decision-making of these complex patients.
Conclusion
In our study, the primary non-response to ts/bDMARDs had an IR of 4.25 (3.4–5.3), being quite similar between RA and PsA, higher in women, and in those that started ts/bDMARDs after 2018. In those with primary non-response, there were 42 subsequent treatment failures with an IR of 26.38 (19.49–35.69). Cycling increases the risk of subsequent failure. This study provides support to consider swapping a better alternative rather than cycling after primary non-response.
