Abstract
Background:
Golimumab (GOL) is a tumor necrosis factor inhibitor that is used for various types of inflammatory arthritis such as rheumatoid arthritis (RA), psoriatic arthritis (PsA), and ankylosing spondylitis (AS). This article is a systematic review of the evidence for the efficacy and safety of golimumab in inflammatory arthritides, specifically RA, PsA and AS.
Methods:
We conducted a search of randomized controlled trails in MEDLINE [PubMed], CENTRAL, Embase, and Current Controlled Trials databases (ClinicalTrials.gov) through 2017 for studies that evaluated golimumab in inflammatory arthritides. We focused on pivotal, phase III trials for this review of the safety and efficacy of the drug. However, as some important information is not available in detail in publications from the phase III studies, additional individual studies pertaining to antidrug antibodies were also included.
Results:
A total of 12, randomized, double-blind, placebo-controlled studies were included in this review of literature. Two trials focused on the GOL response in the PsA population, four trials focused on the GOL response in the AS population, and five trials focused on the GOL response in the RA population. Additional studies that evaluated autodrug antibodies produced in patients using GOL were also included.
Conclusion:
Golimumab was found to be clinically effective and also have a good safety profile in the treatment of RA, PsA, and AS based on data available from large studies.
Keywords
Introduction
Inflammatory arthritides such as rheumatoid arthritis (RA), ankylosing spondylitis (AS) and psoriatic arthritis (PsA) have multiple therapeutic options. Among therapeutic options, antitumor necrosis factor (anti-TNF) therapies remain one of the most beneficial. TNF-α inhibitors decrease inflammation by blocking TNF-α which is a central pro-inflammatory cytokine. 1 There are five TNF-α inhibitors used in clinical practice worldwide: golimumab (GOL), infliximab, adalimumab, etanercept, and certolizumab pegol. All have slightly different structures but presumably similar mechanisms of actions. 2 GOL is a human monoclonal antibody to TNF-α that is available both subcutaneously (SC) and in intravenous (IV) formulations and has received regulatory approval for treatment of PsA, RA and AS. 1 Overall, it may be considered that the TNF inhibitors are more similar than they are different. This would presumably be particularly the case for the most similar structures, such as the human monoclonal antibodies. However, there are some differences in terms of dosing and dose frequency. This review article will focus on the therapeutic data available for golimumab regarding its effects on patients with inflammatory arthritis.
Methods
We conducted an electronic search in MEDLINE [PubMed], CENTRAL, Embase, and Current Controlled Trials (ClinicalTrials.gov) databases through 2017 with focus pertaining to GOL in inflammatory arthritides. The keywords for the search consisted of ‘golimumab’ combined with ‘rheumatoid arthritis’, ‘ankylosing spondylitis’ and ‘psoriatic arthritis’. We then performed a manual selection of studies that were either multicenter cohorts, randomized controlled trials, or systematic reviews. In addition, we performed individual topic search for autodrug antibodies and golimumab. Exclusion criteria included studies not published in the English language, letters, and case reports/case series. Reference lists of all retrieved articles were also searched to try to find additional studies.
Psoriatic arthritis.

ACR, ; DAS28-CRP, ; GOL, golimumab; HAQ-DI, IV, intravenous; PAS175, PBO, placebo; PsA, psoriatic arthritis; SHSc.
Ankylosing spondylitis and nonradiographic axial spondyloarthritis.

AS, ankylosing spondylitis; ASAS, Assessment of Spondyloarthritis International Society; ASAS20, ASAS improved score by 20%; AxSpA, axial spondyloarthritis; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASDAI50, BASDAI improved score by 50%; BASFI, Bath Ankylosing Spondylitis Functional Index; BASMI, Bath Ankylosing Spondylitis Metrology Index; CRP, C-reactive protein; GOL, golimumab; MRI, magnetic resonance imaging; NR, nonradiographic; PBO, placebo; SF-36, Short Form 36; SI, ; SPARCC, sacroiliac, SpondyloArthritis Research Consortium of Canada; VAS, Visual Analog Scale.
Psoriatic arthritis
PsA is a chronic systemic inflammatory disease characterized by skin and nail psoriasis, peripheral arthritis, axial disease, dactylitis and enthesitis. 3 Data reviewed and presented in the Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) for their 2015 treatment recommendations note that TNF inhibitors have proven beneficial across the key domains of PsA disease activity. This was concluded after use of a variety of instruments in evaluating level of disease activity across domains. This section will discuss the effectiveness and safety of GOL in PsA for various aspects of disease, especially skin and joint disease.
Rheumatoid arthritis.
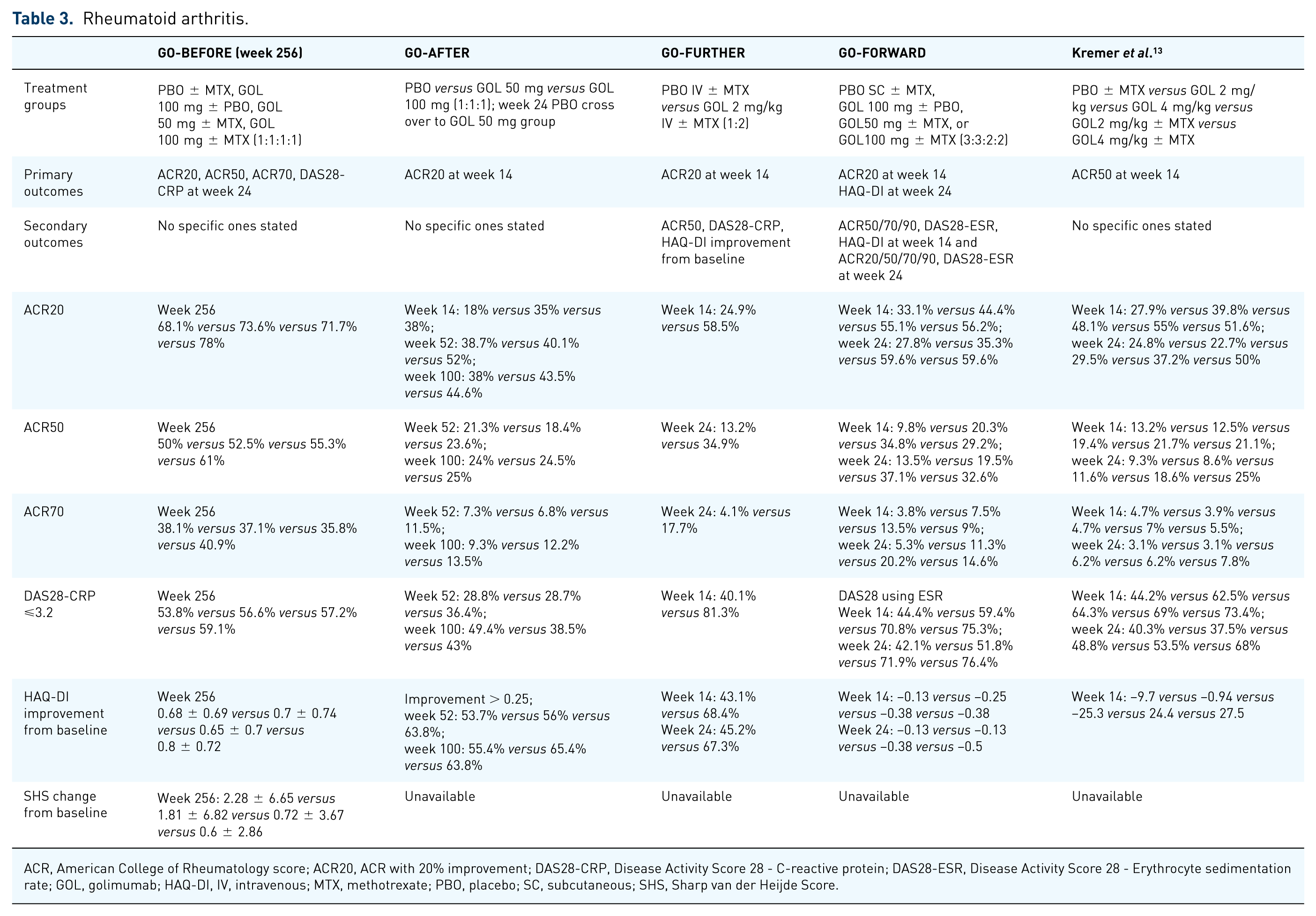
ACR, American College of Rheumatology score; ACR20, ACR with 20% improvement; DAS28-CRP, Disease Activity Score 28 - C-reactive protein; DAS28-ESR, Disease Activity Score 28 - Erythrocyte sedimentation rate; GOL, golimumab; HAQ-DI, IV, intravenous; MTX, methotrexate; PBO, placebo; SC, subcutaneous; SHS, Sharp van der Heijde Score.
Adverse events.

GOL, golimumab; MTX, methotrexate; PBO, placebo; SC subcutaneous.
The two major studies that evaluate GOL in PsA are the GO-REVEAL and the GO-VIBRANT studies. The GO-REVEAL was a double-blind, placebo (PBO)-controlled trial that compared the effects of PBO and two different doses of GOL administered subcutaneously (SC) every 4 weeks (50 mg or 100 mg) in PsA patients. 4 A total of 405 patients with active PsA, defined as three or more swollen/tender joints and active psoriasis, were included in the study and randomly assigned to getting either PBO SC, GOL 50 mg SC, or GOL 100 mg SC every 4 weeks (q4wk). Importantly, at week 24, patients in the PBO group crossed over to the GOL 50 mg group and therefore all patients were on either the 50 mg or 100 mg dose of GOL after week 24. 4 As it has been typical in the design of most studies of TNF inhibitors in PsA, methotrexate (MTX) was permitted but not required for the study. The response rates for the randomized groups at week 256 were 62.8–69.9%, 43.4–50.7% and 30.8–35.6% for American College of Rheumatology score (ACR) 20%/50%/70%. 4 Disease Activity Score 28- C-reactive protein (DAS28-CRP) scores decreased from 4.9–5.0 to <3.2 across treatment groups and Psoriasis Area and Severity Index 75 score (PASI75) was achieved in 60.8–72.2% of the patients that had baseline psoriasis ⩾3% body surface area. 4 The Health Assessment Questionnaire-Disability Index (HAQ-DI) decreased ⩾0.3 for 52–58% of the randomized patients. Only minimal changes in radiographic scores in treatment group were noted; baseline PsA-modified radiographic scores were 0.3 ± 3.8, 0.3 ± 4.2, and 0.1 ± 2.7 in the PBO, 50 mg and 100 mg groups, respectively. 4 Patients in the GOL 50 mg group who did not achieve DAS28-CRP < 2.6 were switched to 100 mg, which showed improvement in DAS28-CRP by 18% and in Psoriasis Area and Severity Index (PASI) scores by 44%. 4 Clinical efficacy appeared to be consistent among patients receiving MTX at baseline and those not on MTX in the study. There were no p values available from the publications pertaining to these data. Since all patients were required to have active PsA in order to enroll in the trial, no definite conclusion regarding possible additive efficacy of GOL and MTX could be made from these data.
Adverse events (AEs): All patients in the PBO group were switched to the GOL group at week 24, so AEs can only be compared between active treatment and PBO for that time period. 4 The most common AE in the GOL groups was upper respiratory infections (URIs) and nasopharyngitis. 4 Infections occurred in 45% of patients who received PBO and then switched to GOL 50 mg after entering the early escape phase, 35% of patients who received PBO and then GOL 50 mg after crossover at week 24, 47% in the GOL 50 mg group, 32% in the GOL 50 mg followed by 100 mg group, and 58% of the GOL 100 mg patients. 4 Serious AEs were reported for 4% of the patients who received PBO and then GOL 50 mg after entering the early escape phase at week 16, 6% of the patients who received GOL 50 mg only, 7% of patients who received GOL 50 mg and then 100 mg, and 3% of patients who received GOL 100 mg. 4
No patient developed active tuberculosis (TB) through week 52. A total of 4% of the GOL-treated patients discontinued the study agent because of an AE, including two (4%) patients who received PBO and then GOL 50 mg after entering the early escape phase at week 16, five (3%) patients who received GOL 50 mg only, and seven (5%) patients who received GOL 100 mg. 4 Injection site reactions occurred in 1 (2%) patient who received PBO and then GOL 50 mg after entering the early escape phase at week 16, 5 (10%) patients who received PBO and then GOL 50 mg after crossover, 11 (8%) patients in the GOL 50 mg group, and 13 (9%) patients who received GOL 100 mg. 4 A total of 21 patients developed malignancies, including 10 patients with nonmelanoma skin cancer. 4 The overall risk of malignancy was not different from general US population but risk of opportunistic infections was higher in the GOL 100 mg group. It is possible that the US Food and Drug Administration approved a lower dose of GOL 50 mg compared with GOL 100 mg because both doses had similar efficacy for PsA but GOL 100 mg had a higher number of AEs.
The GO-VIBRANT trial was a randomized, double-blind, PBO-controlled study that evaluated the safety and efficacy of intravenous (IV) GOL in patients with active PsA. 5 The patients were randomized into IV PBO (n = 239) or IV GOL 2 mg/kg (n = 241) at weeks 0, 4, 12, and 20. 5 Patients otherwise meeting the eligibility criteria especially for active disease were able to continue their baseline MTX (70%) or low-dose prednisone (27%) as long as they were on a stable dose prior to the trial. 6 Results showed that the GOL group had significantly better ACR 20/50/70 responses of 75.1%/43.6%/24.5% compared with PBO 21.8%/6.3%/2.1% at week 14 (p < 0.001). 5 The GOL group also had statistically significant improvement in PASI75 (59.2% versus 13.6%; (p < 0.001)) and greater mean changes at week 14 in HAQ-DI compared with PBO (−0.60 versus −0.12; p < 0.001) at week 14. 5 At 24 weeks, the GOL group continued to have significantly greater improvements in joint and skin disease activity, health-related quality of life (HRQoL), and radiographic progression compared with patients receiving PBO. 5
AEs: there was no significant difference in AEs or serious AEs between the GOL and PBO groups at week 24. 5 Approximately 40.6% patients in the PBO group and 46.3% patients in the GOL group had at least one adverse side effect, with infection being most common. 5 The incidence of serious AEs was also similar, with 3.3% occurring in the PBO group and 2.9% in the GOL group. 5 Two people developed malignancy and both were in the PBO group.
Axial spondyloarthritis, including ankylosing spondylitis
AS is an inflammatory disease that typically presents in young males who present with lower back pain and stiffness. It can be associated with enthesitis, dactylitis, peripheral arthritis, and it can also present with inflammatory bowel disease and uveitis. Diagnosis of axial spondyloarthritis (SpA) defined by the Assessment of Spondyloarthritis International Society (ASAS) criteria includes evidence of sacroiliitis by magnetic resonance imaging (MRI) and at least one SpA feature, or being human leukocyte antigen B27 positive with two or more SpA features. 6 Nonradiographic axial SpA is a new terminology used to classify patients who have a clinical picture of AS but do not exhibit radiographic sacroiliitis. 6 There are four prominent studies that will be discussed in this paper that evaluated GOL in patients with axial SpA.
The GO-RAISE study provided an in-depth analysis of the efficacy and safety of GOL in active AS patients. 1 Inclusion criteria included baseline Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) score ⩾ 4, and a back pain Visual Analog Scale (VAS) score of ⩾4. 1 The patients were then randomized in a 1.8:1.8:1 ratio (138, 140, 78 patients) to receive SC injections of GOL (50 mg or 100 mg) or PBO every 4 weeks for 14 weeks. 1 Primary endpoint of ASAS20 at 14 weeks was achieved in 59.4%, 60.0%, and 21.8% of patient in the GOL 50 mg, GOL 100 mg and PBO patients, respectively (p < 0.001). 1 At 24 weeks, ASAS40 was achieved in 43.5%, 54.3%, and 15.4% of patients, respectively (p < 0.001). 1 The efficacy of GOL was reasserted when 50% of the 41 patients who entered early escape at week 16 achieved an ASAS20 response at week 24. 1 Interestingly, the patients who weighed >87 kg in the GOL 50 mg group and those between 75.15–87 kg in the GOL 100 mg did not have an ASAS20 response statistically significantly different from that of the PBO group, indicating that there may be a weight-based effectiveness to GOL. 1 There was also significant improvement in the physical and mental component including the summary scores of the Short Form 36 (SF-36) Health Survey (both the physical and mental portions had statistically significant improvement in the combined GOL group compared with PBO), the Jenkins Sleep Evaluation Questionnaire score (p < 0.05 in the combined GOL group), and the BASDAI score [>50% improvement; (p < 0.001)] at week 24. 1
AEs: patients in the combined-GOL group and PBO group had similar proportions of patients who had at least one AE at week 16 (77.3% versus 74.0%) and at week 24 (79.9% versus 85.6%). 1 Serious AEs were present in 5.4% of the combined-GOL group and 6.5% of the PBO group which caused approximately nine patients to discontinue the treatment. 1 Serious infections in the GOL 100 mg group included mononucleosis in one patient and chronic otitis media in another; one patient in the PBO group had gastrointestinal inflammation. 1 Injection site reaction occurred in 8.7%, 6.4%, and 2.6% of the patients in GOL 100 mg, GOL 50 mg and PBO groups, respectively, at week 24. 1 There was elevation of aspartate transaminase/alanine transaminase (AST/ALT; one patient in the PBO group, four patients in GOL 50 mg group, five patients in GOL 100 mg group) which caused three of the GOL 100 mg patients to discontinue therapy. 1 However, there was no chronic liver toxicity in any of the patients and the levels normalized in eight/nine patients at week 24. 1 There were no deaths or opportunistic infections. Two patients developed basal cell carcinoma at week 24 (one in PBO, one in GOL 100 mg). 1 Overall, AEs occurred more frequently in patients who were not receiving disease-modifying antirheumatic drugs (DMARDs) at baseline compared with those who were receiving concomitant DMARDs at baseline, regardless of treatment group assignment. 1 This study demonstrated that GOL doses of 50 mg or 100 mg administered SC every 4 weeks were effective and well tolerated. 1
The GO-AHEAD study was a phase III, double-blind, randomized, PBO-controlled trial which evaluated superiority of GOL compared with PBO in patients with active nonradiographic axial SpA (based on the ASAS criteria). 7 The baseline BASDAI and Ankylosing Spondylitis Disease Activity Score (ASDAS) scores for these patients were 6.5 ± 1.5 and 3.5 ± 0.9 respectively. 7 A total of 198 patients were randomized 1:1 to receive GOL 50 mg (97 patients) or PBO (100 patients) SC every 4 weeks. Results showed that both ASAS20 and ASAS40 were statistically significantly higher in the GOL compared with the PBO group (ASAS20 71.1% versus 40%, p < 0.0001; ASAS40 56.7% versus 23%, p < 0.0001). 7 Secondary endpoints such as BASDAI50, ASAS partial remission, and mean change in the SpondyloArthritis Research Consortium of Canada, sacroiliac (SPARCC MRI SI) joint score were significantly improved in the GOL group with p values of p < 0.0001, p = 0.0136, and p < 0.0001, respectively. 7 Actual scores between the GOL and PBO for BASDAI50 at week 16 were 2.93 ± 2.51 versus 4.68 ± 2.75, ASDAS 1.87 ± 1.02 versus 2.8 ± 1.22, SF-36 physical 43.43 ± 10.21 versus 38.33 ± 9.65, SF-36 mental 47.06 ± 11.08 versus 43.08 ± 11.84, Ankylosing Spondylitis quality of life (ASQoL) 5.6 ± 5.16 versus 8.6 ± 5.09, and SPARCC MRI SI score 4.6 ± 7.92 versus 11.71 ± 14.79. 7 This study concluded that the GOL group had significant improvement in symptoms compared with the PBO group in patients with active nonradiographic axial SpA. 7 However, this was a short study which lasted only 16 weeks. 7
AEs: incidence of AEs (47% PBO versus 41.2% GOL) and serious adverse effects (2% PBO versus 1% GOL) were comparable in the PBO and GOL groups, if not lower in the GOL groups. 7 Of the total of three serious AEs (SAEs) reported, two events (back pain and cholelithiasis) occurred in the PBO group and one event (female partner of patient experienced spontaneous fetal death) in the GOL group. 7 There were also no differences between drug-related adverse effects between the PBO and GOL groups (17% versus 13.4%, respectively). 7 No deaths, active TB, malignancies, or serious systemic hypersensitivity reactions were reported during this study. 7
A multicenter, randomized, double-blinded, PBO-controlled trial evaluated Chinese patients with active AS. It involved a total of 213 patients who were randomized to PBO for 0–20 weeks followed by GOL 50 mg for 24–48 weeks (n = 105) or GOL 50 mg for 0–48 weeks (n = 108). 8 Both groups were given SC injections q4wk. 8 The study found that the GOL-treated group had a better ASAS20 response at week 14 and week 24 compared with the PBO group (week 14: 49.1% versus 24.8%, p < 0.001; week 24: 50% versus 22.9%, p < 0.001). 8 The GOL group also had improvements in Bath Ankylosing Spondylitis Functional Index (BASFI) (−1.26 versus 0.11, p < 0.001) and Bath Ankylosing Spondylitis Metrology Index (BASMI) (−0.42 versus −0.19, p = 0.021) scores at week 14, and the mental and physical components of HRQoL and sleep problems at week 24. 8
AEs: A similar percentage of AEs was noted in the GOL versus PBO group at week 16 (31.4% versus 30.6%, respectively) and through week 24 (34.3% versus 32.0%, respectively). 8 Common AEs were URIs and liver transaminase elevations. 8 Two patients discontinued the study, including a patient with ovarian cancer who had only received one dose of GOL and a patient who had developed increased transaminase levels after taking GOL. 9 Serious AEs in the GOL group included enteritis, TB pleurisy, tibial fracture, uveitis and epistaxis. 8 There were no deaths reported during the study. Injection site reaction occurred in three patients receiving GOL 50 mg; all reactions were mild, and no one had to discontinue the study. 8 Authors concluded that GOL was effective and safe in the AS population. 8
Lastly, the GO-ALIVE study was a phase III, randomized, double-blind, PBO-controlled trial in which patients with active AS were randomized in a 1:1 ratio to receive either IV infusions of GOL 2 mg/kg (n = 105) or IV PBO (n = 103) at weeks 0, 4, 12, and then every 8 weeks. 9 Patients were allowed to continue systemic DMARDs such as MTX, sulfasalazine (SSZ), or hydroxychloroquine (HCQ). The study achieved its primary endpoint of ASAS20 at week 16 in the GOL-treated group compared with the PBO group (73.3% versus 26.2% respectively; p < 0.001). 9 Secondary endpoints including ASAS40 (47.6% versus 8.7%; p ⩽ 0.001), improvement in the BASDAI50 (41% versus 14.6%; p < 0.001), and change in the BASFI (−2.4 versus −0.5; p < 0.001) at week 16 were also achieved in the GOL versus PBO group. 9 The GOL group had significant reduction in the signs and symptoms of AS compared with the PBO group. 9
AEs: safety was monitored through week 28 with patient crossover from the PBO to GOL group at week 16. 9 A total of 23.3% of the PBO group and 32.4% of the GOL had more than one AE at week 16. 9 Infections such as nasopharyngitis and URI were most frequent. 9 Two events of SAE occurred in the GOL group, with one patient getting pneumonia and another with pancreatitis. 9 Three patients developed infusion reactions in the GOL group. 9 There were no reported demyelination, opportunistic infection, malignancy, or death through week 28. 9 A total of 21 patients in the GOL group received prophylaxis for latent TB but none of them developed active TB infection. 9 The number of AEs was consistent with other TNF inhibitor therapies with no new safety signals. 9
Rheumatoid arthritis
The GO-BEFORE trial evaluated the effectiveness of GOL in MTX-naïve patients with active RA. 10 A total of 637 were randomized to the following groups: PBO plus MTX, GOL 100 mg plus PBO, GOL 50 mg plus MTX, or GOL 100 mg plus MTX. 10 The patients received either GOL or PBO SC every four weeks and the MTX dose started at 10 mg/week and then increased to 20 mg/week. The primary endpoint of ACR50 at week 24 was not achieved between the combined GOL-MTX groups and PBO-MTX group (38.4% and 29.4%, respectively; p = 0.053). 10 However, a post hoc modified intention-to-treat (ITT) analysis of the ACR50 response (which excluded three untreated patients) demonstrated a significant p value when comparing the combined GOL-MTX groups and PBO-MTX group (38.5% versus 29.4%; p = 0.049) and between the GOL 50 mg plus MTX group (40.5%; p = 0.038). 10
The combined GOL-MTX groups had a significantly better DAS28 score compared with PBO ± MTX for response/remission. 10 Levels of significance for. European League Against Rheumatism (EULAR) good or moderate response for patients with disease activity < 1 year and disease activity < 3 years were p = 0.029 and p = 0.026, respectively. 10 Similarly, Disease Activity Score 28 - Erythrocyte sedimentation rate (DAS28-ESR) remission for patients with disease activity < 1 year and <3 years had p = 0.017 and p = 0.005, respectively, when comparing the combined GOL-MTX groups with PBO. 10 Even though the study did not achieve the prespecified primary endpoint, the modified ITT analysis of the primary endpoint demonstrated that GOL plus MTX had better efficacy than the PBO plus MTX group in reducing RA signs and symptoms. 10
AEs: at least one AE was found in 72.5% (116 of 160) of the PBO ± MTX group, 68.2% (107 of 157) of the GOL monotherapy group, 81.6% (129 of 158) in the GOL 50 mg ± MTX group, and 76.1% (121 of 159) in the GOL 100 mg ± MTX group at week 24. 10 SAE occurred in 7% of the PBO ± MTX group, 3% in GOL monotherapy group, and 6% in both the GOL 50/100 mg ± MTX groups. 10 The most frequent side effects for the GOL ± MTX groups were nausea, upper respiratory tract infection, increased AST level, increased ALT level, dyspepsia, and headache. 10 Malignancies developed in four patients; one patient developed breast cancer in the PBO ± MTX group, the rest from other groups which included Hodgkin’s lymphoma, lip squamous cell skin cancer, and another case of breast cancer. 10
The GO-FORWARD trial was a phase III, double-blinded study evaluating the efficacy and safety of GOL in patients with active RA despite being on MTX monotherapy. 11 This study had an open-label extension up to 5 years after week 52. 11 A total of 417 completed up to the 24-week study period during which all patients were maintained on an MTX dose between 15–25 mg/week. Patients were divided in a 3:3:2:2 ratio to receive PBO SC ± MTX, GOL 100 mg ± PBO, GOL50 mg ± MTX, or GOL100 mg ± MTX and the injections were given SC q4wk. 11 The primary endpoints were ACR20 at week 14 and change from baseline in HAQ-DI at week 24. Secondary endpoints included ACR50/70/90, DAS28-ESR, HAQ-DI at week 14 and ACR20/50/70/90, DAS28-ESR at week 24. 11 They found that ACR20 at week 14 was 33.1% versus 44.4% versus 55.1% versus 56.2% and HAQ-DI at week 24 was 0.13 versus 0.13 versus 0.38 versus 0.50 in the PBO SC ± MTX, GOL 100 mg ± PBO, GOL50 mg ± MTX, and GOL100 mg ± MTX groups, respectively. 11 These values had statistically significant improvement in the GOL groups for the ACR20 and HAQ-DI compared with the PBO group. 11 The addition of GOL to MTX therapy significantly reduced the signs and symptoms of RA and improved physical function. 11
AEs: at week 16 (before opportunity for early escape), AEs developed in 60.9% versus 63.2% versus 68.3% versus 69.7% and SAEs occurred in 2.3% versus 3.8% versus 5.6% versus 9% in the PBO SC ± MTX, GOL 100 mg ± PBO, GOL50 mg ± MTX, and GOL100 mg ± MTX groups, respectively. 11 One patient died during the study by week 24 secondary to ileus and aspiration pneumonia. A total of 92 patients were diagnosed with latent TB at the onset of the study and were treated, and none developed active TB. 11 Injection-site reactions were uncommon but no patients had to stop the study because of that side effect. 11
The radiographic effects and maintenance of GOL in active RA patients was evaluated in the randomized, double-blind, PBO-controlled, phase III GO-FURTHER study. 12 In this study, GOL was given in IV formulation rather than SC. A total of 592 patients with RA defined as ⩾6/66 swollen, ⩾6/68 tender joints, CRP ⩾ 1.0 mg/dl and positive for rheumatoid factor or anticyclic citrullinated protein antibody with active disease despite being on a stable dose of MTX ⩾ 3 months (15–25 mg/week) were evaluated. 12 They were randomized 2:1 to receive IV GOL 2 mg/kg or PBO infusions at weeks 0 and 4 and then every 8 weeks for up to 52 weeks, while continuing their MTX regimen. 12 The study achieved its primary endpoint of statistically significant ACR20 response at week 14 in the GOL group compared with the PBO group (p < 0.001). 12 The GOL ± MTX group had less radiographic progression compared with the PBO group at week 24, with an Sharp van der Heijde Score (SHS) score mean change of 0.03 versus 1.09; p < 0.001 at week 24 and 0.13 versus 1.22; p = 0.001 at week 52. 12 Results at week 14 between GOL versus PBO were the following: DAS28-CRP 81.3% versus 40.1%, p < 0.001; Clinical Disease Acitivty Index (CDAI) score improvement 19.2 ± 12.80 versus 7.6 ± 16.17; p < 0.001; Health Assessment Questionnaire (HAQ) score improvement by 0.25 units was 68.4% versus 43.1%; p < 0.001. 12 Total of 80% of the GOL ± MTX group maintained an ACR20 response at week 52. 12
AEs: the number of AEs in the PBO ± MTX- and GOL ± MTX-treated patients through week 16 (43.7% and 47.3%, respectively) and week 24 (49.2% and 52.9%, respectively) were similar. 12 The most common AEs were URIs, urinary tract infection, and nasopharyngitis. Higher incidence of musculoskeletal disorders was observed in the PBO ± MTX (14.7%) compared with the GOL ± MTX (6.8%) group. 12 Through week 24, SAEs occurred in 2.0% and 4.1% of the PBO ± MTX- and GOL ± MTX-treated patients, of which infections were the most common. 12 No serious opportunistic or TB infections were reported in either group. 12 At week 24, there was one case of breast cancer in the GOL ± MTX group and one case of lung adenocarcinoma in the PBO ± MTX group. 12 No lymphoma or demyelination were reported. Mild infusion reactions occurred in 3.5% in the combined GOL ± MTX group and 0.5% in the PBO ± MTX group. 12 The study concluded that IV GOL had a good safety profile. 12
Another study that evaluated the efficacy and safety of IV GOL consisted of 643 patients with moderate-to-severe RA who had persistent disease activity despite treatment with MTX (15–25 mg/week for >4 weeks). 13 They were randomly distributed in 1:1:1:1:1 ratio to the following groups: IV PBO plus MTX, 2 mg/kg GOL with or without MTX, or 4 mg/kg GOL with or without MTX. 13 Patients were allowed to continue on their baseline nonsteroidal anti-inflammatory drugs and steroid. Approximately 5% of the patients had received either infliximab, etanercept, or adalimumab prior to study participation but none during the study. 13 The patients received infusions every 12 weeks through week 48 and there was an early escape group at 16 weeks for patients with <20% improvement in the swollen and tender joint counts. Results showed that the primary endpoint of ACR50 was not met by week 14 (21% in GOL ± MTX versus 13% in PBO ± MTX; p = 0.051). 13 Even though the ACR50 response increased by week 24 and 48 in the GOL monotherapy group, it was not significantly different compared with PBO. 13 By week 48, the GOL 4 mg/kg plus MTX group had the highest ACR20 and ACR50 responses (70% and 48% respectively; p value not available). 13 However, from a clinical perspective, patients in the GOL (2 mg/kg and 4 mg/kg) plus MTX groups were superior, as they had greater improvement in the DAS28-CRP compared with the PBO ± MTX group (71% versus 44%; p < 0.001). 13 There were similar results for disease remission based on the DAS28-CRP for patients in the combined group receiving GOL ± MTX compared with PBO ± MTX at week 14 and week 24. 13 Clinically meaningful improvement in HAQ-DI was seen in approximately 8% of the PBO ± MTX group versus 25% (p = 0.004) of the GOL group versus 33% (p < 0.001) of the GOL ± MTX group. 13
Although the primary endpoint of ACR50 was not achieved in the study, there was noticeable clinical improvement in the IV GOL plus MTX group in long-term benefits compared with the PBO group, without any significant safety concerns.
AEs: there were no significant differences in AEs between the GOL-treated and the PBO groups at week 24. 13 However, there were more AEs in the GOL ± MTX group compared with the monotherapy GOL group. 13 There were no serious opportunistic infections or TB in either group. 13 Infusion reactions among the GOL-treated patients was 4% and 5% in the PBO ± MTX group at week 24 and week 48. 13 However at week 48, 82% of the GOL-treated patients had AEs compared with 72% of the PBO group. Infections were the most common (URI and bronchitis), occurring in 48% of the GOL group and 41% of the PBO ± MTX group. 13 Two cases of TB reported between week 24 and week 48 in the 4 mg/kg GOL-treated groups. 13 At the end of 48 weeks, a total of three deaths (one in the 2 mg/kg GOL group, one in the GOL 4 mg/kg group and one in the GOL ± MTX group) occurred following myocardial infarction. 13
In the GO-AFTER study, the long-term safety of GOL in patients with active RA (four or more tender, four or more swollen joints) previously on TNF inhibitor was assessed. 14 The effectiveness of GOL was evaluated in the first part of the study, which lasted for 24 weeks, and the study extension evaluated for the safety of GOL through 160 weeks. Patients were initially randomized to receive PBO, 50 mg GOL SC or 100 mg GOL SC injections q4wk. 14 Results from the 24-week analysis showed that the ACR20, DAS28-ESR response and DAS-ESR remission for GOL 50 mg (34%, 46% and 10%, respectively) and GOL 100 mg (44%, 61% and 16%, respectively) were significantly higher than PBO group (17%, 25% and 3%, respectively; all p < 0.05). 14 Patients in the PBO and GOL 50 mg SC q4wk groups who did not achieve ACR20 were switched to GOL 50/100 mg respectively for early escape at week 16. 14 All patients from the PBO group were switched to GOL 50 mg SC q4wk at week 24. A total of 459 of the 461 randomized patients were treated with GOL and 51% of those patients continued treatment through week 160. 14
There was response maintenance from week 24 to week 100 as ACR20 and ⩾0.25 unit HAQ improvement were sustained in 70–73% of PBO patients who had crossed over and 75–81% of responding patients in the GOL 50/100 mg groups (p value not available). 14 Similar results were present at week 160: 63%, 67% and 57% of patients achieved ACR20 response and 59%, 65% and 64% had HAQ improvement ⩾0.25 unit in the PBO, GOL 50/100 mg groups, respectively (p value not available). 14
AEs: weeks 0–16: adverse side effects were experienced in 70%, 61% and 73% of the patients in the PBO, GOL 50 mg and GOL 100 mg groups, respectively. 14 The rates of infections (28%, 27% and 25%), serious infections (2%, 2% and 1%), and serious adverse effects (7%, 5% and 3%) were similar across all groups. 14 More injection-site reactions were noted in the GOL 100 mg (11%) compared with the PBO (3%) or GOL 50 mg (4%) groups. 14 Week 160: a total of 81% in the GOL 50 mg group and 90% of GOL 100 mg group experienced one or more AE, 18% and 25% had SAEs, 5% and 9% had serious infections and 9% and 12% discontinued the study agent due to an AE. 14 The number of deaths through week 160 were 1.73 (0.04 to 9.65), 0.00 (0.00 to 0.94) and 0.62 (0.17 to 1.59)/100 patient-years, respectively, for the PBO, GOL 50 mg and GOL 100 mg groups. Cause of deaths in the GOL group were aggressive lymphoma, cardiovascular events, Congestive heart failure (CHF), pneumonia (PNA).14 The GOL 100 mg group had the highest risk of malignancy of 2.04 (1.09 to 3.49/100 patient-years) with the largest incidence being lymphoma 0.62 (0.17–1.59). 14 The incidence of total malignancies in the treatment group was the same when compared with the general US population using the Surveillance, Epidemiology, and End Results (SEER) database, but the risk of lymphoma in the GOL 100 mg group was increased. 14 Common reasons for study agent discontinuation included unsatisfactory therapeutic effect (43% of discontinued patients) and AEs (28%, including nine patients with worsening RA). 14
This study provided good data demonstrating that patients with active RA who were previously treated with TNF inhibitor could have improvement in signs and symptoms when treated with either GOL 50 mg and GOL 100 mg injections q4wk (approximately 57–67% who received treatment). 14 However, an important limiting factor in this study was that only 50% of the patients were followed up in the long-term-extension study and therefore the trial did not have enough power beyond week 24. 14 Additionally, there were no set criteria for dose escalation and it was decided by the discretion of the investigators.
Antidrug antibodies
Antidrug antibodies have been detected against large protein drugs administered therapeutically to humans, including to the various TNF inhibitors. This phenomenon has been evaluated in multiple GOL studies. The relationship between antidrug antibodies, drug trough levels and therapeutic responses in patients treated with GOL for RA or AS was evaluated in the study by Chen and colleagues. 15 A total of 76 biologic-naïve patients (33 RA, 43AS) were treated with monthly GOL 50 mg SC injections for 24 weeks. 15 At the end of the 24 weeks, serum antidrug antibodies and drug trough levels were tested by the bridging enzyme-linked immunosorbent assay (ELISA) and capture ELISA methods, respectively. A positive result was defined as an antidrug antibody titer > 30 unit/ml in combination with GOL level < 5. 15 They found that antidrug antibodies developed in 15.2% (5/33) of the RA patients and in only 2.3% (1/43) of the AS patients. 15 The following factors may have contributed to the proportionally higher rates of antidrug antibodies in RA patients when compared with other studies (2.1–8.1%): fewer patients using MTX therapy; being on a lower MTX dose; differences in assay analysis; and low sample size. 15 Conversely, the AS group may have found lower antidrug antibodies levels compared with other studies because the bridging ELISA used in this study could not detect antidrug antibodies in the presence of drug. 15
The antidrug-antibody-positive RA patients were found to have low GOL levels, worse EULAR response and a higher DAS28 compared with patients who did not develop the antibodies (EULAR 100% versus 10.7%, p < 0.05; DAS28 100% versus 39.3%, p < 0.05). 15 There was clinical improvement in both the RA and AS treated patients that had normal detectable GOL levels; statistically significant improvement in DAS28 (r = 0.594, p < 0.001) for the RA patients and statistically significant improvement in the BASDAI50 and ASDAS scores for the AS patients. 15 This study concluded that antidrug antibodies were associated with lower GOL levels and worse therapeutic response in both the RA and AS patient cohorts. 15 Thus, drug monitoring could help lead to personalizing therapy strategy in the future.
In another study, combined treatment of GOL with MTX was associated with a lower incidence of antibodies to GOL. During week 24, antibodies to GOL were detected in 27 of the 529 patients, and this number increased to 43 by week 48. 13 Patients who had GOL plus MTX (10 of 299; 3%) had lower proportions of antibody development compared with patients who had GOL monotherapy (17 of 194; 9%) through week 48. 13 The antibody titers were low in all groups at week 24 with the maximum being 1:160 in two GOL-treated patients. 13 Similar to the other studies, this study found that the patients who were negative for autoantibody had higher ACR20 and ACR50 responses compared with those who developed the autoantibodies, however the differences were not statistically significant. 13
These effects of MTX were similarly seen in the GO-BEFORE study and GO-REVEAL study. Patients in the GO-REVEAL study generally developed antibodies in low titer but fewer occurred in patients being treated with MTX (1.8%/10.0% with/without MTX). 4 In the GO-BEFORE study, a total of 6.3% (20 of 315) patients receiving GOL developed antibodies to GOL, of which the majority were in the GOL 100 mg without MTX group [14 of 104 (13.5%)]. 10 Conversely, in the GO-RAISE study, 4.1% of the patients assigned to the GOL groups (without concurrent MTX treatment) developed antibodies to GOL. 1
Some studies have shown that autoantibody development may not make a difference in the clinical response, such as in the GO-VIBRANT trial that reported the GOL group had 44 patients who tested positive for antibodies to GOL but most were low titers, and even the ones with higher titers did not preclude clinical response. 5 The GO-AFTER study reported a low cumulative incidence of antibodies to GOL through week 268 of 8.0%, of which 89.3% had neutralizing antibodies. 14 However, there was no comment regarding what percentage of these patients were on MTX and if developing these antibodies changed their clinical outcome. 14
Discussion
Based on the findings of the current GOL trials, there are sufficient data concluding that GOL is an efficacious drug with a good safety profile for use in PsA, AS, and RA. The main studies of focus for PsA were GO-REVEAL and GO-VIBRANT, both of which were largescale studies comparing a GOL group with a PBO group. The main difference being that GO-VIBRANT used IV GOL compared with GO-REVEAL which used SC GOL. Both studies achieved their primary outcomes of achieving ACR20 response in the GOL groups at week 14 compared with PBO groups, and the GOL groups also had improvement across all domains. The AEs in both studies were similar to the PBO group and did not pose a significantly higher risk with use.
The key studies for AS include the GO-RAISE study, the GO-AHEAD study, the GO-ALIVE study, and a study which focuses on the Chinese population with active AS. All four studies reached their primary endpoints concluding that GOL was an effective therapeutic option for AS patients. Safety profile was also similar between the studies, with infections being one of the most common AEs. However, the infection risk with GOL was not found to be significantly higher than the PBO groups.
The GOL studies in RA patients had mixed outcomes, but overall showed that GOL had improvement in signs/symptoms, functional status and inhibition of radiographic damage. Primary endpoint of ACR20 improvement and functional improvement was achieved in the GO-FORWARD, GO-FURTHER, and GO-AFTER studies. The main difference between these studies being that GO-FURTHER used IV GOL as opposed to SC GOL in the other two studies. Additionally, there was less radiographic progression at week 24 in the GO-FURTHER study.
Two studies that did not achieve their primary endpoints were the GO-BEFORE study and one of the IV GOL studies. In the GO-BEFORE study, the primary endpoint of ACR50 response was not achieved at week 24; however, using modified ITT analysis (which excluded three patients who never received treatment), the improvement in response between the combined GOL-MTX and PBO ± MTX was significant. This raises the issue about the selection of primary endpoint. While ACR50 differences have been suggested to be a good way of comparing two active treatments, ACR20 responses are still the best method to differentiate active treatment from PBO.
It is generally assumed that the TNF inhibitors, including the two different formulations of GOL (IV and SC), are more similar than they are different. There are no head-to-head studies that would allow direct comparison among these agents. It is also not possible to make any comparison of the efficacy and safety of the various agents by assessing results from individual studies of the various TNF inhibitors due to important differences across studies. Some of these key differences include different inclusion criteria, differences in the study populations (e.g. disease activity, duration of disease, etc.) and differences in the geographic areas, and times when the studies were conducted.
Limitations
Some of the studies used in this review were of short duration, such as the GO-AHEAD study, making assessment of the safety outcomes limited. Additionally, patients in the randomized clinical trials typically have high disease activity and therefore may not reflect typical clinic patients. The data for the drug-induced antibodies is also very limited in that only one study out of the ones mentioned above was exclusively specifically designed to evaluate the antibody development. 15 The other results were observations from studies whose primary endpoints were to evaluate the efficacy and safety of GOL.
Footnotes
Funding
Research was supported by the Department of Veterans Affairs
Conflict of interest statement
Dr Arthur Kavanaugh has conducted clinical research studies sponsored by Janssen.
