Abstract
Psoriatic arthritis (PsA) is a heterogeneous, chronic, inflammatory musculoskeletal disease that can lead to peripheral and axial damage and loss of function. A clear difference between PsA and other forms of inflammatory arthritis is the different forms of bone remodeling seen in PSA which incorporates not only increased bone resorption with bone erosions, osteolysis, and loss of bone mineral density but also increased bone formation with periostitis, syndesmophytes, enthesiophytes, and ankylosis. PsA, if diagnosed late, will lead to significant structural damage, the most severe form of which is known as arthritis mutilans, and loss of physical function. Imaging plays a crucial role in diagnosing and monitoring both peripheral and axial conditions associated with PsA. Radiography is currently the main modality used to monitor structural damage in PsA though commonly used scoring systems do not include bony proliferation as a criterion. Besides, radiography is limited in determining the presence and cause of periarticular soft tissue thickening, which may arise from tendinosis, tenosynovitis, synovial proliferation, bursitis, or enthesitis. Recently, much more attention has been paid to determining the imaging characteristics of PsA, which enables more precise identification of disease and severity assessment. Newer imaging technologies also enable variations in normal bone microstructure to be distinguished from disease-related abnormality. This review discusses the current state of innovative imaging modalities in PsA, specifically concentrating on their roles in PsA diagnosis and treatment, improving the early detection of PsA, and identifying patients with skin psoriasis at risk of developing psoriatic arthritis.
Keywords
Background
Psoriatic arthritis (PsA) is a chronic inflammatory joint disease with diverse clinical manifestations including skin and nail alteration, peripheral arthritis, axial disease, dactylitis, and enthesitis. Peripheral synovitis is the most common characteristic of PsA ranging from mild joint inflammation to disabling peripheral arthritis. Preventing structural damage is one of the primary goals of treating PsA patients to optimize health-related quality of life. Different imaging modalities such as radiography, high-resolution peripheral quantitative computed tomography (HR-pQCT), magnetic resonance imaging (MRI), low-dose CT (ldCT), ultrasound (US), and positron emission tomography (PET)-CT (Table 1) can be used to identify and characterize suspected subclinical, early, or established PsA cases, and can provide crucial information regarding treatment. These imaging techniques vary considerably in terms of technology, measurable parameters, spatial resolution, radiation exposure, acquisition time, region of interest, cost, and availability as well as accuracy in identifying disease, predicting clinical outcome, and monitoring treatment response. This review will address all these imaging modalities in turn.
Comparison of different imaging modalities.

, Relative effectiveness of modality. *, easy availability and low cost.
BME, bone marrow edema; DAPSA, disease activity in psoriatic arthritis; FOI, fluorescence-optical imaging; HR-pQCT, high-resolution peripheral quantitative computed tomography; JSW, joint space width; ldCT, low-dose computed tomography; MRI, magnetic resonance imaging; MSOT, multispectral optoacoustic tomography; PET, positron emission tomography; PsA, psoriatic arthritis; STI, soft tissue inflammation, such as at entheses; US, ultrasound.
Radiography and HR-pQCT for peripheral joint structural and microstructural assessment
Radiography is the standard method for assessment of joint disease in PsA since it is affordable, accessible, and safe.1,2 Radiography can help differentiate PsA from other inflammatory arthritis and evaluate the effectiveness of conventional and biologic therapies for PsA. However, commonly used radiographic scoring systems do not include bony proliferation as a criterion.3–6 Radiography is also limited in being a two-dimensional projectional technique rather than a three dimension. 7 HR-pQCT is a technique that enables bone microstructure analysis with high reproducibility in defining and quantifying structural bone changes in patients with inflammatory arthritis, such as bone erosion, new bone formation (enthesiophytes), and joint space width (JSW) (Figure 1).8–10 With its high spatial resolution of 100 or 142 μm, 11 HR-pQCT has higher sensitivity for erosion detection compared to radiography and MRI (the minimal erosion dimensions identified by HR-pQCT, MRI, and radiography were 0.09, 0.14, and 0.66 cm, respectively). 12 It has been used to determine structural and microstructural bone changes at the distal radius, metacarpophalangeal (MCPs) joints, proximal interphalangeal (PIPs) joints, distal interphalangeal (DIPs) joints, and adjacent phalanges.13–17
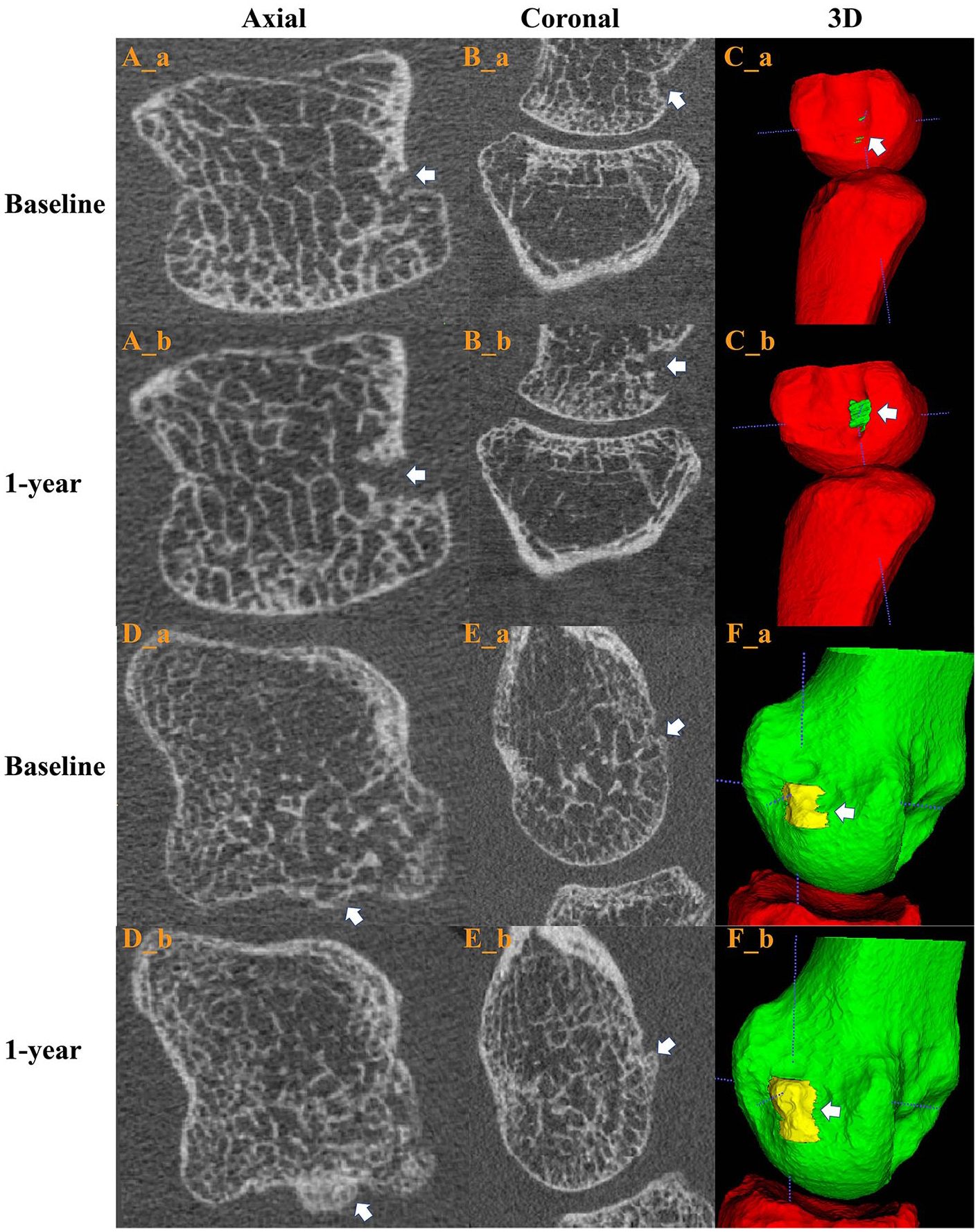
2D and 3D of HR-pQCT images of (A–C) fourth MCP in PsA patient showing erosion progression and (D–F) second MCP showing enthesophyte progression after 1 year. Erosion progression (A–C), baseline axial view (A_a), axial view after 1 year (A_b), baseline coronal view (B_a), coronal view after 1 year (B_b); baseline 3D erosion (C_a), 3D erosion after 1 year (C_b). Enthesophyte progression (D–F), baseline axial view (D_a), axial view after 1 year (D_b), baseline coronal view (E_a), coronal view after 1 year (E_b), baseline 3D enthesophyte (F_a), 3D enthesophyte after 1 year (F_b).
Erosion progression as an indicator of therapeutic efficacy
The sensitivity of radiography for detecting erosions is low. 18 Using HR-pQCT, quantitative measurement of erosion volume can be achieved, 9 using an indirect method to assess volume based on measurements of the width and depth of the erosions. 19 Bone deposition at the margin of erosions (osteosclerosis) with a decrease in erosion depth or width is a feature of either erosion repair or consolidation after effective control of inflammation.19,20 Tumor necrosis factor (TNF) inhibition has been shown, on HR-pQCT, to halt erosion progression after 1 year, but it does not prevent enthesiophyte formation in PsA patients. 21 Interleukin-17 (IL-17) inhibition (IL-17i) arrests the progression of bone erosion after 6 months. 16 Erosive damage typically results from synovitis and osteoclast formation within the synovial microenvironment, mediated by macrophage colony-stimulating factor and RANKL. These factors, which are essential for the growth and differentiation of osteoclasts, are enhanced by pro-inflammatory cytokines such as TNF and IL-17, which further promote osteoclast formation while inhibiting bone formation. Therefore, it is not surprising that targeting cytokines such as IL-23, IL-17, and TNF can slow down erosion progression in PsA.22–24 Progression of erosions on HR-pQCT is significantly less in patients who achieved sustained low disease activity, as judged by disease activity in psoriatic arthritis (DAPSA), after 5 years. 14
New bone formation
Much of the structural change seen in PsA seems to be driven by entheseal inflammation. 25 Similar to erosions, the sensitivity of HR-pQCT for detecting enthesiophytes is much higher than MRI with almost 90% of PsA patients showing enthesiophytes on HR-pQCT compared to only 30% on MRI. 16 HR-qCT has also shown that PsA-related inflammation leads to bone damage (erosions and enthesiophytes) and trabecular bone loss of the second and third metacarpal head in PsA patients. 15 The detection of entheseal new bone formation on HR-pQCT helps predict the development of PsA in psoriasis (PsO) patients.26–28 Utilizing an A1 integrated neural network based on joint shape (including erosions and bone spurs) can help differentiate between rheumatoid arthritis (RA), PsA, and healthy controls (HC). 29 Enthesiophytes are a dominant structural feature in established PsA, though largely absent in RA, 25 and are associated with poorer physical function. 28 Although IL-17i arrests the progression of both bone erosion and enthesiophyte formation after 6 months, 16 long-term efficacy in preventing structural progression is uncertain. Inflammation occurring at entheseal sites triggers a cascade of events, stimulating mesenchymal cells to proliferate and differentiate into osteoblasts responsible for bone formation. This differentiation process from mesenchymal cells to osteoblasts is mediated by inflammatory mediators, among which Prostaglandin E2 (PGE2), IL-17A, and IL-22 have been demonstrated to enhance local bone formation as part of a reparative response to damage.28,30,31 This response involves the expression of mediators such as Wnt proteins and bone morphogenetic proteins, which effectively induce osteoblast differentiation and new bone deposition. 32 Therefore, blocking IL-17A may play a crucial role in preventing new bone formation in PsA.
Joint space width (JSW)
Joint space narrowing (JSN) is more strongly associated with physical disability than erosion severity in PsA patients. 33 JSN, due to lysis of articular cartilage, negatively impacts functional status more than erosions, making it a valid target for treatment. 33 Reproducible, high-throughput, fully automated techniques for evaluating metacarpophalangeal joint (MCPJ) JSW using HR-pQCT have also been developed (Figure 2). 34 In vivo 3D quantification of joint space morphology may improve early detection of joint damage in rheumatological diseases. 35 JSW measurement was associated with Sharp/van der Heijde score in the second and third MCP joints 36 and was reliable in longitudinal studies. 37 HR-pQCT JSW parameters are more sensitive than radiographic JSW parameters for detecting joint damage in PsA patients. 13 Deterioration in JSN is positively associated with the degree of inflammation, with patients who received biologic disease-modifying antirheumatic drugs treatment exhibiting less pronounced JSN. 13

HR-pQCT images of (A) a normal joint and (B) a damaged joint with altered joint space indexes and angulation. (a) 3D views. (b–d) Cross-sectional views.
Summary
HR-pQCT allows for the evaluation of bones at the microstructural level, enabling the quantification of structural pathological features (such as erosion). HR-pQCT has been used to determine structural and microstructural bone changes involved in PsA. Low accessibility and a small field of view are the main limitations of HR-pQCT. Also, while quantification of erosion, enthesiophytes, and JSW holds promise as outcome measurements, one current challenge is the heterogeneity of methods used to identify and quantify these parameters. Fortunately, automatic erosion detection tools 38 and an open-source software tool are available for the identification and quantification of bone erosions. 39 A training tool has recently been made available to provide users with guidance on identifying pathological cortical breaks on HR-pQCT images for erosion analysis. 40 Also, highly sensitive acquisition process makes HR-pQCT prone to motion artifact, which may compromise accuracy. 41 HR-pQCT also cannot evaluate synovitis or osteitis.
Magnetic resonance imaging
High-resolution MRI can detect and quantify inflammatory synovitis, tenosynovitis, and osteitis as well as enthesitis, bone erosions, bone proliferation, and JSN. 42 Dynamic contrast-enhanced MRI enables the perfusion of synovial proliferation to be accurately quantified as a measure of synovial activity. 43
MRI-detected bone edema, erosion, and proliferation are more severe in disabling arthritic PsA, indicating that bone edema (osteitis) is often a precursor to erosion in PsA. 44 The combination of arthralgia and MRI-detected synovitis helps identify psoriatic patients at high risk of developing PsA. 45 It is recommended that clinical trials incorporate MRI inflammation as an inclusion criterion, use MRI inflammation as a key endpoint, and adopt validated MRI outcomes. 46
MRI and peripheral manifestations of PsA
The Outcome Measures in Rheumatology (OMERACT) Psoriatic Arthritis Magnetic Resonance has developed a scoring system, Psoriatic Arthritis Magnetic Resonance Imaging Score (PsAMRIS), which includes synovitis (score 0–3), flexor tenosynovitis (score 0–3), bone edema score (score 0–3), bone erosion (score 0–10), bone proliferation (score 0 or 1), and periarticular inflammation (score 0 or 1), for assessing inflammatory and structural changes at the MCP, PIP, and DIP joints of PsA patients. 47 This system is currently among the most effective tools for evaluating inflammatory and structural alterations in PsA patients PsA. 48 Patients with PsA receiving abatacept demonstrated improvements in PsAMRIS synovitis and tenosynovitis subscores. 49 Early disease interception by IL-17i in PsA improves skin symptoms, pain, total PsAMRIS, and synovitis subscore. 50 An MRI scoring system for heel enthesitis in PsA patients, OMERACT MRI heel enthesitis scoring system (HEMRIS), has been established, which categorizes enthesitis pathology on MRI into inflammatory and structural changes. 51
MRI and axial PsA
In total, 25%–70% of PsA patients present with axial disease, characterized by inflammation and post-inflammatory structural changes in the sacroiliac joints (SIJ) or the spine on imaging.52–54 MRI is the most sensitive means of diagnosing axSpA, offering higher resolution and visualization than radiography of active inflammation at the entheses, discovertebral, costovertebral, and facet joints as well as structural changes (erosions, fat metaplasia, syndesmophytes, and ankylosis). While MRI generally has lower resolution than CT and as a result slightly poorer sensitivity in identifying axial PsA (axPsA) structural lesions, it is the most sensitive technique in identifying the inflammatory component of sacroiliitis, spondylitis, and enthesitis.46,55,56 MRI, as a result, is the most sensitive technique for detecting early disease57,58 (Figure 3).
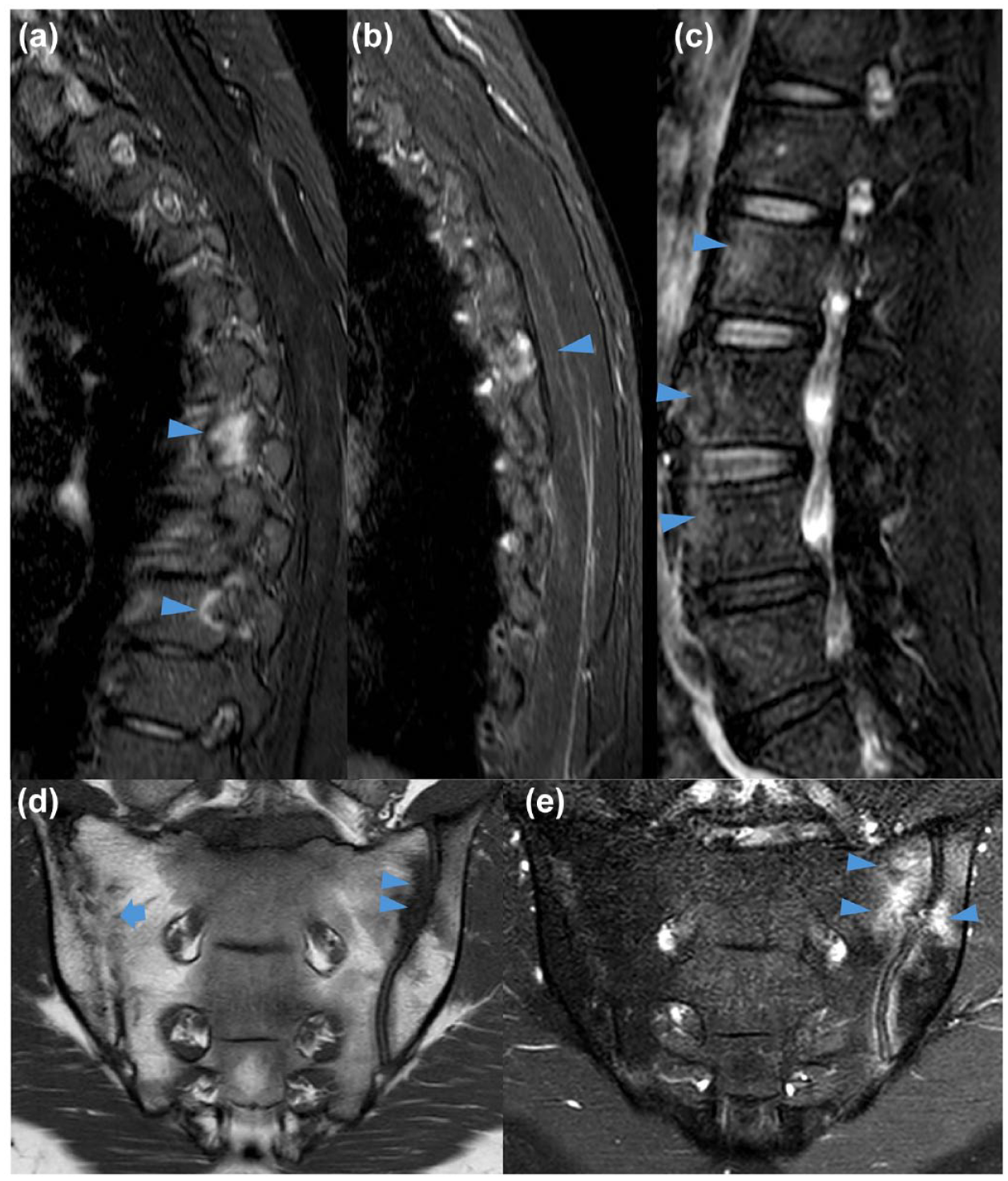
Spine MRI with sagittal Short Tau Inversion Recovery sequences T2-weighted fat-suppressed sagittal MR image showing active inflammation along the spine (a–c), erosive changes (d), and osteitis around the sacroiliac joint (e) in PsA patients.
There is no universally accepted consensus on the role of MRI in the diagnosis of axPsA. The most common clinical features at disease onset for PsA patients with the axial disease are as follows: oligoarthritis (30%), inflammatory back pain (IBP) (22%), enthesitis (18%), polyarthritis (16%), gluteal pain (8%), dactylitis (3%), and DIP symptoms (3%). 59 As IBP is not a sensitive indicator of early sacroiliitis, the more judicious use of MRI is recommended. 60 MRI-detected sacroiliitis in PsA patients is associated with restricted spinal movement and longer disease duration. 55 The first randomized controlled trial in PsA patients with axial disease demonstrated significant improvement in both clinical outcomes as well as Berlin MRI spine and SIJ scores over 4 months in patients treated with secukinumab compared to placebo. 61 The presence of inflammatory changes in the posterior elements of the spine and post-inflammatory changes such as corner fat lesions changes tended to be associated with a better clinical outcome in patients receiving secukinumab. 62
Whole-body MRI
Whole-body MRI (WB-MRI) can evaluate the systemic extent and burden of rheumatic disease,63,64 especially in areas that are difficult to assess clinically65,66 (Figure 4). Recently, the OMERACT MRI in Arthritis Working Group developed and validated a scoring system (OMERACT MRI Whole-Body Score for Inflammation in Peripheral Joints and Entheses) to gauge the systemic effect of SpA treatment.67,68 Significant reduction in peripheral and axial inflammation was shown with adalimumab and golimumab treatment.69,70 WB-MRI can evaluate the distribution and changes over time of inflammation in peripheral and axial sites and can distinguish between inflammatory pain arising from joints and entheses and non-inflammatory sources of pain. 71 Although large field-of-view imaging for WB-MRI is advantageous in being able to present a whole-body perspective, there is an inevitable trade-off in resolution with resultant limitations in evaluating erosions, JSN, ankylosis, synovial volume, and synovial perfusion.

WB-MRI series showing synovitis and enthesitis. (a) T1 SE and (b) T2W fat-suppressed MR from WB-MRI series showing synovitis of the left shoulder joint (arrowhead) with enthesitis of right greater trochanter (arrowhead); selected small cropped (c, d) T1W fat-suppressed post-contrast and (e, f) T2W fat-suppressed images from same whole-body MR examination as a previous image showing synovitis of the shoulder joint (arrowhead); enthesitis of lateral femoral condyle at lateral collateral ligament insertion (arrowhead); enthesitis of both greater trochanters (arrows); and enthesitis of anterolateral tibia at insertion of iliotibial band (long arrow).
Summary
MRI has undoubtedly greatly improved the early detection of sacroiliitis and other manifestations of axPSA as well as enabling an accurate recognition and assessment of peripheral PsA. The main factors currently limiting the more widespread usage of MRI are cost and availability. That said, many of the numerous advantages provided by MRI in the assessment of PsA can potentially be provided by ldCT examination. Besides, bone marrow changes observed in MRI may lack specificity for axSpA and can complicate interpretation.72–74 CT can be a reasonably sensitive and highly specific alternative to MRI and can be a useful added investigation if MRI appearances are equivocal. 56
Low-dose CT
The standard CT reconstruction mechanism has “back-filtered projection.” Recently, CT manufacturers have introduced “iterative reconstruction,” which includes artificial neural networks to reduce noise while maintaining spatial resolution. Low-dose CT techniques enable the acquisition of CT data at radiation doses comparable to radiography. SIJ ldCT can be obtained with an effective radiation dose of 0.11 mSv, equivalent to that of a chest radiograph. 56 Image quality is not as good as standard CT examination, being more susceptible to noise, though is still perfectly sufficient for most diagnostic purposes.
ldCT enables high-resolution depiction of bone erosions, sclerosis, ankylosis, and joint space narrow and is considered the gold standard for assessing structural damage. 75 Comparing radiography, ldCT, and MRI for identifying sacroiliitis in axSpA, ldCT had higher sensitivity than radiography but lower sensitivity than MRI though had higher specificity than both radiography and MRI. 70 There is a growing trend to replace radiography with ldCT or MRI for diagnosing sacroiliitis in axSpA.
Erosions and ankylosis are the most specific imaging findings for the diagnosis of sacroiliitis (Figure 5). However, erosions located in the peripheral aspects of the joint are less specific while sclerosis is a nonspecific finding. 76 While MRI is the most sensitive imaging technique due to its ability to detect osteitis, it lacks specificity compared to ldCT as it cannot demonstrate small or medium-sized erosions as well as ldCT. 56 Advances in dual-energy CT may potentially overcome these limitations, with the potential to detect bone marrow edema, though dual-energy CT currently does not lend itself to ldCT imaging.
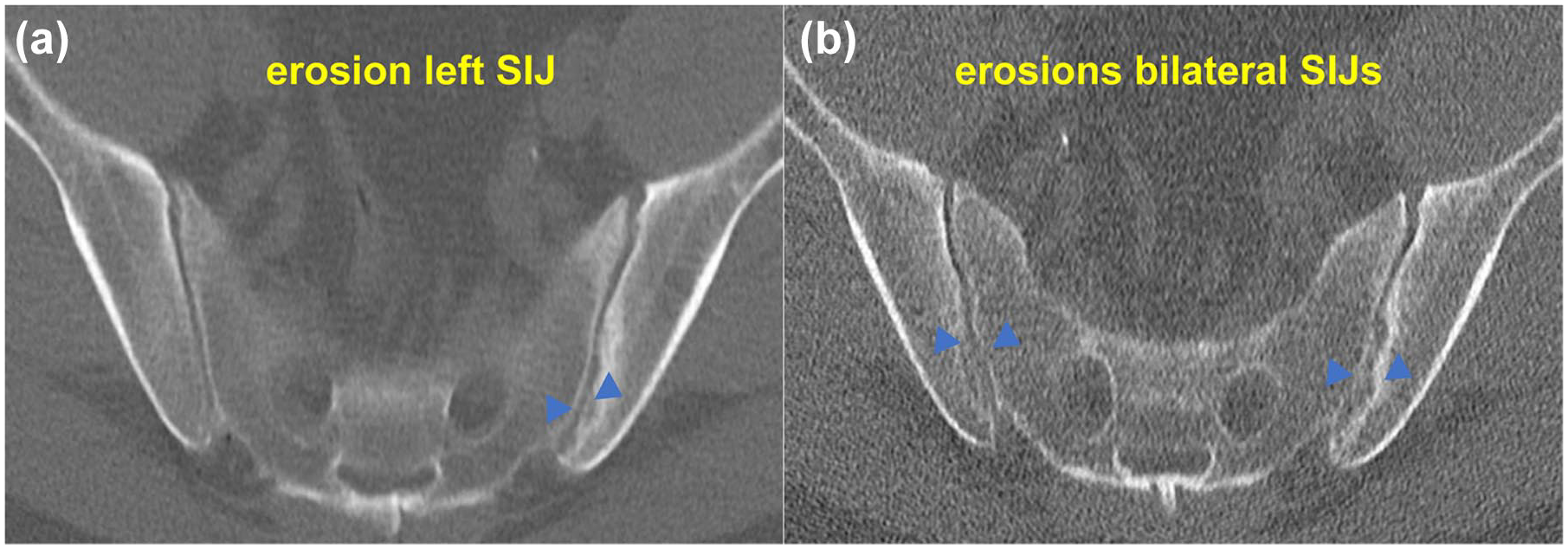
Low-dose CT image showing structural erosive change unilaterally (a) and bilaterally (b) in PsA patients.
By reducing the radiation dose of CT imaging to 4 mSv, which is an acceptable level tolerable for imaging of the whole spine, it becomes possible to study structural lesions throughout the spine. ldCT was recently used to study syndesmophyte progression in AS patients. 77 In axSpA, the ldCT spine is more sensitive than radiography for detecting structural lesions and disease progression mainly because it can more clearly visualize the thoracic spine, which is obscured by rib superposition on radiography, and it can more effectively detect facet joint ankylosis and as well as costovertebral and costotransverse arthropathy. 73 Including assessment of facet joints is particularly important as ankylosis leads to functional impairment.77,78 Facet joints in the upper thoracic region cannot, however, be evaluated fully on ldCT due to poor signal-to-noise issues and may appear spuriously ankylosed. To date, there have been no ldCT-based studies examining structural damage to the spine and SIJ in PsA.
Summary
ldCT demonstrates higher sensitivity and specificity compared to radiography and higher specificity compared to MRI. Further exploration is required to determine the usefulness of ldCT in PsA disease identification and progression.
Ultrasound
As a rapidly iterative imaging technique, US is particularly suitable for evaluating the skin and musculoskeletal areas in PsA with high resolution, such as joints, enthesis, tendons, skin, and nails.79,80 It is currently considered to be a constituent component of the overall assessment of PsA patients, as it can identify pathological blood flow and localized active inflammation (Figure 6). With higher sensitivity in detecting intra-articular and extra-articular inflammation and damage than clinical examination, US does not require contrast agents and does not involve ionizing radiation. 81 US is a cost-effective and non-inferior method for identifying and characterizing early PsA sacroiliitis and has good sensitivity for the detection of inflammatory changes.82–85

US shows severe synovial hypertrophy (a, b), acute tenosynovitis of the peroneal tendons (c, d), and active enthesitis with chronic structural bone damage (e, f). (a, b) US of the ring finger (longitudinal scan) shows (a) severe synovial hypertrophy (*) of the proximal interphalangeal joint, with (b) active hyperemia indicating active arthritis. There is a bone erosion (#) at the base of the middle phalanx. (c, d) US of the lateral side of the ankle (transverse scan) shows (a) a tendon sheath synovitis and fluid (*) with (b) increased power Doppler signal around the peroneal longus and peroneal brevis tendons. This indicates acute tenosynovitis of the peroneal tendons. (e, f) US of the Achilles entheseal insertion (longitudinal scan) shows (a) an increased thickness of the tendon insertion (*) and (b) increased power Doppler signal and bone erosion (#) of the calcaneal enthesis. These findings are compatible with active enthesitis superimposed on chronic structural bone damage.
Recently, rheumatologists have become increasingly interested in using the US for objective assessment of PsA diagnosis and treatment. 86 US can detect subclinical enthesopathy and nail disease in PsO patients with increased PsA risk or those without musculoskeletal disorders, and identify subclinical synovitis in early PsA.79,87,88 US-detected synovio-enthesitis had a significant association with subclinical PsA, especially in the lower limbs and US screening may potentially improve the accuracy of PsA detection.89–91 For PsA management, research concerning the function of the US also prioritizes evaluating disease activity, which encompasses the creation of combined activity indices. 86
US is a sensitive method for detecting peripheral PsA inflammation and structural lesions. 92 US studies have shown that dactylitis can involve various anatomical structures, including joint synovitis, soft tissue edema, extensor peritendinitis, flexor tenosynovitis, and thickened and hypervascular flexor pulleys.93–95 US is more effective in correctly identifying enthesitis than clinical examination and may have practical value in detecting enthesitis in patients with PsA and concomitant fibromyalgia.96–99 US is also employed as a criterion to assess the response to treatment. Secukinumab can rapidly reduce US-measured synovitis, suggesting that IL-17i has a direct effect on synovial tissue in patients with PsA. 100 US inflammation score after apremilast and adalimumab therapy showed significant improvement in tenosynovitis and entheses inflammatory thickness compared to methotrexate treatment.101–103
Summary
The US is a reliable and powerful tool that can assess joints, tendons, entheses, skin, and nails in PsA patients, reflecting morphological structural changes and inflammatory activity. However, US, due to the acoustic window, certain areas of a joint are difficult to visualize, such as the radial or ulnar quadrants of the third MCP. Besides, US detection of erosion has false positives and false negatives, as the limited US resolution is unable to detect cortical lesions with a width less than 2 mm and one may falsely identify irregular surface features as bone erosions. Due to its inability to penetrate bone, it has a lower sensitivity for detecting erosions compared to CT and MRI and cannot detect osteitis. 104
Positron emission tomography-computed tomography
PET-CT is a promising alternative to WB-MRI for whole-body disease activity detection, as it provides a metabolic as well as an anatomic assessment.105,106 Radioactive tracer accumulation enables metabolic activity to be quantified. One recent study showed that maximum standardized uptake value scores had a good correlation with inflammatory SpA-related sacroiliitis.107,108 The frequency of abnormal SIJ uptake on 18F-NaF PET was higher than that of inflammatory and structural sacroiliitis on MRI and CT.107,108
PET-CT imaging has the potential for comprehending the source of pain in inflammatory arthritis as well as providing a minimally invasive and objective means of assessing severity and response to treatment in RA and PsA. For example, in a PsA patient, a good correlation was found between sites of arthritic pain and regions of increased fluorodeoxyglucose (FDG) uptake. 109 Increased [18F]fluoride uptake, which is a marker of bone deposition, was observed in the entire distal phalanx including the periosteum and entheses, supporting the concept of an integrated nail and joint apparatus leading to a wide area of abnormal bone metabolism in PsA patients. 110 Another 18F-NaF PET-CT study showed new bone formation can be observed at peripheral joints and entheses and in the axial skeleton in PsA patients, most of whom did not exhibit clinical symptoms, as a marker of PsA disease activity. 111
18F-FDG uptake, which is a measure of glucose metabolism and hence inflammation, in inflamed joints was significantly increased compared to non-inflamed joints in PsA patients. 112 18 F-FDG uptake patterns corresponded to areas of synovitis, tenosynovitis, enthesitis, and nail dystrophy. 112 PET-CT examinations are nearly always whole-body examinations. 18F-FDG PET can show a moderate correlation with DAPSA assessment. 113
PET-CT imaging has the potential to deploy different radioisotopes as markers of different disease processes, particularly if there is quick access to a nearby cyclotron as the half-life of many radioisotopes is short. Fibroblast activation protein (FAP) PET-CT imaging may improve risk assessment for joint damage in PsA patients and provide new options for treat-to-target strategies. 114 Similarly, 68Ga-FAPI-04 PET-CT-detected fibroblast activity may be a potential biomarker for identifying high-risk patients transitioning to PsA. 115
Summary
PET/CT imaging has a wide potential, particularly with the use of biomarkers to recognize different types of metabolic activity. It is currently limited by relatively low spatial resolution as well as low signal issues, limited availability, and high cost. The radiation dose for PET/CT examination is not low, limiting its more widespread usage, particularly in younger patients and those in whom repeated examination is considered necessary.
Other novel imaging modalities for PsA: Fluorescence-optical imaging
Fluorescence-optical imaging has recently been introduced as an additional imaging technique for diagnosing systemic inflammatory joint diseases, revealing microcirculatory disorders in the hands. Inflammation leads to vasodilation, increased blood flow, increased capillary permeability, and interstitial diffusion at affected sites with the injected fluorescent dye accumulating in areas of inflammation, leading to signal enhancement. 116 Previous studies have shown the suitability of FOI for monitoring treatment in early inflammatory arthritis, and it is also applicable for detecting hand inflammatory skin changes in PsA.116–118 FOI is a rapid, reliable technique that is comparable to 1.5 T MRI and US for detecting synovitis and tenosynovitis. 119 FOI could potentially be a useful tool for identifying patients with mild hand and wrist synovial inflammation, which could be applied in clinical practice to detect early synovitis with greater confidence. 120 FOI may also help in identifying nail involvement and DIP joint arthritis in patients with PsO and/or PsA. 119 Nail region fluorescence has high specificity for PsA, which might be very helpful in distinguishing seronegative arthritis. 118 FOI is useful for detecting early hand inflammation in suspected PsA patients, and combining clinical manifestations with US ± FOI results might increase accuracy in confirming a diagnosis of PsA. 121 Using FOI, most psoriatic patients with or without PsA could be correctly classified based on the physician’s diagnosis with the additional benefit of being able to detect dermal and subcutaneous skin inflammation. 116 FOI can also identify early PsA and distinguish between acute and chronic disease, potentially acting as a triage to select psoriatic patients who might benefit from additional treatment, 122 as well as being a good early imaging biomarker to identify transition of psoriatic patients to PsA. 123 However, the assessment of FOI was limited to the hands, which is a limitation of it. 123
Multispectral optoacoustic tomography
Multispectral optoacoustic tomography (MSOT) combines the high contrast of optical imaging and low cost, versatility, and high spatial resolution of US imaging, achieving high sensitivity and high-resolution imaging of biological tissues. 124 MSOT is capable of performing molecular and functional imaging of joints while collecting anatomical data. 125 Unlike the US, MSOT does not rely on velocity and direction to detect blood flow, but measures oxygenated or de-oxygenated hemoglobin directly and improves accuracy by detecting changes in these constituents at a tissue level.126–129 One recent research reported the application of MSOT in the diagnosis of PsA. 130 With MSOT, it was possible to visualize increased vascularity around finger joints without the use of contrast agents. The degree of oxygen saturation in synovial structures was directly proportional to the degree of inflammation. 130 Besides, in patients without radiographic signs of PsA, an increase in optoacoustic signal intensity could also be detected, indicating increased blood flow and showing how MSOT may be helpful in the early diagnosis of PsA. 130 Another study used MSOT to help distinguish joints with and without inflammation as an adjunct to clinical and US assessment. 131
MSOT could be highly effective in identifying the earliest changes associated with enthesitis. 132 However, further studies are required on PsA patients across different stages of disease activity.
Discussion
Imaging is fundamental for the direct observation of axial manifestations, making it a valuable and promising assessment tool, as recognized by GRAPPA members. 133 While conventional radiography and CT are useful for detecting structural changes, they fall short in assessing active inflammation. MRI stands out as a superior modality, capable of providing detailed visualization of inflammation in both the SIJs and the spine in patients with PsA. 134 This makes MRI particularly advantageous for diagnosing and monitoring the disease, offering insights that are critical for effective patient management. Indeed, MRI has been used as an outcome measure in the MAXIMISE (Managing AXIal Manifestations in psorIatic arthritis with SEcukinumab) trial. PsA patients who received secukinumab achieved substantial improvements in clinical and Berlin MRI scores at the spine and SIJ by week 12, and these gains were maintained up to week 52. 135 Another analysis of the MAXIMISE trial identified additional inflammatory and post-inflammatory changes indicative of axPsA and showed a trend toward improved clinical outcomes for patients treated with secukinumab through spinal MRI. 136 Furthermore, the STAR study will prospectively assess efficacy outcomes in PsA patients with MRI-confirmed axial inflammation, aiming at evaluating the ability of guselkumab to reduce both axial symptoms and inflammation in patients with active PsA, including axial inflammation, as confirmed by MRI. 137 US exhibits high sensitivity in detecting, characterizing, and monitoring pathologies related to PsA, including synovitis, enthesitis, tenosynovitis, and dactylitis. 138 A consensus-based US scoring system for dactylitis has been developed, showing acceptable interobserver reliability and excellent intraobserver reliability. 139 In addition, small entheses in the fingers were identified and histologically validated. 139 Doppler flow US, when combined with CT images, can also effectively visualize abnormal blood flow in the SIJs and other joints affected by inflammatory arthritis. 140 A study showed that patients with PsA treated with secukinumab, including those who switched from placebo, had consistent improvements in both clinically and US-assessed enthesitis and synovitis, with sustained clinical benefits through week 52. 141
Conclusion
Imaging plays a crucial role in the diagnosis of peripheral and axial lesions in PsA. Early and accurate identification of peripheral arthritis, sacroiliitis, enthesitis, synovitis, and tenosynovitis impact diagnosis and treatment. Significant progress has been made in improving the reliability and accuracy of disease detection through different imaging modalities. Radiography is the standard method for structural assessment of joint disease in PsA though is limited in being a two-dimensional projection technique with only modest sensitivity and responsiveness. Technological advancements in high-resolution imaging fields (including HR-pQCT, MRI, US, and ldCT), as well as functional and molecular-based imaging techniques (such as PET-CT, FOI, and MSOT), have the potential to explore in much greater depth the pathophysiologic processes accompanying PsA which, in turn, will lead to earlier, more specific diagnoses, as well as better prediction and monitoring of treatment response. However, it is important to acknowledge the practical applications and current limitations of these new technologies. While they hold promise for advancing PsA management, challenges such as high costs, limited availability, and the need for specialized expertise must be addressed to fully integrate these methods into clinical practice.
