Abstract
Psoriatic arthritis (PsA) is an inflammatory joint and entheseal disease associated with significant personal and public health burden. PsA has a prevalence of up to 1%, affecting ~20% of people suffering with psoriasis. PsA is frequently accompanied by metabolic syndrome (MetS), and both conditions are characterised by a chronic pro-inflammatory state, with several key cytokines in PsA (interleukin (IL)-17 and IL-23) also elevated in those with MetS. This narrative review aims to provide an update on MetS in PsA, focusing on its prevalence, pathogenesis, prognosis, treatment interactions and future therapeutic options. MetS is particularly prevalent in PsA compared to other inflammatory arthritides. Cohort studies indicate a higher risk of PsA in individuals with obesity, while Mendelian randomization studies link childhood obesity, insulin resistance, and dyslipidaemia to PsA. Weight loss interventions have been shown to reduce disease activity in PsA. Additionally, MetS negatively impacts the efficacy of tumour necrosis factor inhibitor (TNFi) drugs in treating PsA. Drugs given for PsA may also affect the conditions constituting MetS. Leflunomide has been shown to reduce body weight but also increase blood pressure. TNFi drugs lead to weight gain but reduce cardiovascular risk. Janus kinase inhibitors increase lipid levels and cardiovascular risk among high-risk groups. Anti-IL-17 and anti-IL-12/IL-23 drugs may cause a short-term increase in cardiovascular risk, although the long-term effects have yet to be established. Weight loss represents an unexplored avenue for disease modification in PsA, alongside a plethora of general health benefits. Dietary and exercise modifications are the cornerstone of weight management but vary substantially across individuals. Novel therapies to treat weight loss, such as glucagon-like peptide 1 agonists and sodium–glucose cotransporter 2 inhibitors, may prove useful alongside disease-modifying therapies for those with PsA and MetS and should be investigated as potential therapeutic adjuncts.
Plain language summary
Psoriatic arthritis (PsA) is a condition that causes joint pain and swelling, which can lead to permanent damage to the joints over time. This condition affects 1 in 100 people and 1 in 5 people with psoriasis. Metabolic syndrome (MetS) is a condition where a person has a combination of high body fat, high blood pressure, high blood sugar and/or high cholesterol. MetS is more common in someone with PsA. If a person has MetS, they are more likely to get PsA. If a person with PsA has MetS, they have worse joint problems and their joint problems do not improve as much with certain treatments. Many of the drugs given for PsA can affect MetS. This can include increasing or decreasing body weight, increasing blood pressure and increasing or decreasing the risk of having a heart attack or stroke. Different medications affect these risks in different ways. Weight loss helps people with PsA improve their joint problems. However, some people find losing weight harder than others. Medications that can help people lose weight, could be useful to improve joint problems in PsA. Future studies should see if these medications can be useful in people with PsA and MetS.
Keywords
Introduction
Psoriatic arthritis (PsA) is a long-term inflammatory disease affecting the joints and entheses. PsA affects 20% of people who have psoriasis (PsO). 1 The overall prevalence of PsA is estimated to be between 0.1% and 1%, and uncontrolled disease can cause permanent joint damage and deformity.1,2 PsA, particularly active PsA, is associated with poorer functional status, reduced employment (20%–50% unemployment rate) and greater productivity impairment at work.3–7
PsA is underpinned by a chronic inflammatory state. Obesity and metabolic syndrome (MetS) are also characterised by a chronic low-grade inflammatory state, due to the production of pro-inflammatory cytokines by visceral fat tissue. 8 Large cohort studies have linked obesity and MetS to an increased incidence of psoriatic disease, supported by strong shared pathophysiological mechanisms (see Table 1).2,8–18
AMPK, AMP-activated protein kinase; IFN-γ, interferon-gamma; JAK-STAT, Janus kinase-signal transducer and activator; MAPK, mitogen-activated protein kinase; NF-κB, nuclear factor kappa B; PI3K, phosphatidylinositol 3-kinase; PsA, psoriatic arthritis; PsO, psoriasis; TGF-ß, transforming growth factor-β; TNF-α, tumour necrosis factor alpha.
Shared pathways between PsA/PsO and MetS highlighted in bold.
MetS is a condition characterised by a combination of visceral obesity, hyperlipidaemia, hypertension and/or dysglycaemia, as classified by the 2009 International Diabetes Federation (IDF) Task Force Joint Interim Statement or the 2001 National Cholesterol Education Program Adult Treatment Panel III (ATPIII) criteria (see Box 1).8,19,20 Obesity is a driving factor for this condition, with increased adiposity associated with reduced insulin sensitivity, deranged lipid profile and low-grade, chronic systemic inflammation. The aetiology for obesity is complex and multi-factorial, influenced by genetic and environmental factors (e.g. food environment and sleep quality). 8
Systemic inflammation secondary to MetS has been proposed as part of the mechanism driving disease activity in different inflammatory arthritides, including rheumatoid arthritis (RA) and PsA. MetS is particularly prevalent in PsA, with concomitant MetS over 1.6 times more likely when compared to RA (OR 1.66 [95% CI 1.54–1.79]) or PsO (OR 1.61 [95% CI 1.49–1.73]). 21 Obesity is a recognised risk factor for PsA, and weight loss has been shown to independently reduce disease activity in PsA. 22
By 2030, 57.8% of the global population is predicted to be overweight or obese (body mass index (BMI) ⩾25). 8 A BMI of 30 or greater has been shown to have at least a 50% increase in all-cause mortality in otherwise healthy Europeans at 5 years, with an increased risk of ischaemic heart disease, cerebrovascular accident and malignancy. 23 Treatment of obesity and MetS is essential to reduce healthcare costs and excess mortality and morbidity. 8
This narrative review aims to provide an update on MetS in PsA, focusing on its prevalence, pathogenesis, prognosis, treatment interactions and future therapeutic options. We focus specifically on PsA rather than psoriatic disease as a whole. Although we cite some evidence on the relationship between PsO and MetS, this is not a comprehensive review of PsO and MetS.
Prevalence of MetS in PsA
MetS is prevalent in people with PsA, with estimates ranging from 29% to 46%, much higher than the global prevalence of MetS in the general population of 12.5%–31.4% (see Table 2).21,24,25 Comorbidities in PsA tend to be dominated by the cardiometabolic components of MetS, with a meta-analysis by Gupta et al. finding hypertension (34%), obesity (27%) and hyperlipidaemia (24%) to be prevalent in PsA. Interestingly, diabetes mellitus was less prevalent at 12.9%, although this could be related to other factors such as participant age. Obesity is also more common in PsO, particularly in those with severe disease. 26 Cardiovascular disease is common in PsA, with a prevalence of 19.4%. 24 Urruticoechea-Arana et al. found a higher proportion of people with PsA and MetS at 23.5–62.9% during their literature review. Analysing their own cohort, they also found that 30.6% met the definition for MetS and a high proportion had cardiometabolic derangement, with around two-thirds having hypertension, two-fifths having hyperglycaemia and one-third having hypertriglyceridaemia. 27 The discrepancy between the prevalence of these disorders in these studies is likely to be secondary to demographic/clinical factors such as age, biological gender, diet composition, exercise, sleep, smoking and alcohol intake.
HDL, high-density lipoprotein; MetS, metabolic syndrome; PsA, psoriatic arthritis.
Grey shadings to show no result given.
PsA has a higher prevalence of MetS when compared to other autoimmune disorders, such as RA, PsO and axial spondylarthritis (axSpA), despite all being characterised by chronic inflammation.21,24,28 For example, a meta-analysis by Loganathan et al. found a higher prevalence of MetS in PsA (46%) when compared to RA (31%) and PsO (34%). Interestingly, there were similar incidences for the components of MetS (obesity, dyslipidaemia, insulin resistance, hypertension) across all three diseases, suggesting that participants with PsA tend towards multimorbidity compared to those with RA or PsO. 21 This same picture emerges in axSpA, with higher levels of hypertension (34% vs 23%), hyperlipidaemia (24% vs 17%), obesity (27% vs 14%), diabetes mellitus (12.9% vs 6%) and cardiovascular disease (19.4% vs 12%) in PsA when compared to axSpA, from two separate meta-analyses.24,28 Despite this higher prevalence of cardiometabolic risk factors, major adverse cardiovascular events (including angina, myocardial infarction, stroke) and other significant cardiovascular diseases (heart failure, peripheral vascular disease) were found to be of a similar prevalence between PsA and axSpA.24,28 Non-alcoholic fatty liver disease (NAFLD) is more common in PsA, particularly with concomitant MetS. 6
When specifically examining body composition in PsA, higher proportions of subcutaneous, visceral, and abdominal adiposity, alongside reduced muscle mass, have been reported.29,30 Visceral adipose tissue, in particular, is recognised as pro-inflammatory and is associated with increased cardiovascular risk. 8
The prevalence of MetS in PsA, compared to similar autoimmune inflammatory disorders, suggests MetS plays a significant role in the pathophysiology of the disease.
Influence of MetS on psoriatic disease risk
The pathophysiology of PsA is influenced by a combination of genetic (such as HLA-B and -C genes) and environmental factors (biomechanical stress, obesity and smoking). 2 Several cytokines and immune factors are involved in the pathophysiology of PsA, including interleukin (IL)-17, IL-23, IL-12, tumour necrosis factor-alpha (TNFα) and interferon-gamma (IFNγ).2,15,16 Damage-associated molecular patterns and pathogen-associated molecular patterns lead to macrophage and dendritic cell activation, with downstream CD8 cell activation and the production of IL-23 and other cytokines, including TNFα and IFNα.2,15 IL-23 stimulates the clonal expansion of Th17 lymphocytes, which produce IL-17. 15 IL-17 and other cytokines lead to cell recruitment and activation of inflammatory (M1) macrophages, keratinocytes, fibroblasts and chondrocytes, leading to synovial hyperplasia and neovascularisation; this eventually leads to joint destruction and deformity if left unchecked. 2 Obesity-mediated inflammation is driven by visceral adipose tissue. 8 Unlike its subcutaneous counterpart, this tissue releases pro-inflammatory adipokines, including leptin, TNFα, IL-6 and IL-18 and also suppresses the release of anti-inflammatory adipokines from subcutaneous fat.8,12
The potential mechanisms underpinning the link between obesity and psoriatic disease are explored in Figure 1.2,8,12–18,31 Leptin drives monocyte and granulocyte activation and, in turn, increases the number of M1 macrophages. Leptin also affects T lymphocyte differentiation, favouring Th1 CD4 cells and downregulating regulatory T (Treg) cells. Finally, leptin increases the release of pro-inflammatory cytokines, including IL-6, IL-12, IL-17 and TNFα. 31 TNFα is a proinflammatory cytokine highly implicated in the pathophysiology of obesity and PsA/PsO.2,12 TNFα increases the proportion of M1 macrophages, creating a pro-inflammatory state. 15
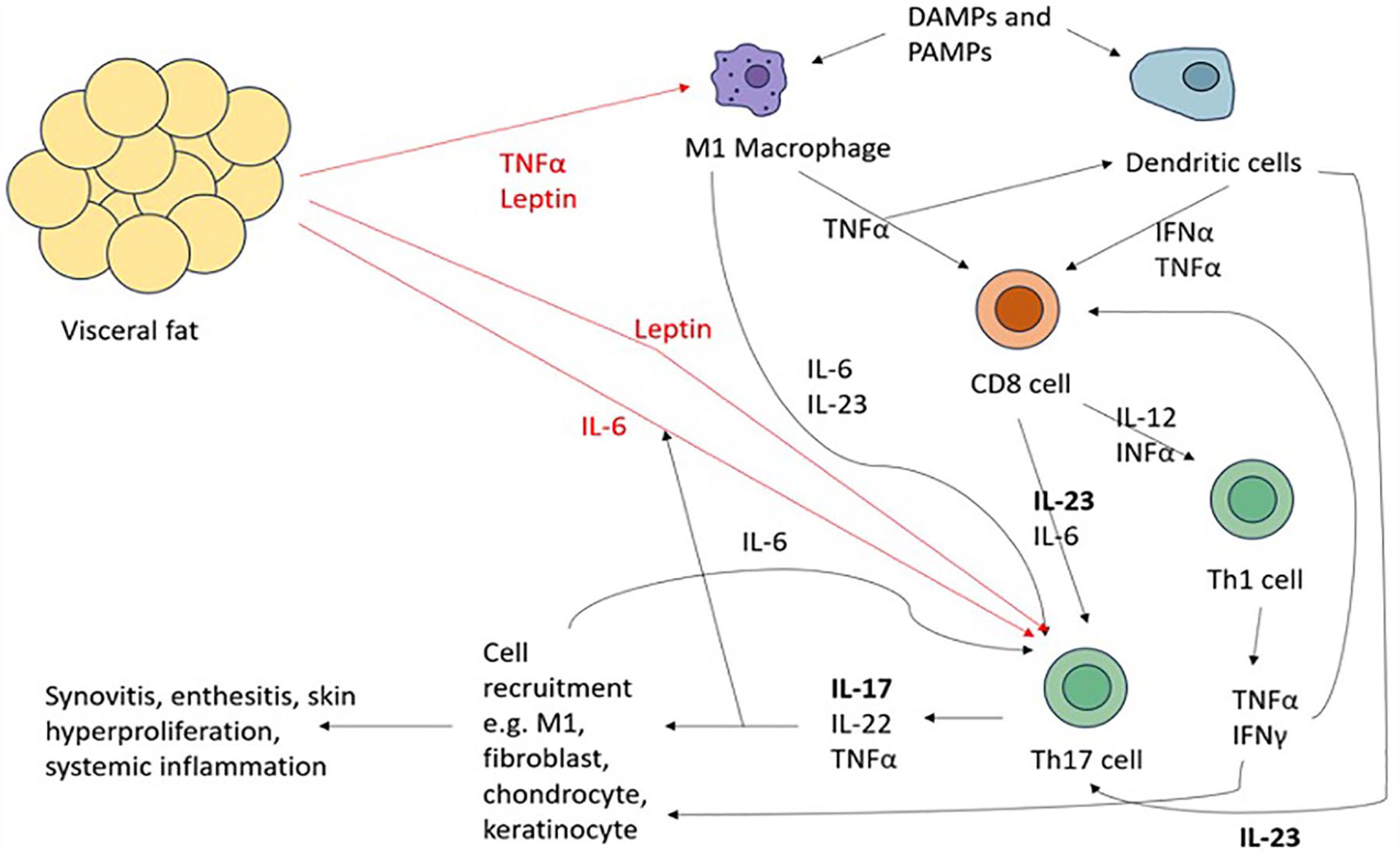
IL-17 and IL-23 are recognised as important drivers of psoriatic disease, with both cytokines recognised to be elevated in people who have obesity compared to controls.2,32,33 IL-23 (and, to a lesser extent, IL-6 and TNFα) stimulate the differentiation of T cells into Th17 cells. Th17 cells produce IL-17, leading to a cascade of proinflammatory mechanisms resulting in skin and joint disease.2,15,18 As mentioned above, IL-6 has been shown to stimulate the production of Th17 cells and, therefore, IL-17. Furthermore, adipocyte IL-6 has been shown in vitro to be upregulated by IL-17, proposing a potential positive feedback mechanism. 18
Beyond the cellular and immune similarities between both disorders, biomechanical overload due to excess body weight could be another potential disease mechanism, secondary to entheseal microtrauma. 2
The causative role of obesity in the development of psoriatic disease has been examined by Maglio et al. 9 In this study, authors compared a group of participants without PsO/PsA who underwent bariatric surgery (n = 1991) versus a similar group who did not (n = 2018). Both groups were followed up over a mean of 18 years to compare the incidence of several outcomes, including the development of PsO and PsA. The results show that bariatric surgery was statistically significantly associated with reduced PsO (71 vs 103 events, adjusted HR 0.65 [95% CI: 0.47–0.89]) but non-significantly reduced for PsA (20 vs 26, adjusted HR 0.71 [95% CI: 0.38–1.33]). However, the number who developed PsA was small, meaning the statistical power may not have been sufficient. The chronicity of a person’s obesity seems to play a significant role, with a longer duration of obesity independently associated with a higher risk of PsO. 9 A similar study by Egeberg et al., in a cohort of people receiving gastric bypass (n = 12,364) and gastric banding (n = 1071), demonstrated a statistically significant reduced risk of PsO, severe PsO and PsA with gastric bypass but not gastric banding in patients post-surgery when compared to pre-surgery. The sample size for gastric banding was markedly smaller when compared to gastric bypass, which may explain why gastric banding was not found to be statistically significant. 34 In those with a history of PsO, BMI ⩾25 kg/m2 has been associated with an increased risk of developing PsA, with a reduction of BMI conferring a reduction in PsA risk. 35
Coupled with the increased prevalence of MetS in PsA, these studies suggest that the incidence of psoriatic disease is likely to have some association with MetS. In addition, large cohort studies have linked obesity in PsO patients and the general population with an increasing incidence of PsA, with obesity tending to predate the diagnosis of PsA.10,11,36 This may be due to the association of obesity with IL-17/IL-23, a key pathogenic mechanism in PsO/PsA.2,18
Several mechanisms could underlie an increased risk of PsA in MetS. As described above, the low-level inflammatory state associated with obesity is thought to increase the systemic inflammatory burden. 8 As similar immune mechanisms underpin obesity-mediated inflammation and psoriatic disease, stimulation via these pathways may contribute to the development of PsA/PsO.2,8 Mendelian randomization studies have suggested childhood obesity to be independently associated with an increased risk of PsA, potentially despite normalising BMI in adulthood. 37 Insulin resistance and impaired glycaemic control have also been independently associated with PsA in Mendelian randomization studies. 38 Lipids have been demonstrated to have a role in PsO, with Mendelian randomization studies showing genetically mimicked inhibition of proprotein convertase subtilisin/kexin type 9 (PCSK9) being associated with a reduction in PsO (this association is yet to be tested in PsA). 39 These disease processes all characterise the cardiometabolic dysfunction associated with MetS and may explain why those with multiple cardiometabolic morbidities seem particularly at risk of PsA when compared to diseases like RA. 21
Influence of MetS on PsA prognosis
Obesity is understood to alter the immunological mechanisms underpinning disease response. In a study by Bapat et al. in mouse models of atopic dermatitis, obesity was shown to alter the composition of T-cell populations. Obesity heightened the inflammatory response, with obese mice demonstrating a significantly greater number of CD4 T cells, regulatory T cells and CD8 T cells when compared to lean mice. Further analysis showed a moderate increase in Th2 cytokines (IL-4, IL-13) and a proportionally larger increase in Th17 cytokines (IL-17A, IL-17F). Human research has also demonstrated similar findings in atopic disease. In this same study, an IL-4/IL-13 antagonist was found to improve disease in lean mice but worsen disease in obese mice. On flow cytometry, after IL-4/IL-13 blockade, IL-4, IL-13, IL-17A and IL-17F were all reduced in the lean mice, but all increased or static in the obese mice. It was hypothesised by Bapat et al. that due to the alternate inflammatory pathway in obese mice, blockade of Th2 cytokines only exacerbated the already active alternative pathway, leading to a worsening of the disease. 40
The mechanism underpinning this is thought to be related to reduced Th2-cell PPAR-gamma activity in obese mice. When flow cytometry was performed on a group of PPAR-gamma knock-out mice, they demonstrated a similar picture to the obese mice (generally increased inflammatory response with no Th2 predominance), with worsening reproduced with IL-4/IL-13 therapy in the PPAR-gamma knock-out mice. Finally, treatment with PPAR-gamma agonists (rosiglitazone) in obese mice was associated with a significant improvement in atopic dermatitis, although this was not reproduced in PPAR-gamma knock-out mice. On flow cytometry, treatment with rosiglitazone in the obese mice led to a picture resembling the lean mice (Th2 predominance), with these mice now responding positively to IL-4/IL-13 antagonism. 40
The above study highlights that obesity can affect the immunopathogenic response to disease, including the response to therapy. Obesity is also thought to have disease effects in PsA, including disease severity and response to biological therapy.22,41 IL-17 is heavily implicated as a key antibody driving the pathophysiological process in PsA. 2 If obesity is a key driver for the proliferation of Th17 cells and the production of IL-17, IL-23 and TNFα, this could explain the mechanism behind obesity-related disease onset or worsening.2,15,18 Understanding how these immune processes interact in PsA with MetS creates the potential for new drug targets to help both conditions.
The reduction of BMI in people with PsA has been associated with an improvement in disease activity. In a 6-month, interventional open-label trial, Klingberg et al. examined the effect of a very low-energy diet on disease activity, with weight loss leading to statistically significant improvements in several disease activity and quality of life measures. Although the study design is limited by its open-label approach and lack of a comparator group, the results are encouraging. 22 These improvements were maintained at 24-month follow-up. 42
Obesity has been reported to reduce the effect of disease-modifying therapy in PsA, particularly tumour necrosis factor inhibitor (TNFi) drugs. TNFi efficacy is adversely associated with MetS, obesity and hypertension, specifically in the achievement of remission and overall drug persistence.41,43–46 Furthermore, weight loss interventions delivered alongside TNFi have been demonstrated to increase the likelihood of achieving a minimal disease activity (MDA) state, with higher levels of weight loss correlating to higher chances of MDA. 47 The exact mechanism underlying this phenomenon in PsA is unclear, although it is speculated to be related to the production of TNFα by adipose tissue.2,41,47
MetS and PsA are pro-inflammatory conditions that increase the risk of major adverse cardiac events (MACE).8,48 A composite analysis of a cohort of patients with RA/axSpA/PsA with or without MetS found that MetS was independently associated with an increased risk of hospitalisation or death from all causes and cardiovascular causes. These patients also had higher left ventricular (LV) heart mass and a higher risk of malignancy and LV diastolic heart dysfunction. Although these data represent a composite of PsA with other inflammatory arthropathies, the overall picture suggests a worse prognosis in PsA with MetS. 49 However, some studies have shown conflicting data; for example, plaque burden is similar in PsA with or without MetS, implying this process may be more heavily influenced by the rheumatological inflammatory disease and not the MetS inflammatory disease. 50
The influence of disease-modifying anti-rheumatic drugs on MetS
Given the association between MetS and PsA, disease-modifying treatments should ideally aim to address both the inflammatory/autoimmune disease and the cardiometabolic comorbidity. Conventional disease-modifying rheumatic drugs (DMARDs) unfortunately cannot always address both. Unsurprisingly, prednisolone is associated with excess weight gain, with methotrexate showing a smaller but still moderate increase in body weight. 51 Leflunomide and apremilast have both been associated with weight loss, although there is conflicting evidence regarding the presence of any cardiometabolic or prognostic benefit from this weight loss. For example, Baker et al. demonstrated increased mortality in those who lost weight while taking leflunomide.51–55 It is unclear whether this weight loss is because of a loss of fat mass or lean tissue.
Although leflunomide has been shown to reduce body weight, it is also associated with an increased blood pressure and heart rate from baseline, including blood pressures that fall into the hypertension range (140/90 mmHg).56,57 Leflunomide has been shown to significantly increase systolic and diastolic blood pressure at 6 months of treatment when compared to methotrexate therapy. In addition, the odds of being diagnosed with hypertension are also increased after leflunomide initiation. 56 This response is sustained, with a study by Ishaq et al. finding pre-hypertensive and hypertensive rates higher in leflunomide when compared with methotrexate at 1-year follow-up (although this was not tested for statistical significance). 58 The exact mechanism for this is unknown, with proposed mechanisms including an elevated sympathetic neurological response. 57
TNFi drugs, in addition to being associated with a poorer treatment response in people with a higher body mass, may also lead to weight gain, particularly with adalimumab and etanercept.45,59 In particular, a meta-analysis by Patsalos et al. found an increase in BMI in people on TNFi therapy, particularly in those with PsO (only one study in patients with PsA was identified in this meta-analysis). 59 As mentioned above, TNF is a pro-inflammatory cytokine released from visceral fat. 8 Similar to leptin, high levels of TNFα are thought to have a negative feedback effect on body weight, with proposed mechanisms including appetite suppression and muscle catabolism (although low serum levels may promote obesity).8,59,60 Currently, it is unclear whether the reported weight gain is a gain in lean (i.e. muscle) mass, subcutaneous or visceral fat mass. 59 While muscle mass does not contribute to an inflammatory state, a gain in visceral fat mass would contribute to the systemic inflammatory burden, MetS and potentially psoriatic disease activity.2,8,59 PsA is associated with a reduction in muscle mass and excess adiposity; if weight gain was a result of muscle mass, this would likely have positive metabolic effects.29,30,59 Further studies should further elucidate the relationship between visceral adiposity and TNFi therapy. The relationship between TNFi and the incidence of MACE is controversial. However, several systematic reviews and large-scale cohort studies have suggested that TNFi drugs lead to a reduction in the incidence of cardiovascular events in RA, PsA and axSpA.61–63
Historically, there have been concerns about methotrexate being associated with chronic, progressive liver fibrosis. However, recent evidence suggests that low-dose methotrexate use is not associated with liver fibrosis.6,64–67 There are several factors contributing to this change in opinion, partly due to a previously limited understanding of the role of NAFLD in liver fibrosis.65,66 The presence of MetS and NAFLD was a likely confounder in these early studies. 66 Although there is now more confidence that liver fibrosis is not a result of methotrexate use, whether its use in people with a history of NAFLD may be associated with a faster progression to fibrosis remains unclear. 65
Janus kinase inhibitors (JAKi) have been associated with lipid derangement. A meta-analysis by Li et al. found that in patients with RA treated with JAKi, high-density lipoprotein and low-density lipoprotein (LDL) levels increased from the pre-treatment baseline. Although IL-6 signalling may be implicated, the mechanism underpinning this is unclear. 68 JAKi are cautioned in those with a history of cardiovascular disease and venous thromboembolism (VTE) due to the findings of the ORAL Surveillance trial. The trial was a post-authorisation, open-label, non-inferiority safety trial which recruited participants at high cardiovascular risk (over 50 years old with rheumatoid arthritis and at least one cardiovascular risk factor), who were randomised to 5 mg twice daily of tofacitinib, 10 mg twice daily of tofacitinib or a TNFi. There was a numerical increase in MACE risk with tofacitinib at 5 and 10 mg doses and a statistically significant increase in the risk of pulmonary embolism and VTE at 10 mg but not 5 mg doses. 69 However, several meta-analyses of randomised controlled trials and large cohort studies have found no increased risk of VTE or MACE when comparing JAKi against control in inflammatory rheumatic and skin diseases.70–79 The long-term implications of JAKi therapy on cardiovascular risk are therefore unclear. In particular, the lipid and cardiovascular effects of JAKi have not been fully explored in PsA, in which people are more prone to MetS and cardiovascular multi-morbidity when compared to other rheumatological disorders.
Concerns around MACE are also present for IL-12/23 inhibitors, such as ustekinumab. In contrast to TNFi, IL-17/IL-12/23 drugs have not been shown to influence body weight but are associated with an increased risk of MACE.80–82 Two large cohort studies have found an increased risk of MACE in people initiating ustekinumab with PsO (and high cardiovascular risk) and in people in PsA, particularly in the first 6 months of treatment.81,82 There was also an elevated risk of MACE in PsA in people treated with IL-17 inhibitor drugs (e.g. secukinumab). 81 The exact role of IL-17 and IL-23 in the pathogenesis of atherosclerosis is not fully understood; however, it is thought to drive plaque formation and contribute to plaque stability. Therefore, inhibition of the IL-17 pathway in those with a significant plaque burden may precipitate atherosclerotic plaque rupture (leading to myocardial infarction or cerebrovascular accident) in early treatment.81,82
The management of MetS in PsA
Dietary interventions and physical exercise form the cornerstone of weight management. 8 However, the effectiveness of these interventions varies substantially depending on the individual and is highly influenced by genetics, appetite and food environment, among other factors.8,83 This creates several barriers when treating with lifestyle-based weight loss interventions alone. In PsA, concerns surround physical exercise due to the potential for mechanical stress to worsen enthesitis. 2 Although the early evidence suggests the long-term benefits of exercise on symptoms outweigh the risk of enthesitis, active joint, entheseal or skin disease create barriers to exercise program adherence.84,85 Reported physical activity in people with PsO and PsA is lower than in the general population. 85 As body composition in PsA generally consists of a higher ratio of adiposity and a lower ratio of muscle mass, the identification and individualised management (e.g. tailored exercise and dietary plans) of high-risk individuals should be emphasised.29,30 Physiotherapists, occupational therapists and dieticians are crucial to the holistic care of those with PsA, particularly in the context of MetS and obesity. 86
Glucagon-like peptide 1 (GLP-1) agonist drugs (e.g. semaglutide) are anti-diabetic drugs targeting the GLP-1 receptor on pancreatic islet cells. In addition to improved diabetic control via increased endogenous insulin production, GLP-1 agonists improve BMI and several cardiometabolic parameters. 87 The effect on body habitus is extra-pancreatic, primarily via hypothalamic-mediated appetite suppression and increased satiety. 88 This alteration to energy balance leads to an improvement in body weight, lipids, C-reactive protein, and blood pressure in type 2 diabetes mellitus (T2DM). 89 The cardiovascular benefits of GLP-1 agonists are well recognised in T2DM.90,91 However, a recent multicentre randomised controlled trial of semaglutide versus placebo demonstrated a reduction in cardiovascular events in a large cohort of patients with obesity and pre-existing cardiovascular disease without a diagnosis of diabetes mellitus. In addition, the semaglutide group had improved body weight, waist circumference, HbA1c, blood pressure, lipids and C-reactive protein compared to placebo. This study by Lincoff et al. highlights the effectiveness of these drugs for indications outside of T2DM. 92 Semaglutide has been approved for chronic weight management by the US Food and Drug Administration (FDA) in 2021 and the European Medicines Agency in 2022.93,94
Sodium–glucose cotransporter 2 (SGLT2) inhibitors target the SGLT2 glucose transporters in the proximal renal tubule, reducing glucose reabsorption and therefore improving glycaemic control. 95 SGLT2 inhibitors have been shown to lead to weight loss, reduce the risk of cardiovascular death, improve cardiovascular outcomes and reduce the incidence of heart failure, in those with and without T2DM.95,96 The cardiovascular benefits of SGLT2 inhibitors have been demonstrated to occur independently of glycaemic control, with other potential contributory mechanisms including a reduction in BMI and adipokine production, and improved blood pressure, liver steatosis, parasympathetic tone and natriuresis. 96 SGLT2 inhibitors have been shown to tackle several aspects of MetS, including body mass and fat composition, insulin resistance and hypertension.95,96 The use of SGLT2 inhibitors as a disease-modulating therapy has not been explored in PsA. A large cohort study by Ma et al. found no significant impact on the risk of developing PsO with the use of SGLT2 inhibitors and even elevated risk of developing PsO in those over 60, of female sex, with renal disease and hypertension. However, these results were not adjusted for MetS, BMI/obesity or disease severity (PsO or T2DM). The cohort used in this study was Taiwanese, which may not be representative of other populations’ responses to SGLT2 therapy. 97 The effect of SGLT2 inhibitors on PsO and PsA should be carefully examined to see whether these could be potential disease-modifying agents or could potentially worsen psoriatic skin disease.
PCSK9 inhibitors are a class of drugs used for resistant/familial hypercholesterolaemia. PCSK9 inhibition leads to an overexpression of LDL receptors, decreasing serum LDL and improving serum lipid composition. PCSK9 inhibitors have been shown to decrease serum LDL and the rate of MACE. 98 Dyslipidaemia is a major component of MetS and patients with PsO have been shown to have higher serum and dermal PCSK9 levels when compared to controls. 99 Mendelian randomization studies have shown PCSK9 inhibition to be associated with a decreased risk of PsO. 39 The effect of PCSK9 inhibitors on the incidence and prognosis of PsO/PsA has yet to be tested in a real-world cohort.
Given that GLP-1 agonist drugs have been demonstrated to improve obesity and, in turn, MetS, this raises the question of whether GLP-1 agonists can improve disease activity and cardiovascular outcomes in those with PsA. Unfortunately, no large studies have examined this, although promising case reports of treatment effects in PsO exist. 100 The mechanism underlying GLP-1 agonist therapy is potentially disease-modifying (in addition to other benefits from weight loss). Preliminary clinical trials should be performed to detect whether GLP-1 or other weight-modifying agents could be used as an adjunctive treatment alongside conventional/biological DMARD therapy to improve disease activity or prognosis in those with PsA and MetS.
Conclusion
The intersection of MetS and PsA presents a complex challenge due to their shared inflammatory pathways and the exacerbation of PsA by MetS-related conditions such as obesity. The prevalence of MetS in PsA is notably higher compared to other autoimmune disorders, highlighting the significant impact of metabolic health on psoriatic disease. Addressing MetS in PsA involves a multifaceted approach, including lifestyle modifications and pharmacological interventions. Dietary and exercise interventions, though effective, are highly individualised and face adherence challenges. Emerging therapies such as GLP-1 agonists and SGLT2 inhibitors show promise in managing both metabolic and inflammatory aspects of PsA, but further research is needed to establish their efficacy and safety in this specific context. Understanding the bidirectional influence of MetS and PsA is crucial for developing comprehensive treatment strategies. By integrating MetS therapeutics with traditional DMARDs, there is potential to significantly improve disease outcomes and quality of life for individuals with PsA. Future studies are needed to explore these combined approaches, which represent an exciting frontier in the management of PsA complicated by MetS.




