Abstract
Sodium–glucose cotransporter 2 inhibitors demonstrate significant cardiovascular benefits in the management of heart failure; however, adverse effects such as urinary tract infection require careful clinical vigilance. This report describes a case of an 80-year-old female patient with heart failure who developed a urinary tract infection and subsequent septic shock following the initiation of dapagliflozin. The patient had been receiving long-term heart failure therapy, with dapagliflozin added 2 months prior to presentation. She presented with fever and developed hypotension (73/40 mmHg) within 30 min of hospital admission, leading to a diagnosis of septic shock. Management included antimicrobial therapy, vasopressor support, and fluid resuscitation, resulting in hemodynamic stabilization and resolution of the infection. The patient was discharged following clinical improvement. This case underscores the critical importance of vigilant monitoring, proactive patient education, and individualized risk–benefit assessment when prescribing sodium–glucose cotransporter 2 inhibitors to older patients with heart failure who are at high risk. It also provides valuable insights for the development of risk-mitigation strategies.
Keywords
Background
Sodium–glucose cotransporter 2 (SGLT2) inhibitors have been shown to reduce the risks of cardiovascular death and all-cause mortality as well as the worsening of heart failure and hospitalization, and they are now widely used in patients with heart failure.1–3 Dapagliflozin, an SGLT2 inhibitor, has been approved for the treatment of heart failure. With increasing clinical use, adverse drug reactions (ADRs) associated with dapagliflozin have received growing clinical attention. Commonly reported adverse reactions include diabetic ketoacidosis, acute kidney injury, and urinary tract infection (UTI).4–6 Among these, UTIs are common adverse effects of SGLT2 inhibitors; however, cases of septic shock resulting from UTIs are relatively rare. This article reports a case of septic shock caused by UTI following the use of an SGLT2 inhibitor, with the aim of increasing clinical awareness and providing references for the management and prevention of such serious ADRs in vulnerable populations.
Case report
The reporting of this study conforms to the Case Report (CARE) guidelines. 7 The patient provided written informed consent for treatment.
The patient was an 80-year-old female with a body mass index (BMI) of 28.76 kg/m2. She had a medical history of hypertension for more than 30 years, with blood pressure controlled at 130–140/60–90 mmHg; hyperlipidemia for more than 10 years; atrial fibrillation for more than 5 years; and chronic heart failure for more than 5 years, with a left ventricular ejection fraction (LVEF) of 69%. She had undergone left knee joint replacement surgery 6 years ago. She denied a history of diabetes mellitus. Her medication history included 100 mg sacubitril/valsartan twice daily, 20 mg atorvastatin calcium once daily, 20 mg furosemide once daily, 20 mg spironolactone once daily, 5 mg bisoprolol once daily, and 25 mg hydrochlorothiazide once daily. Two months before presentation, 10 mg dapagliflozin once daily was initiated for the treatment of heart failure, and hydrochlorothiazide was discontinued.
The patient presented to a community hospital with a 3-day history of fever. Upon arrival, her temperature was 39.6°C, blood pressure was 128/95 mmHg, and heart rate was 100 beats per minute (bpm). Laboratory examination revealed an elevated white blood cell count of 15,700/μL (normal range, 3500–9500/μL), an elevated C-reactive protein (CRP) level of 177.47 mg/L (normal range, 0–10 mg/L), and an elevated B-type natriuretic peptide level of 1013.2 pg/mL (normal range, 0–100 pg/mL). Chest computed tomography (CT) revealed multiple bilateral pulmonary nodules, emphysema in the anterior segment of the right upper lobe, an enlarged cardiac shadow, calcifications of the aortic and coronary artery walls, bilateral adrenal gland thickening, and mild perinephric exudative changes surrounding the left kidney. Electrocardiography (ECG) showed atrial fibrillation. The patient was treated with 0.4 mg deslanoside administered via infusion pump for ventricular rate control, 100 mg dexibuprofen administered rectally for fever reduction, and 500 mL Ringer’s solution for fluid replacement. Subsequently, she was referred to a higher-level medical institution.
Upon transfer to our institution, the patient exhibited a temperature of 37.6°C, blood pressure of 114/70 mmHg, and heart rate of 185 bpm. She was administered 2 g cefoperazone-sulbactam intravenously (IV). Approximately 30 min later, she developed a persistent, distending headache, with blood pressure dropping to 67/46 mmHg, heart rate decreasing to 143 bpm, and peripheral oxygen saturation remaining at 97% on supplemental oxygen via nasal cannula at 3 L/min. Metaraminol (0.48–2.38 µg/kg/min) and 250 mL of normal saline were administered to increase blood pressure. Abdominal CT revealed inflammatory changes surrounding the left kidney and proximal ureter, suggestive of infectious pathology, concurrent with right nephrolithiasis (Figure 1). Urinalysis was positive for leukocyte esterase (2+) and nitrite and revealed numerous bacteria. The sequential organ failure assessment (SOFA) score was 4. 8 She was diagnosed with septic shock secondary to UTI, and dapagliflozin was discontinued. Laboratory parameters are detailed in Table 1.

Abdominal computed tomography (CT) on admission. CT showing right nephrolithiasis and swelling of the left kidney.
Blood test results.

WBC: white blood cells; HGB: hemoglobin; Plt: platelets; ALT: alanine transaminase; AST: aspartate transaminase; LDH: lactate dehydrogenase; Alb: albumin; BUN: blood urea nitrogen; Cr: creatinine; K: potassium; Na: sodium; NT-proBNP: N-terminal pro-brain-type natriuretic peptide; CRP: C-reactive protein; PCT: procalcitonin.
Following admission, antimicrobial therapy was escalated to imipenem-cilastatin sodium (0.5 g, q6h). By day 3, her SOFA score improved to 1, and hemodynamic stability was achieved, allowing discontinuation of metaraminol. On day 5, the imipenem-cilastatin regimen was adjusted (0.5 g, q8h). Progressive hypoalbuminemia during hospitalization prompted albumin supplementation (10 g, q12h). A gout flare on day 9 resolved with colchicine therapy (0.5 mg, bid orally). By day 12, infection control permitted de-escalation to cefoperazone-sulbactam (2 g, q8h). Throughout hospitalization, urine and blood cultures remained negative. ECG revealed left atrial enlargement, mild mitral stenosis with focal calcification at the base of the posterior leaflet, increased flow velocity at the aortic valve orifice, mild tricuspid regurgitation, and mild pulmonary hypertension. The patient requested discharge on day 14 and was transitioned to oral cefixime capsules (0.1 g, bid) for continued antimicrobial coverage. Additionally, 20 mg furosemide twice daily was prescribed post-discharge. Key laboratory trends during hospitalization are summarized in Figures 2 and 3.
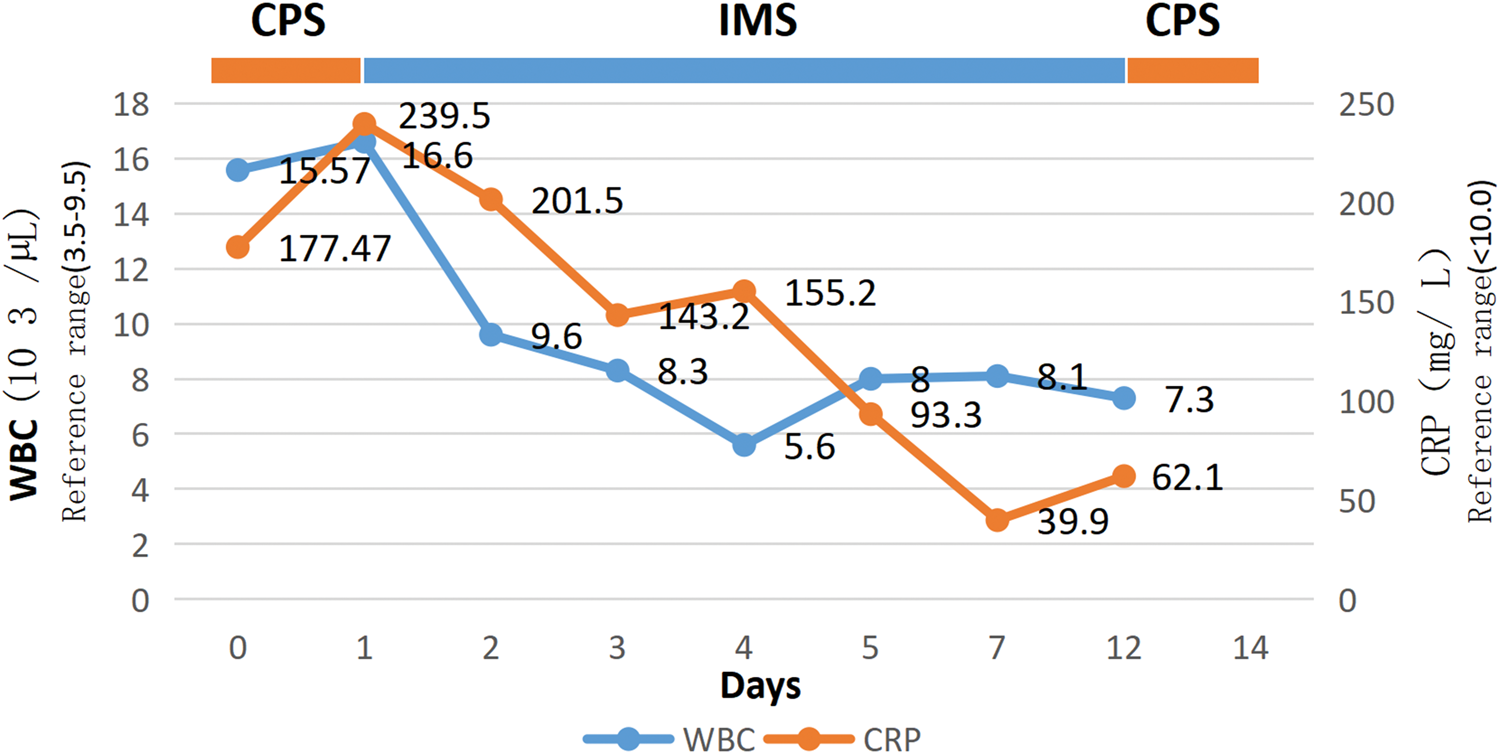
Changes in inflammatory indicators of patients after visiting the hospital. CRP: C-reactive protein; WBC: white blood cell.
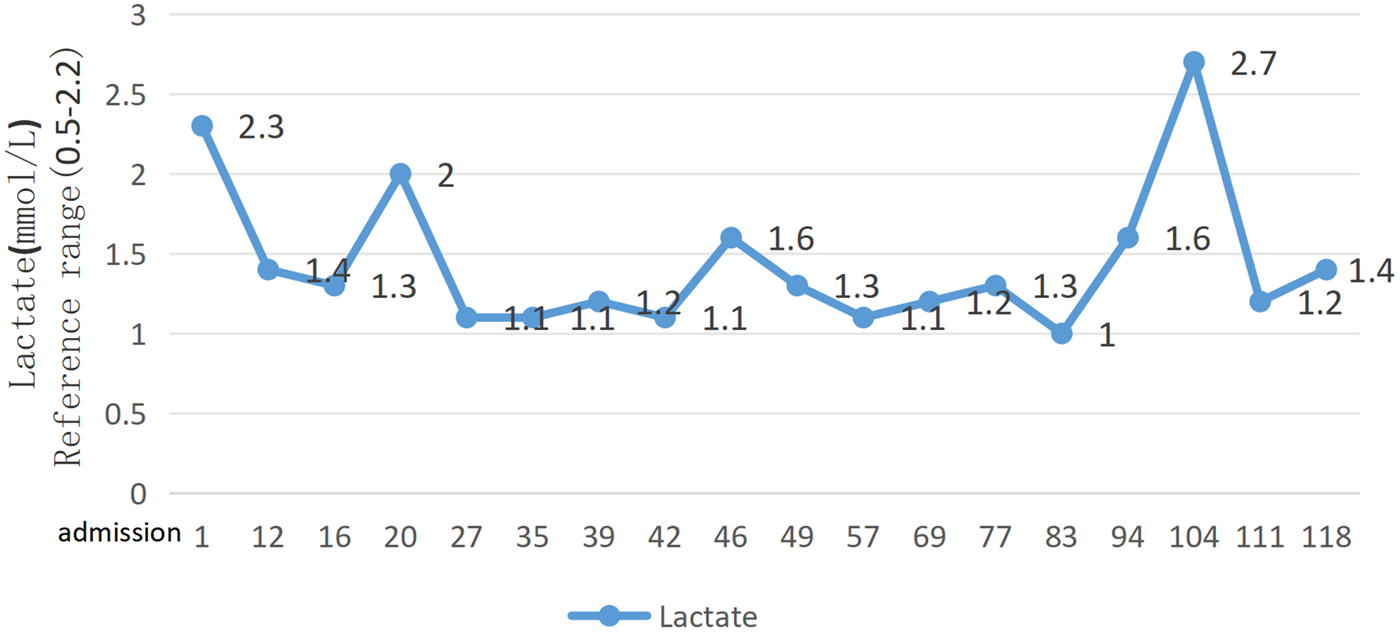
Changes in lactate levels of patients after visiting the hospital.
Dapagliflozin was discontinued during hospitalization. Post-discharge follow-up revealed nonreinitiation of dapagliflozin, and the patient was subsequently rehospitalization for acute heart failure exacerbation 2 weeks later. One month post-discharge, 10 mg dapagliflozin was cautiously reintroduced because of recurrent heart failure episodes, highlighting the essential cardiovascular benefits of this drug class despite infection risk, with close monitoring for urinary tract complications. The clinical pharmacist conducted follow-up and provided medication education, emphasizing the importance of hygiene and safety precautions while taking dapagliflozin. The patient was instructed to seek immediate medical attention if symptoms such as urinary frequency, urgency, or dysuria occurred. After 6 months of follow-up, the patient did not experience any recurrent UTI.
Discussion
Mechanisms of SGLT2 inhibitor–associated UTI
The use of SGLT2 inhibitors in the treatment of heart failure is becoming increasingly widespread; however, their adverse effects, including acute kidney injury, ketoacidosis, and UTI, are also receiving greater attention.4–6 UTI is one of the most common adverse reactions. In this patient, the Naranjo scale 9 to assess the likelihood of an ADR yielded a score of 3, indicating a possible relationship between dapagliflozin and the development of UTI. The possible mechanism underlying SGLT2 inhibitors–associated UTI involve the reduction of renal glucose reabsorption due to inhibition of SGLT2, resulting in elevated glucose concentrations in the urine (glycosuria). This hyperglycemic environment provides a nutrient-rich medium for bacterial growth and colonization. 10 Additionally, persistent glycosuria may impair the natural defense mechanisms of the urinary tract. 11
Risk factors for SGLT2 inhibitor–associated UTIs
Clinical studies have identified various factors associated with an increased risk of UTIs in patients treated with SGLT2 inhibitors. 1. Demographic characteristics. Female sex is a well-established independent risk factor, as the short and straight anatomical structure of the female urethra facilitates the upward migration of bacteria. Older patients, particularly postmenopausal women, are at a significantly increased risk of infection because of impaired local defenses resulting from urethral mucosal atrophy and reduced estrogen levels. 12 2. Underlying diseases. Patients with chronic kidney disease experience impaired renal function, which leads to abnormal urine excretion and an increased risk of bacterial retention. 13 Obese patients (BMI ≥ 28 kg/m2) may have an increased risk of infection due to alterations in urinary tract anatomy and reduced immune function. 14 Moreover, patients with kidney stones, whether unilateral or bilateral, have a higher probability of systemic infection due to disrupted urodynamics, chronic urinary tract inflammation, and compromised mucosal integrity. 15 3. Other factors. The use of immunosuppressants directly impairs the body’s ability to resist infection and increases susceptibility to bacterial infections. 16
In this case, the patient presented with multiple high-risk factors, including advanced age, female sex, obesity, and kidney stones, placing her in a high-risk group for UTI. These factors also explain the potential for infection following the use of dapagliflozin.
Controversy over the risk of UTI with SGLT2 inhibitors in patients with heart failure
The association of SGLT2 inhibitors and the increased risk of UTI in patients with heart failure remains controversial. A meta-analysis by Staplin et al. 17 indicated that patients with heart failure using SGLT2 inhibitors had a higher risk of UTI than with placebo. Similarly, Soleimani et al. 18 reported that genitourinary infections were more common in patients with heart failure receiving SGLT2 inhibitors; however, subgroup analysis revealed that empagliflozin had a significantly higher risk than placebo, whereas dapagliflozin did not show significant difference. However, a study by Cai et al. 19 showed no increased risk of UTI in patients with chronic heart failure treated with dapagliflozin. Similarly, Borovac et al. 20 did not demonstrate a clinically meaningful excess risk of UTIs with SGLT2 inhibitors. Nevertheless, these studies did not further stratify patients by risk factors such as obesity, advanced age in women, or urolithiasis. Patients with high-risk phenotypes (older female, obesity, and urolithiasis) may diverge from the average trial risk, highlighting the need for further investigation.
Suetani et al. 21 reported a case of a 69-year-old immunocompromised woman with heart failure who developed septic shock secondary to UTI after treatment with dapagliflozin. Following discontinuation of dapagliflozin, the patient experienced no recurrence of UTI or worsening of heart failure during a 10-month follow-up. Both that case and the present case suggest that high-risk populations receiving SGLT2 inhibitors must remain vigilant regarding the risk of severe infections. Notably, in the present case, after restarting dapagliflozin and receiving comprehensive medication guidance, the patient did not experience any UTIs during approximately 6 months of follow-up. This outcome has significant clinical implications. For patients who can derive substantial cardiovascular benefits from SGLT2 inhibitor therapy, the potential risk of infection should not lead to arbitrary discontinuation of the medication. Instead, proactive risk management measures should be implemented. Clinicians must thoroughly assess patients’ infection risk before initiating treatment, increase monitoring during therapy, and reduce the likelihood of infections through systematic patient education.
Strategies for the prevention and management of SGLT2 inhibitor–associated UTI
Combined with the special pathophysiological status of patients with heart failure, the prevention of UTI requires individualized adjustments in addition to routine measures, in order to balance the needs of infection prevention and heart failure management. Key practice points include the following.
Prior to treatment initiation, a comprehensive patient risk assessment should be conducted. This includes systematic screening for potential urinary system abnormalities (e.g. detection of stones, obstructions, and anatomical anomalies detected via ultrasound) and clear identification of high-risk populations, including older females, individuals with obesity, those with poorly controlled blood glucose levels, and patients with a history of recurrent UTIs. During treatment, patient education must be emphasized. Patients should be instructed to promptly recognize early signs of infection, such as frequent urination and urgency, and immediately report the symptoms. Concurrently, individualized hydration plans should be developed based on heart failure status, and personal hygiene management should be reinforced. In the event of an infection, timely etiological testing should be performed immediately. Management should be stratified according to the severity of the infection. Medication should be temporarily suspended for upper urinary tract or systemic infections, whereas treatment may continue under close monitoring for uncomplicated lower UTIs. All decisions must be guided by individualized risk–benefit assessments. After infection control is achieved, the benefits and risks of treatment should be reevaluated, and for patients with clear clinical benefit, therapy may be restarted under enhanced monitoring.
This case report has several limitations. First, although the patient was at high risk for UTI, no urinalysis was performed prior to the initiation of dapagliflozin. Therefore, it remains unclear whether a UTI was present before treatment. Second, the follow-up period was relatively short, and extended monitoring would be necessary to adequately assess the risk of UTI following reinitiation of dapagliflozin.
In conclusion, the use of SGLT2 inhibitors may lead to UTI accompanied by septic shock, particularly in high-risk individuals such as older, obese females. This case highlights the critical importance of individualized risk assessment before initiating SGLT2 inhibitor therapy. Clinicians should not only weigh the substantial cardiovascular benefits against potential infection risks but also implement proactive strategies, including comprehensive patient education and close monitoring, to effectively mitigate the risk of UTI.
Footnotes
Acknowledgments
We thank DeepSeek Chat (V3) for assistance with language polishing and editing of the manuscript.
Author contributions
M-LW designed this study. M-LW and Y-TT wrote the original draft. Y-HZ, X-HL, G-HC, and L-MY conducted the literature review and revised the manuscript. All authors contributed to the article and approved the submitted version.
Declaration of financial/other relationships
The authors declare that there is no conflict of interest.
Data availability statement
All of the data supporting our findings are contained within the manuscript.
Ethics statement
Ethical approval was waived by the Ethics Committee of Beilun People’s Hospital for this study. The study was conducted in accordance with the principles of the Declaration of Helsinki. The patient’s daughter provided the written informed consent to participate in this study. Written informed consent was obtained from the patient’s daughter for the publication of any potentially identifiable images or data included in this article.
Funding
This work was supported by the Foundation of Hospital Pharmacy of Zhejiang Pharmaceutical Association (No. 2022ZYY37).
