Abstract
Background:
Obesity has been proposed as a risk factor for the development of rheumatoid arthritis (RA). Glucagon-like peptide-1 receptor agonists (GLP-1RAs) are increasingly prescribed for weight reduction and glycemic control and have been shown to exert immunomodulatory effects. However, the association between GLP-1RA use and the onset of RA remains unclear.
Objectives:
To investigate the association between GLP-1RA exposure and new-onset RA in a large population-based study.
Design:
Retrospective, population-based case–control study.
Methods:
We analyzed data from a nationwide health provider database. All adults diagnosed with RA were matched with controls (1:5) by age, sex, and socioeconomic status. The primary exposure was GLP-1RA use within the 10 years preceding RA diagnosis. Multivariable logistic regression models were used to estimate the association between GLP-1RA use and new onset RA, adjusting for age, body mass index (BMI), smoking status, and diabetes mellitus (DM). Duration of GLP-1RA exposure was stratified according to length of exposure (⩽6 vs >6 months).
Results:
The study included 4535 RA cases and 22,675 matched controls. In univariate analyses, subcutaneous semaglutide and liraglutide were significantly associated with new onset RA, while dulaglutide showed a non-significant trend. These associations remained significant in multivariable models adjusted for potential confounders. Higher BMI categories and DM were independently associated with new onset RA. When GLP-1RA exposure was stratified according to length of exposure (⩽6 vs > 6 months), shorter exposure, but not longer exposure, was associated with new onset RA.
Conclusion:
GLP-1RA use was associated with new onset RA. However, prolonged treatment appeared to attenuate this association, potentially reflecting the beneficial effects of GLP-1RAs on BMI and glycemic control, which are independently associated with new onset RA.
Plain language summary
Introduction
Rheumatoid arthritis (RA) is a relatively prevalent chronic inflammatory autoimmune disease characterized by persistent joint inflammation leading to cartilage degradation and bone erosion. 1 RA pathogenesis involves complex interaction between genetic predisposition and environmental triggers that activate both the innate and adaptive immune responses leading to chronic inflammation. 2
Whether obesity, a modifiable environmental factor, predisposes to RA development, is controversial. Population-based case–control studies have reported positive associations with odds ratios (OR) ranging from 1.4 to 3.7.3,4 However, these findings were not consistently replicated. Some studies demonstrated associations in specific RA subpopulations, such as anti-cyclic citrullinated peptide-negative female patients and women diagnosed with RA prior to the age of 55.5,6 One study reported an association in men but not in women. 7 Conversely, two prospective cohort studies found no association between obesity and RA risk.8,9
Glucagon-like peptide-1 receptor agonists (GLP-1RAs) are primarily indicated for type 2 diabetes, but also for weight reduction in patients with obesity.10,11 The use of these agents has markedly increased in recent years, a trend attributed, at least in part, to their efficacy in promoting weight loss. 12 Mechanistically, GLP-1RAs mimic the physiological actions of the endogenous hormone GLP-1 released from the gastrointestinal tract postprandially. 10 GLP-1RA’s effects include increased glucose-dependent insulin secretion, suppression of glucagon secretion, delayed gastric emptying, and greater satiety, contributing to reduced energy intake and, consequently, weight reduction (Figure 1).10,13 Evidence from clinical trials supports this, demonstrating that GLP-1RA therapy can induce clinically significant weight loss, typically within the range of 5%—10% from baseline body weight.10,14

Physiological role of GLP-1 and its impact on body weight.
Beyond their recognized effects on endocrine function and body weight, GLP-1RAs are increasingly recognized as immunomodulators. Evidence regarding the association between GLP-1RA usage and RA is conflicting. On one hand, case reports have linked liraglutide use (e.g., GLP-1RA) to polyarthritis, suggesting the possibility of medication-triggered inflammatory response. 15 A recent network meta-analysis evaluating over 290,000 participants from 435 randomized trials identified a potential safety signal linking certain GLP-1RAs with an increased incidence of arthritis-like events. 16 While these findings raise concern about possible immune or inflammatory effects, the study’s limitations, including inconsistent event reporting, lack of standardized definitions, and arthritis not being a prespecified outcome, prevent definitive conclusions. Conversely, other studies indicated potential benefits. For instance, exenatide, another GLP-1RA, has demonstrated anti-inflammatory effects on fibroblast-like synoviocytes, implying a possible mechanism for mitigating RA symptoms through the modulation of inflammatory pathways. 17 Furthermore, a systematic review suggested that GLP-1RAs might reduce RA disease activity, possibly mediated by significant weight loss and distinct immunomodulatory effects independent of weight reduction. 18
This apparently paradoxical body of evidence, recently reviewed by Bilgin et al., 19 suggests that GLP-1RAs may possess dual immunomodulatory properties, capable of eliciting either pro-inflammatory or anti-inflammatory responses. To address this uncertainty, our study aimed to investigate the association between GLP-1RA use and new onset RA by employing a two-sided association test.
Methods
Study design
This case–control study was conducted using the Leumit Health Services (LHS) computerized database. LHS database has been previously described. 20 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement for case–control studies. 21
Participants
We identified all adult (ages ⩾18 years) RA patients in the LHS database up to July 31, 2025. RA patients were identified using the International Classification of Diseases, Ninth Revision (ICD-9) diagnostic code “714.0.” To enhance diagnostic accuracy and ensure the inclusion of confirmed RA cases, RA patients were included in the study only if they had either a documented treatment with a conventional synthetic disease-modifying antirheumatic drug (csDMARD) post RA diagnoses or diagnosis being documented by rheumatologist (Figure 2). Each RA was matched with five controls based on age, gender, and socioeconomic status. Controls were selected from within the LHS database as patients without RA diagnosis. The study size was determined by the number of eligible patients available in LHS database during the study period. All adult patients who met the predefined inclusion criteria were included, and each case was matched with five controls from the same source population. No sample calculations were performed as the study included all eligible patients, aiming to minimize potential selection bias.
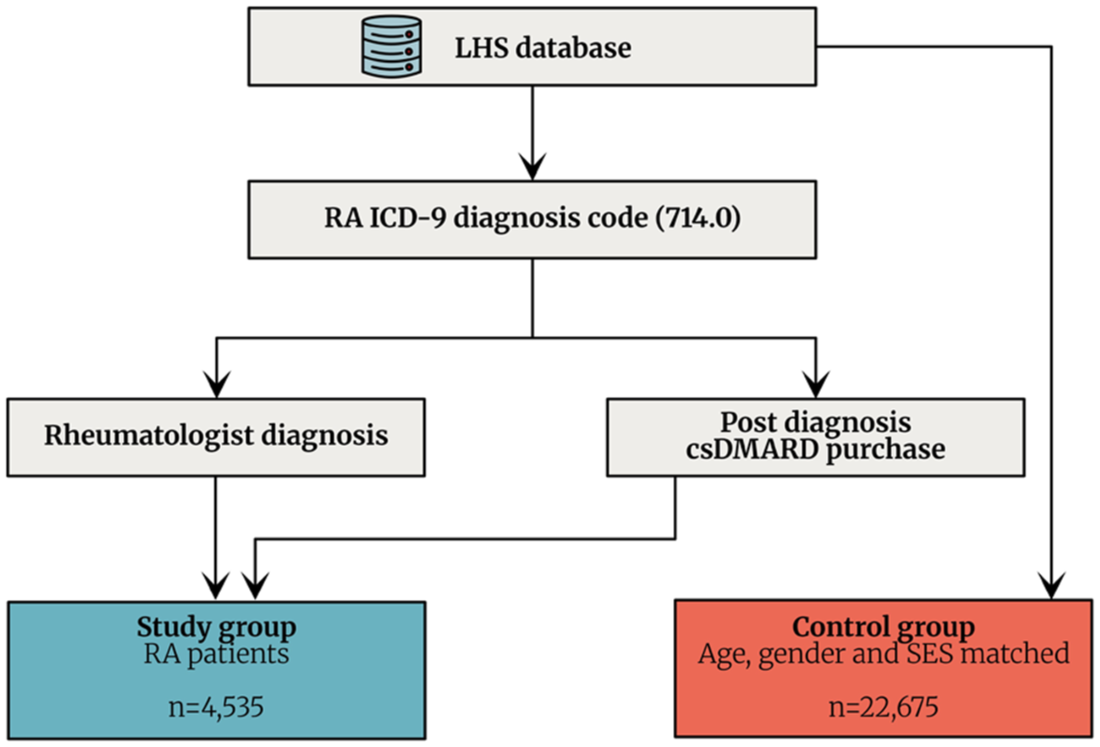
Study population.
Outcomes
The primary outcome was new onset of RA, documented as a chronic diagnosis in the patient’s electronic health record, encoded by its ICD-9 code, and fulfilling the additional criterion of csDMARD consumption or diagnosis documentation by a rheumatologist.
Exposures
The primary exposure variable of interest was the use of GLP-1RA within a 10-year period preceding RA diagnosis. The following GLP-1RAs were included in the analysis: liraglutide, dulaglutide, and semaglutide. Exposure data were ascertained from the healthcare database based on prescription records and confirmed drug purchases, ensuring that patients had both a physician-issued prescription and evidence of actual acquisition of the medication. The 10-year exposure window was chosen to balance between biological relevance and temporal proximity to RA onset. On one hand, this duration allows sufficient time for the pathogenic processes underlying RA, such as immune dysregulation and autoantibody formation, to evolve following exposure to potential modifying factors like GLP-1RAs. On the other hand, it is not so extended as to risk capturing remote exposures that may no longer be linked to disease development. Exposure length was further categorized as <6 months or ⩾6 months.
Other variables
The following variables were collected from the LHS database, including demographic characteristics (age, gender, marital status) and smoking status (classified as active smoker, past smoker, or non-smoker). General comorbidities included ischemic heart disease, diabetes mellitus (DM), chronic obstructive pulmonary disease (COPD), chronic kidney disease, cerebrovascular accident, liver cirrhosis, deep venous thrombosis, osteoporosis, hypothyroidism, inflammatory bowel disease (IBD), and fibromyalgia. Malignancy history was also recorded, including breast, colon, lung, pancreatic, prostate, and skin (melanoma) cancers. All comorbidities were identified using relevant ICD-9 diagnostic codes.
Bias
To minimize potential sources of bias, cases and controls were derived from the same source population and were matched based on age, gender, and socioeconomic status; RA diagnosis and GLP-1RA exposure were stringently defined to reduce misclassification, and multivariable analyses were used to adjust for relevant confounders. Additionally, all eligible RA patients were included to minimize potential selection bias.
Statistical analysis
Baseline characteristics were presented as frequencies and percentages for categorical variables and means and standard deviations or medians and interquartile ranges for continuous variables, as appropriate. Differences between baseline variables were tested with Fisher’s exact test for categorical variables and two-sample t test or the Mann–Whitney test for continuous variables, as appropriate. The association between GLP-1RA usage and new onset RA was assessed using univariate logistic regression analysis. Multivariable logistic regression analysis was subsequently employed to adjust for potential confounders. Missing data were handled using complete-case analysis; no imputation was performed, and analyses were based on available data for each variable. Statistical analyses were conducted using R version 4.4 (The R Foundation for Statistical Computing, Vienna, Austria). All statistical tests were two-sided, with a p-value <0.05 considered statistically significant.
Ethical approval
Ethical approval for this study was obtained from the Institutional Review Board of LHS.
Results
A total of 4535 RA patients and 22,675 matched controls were included in the analysis. Baseline characteristics of the study population are summarized in Table 1. RA patients had slightly higher BMI compared to controls (28.5 ± 5.7 vs 28.0 ± 5.7, p < 0.001). They also had a higher prevalence of several comorbidities, including DM (18.7% vs 15.6%, p < 0.001), COPD (7.1% vs 4.8%, p < 0.001), osteoporosis (12.5% vs 9.6%, p < 0.001), hypothyroidism (8.9% vs 7.1%, p < 0.001), IBD (1.3% vs 0.6%, p < 0.001), and fibromyalgia (9.7% vs 2.5%, p < 0.001). No significant differences were observed for other malignancies, although prostate cancer was slightly less frequent among RA patients (0.33% vs 0.60%, p = 0.03). Additional characteristics are presented in Table 1.
Baseline characteristics.

Bold styling indicates clinical significance.
Anti-CCP, anti-cyclic citrullinated peptide; BMI, body mass index; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; RA, rheumatoid arthritis; SES, socioeconomic status.
After characterizing baseline comorbidities, we next examined the association between GLP-1RA exposure and subsequent RA diagnosis. The associations between the use of GLP-1RAs within the 10-year period preceding the diagnosis of RA are presented in Table 2. Liraglutide use was significantly associated with new onset RA, with 2.12% of RA cases having prior exposure compared to 1.33% of controls (odds ratio (OR) 1.61, 95% confidence interval (CI) 1.26–2.03; p < 0.001). Similarly, semaglutide exposure was more frequent among RA cases (1.79%) than controls (1.06%), corresponding to an OR of 1.69 (95% CI 1.30–2.19; p < 0.001). For dulaglutide, an increased rate of exposure was also observed among RA cases (0.75%) compared to controls (0.55%), although this difference did not reach statistical significance (OR 1.36, 95% CI 0.90–2.01; p = 0.110).
GLP-1RA use within a 10-year period preceding the diagnosis of RA.

CI, confidence interval; GLP-1RA, glucagon-like peptide-1 receptor agonists; RA, rheumatoid arthritis.
To further evaluate the association between GLP-1RA use and new onset RA while accounting for potential confounders, multivariable conditional logistic regression analyses were performed (Figure 3). After adjustment for age, sex, smoking status, BMI, DM, and socioeconomic status, liraglutide (adjusted odds ratio (aOR) 1.58, 95% CI 1.22–2.04; p < 0.001) and semaglutide (aOR 1.64, 95% CI 1.26–2.16; p < 0.001) remained significantly associated with new onset RA, while dulaglutide showed a non-significant trend in the same direction (aOR 1.31, 95% CI 0.86–1.99; p = 0.12). A clear dose–response relationship was observed between BMI and new onset RA, and DM remained independently associated with new onset RA. When GLP-1RA exposure was stratified by duration, the association was confined mainly to shorter treatment periods (⩽6 months), suggesting a transient pro-inflammatory effect that may be offset by the long-term metabolic benefits of continued therapy.

Multivariate analysis of GLP-1RA use up to 10 years prior to RA diagnosis. Forest plots displaying multivariable aORs and 95% CIs for RA according to GLP-1RA use. (a) Model adjusted for BMI category, diabetes mellitus diagnosis, age, sex, smoking status (current, past, or never), and socioeconomic status category. (b) Same. Model stratified by cumulative exposure, comparing ⩽6 and >6 months of GLP-1RA purchases.
Discussion
This large retrospective study, performed in a national healthcare provider, demonstrated that exposure to GLP-1RAs, particularly semaglutide and liraglutide, was significantly associated with new onset RA even after adjustment for major confounders, including age, smoking status, BMI, DM, and socioeconomic status. The effect was strongest during the first 6 months of therapy, while longer exposure was not associated with new onset RA. Dulaglutide showed a non-significant trend in the same direction, suggesting a possible class effect.
While GLP-1RAs were primarily developed as anti-diabetic drugs and later were used and indicated for weight loss in patients with obesity, they were also found to have potential immunomodulatory effect (Figure 4). A recent study demonstrated that lixisenatide, selective GLP-1RA, has anti-inflammatory and cytoprotective effects in interleukin (IL)-1β-stimulated RA fibroblast-like synoviocytes. 17 Lixisenatide treatment significantly reduced the production of key proinflammatory cytokines including tumor necrosis factor alpha (TNF-α), IL-6, and IL-8, suppressed the expression of matrix metalloproteinases, and ameliorated mitochondrial dysfunction and oxidative stress while inhibiting activation of central inflammatory signaling pathways such as JNK, AP-1, and nuclear factor kappa B (NFκB), thereby supporting the hypothesis that GLP-1RAs may exert disease-modifying effects through direct immunomodulation of synovial cells. Beyond their effects on fibroblast-like synoviocytes, GLP-1RAs also exhibit broad immunomodulatory activity through direct interaction with immune cells expressing the GLP-1 receptor, including monocytes, macrophages, T and B lymphocytes. These agents attenuate innate immune responses by suppressing the production of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, inhibiting NF-κB signaling, and promoting the polarization of macrophages toward an anti-inflammatory M2 phenotype 22 (Figure 4). GLP-1RAs also act on the adaptive immune system by inhibiting T cell proliferation and the secretion of effector cytokines (e.g., IFN-γ) while promoting enhanced regulatory T cell function. In addition, GLP-1RAs downregulate adhesion molecules on endothelial cells, including vascular cell adhesion protein 1 (VCAM-1) and intercellular adhesion molecule 1 (ICAM-1), thereby reducing leukocyte recruitment and subsequent inflammation. 22 Notably, a recent Mendelian randomization study demonstrated a significant association between GLP-1 receptor expression and reduced risk of RA, providing further evidence that the immunomodulatory effects of GLP-1RAs extend further beyond their metabolic benefits. 18
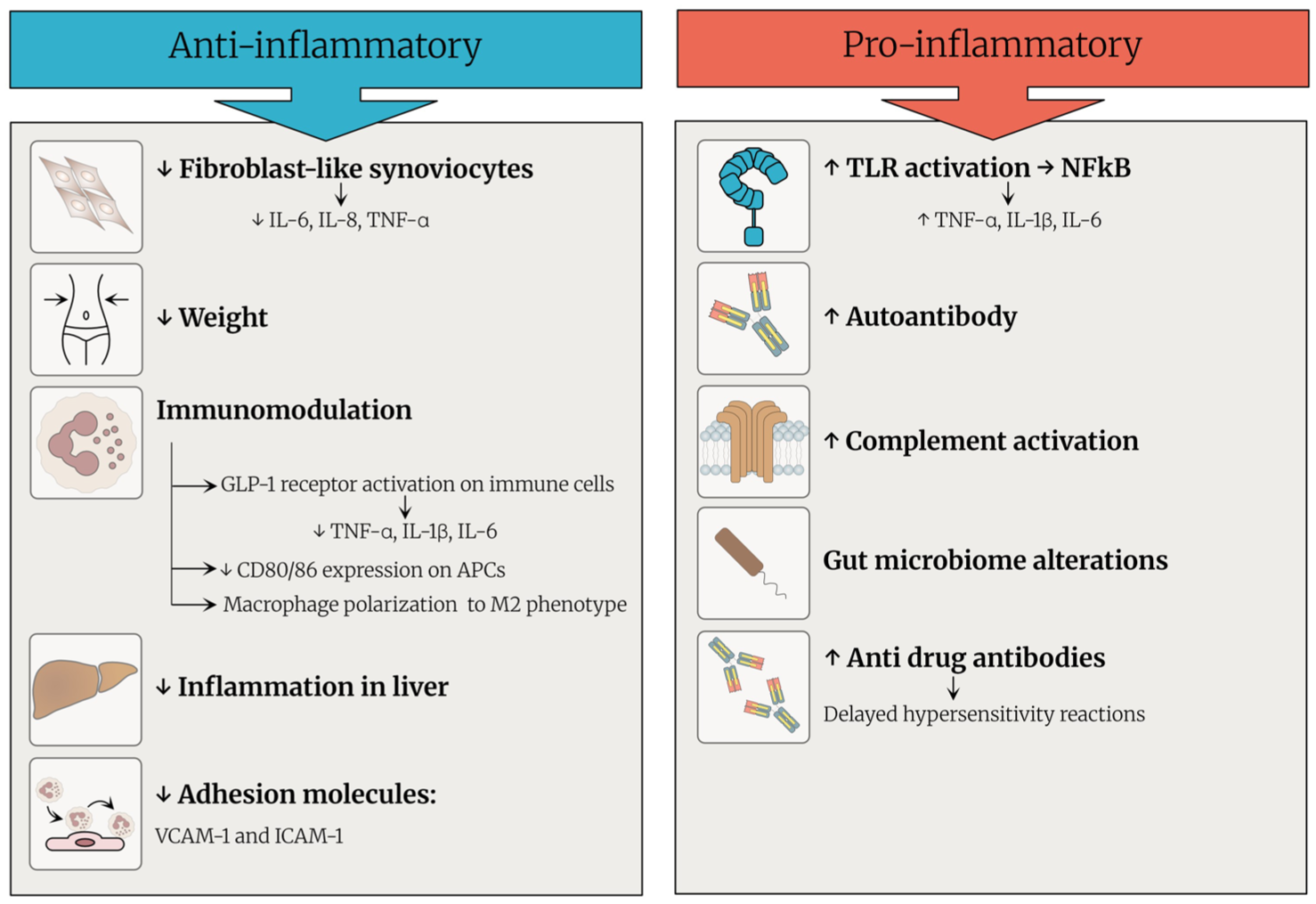
Pro- and anti-inflammatory properties of GLP-1 receptor agonists.
However, our results, together with emerging clinical reports, suggest that GLP-1RAs may also exert transient pro-inflammatory or immune-activating effects (Figure 4). One proposed mechanism involves the activation of innate immunity via Toll-like receptors (TLRs). Subsequently, activated TLRs may trigger the NFκB pathway resulting in the production of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, all of which play pivotal roles in autoimmune inflammation, including RA. 23 Furthermore, GLP-1RAs may impact adaptive immune responses by stimulating autoantibody production, as demonstrated in drug-induced lupus erythematosus (DIL), when anti-dsDNA, anti-histone, and anti-mitochondrial antibodies were found after semaglutide administration. 24 Furthermore, immune complex-mediated injury resulting from activation of the complement system, as indicated by decreased serum complement levels in affected patients, may be involved. GLP-1RAs have also been documented to form anti-drug antibodies, potentially triggering delayed hypersensitivity reactions and vasculitis. 25 By altering gut microbiota, GLP-1RAs may further amplify inflammatory signaling cascades by influencing cross-talk between the microbiome and immune system. 26 In addition to mechanistic insights, clinical reports of autoimmunity caused by GLP-1RA support these findings. One case described a 76-year-old woman who developed multiorgan DIL following semaglutide use, characterized by debilitating fatigue, petechial rash, elevated liver enzymes, and positive ANA, anti-dsDNA, and anti-histone antibodies with low complement levels, consistent with systemic autoimmune activation. 24 Another report documented a case of leukocytoclastic vasculitis attributed to subcutaneous semaglutide in a 73-year-old man, presenting with palpable purpura and histopathological findings of small-vessel immune complex deposition and neutrophilic infiltration, which resolved upon drug cessation. 25
Our findings indicate a complex bidirectional relationship between GLP-1RA exposure and new onset RA. Early treatment phases may transiently promote immune activation and inflammation, whereas sustained therapy, by reducing BMI, improving insulin sensitivity, and lowering systemic inflammation, may mitigate this effect. The temporal pattern observed in our study, with an association between GLP-1RAs and new onset RA during the first 6 months of GLP-1RA therapy followed by attenuation with prolonged exposure, supports the hypothesis that the protective effects of GLP-1RAs may be primarily mediated through metabolic improvements, particularly weight reduction, which require time to manifest and counteract any early pro-inflammatory effects. This interpretation aligns with clinical observations suggesting that while GLP-1RAs demonstrate direct anti-inflammatory properties in preclinical models, their clinical benefits in established RA appear to be strongly correlated with weight loss and metabolic improvements. 19 The observation that clinical improvements in RA disease activity among patients treated with liraglutide were associated with reductions in body weight, inflammatory markers, and metabolic parameters further supports this mechanism. 19 Conversely, the early association observed in our cohort may reflect transient immune activation before sufficient weight loss and metabolic benefits have occurred to exert their protective anti-inflammatory effects. This biphasic response shed light on the complex interplay between direct immunomodulatory actions and indirect metabolic-mediated effects of GLP-1RAs in autoimmune disease pathogenesis, which could explain some of the differences seen in studies examining the effect of GLP1-RA use and immune disease.15–18 Furthermore, the clear dose–response association between BMI and new onset RA observed in our cohort strengthens the evidence that adiposity contributes to RA pathogenesis.
Reverse causality or confounding by indication may have affected the associations found in our study, particularly since obesity, metabolic syndrome, and DM, the primary indications of the use of GLP-1RA, were identified as independent risk factors for RA in previous studies.3,4,7 Both DM and BMI were found to have statistically significant association with RA in the multivariate model, with a gradual dose–response relationship observed for BMI. This finding is consistent with growing evidence pointing to metabolic dysregulation as a cause of systemic autoimmunity.27–29 Pro-inflammatory cytokine patterns, insulin resistance, and persistent hyperglycemia could facilitate immune activation and joint inflammation, causing RA in susceptible individuals. Despite our model’s adjustment for several confounding factors, residual confounding related to unmeasured components of the metabolic syndrome, such as visceral fat, adipokine dysregulation, or low-grade systemic inflammation, may still be present and underlie some of these observed associations. Further studies with in-depth metabolic profiling and inflammatory biomarkers are required to determine the mechanistic pathways through which DM and RA are associated.
Our findings are timely given the rapid global rise in GLP-1RA prescriptions, both for diabetes and obesity management. An increased incidence of RA or related autoimmune diseases, even if limited to early treatment phases, could have meaningful public health implications.15,16,30,31
This study has limitations inherent to its observational retrospective design. Causality cannot be inferred, and residual confounding may persist despite rigorous matching and adjustment. As all eligible patients who met the predefined inclusion criteria were included, no sample size calculations were performed. Paucity of serological data did not allow sufficient power for subgroup analysis, and we were not able to adjust for glucocorticoid or immunomodulatory medication use. Nonetheless, the large sample size and consistent findings across multiple GLP-1RAs support the robustness of the observed associations.
Conclusion
The use of GLP-1RAs, particularly semaglutide and liraglutide, was associated with new onset RA, most evident during the initial months of treatment. This finding supports a potential short-term pro-inflammatory effect that may be counterbalanced by the long-term metabolic and anti-inflammatory benefits of sustained therapy. Prospective mechanistic and longitudinal studies are warranted to clarify these dual immune-metabolic effects and to guide safer therapeutic use, particularly during the early months of treatment.
Supplemental Material
sj-doc-1-tab-10.1177_1759720X261425441 – Supplemental material for The association between glucagon-like peptide-1 receptor agonist and rheumatoid arthritis: a population-based case–control study
Supplemental material, sj-doc-1-tab-10.1177_1759720X261425441 for The association between glucagon-like peptide-1 receptor agonist and rheumatoid arthritis: a population-based case–control study by Ariel Israel, Fadi Hassan, Eugene Merzon, Jalal Kurtam, Jamal Awad, Mai Assalia, Ilan Green, Shlomo Vinker and Mohammad E. Naffaa in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
During the preparation of this work, ChatGPT use was limited to grammar checks. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
