Abstract
Background:
The Ankylosing Spondylitis (AS) Disease Activity Score (ASDAS) is largely used for assessing disease activity in patients with AS.
Objectives:
We aimed to investigate the predictability of ASDAS on drug survival in patients with low Bath AS Disease Activity Index (BASDAI) during biologic therapy.
Design:
Using data from multi-center, prospective, observational prospective cohort, Korean College of Rheumatology Biologics and Targeted Therapy (KOBIO) registry.
Methods:
The study population consisted of patients enrolled in the KOBIO registry from December 2012 to December 2018. The baseline demographic data and variables such as extra-articular manifestations, HLA-B27 positivity, family history of spondyloarthritis, ASDAS C-reactive protein (CRP), BASDAI, and Bath AS Functional Index scores were collected from the database. The disease activity indices were followed yearly after initiating a tumor necrosis factor (TNF) inhibitor (TNFi). Disease activities were defined as high (ASDAS-CRP ⩾ 2.1, BASDAI ⩾ 4) and low (ASDAS-CRP < 2.1, BASDAI < 4).
Results:
Data from 1773 patients were analyzed. Among 269 patients with low BASDAI at baseline, 151 (56.1%) patients had high ASDAS-CRP, yet in 142 patients with low ASDAS-CRP at baseline, only 24 (16.9%) patients had a high BASDAI. High ASDAS-CRP captured more patients who had initiated or switched to a TNFi than those with high BASDAI (92.5% versus 84.8%, respectively, p < 0.001). Moreover, among AS patients with low BASDAI after 1 year of therapy, drug persistence in the following year was significantly lower in patients with high ASDAS than in those with low ASDAS (68.7% versus 82.5%, p < 0.001).
Conclusion:
ASDAS-CRP not only has its advantages over BASDAI in assessing disease activity but also low ASDAS-CRP at 1 year can be a marker of long-term drug survival of TNFi therapy.
Introduction
Ankylosing spondylitis (AS) is a chronic inflammatory disease of unknown etiology, which mainly affects the spine and sacroiliac joints but also extra-spinal and extra-articular areas. 1 Disease activity leads to severe ankyloses and functional impairment. Thus, disease monitoring including patient-reported outcomes, clinical findings, laboratory tests, and imaging is important. 2
In measuring disease activity in patients with AS, the Bath AS Disease Activity Index (BASDAI) is commonly used in daily clinical practice for patients and is also the key indicator used for National Health Insurance reimbursement in Korea. The BASDAI contains six questions regarding fatigue, back pain, peripheral joint symptoms, enthesitis, and morning stiffness. 3 The Ankylosing Spondylitis Disease Activity Score (ASDAS) developed by the Assessment of SpondyloArthritis International Society 4 includes selected patient-reported outcomes within the BASDAI and also contains laboratory test results, such as C-reactive protein (CRP) levels or erythrocyte sedimentation rate (ESR), weighted in the index. Using ASDAS, disease activity status is classified as inactive (<1.3), low (⩾1.3, <2.1), high (⩾2.1, ⩽3.5), or very high (>3.5). 5
Although ASDAS has been endorsed6,7 and demonstrated as an index for assessing disease activity after TNF inhibitor (TNFi) therapy and its association with structural damage,8,9 no large-scale study has focused on its utility along with BASDAI in patients with AS after long-term biologic therapy. Using a nationwide biologic registry, this study aimed to explore the discordances between the ASDAS and BASDAI at the initiation of biologic therapy and thereafter, plus investigate the clinical outcomes in patients showing such discrepancies.
Methods
Data collection
This study utilized data from the Korean College of Rheumatology Biologics and Targeted therapy (KOBIO) registry. The KOBIO registry is a multi-center, prospective, observational cohort that prospectively collects clinical manifestations, laboratory and radiologic findings, treatment outcomes, and safety profiles in patients with AS, rheumatoid arthritis, and psoriatic arthritis treated with biologics. 10 The KOBIO registry enrolled patients at the time point of initiating a new biologic agent or switching to another agent. The following agents were included in the analysis: etanercept, infliximab (originator and biosimilar), adalimumab, and golimumab. Treatment decisions were made at the discretion of the attending physician. Data collection was done at enrollment and yearly thereafter. Adverse events, disease activity, medications, etc. were documented at each follow-up. If the subject switched or stopped the biologic agent, the data were obtained at the time point of the event. The biologics can be prescribed to patients with AS who had an inadequate response (BASDAI ⩾ 4) despite treatment with two NSAIDs within a 3-month period according to the National Health Insurance System.
The study population consisted of patients with AS enrolled from December 2012 to December 2018. Patients were required to meet the modified New York criteria for AS at enrollment to the KOBIO registry. Baseline demographic data and clinical variables such as extra-articular manifestations, HLA-B27 positivity, family history of spondyloarthritis, ASDAS-CRP, the BASDAI, and Bath Ankylosing Spondylitis Functional Index (BASFI) values were collected from the database. The disease activity indices and the status of biologic agent use were followed yearly after the initiation of biologic therapy. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 11 (Supplemental Material).
Definition of patient subgroups
The patients were grouped according to their disease activity status. The cutoff level for high disease activity was ⩾2.1 for ASDAS-CRP and ⩾4 for BASDAI, and the low disease activity cutoff was <2.1 for ASDAS-CRP and <4 for BASDAI, respectively. One-year ASDAS-CRP or BASDAI responders were defined as biologics-treated patients who achieved a low disease activity at their 1-year follow-up.
Statistical analysis
Statistical analyses were performed using SPSS 26.0 (SPSS Inc., Chicago, IL, USA). Missing data were not imputed. Pearson’s correlation coefficient was used for correlation analysis. For the categorical variables, the significance of the differences across the groups was analyzed using the χ2 test or Fisher’s exact test. Continuous variables are expressed as the mean ± standard deviation (SD) or median (range) and analyzed using the two-sample t-test or the Mann–Whitney test. A p value of <0.05 was considered to indicate statistical significance.
Results
Baseline characteristics and discrepancies between ASDAS-CRP and BASDAI
Data from 1773 registered patients were analyzed. The mean (±SD) age was 38.5 ± 13.1 years, and 76.8% of the subjects were males. The baseline characteristics are summarized in Table 1. The most prescribed drug was adalimumab (n = 703, 39.7%), followed by golimumab (n = 387, 21.8%), infliximab biosimilar (n = 264, 14.9%), and etanercept (n = 135, 7.6%). All patients initiated or switched to a biologic agent at baseline; 22.6% of the patients were ⩾ second-line users. The Pearson’s correlation coefficient between ASDAS-CRP and BASDAI was 0.71 (p < 0.001) (Supplemental Figure 1). When the patients were divided into two groups based on BASDAI at baseline (low versus high), the clinical and laboratory features differed largely between the two (Table 1).
Baseline characteristics of patients based on BASDAI.

BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BMI, body mass index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IBD, inflammatory bowel disease; SpA, spondyloarthritis.
Data are expressed as mean ± SD or n (%).
p Values were calculated by the Mann–Whitney test or chi-square test.
At baseline, 269 (15.2%) patients had a low BASDAI. At the same time, 56.1% of these patients had a high ASDAS-CRP (Table 2). By contrast, among 142 (8.0%) patients with a low ASDAS-CRP at baseline, 16.9% of the patients had a high BASDAI. On the contrary, only 8.5% of the patients had a high ASDAS-CRP despite a low BASDAI at baseline. The kappa value was 0.52 between ASDAS-CRP and BASDAI (p < 0.001). A high ASDAS-CRP was more sensitive than a high BASDAI in detecting patients who initiated TNFi or switched to another agent (high ASDAS-CRP, 92.5% versus high BASDAI, 84.8%, χ2 = 44.4, p < 0.001).
Disease activity status at baseline stratified by BASDAI and ASDAS.
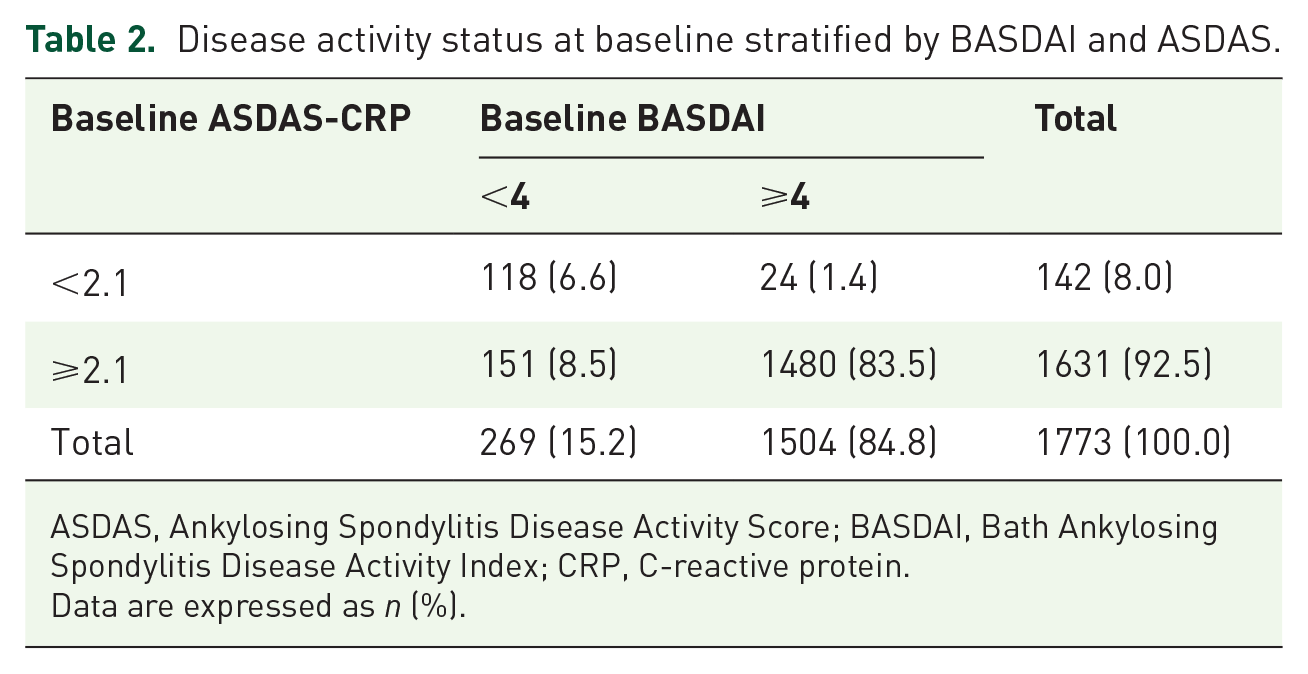
ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein.
Data are expressed as n (%).
Characteristics of patients with low BASDAI at baseline and follow-up
We next analyzed the characteristics of patients with a low BASDAI at baseline along with their ASDAS-CRP status (Supplemental Figure 2 and Supplemental Table 1). The high ASDAS-low BASDAI subgroup at baseline had a greater male:female ratio and higher baseline ESR and CRP levels and BASFI values than the low ASDAS-low BASDAI subgroup (Table 3).
Characteristic of patients with a low BASDAI at baseline based on ASDAS.

ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; BASFI, Bath Ankylosing Spondylitis Functional Index; BMI, body mass index; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; IBD, inflammatory bowel disease; SpA, spondyloarthritis.
Data are expressed as mean ± SD or n (%).
p Values were calculated by the Mann–Whitney test or chi-square test.
On follow-up of patients with a low BASDAI at baseline, there was no difference in the ASDAS-CRP, BASDAI, or BASFI values up to 5 years between the high and low ASDAS subgroups at baseline. Although most patients reached low disease activity (both ASDAS-CRP and BASDAI) on follow-up, 8.3% of the patients in the first year and 14.3% of the patients in the fourth year of follow-up maintained high disease activity. Moreover, over 10% of the patients continued to have a high ASDAS-CRP value despite a low BASDAI in the fourth and fifth year of follow-up (Figure 1).

Disease activity changes in patients with low BASDAI at baseline during follow-up.
Predictability of treatment response in the first-year ASDAS-CRP in BASDAI responders
We examined ASDAS, BASDAI, and the drug retention rate at 2 years stratified by the 1-year ASDAS or BASDAI response. Both 1-year ASDAS responders and 1-year BASDAI responders had a significantly higher percentage of patients with BASDAI < 4.0 and ASDAS <2.1 than non-responders, and a better drug retention rate at 2 years (Supplemental Table 2). Next, all patients were divided into two groups based on their ASDAS-CRP at 1 year: ASDAS-CRP <2.1 (i.e. ASDAS responders) and ASDAS-CRP ⩾2.1 (i.e. ASDAS non-responders). We found that ASDAS non-responders despite having a low BASDAI (<4.0) at 1 year of TNFi treatment had a poor clinical response thereafter (Supplemental Table 3). Upon tracking subsequent changes in ASDAS-CRP and BASDAI in the patients, the ASDAS response in the BASDAI responders was closely linked to a good treatment response afterward. In patients with a BASDAI response at 1 year, both the ASDAS-CRP and BASDAI values at follow-up were higher in the ASDAS-CRP non-responders than in the ASDAS-CRP responders (second year to the fifth year). The yearly changes in ASDAS-CRP and BASDAI values in the two groups are shown in Supplemental Table 4.
To verify the usefulness of ASDAS-CRP at 1 year, we compared the switching rate of biologics between ASDAS responders and ASDAS non-responders in BASDAI responders (Supplemental Table 5 and Figure 2). There was a significant difference in the switching rate for the following year: 27/681 (4.0%) of the ASDAS responders and 13/131 (9.9%) of the ASDAS non-responders switched to another agent (χ2 = 8.33, p < 0.01). In terms of drug survival, 82.5% of the ASDAS responders and 68.7% of the ASDAS non-responders retained their biologic agent (χ2 = 13.27, p < 0.001) for the following year. This difference was not found in the third to the fifth years (Figure 2). Taken together, the 1-year ASDAS-CRP was able to help predict the retention of biologic therapy in the following year in BASDAI responders.

Drug survival of biologic therapy in BASDAI responders stratified by ASDAS responders and non-responders (*p < 0.01).
Discussion
Disease activity indices have become essential tools not only for assessing patient status but also for modulating ongoing therapy. We demonstrated that the ASDAS-CRP and BASDAI values correlated well in patients with AS starting biologic therapy. However, 8.5% of the patients had high ASDAS-CRP despite a low BASDAI at baseline. More patients had a higher disease activity according to ASDAS-CRP compared with BASDAI when TNFi was started or switched to another agent. Among patients with a low BASDAI at baseline, the treatment response to biologics was not different from patients with high or low ASDAS at baseline. However, in the BASDAI responders at 1 year, the disease activity indices at follow-up differed significantly between ASDAS responders and non-responders. The rate of switching or drug survival of the initial biologic agent was significantly different for the following year.
The ASDAS has been utilized and validated in many studies.9,12,13 ASDAS was better associated with structural progression 8 and inflammatory, cartilage, and bone biomarkers than BASDAI. 14 On the other hand, some studies favored BASDAI over ASDAS.15,16 A multicenter study in Spain reported that the BASDAI had better validity than ASDAS, disclaiming the advantages of ASDAS over BASDAI. 15 Another study demonstrated that the ASDAS showed low internal consistency. 16 There is also a controversy regarding the ASDAS cutoff point. A study showed that ASDAS-CRP and BASDAI values that indicate a patient-acceptable symptom state are <2.3 and <4.1, respectively. 17 From this point of view, the ASDAS cutoff point of 2.1 for low disease activity is relatively more stringent than the BASDAI cutoff point of 4. Symptoms represent the patient’s perspective of active disease, whereas ASDAS, which incorporates laboratory indicators of inflammation, reflects disease activity from the physician’s perspective. 18 Recently, a quick quantitative CRP testing was applied to assess disease activity in axial spondyloarthritis (ASDAS-qCRP), allowing for immediate decision-making in clinical practice with the goal of treat-to-target. 19 BASDAI is preferred in clinics because it is less time-consuming than ASDAS-CRP. However, ASDAS-qCRP, which is simple to perform and includes CRP, may become a new tool used in daily clinical practice.
Moreover, the ASDAS could also help in assessing the response to therapy. 2 Our study demonstrates that ASDAS-CRP could capture more patients at the time of starting biologic treatment. Clinical trials of biologics have also shown that the proportion of patients with a high ASDAS is greater than that of patients with a high BASDAI. 20 Fagerli et al. compared ASDAS and BASDAI in selected patients treated with a TNFi. In that study, 16.6% of the patients had a high ASDAS and a low BASDAI, 21 which was similar to our results. Overall, 8.5% of patients in our cohort had a low BASDAI and a high ASDAS-CRP, while more than half of the patients with a low BASDAI had a high ASDAS-CRP at baseline. A previous study showed that 38.8% of AS patients with a low BASDAI still had a high ASDAS-CRP at 3 months after starting TNFi therapy. 22 A Chinese cohort study demonstrated that the performance of ASDAS was better than BASDAI in patients with high inflammatory markers while the performance of both indices was comparable in patients with low inflammatory markers. 23 Along with the data of our study, this indicates that ASDAS-CRP is very helpful in identifying patients with high inflammatory markers.
Our study also showed that the ASDAS responses at 1 year could predict drug persistence during follow-up among patients with a low BASDAI at 1 year. A previous study demonstrated that a high ASDAS was linked to responsiveness to TNFi, whereas a high BASDAI was associated with treatment discontinuation. 9 In a retrospective cohort study, despite TNFi therapy for 3 months, patients with a low BASDAI/high ASDAS had a higher risk of discontinuing biologics at 9 months than those with a low BASDAI/low ASDAS. 22 However, the ASDAS at 3 months after biologic therapy may be a timepoint too early to predict the long-term response to therapy. A recent study demonstrated a treat-to-target strategy to achieve ASDAS <2.1 was more beneficial in efficacy outcomes of ASDAS low disease activity and ASAS 40 than a usual care strategy in patients with axial spondyloarthritis that were biologic-naïve. 24 Overall, the ASDAS provides important clinical information about patients with AS beyond complementing BASDAI.
Despite that our study was based on a nationwide registry, there are some limitations. The study population was mainly based on patients that initiated biologic therapy, thus skewed to subjects with high disease activity at baseline as measured by BASDAI. Therefore, our results could not encompass the utility of ASDAS in patients with low disease activity persistently. Second, some CRP values were missing in a few patients at follow-up visits. Meanwhile, CRP levels may be associated with the HLA-B27 status. 25 Nevertheless, the proportion of positive HLA-B27 was comparable between high and low disease activity groups in this study. Last, we were not able to investigate the consequences of structural damage in patients with discrepancies in ASDAS and BASDAI.
Conclusion
In summary, the ASDAS-CRP captured more patients who were about to initiate biologic therapy than BASDAI did. Moreover, the ASDAS responders among the BASDAI responders at 1 year had better persistence in biologic therapy than the ASDAS non-responders. Therefore, ASDAS could provide further valuable information to physicians who treat patients on biologic therapy.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X231201714 – Supplemental material for The predictability of ASDAS on drug survival in patients with ankylosing spondylitis on biologic therapy: data from the KOBIO registry
Supplemental material, sj-docx-1-tab-10.1177_1759720X231201714 for The predictability of ASDAS on drug survival in patients with ankylosing spondylitis on biologic therapy: data from the KOBIO registry by Jinhyun Kim, Min Jung Kim, Geun Young Oh, Sun Kyung Lee, Taeeun Kim and Kichul Shin in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
