Abstract
Objectives
To investigate the relationships between inflammation, nocturnal back pain and fatigue in ankylosing spondylitis (AS) and the impact of 12 weeks' etanercept treatment versus sulfasalazine or placebo.
Methods
Data were combined from four clinical trials for patients with AS who received at least one dose of etanercept, sulfasalazine or placebo and had at least one postbaseline assessment value. Linear regression was performed (controlling for site, protocol and demographics), to explore the relationship between inflammation (C-reactive protein [CRP]), nocturnal back pain (visual analog scale [VAS] 0–100 mm) and fatigue (VAS 0–100 mm Bath AS Disease Activity Index fatigue item).
Results
Out of 1283 patients (etanercept,
Conclusions
AS patients treated with etanercept demonstrated superior improvement in nocturnal back pain and fatigue versus sulfasalazine or placebo. Decrease in nocturnal back pain can improve fatigue. Assessing treatment response using CRP levels alone may be misleading without also examining patient-reported outcomes such as back pain and fatigue.
Introduction
Ankylosing spondylitis (AS) is characterized by inflammation of the sacroiliac joints and spine, with nocturnal back pain and fatigue being chief complaints; AS is associated with impaired functioning and reduced health-related quality of life.1,2 Nocturnal back pain is a primary symptom of AS, with 62–63% of patients also experiencing severe fatigue.3,4 At the cellular level, measurements of inflammation biomarkers such as C-reactive protein (CRP) are used in diagnosis and are components of outcome assessments and predictors of treatment response.5–7 Relationships between inflammation, back pain and fatigue have not been well established.
The AS Disease Activity Score (ASDAS) utilizes both back pain and CRP in its algorithm to help assess the overall AS disease state at any given timepoint. 8 Independently, back pain and CRP levels at the start of treatment are strongly correlated with improvement in treatment response (ASDAS) and a 20% improvement in the Assessments of the SpondyloArthritis International Society criteria (ASAS).9,10 In addition, CRP level at the start of treatment has been strongly associated with a 50% improvement in the Bath AS Disease Activity Index (BASDAI), of which back pain is a component. 11 Elevated CRP levels at baseline have been associated with an effective response to anti-tumor necrosis factor (TNF) therapy, achievement of remission by various functional and disability component indices and continuation of therapy.5–7
As an independent variable, fatigue has been under-studied in AS; it has mainly been researched as an item of composite measures such as the BASDAI.4,5 The aetiology of fatigue in AS remains elusive, yet a number of studies have shown that nocturnal back pain and disease activity are strongly related to fatigue in patients with AS.3,4,12–15 Despite these findings, fatigue continues to be routinely overlooked in clinical practice as a treatable AS symptom, even though it has been reported as the third most common complaint.4,14 Although CRP, back pain and fatigue are used independently and as variables in composite measures to assess AS disease state and treatment efficacy, the relationship between CRP and back pain has not been well characterized.
Nonsteroidal anti-inflammatory drug (NSAID) therapy is the first-line treatment for AS and reports are varied regarding its effect on pain and fatigue. Patients have reported that NSAID therapy has alleviated their fatigue symptoms, but that it had a greater effect on pain and functional impairment than on fatigue.1,16 Disease-modifying anti-rheumatic drugs are other conventional treatments that may be associated with greater improvements in fatigue compared with other traditional therapies, although their effect on pain is variable and largely ineffective in those with axial disease.14,17 Biologic therapies, such as the fully-human TNF-soluble receptor, etanercept, tend to be more effective in improving the signs and symptoms of AS (including inflammation, nocturnal back pain and fatigue) in controlled studies.18–22
In this novel analysis, using data derived from several controlled clinical trials of etanercept in patients with active AS, relationships between inflammation (measured by CRP levels), nocturnal back pain and fatigue were investigated, and the impact of 12 weeks’ etanercept treatment was evaluated. Understanding these relationships may help clinicians in making evidence-based decisions and maximize treatment benefits for patients with AS.
Patients and methods
Study population and design
Summary of four clinical trials of ankylosing spondylitis (AS), undertaken in patients treated with etanercept (ETN), sulfasalazine (SSZ) or placebo, included in the current pooled analysis.

BIW, biweekly; s.c., subcutaneously; QW, once weekly; p.o., orally; BID, twice daily; VAS, visual analog scale; BASFI, bath ankylosing spondylitis functional index; BASDAI, bath ankylosing spondylitis disease activity index; NSAIDs, nonsteroidal anti-inflammatory drugs.
The study designs were qualitatively consistent across trials. A week-12 endpoint was selected for the pooled analysis as it was shared by all four trials. Data for the etanercept 50-mg weekly and 25-mg twice-weekly dosing groups were combined to create one etanercept treatment group for analysis.
These four studies had been conducted in compliance with the Declaration of Helsinki, the International Conference on Harmonization Good Clinical Practice guidelines and all applicable local/country specific regulations. Before the start of each study, independent ethics committees or institutional review boards in each country reviewed and approved each study, and written informed consent was received from all participants.
Symptom assessment
Nocturnal back pain was assessed using a single-item visual analog scale (VAS), with scores ranging from 0–100 mm, where 0 mm represented no pain and 100 mm represented worst possible pain. Fatigue was assessed using the fatigue item of the BASDAI, with scores ranging from 0–100 mm on a VAS (0 mm represented no fatigue; 100 mm represented the worst possible symptoms of fatigue). 26 CRP levels were assessed as an objective measure of inflammation using the standard procedures of centralized laboratories for each clinical trial. The presence of the human leukocyte antigen (HLA)-B27 allele is strongly associated with a diagnosis of AS and was assessed using the standard procedures of centralized laboratories (polymerase chain reaction) for each clinical trial. Nocturnal back pain, fatigue and CRP levels were assessed at baseline and at week 12.
Statistical analyses
Descriptive statistics and the correlations (Pearson’s correlation coefficient) between variables were calculated for baseline, week 12 and change from baseline to week 12. Linear regression was performed, controlling for pooled site, protocol and demographic characteristics (i.e., age, sex and disease duration), to explore the relationship between nocturnal back pain and fatigue. The statistical tests included paired
Results
In total, 1283 patients (etanercept, Relationship between change in nocturnal back pain (visual analog scale [VAS] 0–100 mm) and change in fatigue (VAS 0–100 mm) from four pooled clinical trials in patients with ankylosing spondylitis (treatments in the four trials included: etanercept 25–50 mg subcutaneously once or twice weekly; sulfasalazine 1.5 g orally twice daily; placebo twice weekly). Baseline demographic and disease characteristics of four pooled clinical trials in patients with ankylosing spondylitis (AS; treatments in the four trials included: etanercept 25–50 mg subcutaneously once or twice weekly; sulfasalazine 1.5 g orally twice daily; placebo twice weekly). Data presented as One-way analysis of variance with treatment as a factor; bexact Mantel–Haenszel χ2-test after combining black and other; cexact Mantel–Haenszel χ2-test. BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; VAS, visual analog scale; CRP, C-reactive protein; HLA-B27 Pearson’s correlation coefficients for nocturnal back pain (visual analog scale [VAS] 0–100 mm) and fatigue (VAS 0–100 mm) at baseline, week 12 and change from baseline. Data analyzed from four pooled clinical trials in patients with ankylosing spondylitis (treatments in the four trials included: etanercept 25–50 mg subcutaneously once or twice weekly; sulfasalazine 1.5 g orally twice daily; placebo twice weekly).


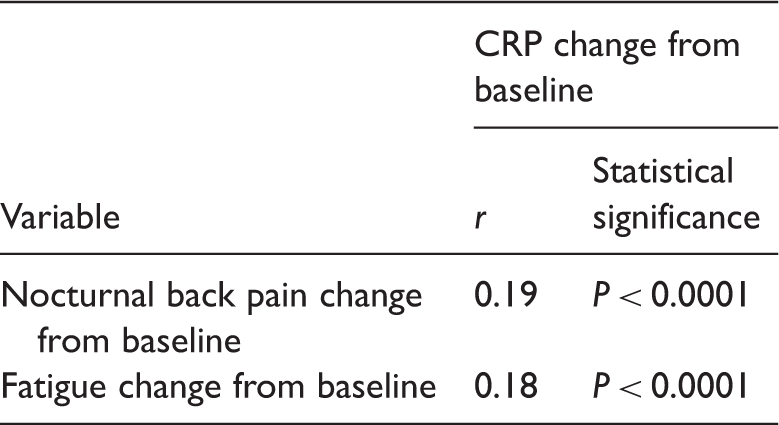
Pearson’s correlation coefficients for change from baseline in nocturnal back pain (visual analog scale [VAS] 0–100 mm), fatigue (VAS 0–100 mm) and C-reactive protein (CRP) levels from four pooled clinical trials in patients with ankylosing spondylitis (treatments in the four trials included: etanercept 25–50 mg subcutaneously once or twice weekly; sulfasalazine 1.5 g orally twice daily; placebo twice weekly).
Etanercept provided significantly greater improvements from baseline to week 12 in pain and fatigue than either sulfasalazine or placebo (Figure 2). Nocturnal back pain was improved by 58% in the etanercept group, compared with 38% and 16% in the sulfasalazine and placebo groups, respectively ( Effects of treatment on symptoms of nocturnal back pain (visual analog scale [VAS] 0–100 mm) and fatigue (VAS 0–100 mm) at week 12 from four pooled clinical trials in patients with ankylosing spondylitis (treatments in the four trials included: etanercept [ETN] 25–50 mg subcutaneously once or twice weekly; sulfasalazine [SSZ] 1.5 g orally twice daily; placebo twice weekly). 
Discussion
Fatigue is a major common symptom experienced by the majority of patients with AS; this burden of disease leads to a significantly larger impact on their lives than in the general population.1,13,27,28 Fatigue has been shown to be severe (BASDAI fatigue item ≥50 mm) in 62–63% of patients with AS and has been reported as the third most common symptom after pain and stiffness.3,16
Data presented in the current study, from a pooled population of patients with AS, show nocturnal back pain – one of the major symptoms of AS – to be strongly correlated with fatigue. This is most likely because of nocturnal pain disrupting restorative and high-quality sleep, thus contributing to the severity of fatigue during waking hours. In the current study, improvement in nocturnal back pain with treatment was associated with reduced fatigue.
Improvements in both nocturnal back pain and fatigue were shown to have weak, yet statistically significant, relationships with improvement in inflammation, most likely revealed by the large sample size in the current study. These results complement other findings of weak relationships between fatigue and inflammation and between back pain and inflammation in patients with AS.3,16,29
Etanercept was shown to be superior to sulfasalazine in reducing nocturnal back pain and this finding was mirrored by a similar decrease in fatigue. In addition, etanercept provided improvement in fatigue, even after controlling for its effects on nocturnal back pain.
This post hoc analysis was limited in utilizing only one question/measure of fatigue (from the BASDAI scale), which may not have provided the full extent and details of the patients’ fatigue. 26 In addition, the short observation period prevented any long-term analysis of changes in fatigue. The original studies from which the data for this study were drawn were not intended to evaluate the impact of AS on the effects of fatigue on mood and family interactions. Sleep studies, hygiene and logs were not collected, nor were other mitigating factors addressed (such as level of education, consumption of caffeine-containing drinks and work [shift work/night work or manual labour]). Additional studies are warranted to more comprehensively assess the aetiology and consequences of fatigue in patients with AS.
Although studies have reported relationships between various patient-reported measures and fatigue, we believe this is the first analysis that has shown nocturnal back pain to be predictive of fatigue in this population of patients with AS. Patients treated with etanercept demonstrated a superior improvement in both nocturnal back pain and fatigue compared with sulfasalazine and placebo. Laboratory measures of disease inflammation (CRP levels) were not related to changes in nocturnal back pain or fatigue, indicating that clinicians should not rely solely on CRP levels to assess disease activity. Attention should also be focused on the patient’s report of nocturnal back pain, especially given its link to fatigue and the possible subsequent effects on daily life.
Footnotes
Declaration of conflicting interest
Potential conflicts of interest include the following: M Hammoudeh is a clinical investigator of ankylosing spondylitis clinical trials sponsored by Pfizer Inc; DJ Zack is a former employee of Amgen; VM Stewart and AS Koenig are current employees of Pfizer Inc; Previously, W Li was an employee of PharmaNet/i3, who were paid contractors to Pfizer Inc in the development of this manuscript. All authors participated in the development of this report and approved the final content for publication.
Funding
The studies included in this analysis were funded by Wyeth Pharmaceuticals, Inc., which was acquired by Pfizer Inc. in October 2009. Medical writing support was provided by Stephanie Eide of UBC Scientific Solutions and funded by Pfizer Inc.
