Abstract
Background:
Major improvements in the management of rheumatoid arthritis (RA) have made clinical remission an achievable and desirable goal but, despite the relevance gained by a profound disease suppression, many patients with RA still miss clinical remission due to several factors influencing disease activity, including treatment adherence.
Objective:
To evaluate the effect of adherence to conventional synthetic disease-modifying anti-rheumatic drugs (csDMARDs) on the achievement of clinical remission in a cohort of patients with new-onset inflammatory arthritis.
Study design:
A prospective cohort study was conducted using the ELECTRA database, which consists of clinical data from patients followed at the IRCCS Policlinico San Matteo Foundation (Pavia, Italy), linked to regional administrative healthcare databases.
Methods:
We enrolled patients with new-onset active disease between January 2006 and December 2013 and followed them until their first clinical remission or end of follow-up (December 2015). To assess the association of csDMARD adherence with clinical remission, we estimated the csDMARD proportion of days covered (PDC) during follow-up. PDC was added to the main clinical adjustment covariates as a time-dependent variable in a proportional hazard Cox regression model.
Results:
The cohort included 324 patients with a mean (SD) age of 58 (13.9) and predominantly female (74.5%). A total of 219 patients (67.6%) achieved clinical remission during follow-up and 85 (26.2%) in the first 6 months (early clinical remission). Cox regression models showed that a 10% increment of PDC increased the probability of achieving clinical remission by 10% (p < 0.001) and the probability of early clinical remission by 21% (p = 0.03).
Conclusion:
Patients at disease onset with higher adherence to csDMARDs were more likely to achieve clinical remission and early clinical remission. Our study highlighted the importance of close monitoring of patients to increase their likelihood of following therapeutic indications and achieving favorable disease outcomes, such as lower disability.
Background
Major improvements in the management of rheumatoid arthritis (RA) have made clinical remission an achievable and desirable goal1,2: in fact, reaching a deep control of disease activity has been consistently associated with better disease-specific and long-term outcomes, such as functional disability.3,4 Despite the relevance gained by a profound disease suppression, however, many patients with RA still miss clinical remission due to several factors influencing disease activity, including treatment adherence. Medication adherence can be defined as the degree to which a patient’s behavior, related to taking medications, reflects the recommendation of the healthcare provider. Adherence includes several different aspects, such as persistence of treatment, initiation adherence, and execution adherence, and it is known to be poor in chronic medical conditions, with about 50% of subjects being non-adherent to the prescribed treatment. 5 Patients with RA make no exception: several studies have assessed adherence to conventional synthetic and biologic disease-modifying anti-rheumatic drugs (cs and b-DMARDs), reporting rates ranging from underuse (22%) to overuse (7%).6,7 In particular, the attitude toward medication intake seems to exert influence on disease activity, with higher adherence leading to better disease control8–10 and lower disability. 11 Several interventions have been attempted to try and improve adherence in RA, including patient education 12 and new technologies,13,14 mostly with suboptimal results.
While most of the available evidence on medication adherence refers to longstanding RA, even subjects with recent onset disease might take treatment with discontinuity, 15 and it has been demonstrated that this might finally lead to increased healthcare costs. 16 Despite the relevance of this issue, information on the adherence in the setting of early and tailored treatment of RA, a type of approach that is strongly supported by the current recommendations, 1 is still scarce, and this also applies to the context of Early Arthritis Clinics (EACs).
Adherence to medication can be assessed by different means, including patient-reported measures, tablet counts, and electronic monitoring devices. In this context, administrative healthcare databases (AHD) constitute a useful tool to measure medication intake by measuring pharmacy refills. The advantages of AHD include the possibility to assess large samples of patients with complete data for long periods, although their main limitation is represented by the poor information on clinical features. 17 To overcome this limitation, the AHD can be linked with clinical databases, to obtain more detailed clinical variables.
The purpose of this study was to evaluate the effect of adherence to csDMARD on the achievement of clinical remission in a cohort of patients with new-onset inflammatory arthritis treated according to a treat-to-target strategy, in the specific setting of an EAC, by a record linkage of clinical databases and AHD. Moreover, we focused on the effect of adherence to csDMARD on early clinical remission (<6 months from diagnosis).
Patients and methods
Study design and setting
This is a prospective cohort study performed using the ELECTRA database. The reporting of this study followed the STROBE guidelines. 18 This database included all consecutive patients with new-onset RA or undifferentiated arthritis (UA) from the EAC of the IRCCS Policlinico San Matteo Foundation, Pavia, Italy19,20 during the period 2004–2015. The EAC was instituted to include consecutive patients, referred by general practitioners, with new-onset inflammatory arthritis (symptom duration <12 months) from the Pavia district (Italy). Details about diagnostic criteria as well as inclusion and exclusion criteria were reported elsewhere, the EAC was the first early arthritis clinic instituted in Italy and in this setting the principles of treat-to-target and tight control were applied to all patients. 19 Clinical data, such as disease activity, prescribed drug therapy [with relative dose, prescribed daily dose (PDD)] and laboratory tests results on the recruited patients were recorded in the ELECTRA database. This information was completed with data from the AHD of the Pavia district using a linkage based on a single anonymized identification code. The AHD recorded all healthcare access in the Pavia population covered by the National Health Service (NHS). The system of AHD included 1 the following: an archive of demographic data 2 ; an archive of all hospital discharge forms (HDFs) including information on diagnoses and procedures coded according to the International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) and Disease-Related Group 3 ; a database providing information on outpatient NHS-refundable drug delivery, coded according to the Anatomical Therapeutic Chemical classification system, including also the ‘autorizzazione immissione in commercio’ (AIC) code 4 ; an archive containing all the certifications of chronic diseases for the exemption from copayment 5 ; an archive of outpatient services (e.g. diagnostic test, consultations). 19
Participants
We enrolled patients aged 18 years and older, with RA or UA and with disease onset between 2004 and 2015, included in the ELECTRA database. We excluded patients (i) with disease onset before January 2006 to ensure 2 years for the measurement of other risk factors; (ii) with disease onset after December 2013 to guarantee at least 2 years of follow-up; (iii) with prescription of glucocorticoids in the 2 years before disease onset, to reduce the likelihood of including cases with a diagnostic delay; and (iv) in remission at inclusion date.
The included patients remained in the cohort until they achieve the first among disease remission (see Predictors and outcome paragraph), 31st December 2015, emigration, death, or loss to follow-up visits.
Predictors and outcome
For each patient, all prescriptions of csDMARDs during follow-up were extracted. To calculate the coverage of each prescription, we considered the AIC code to identify the number of milligrams of active substance contained in each drug box. The number of milligrams was multiplied by the number of drug boxes that were purchased by the subject, and this quantity was divided by the defined daily dose (DDD). In case of overlapping of some prescriptions, the covered days were considered only once. The cumulative proportion of follow-up covered by DMARDs treatment (PDC) during follow-up was obtained by dividing the number of days covered by the follow-up days and multiplying this ratio by 100. The coverage for each AIC is reported in Supplemental Material S1.
The outcome of interest was the achievement of clinical remission, defined by Disease Activity Score-28 (DAS28) < 2.6, at any time during the follow-up and, as a separate secondary outcome, during the first 6 months.
Covariates
To characterize the recruited patients, we considered their sociodemographic and clinical profile through the following variables: sex, age, DAS28 at recruitment, and the Charlson Comorbidity Index (CCI). The last one was estimated from HDFs in the 2-year period before recruitment. This index was dichotomized into two classes (CCI = 0 and CCI > 0). The study design is shown in Figure 1. The ICD-9-CM codes used to estimate CCI are reported in Supplemental Material S2.

Study design.
Statistical methods
Continuous variables were reported as mean [±standard deviation (SD)] or as median (interquartile range) for non-normally distributed data. Categorical variables were reported as absolute and relative frequencies. To assess the association between the proportion of days covered (PDC) and disease remission, a proportional hazard Cox regression model was performed. We included in this model PDC as a time-dependent variable and clinical covariates measured at recruitment as fixed adjustment variables. Association estimates were reported as hazard ratios (HRs) and relative 95% confidence intervals. An analysis taking into account the subset of patients diagnosed with RA was also performed. To evaluate the effect of PDC on early clinical remission (<6 months from diagnosis), a Cox regression model with robust variance was performed to directly estimate the relative risk (RR) in the presence of an unrare outcome. 20
In this study, we did not use the PDD to estimate the PDC because of missing data. A sensitivity analysis was performed to verify a possible misclassification bias introduced using PDC based on DDD. In brief, for each patient, we calibrated the cumulative DDD with the cumulative PDD, considering the portion of follow-up in which the corresponding PDD was available. For each stratum defined by DMARD type (methotrexate and hydroxychloroquine) and the length of the intervals between visits (using the median time of 150 days as cutoff), we performed a linear model with cumulative PDD as an outcome and cumulative DDD as a regressor. We used the estimated linear model coefficients as weights to calibrate the PDC–DDD of each patient. In particular, we implemented a Cox regression model using the time-dependent PDC–DDD multiplied by the weight as a regressor.
All hypothesis tests were two-sided and p values for statistical significance were set at 0.05. All the analyses were performed using R statistical software (Foundation for Statistical Computing, Vienna, Austria) and Statistical Analysis System Software (version 9.4; SAS Institute, Cary, NC, USA).
Results
The cohort included 324 incident cases of inflammatory arthritis, enrolled between 2006 and 2013, with a mean age of 58 (13.9) and predominantly female (74.5%). A total of 219 patients (67.6%) achieved clinical remission during follow-up and 85 (26.2%) achieved early clinical remission. Patients enrolled in the cohort generally had good health conditions (CCI = 0 in 91.7% of patients) but with high disease activity (median DAS28 at recruitment equal to 4.6). Characteristics of enrolled patients are shown in Table 1. As a result of patients with UA initially receiving hydroxychloroquine, 37.7% of the cohort was treated with this drug. During the follow-up, 80 patients changed csDMARDs, of which 49/80 were on initial treatment with hydroxychloroquine and 28/80 on methotrexate. Methotrexate (42%), hydroxychloroquine (26%), and sulfasalazine were the most commonly prescribed second-line csDMARDs. Seven patients received bDMARDs during the follow-up.
Description of the study cohort at recruitment and during follow-up.

ACPA, anti-citrullinated protein antibody; CRP, C-reactive protein; DAS28, Disease Activity Score-28; DMARD, disease-modifying anti-rheumatic drug; ESR, erythrocyte sedimentation rate; GH, general health; IQR, interquartile range; MTX, methotrexate; PGA, Physician Global Assessment; RA, rheumatoid arthritis; SD, standard deviation; VAS, visual analogue scale.
The association between PDC and RA remission, obtained by the Cox regression model, as well as those obtained by sensitivity analysis, is shown in Table 2.
Results of Cox regression models.
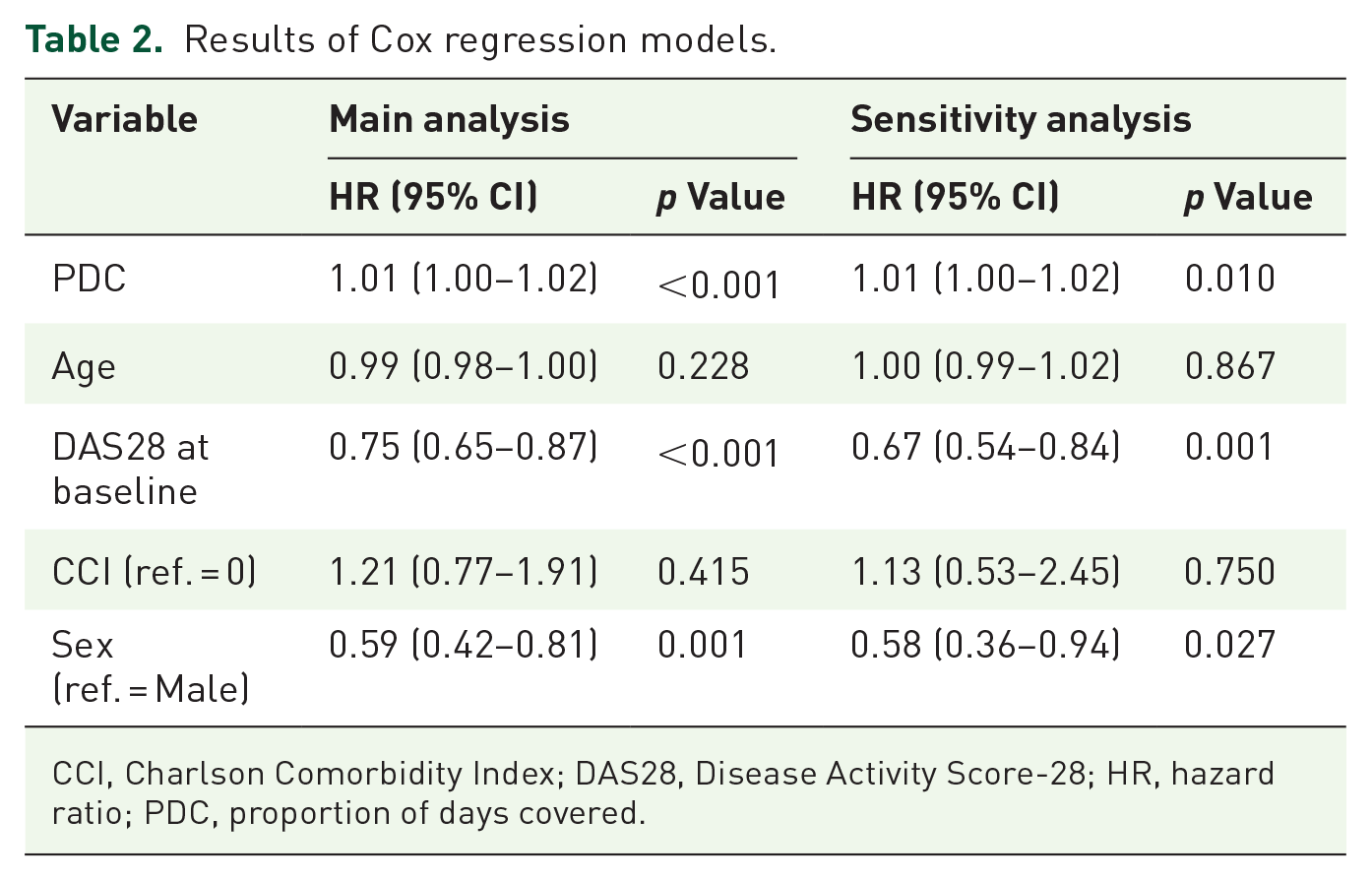
CCI, Charlson Comorbidity Index; DAS28, Disease Activity Score-28; HR, hazard ratio; PDC, proportion of days covered.
From the main analysis, PDC was significantly associated with disease remission with an HR of 1.01 (95% CI: 1.00–1.02, p < 0.001). It means that an increment of 10% in PDC is associated with an increment of 10% in the probability of achieving clinical remission. Female sex and DAS28 at recruitment were negatively associated with the outcome with an HR of 0.59 (95% CI: 0.42–0.81, p = 0.001) and an HR of 0.75 (95% CI: 0.65–0.87, p < 0.001), respectively. Similar results were obtained from sensitivity analysis. The weights calculated to calibrate the PDC–DDD were obtained by considering a cohort of 148 patients for whom PDD data were available. Each subject was classified into one of four strata, each of which had an associated weight. The strata with their respective weights were as follows: 0.40 for hydroxychloroquine and time between visits ⩾150 days; 0.69 for methotrexate and time between visits ⩾150 days stratum; 0.74 for hydroxychloroquine and time between visits <150 days stratum; and 0.82 for methotrexate and time between visits <150 days stratum.
When we considered only the subset of subjects diagnosed with RA, the results remained consistent, with an increase in PDC significantly associated with achieving remission [HR (95% CI): 1.01 (1.01–1.02), p < 0.001].
The analysis related to the early clinical remission confirmed the main analysis results with a RR of 1.02 (95% CI: 1.00–1.03, p = 0.03) for PDC. It means that an increment of 10% in PDC is associated with an increment of 21% in the probability of achieving early clinical remission. Female sex and DAS28 at recruitment were still significantly and negatively associated with the outcome.
Discussion
Clinical remission represents the ideal treatment goal in early RA, allowing better functional and radiographic outcomes,3,21,22 as well as greater survival, especially when reached early. 4 Since remission has become achievable with modern treatment strategies for RA, a greater interest in its predictors has been raised, to design personalized strategies from the early stages of the disease.
In this study, we analyzed a population of early inflammatory arthritis, including RA and UA, starting treatment with csDMARDs, whose overall features are in line with similar early arthritis populations. 23 To exclude subjects with a significant diagnostic delay, we excluded patients receiving glucocorticoids before disease onset. Our cohort was treated and followed according to a treat-to-target and tight control protocol, in the setting of an EAC, and concurrent information on prescriptions in the AHD was available, which allowed us to assess the overall csDMARD intake. The median PDC in the first year was 70% (data not shown), and this is in line with what could be expected in a chronic condition. 5
The PDC, as a measure of overall adherence, emerged as a significant predictor of clinical remission. The solidity of this finding was confirmed by the sensitivity analyses, taking the effect of different drugs and follow-up into account and considering a more selective outcome of early clinical remission. Several previous investigations have evaluated the impact of medication adherence on disease activity, reporting an overall effect in line with our findings. In a study performed by linking the Veteran Affairs Rheumatoid Arthritis database, constituted of patients with RA with a mean disease duration of 9 years, with the Veterans AHD, Cannon et al. 9 demonstrated a lower mean DAS28 in highly adherent patients, compared to the poorly adherent ones. In an analysis based on the Kyoto University Rheumatoid Arthritis Management Alliance cohort, the impact of self-reported adherence to cs-DMARDs was assessed in strata of patients defined by disease duration, although a group of DMARD-naïve patients was not available. Patients were included in the early RA group with a disease duration of less than 4.6 years; in this stratum, moderately and poorly adherent patients had an increased risk of flare at 1 year compared to the highly adherent ones (HR of 2.61 and 2.49, respectively), while this relationship was not evident in patients with longer disease duration, 24 confirming the relevance of medication adherence from the early stages. This seems to be of particular importance also in light of the finding, in a different population, of a positive effect of adherence on disease activity in new users of csDMARDs, which was not replicated in patients already taking csDMARDs. 25
In the more specific setting of early RA, the effect of adherence on clinical remission had previously been reported in an observational study, in which treatment persistence was self-reported by the patient during a structured interview and treated as a dichotomous outcome (non-persistence was defined when treatment was interrupted for at least 7 days). In this setting, persistence predicted sustained clinical remission, defined through the DAS28. 11 In the same cohort, moreover, poor self-reported adherence and persistence were significant predictors of flare within 6 months in patients that had already achieved clinical remission. 10
In our study, we tried to address all the aspects of adherence to medication (initiation, execution, and persistence) by assessing the PDC and performing sensitivity analyses including the PDD. We adopted an objective and comprehensive measure of medication acquisition by the patients, and, although the AHD tracks acquisition rather than consumption, we are confident to have provided a reliable measure of treatment adherence. While adherence in early RA has been previously defined mainly by dichotomous or categorical measures, in some cases lacking a full validation, we are also providing one of the first evaluations by a continuous measure. By these means, we were able to define the impact of behavior toward treatment at all levels of intake, ranging from very low to almost continuous intake.
In comparison to previous studies, moreover, to our knowledge this is the first analysis on the impact of adherence on disease activity in an early RA population followed and treated according to a standardized protocol, such as that applied in an EAC, reflecting current management recommendations. While the fact of including both patients with RA and UA might be regarded as a limitation, representing both groups supports the generalizability of our findings to the overall setting of early arthritis. Moreover, as differential diagnosis was carefully performed at inclusion, our cohort is composed of subjects with early onset persistent inflammatory arthritis, also reflecting a more modern concept of RA.
In our analysis, also a higher baseline disease activity and female gender determined the failure to subsequently reach clinical remission, which is consistent with previous reports, 26 while the impact of comorbidities was not significant, although a limited burden of comorbid conditions should be noted.
Our analysis carries some limitations. First, the results obtained in the Italian setting, where a universalistic healthcare system is available, are not generalizable to other countries, in which the socioeconomical status might have a greater impact on adherence. Measuring the prescription in the AHD implies an assessment of pill refill, but it does not measure the real drug intake by patients, as well as the precise dosage. Since only patients presenting at follow-up visits, likely experiencing a more severe disease, are taken into consideration, the final effect of the PDC might be underestimated. Moreover, we were not able to assess other relevant outcomes (such as sustained remission or disability) due to incomplete data in the clinical dataset and the limited time of observation. However, the strength of this study relies on the availability of joint clinical and administrative data, assuring detailed information on clinical features together with the completeness of the AHD, and on the inclusion of a homogeneous group of DMARD-naïve patients treated according to the current recommendations.
The current model of RA treatment includes a timely diagnosis, the prompt introduction of DMARDs, and an intensive follow-up. 1 All of these paradigms have been implemented following the results of strategic clinical trials and observational research based on the EACs.27,28 Although this type of approach has been strongly supported over the last two decades, in everyday clinical settings there are still barriers impeaching its full application, with several reports describing incomplete compliance to these principles by physicians treating RA.29–31 As the strategies whose efficacy has been so strongly shown in a research setting are only partially implemented by rheumatologists, and, at the following step, patients adhere only partially to rheumatologists’ indication, we can understand how the benefits demonstrated in a controlled environment are delivered to the patients with relevant attrition. This seems to apply particularly to early disease, where poorer treatment intake corresponds to poorer outcomes.
Conclusion
The results of our study provide further evidence to support the attempts for implementation of adherence in early RA. Further interventional studies will be needed to verify the impact of strategies to implement adherence on relevant clinical outcomes, including long-term aspects such as disability.
Supplemental Material
sj-doc-1-tab-10.1177_1759720X231194179 – Supplemental material for Impact of csDMARDs adherence on clinical remission in patients with new-onset inflammatory arthritis: a prospective cohort study from the ELECTRA database
Supplemental material, sj-doc-1-tab-10.1177_1759720X231194179 for Impact of csDMARDs adherence on clinical remission in patients with new-onset inflammatory arthritis: a prospective cohort study from the ELECTRA database by Anna Zanetti, Antonella Zambon, Carlo A. Scirè, Serena Bugatti, Carlomaurizio Montecucco and Garifallia Sakellariou in Therapeutic Advances in Musculoskeletal Disease
Supplemental Material
sj-docx-2-tab-10.1177_1759720X231194179 – Supplemental material for Impact of csDMARDs adherence on clinical remission in patients with new-onset inflammatory arthritis: a prospective cohort study from the ELECTRA database
Supplemental material, sj-docx-2-tab-10.1177_1759720X231194179 for Impact of csDMARDs adherence on clinical remission in patients with new-onset inflammatory arthritis: a prospective cohort study from the ELECTRA database by Anna Zanetti, Antonella Zambon, Carlo A. Scirè, Serena Bugatti, Carlomaurizio Montecucco and Garifallia Sakellariou in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
