Abstract
Background:
Psoriatic arthritis (PsA) is a chronic inflammatory condition predominantly affecting the peripheral joints. However, some patients with PsA can experience axial involvement which is manifested with back pain and associated with increased burden of illness.
Objectives:
The aim of this post hoc analysis was to determine the efficacy of ixekizumab (IXE) up to 52 weeks in reducing axial symptoms in PsA patients, presenting with axial manifestations.
Design:
This was a post hoc analysis of two pooled phase III clinical trials.
Methods:
Patients with axial manifestations, from two placebo-controlled, randomized, double-blind, phase III trials (SPIRIT-P1 and SPIRIT-P2), were defined as Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) Question 2 (Q2; back pain)] total score ⩾4 and average of BASDAI Q5 + Q6 (morning stiffness) ⩾4 at baseline. For this post hoc analysis, the efficacy of IXE was evaluated at weeks 16, 24, and 52 using separate BASDAI questions (including back pain and morning stiffness), total BASDAI and modified BASDAI (mBASDAI; without Q3), Ankylosing Spondylitis Disease Activity Score (ASDAS), and 50% improvement in BASDAI (BASDAI50) response. Treatment comparisons were performed using logistic regression and analysis of covariance model for categorical and continuous end points, respectively.
Results:
In the post hoc analysis among PsA patients with axial manifestations at baseline (N = 313), improvements in back pain and morning stiffness at weeks 16 and 24 were significantly greater in patients receiving IXE versus placebo (both p < 0.001). Improvements in BASDAI individual scores and total scores, mBASDAI, and ASDAS were significantly greater in patients receiving IXE compared with placebo. Similarly, significantly more IXE-treated patients achieved BASDAI50 at weeks 16 and 24 versus placebo. The effect of IXE was sustained at week 52. Similar effects were observed in sensitivity analyses subgroups.
Conclusion:
IXE is effective in improving axial symptoms in patients with active PsA presenting with axial manifestations.
Plain Language Summary
Introduction
Psoriatic arthritis is a chronic, immune-mediated condition with heterogeneous manifestations including peripheral arthritis, axial arthritis, enthesitis, dactylitis, and skin and nail psoriasis. However, some patients with PsA can also have axial involvement, which usually manifests with inflammatory back pain 1 and can contribute to an increased burden of illness. 2
The prevalence of axial manifestations in patients with PsA is thought to depend on disease duration. It has been reported to occur in 5–28% of patients with early-stage PsA and 25–70% of patients with long-standing PsA.3,4 Risk factors for the development of axial disease in patients with PsA include being positive for human leukocyte antigen (HLA)-B27, the presence of peripheral joint radiographic damage, and an increased erythrocyte sedimentation rate. 2 At present, there is no defined criteria to classify patients with PsA and axial involvement, and much of what is known about treatment has been derived from studies in radiographic-axial spondyloarthritis (r-axSpA). 5 The 2022 Group for Research and Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) treatment guidelines for PsA recommend the use of interleukin (IL)-17 inhibitors (IL-17i), tumor necrosis factor inhibitors (TNFi), and Janus kinase inhibitors (JAKi). 6
Ixekizumab (IXE) has demonstrated efficacy in multiple phase III trials in PsA, r-axSpA, and non-radiographic axial spondyloarthritis (nr-axSpA).7–11 The aim of this post hoc analysis was to determine the efficacy of IXE up to 52 weeks in reducing axial symptoms in patients with active PsA and axial manifestations.
Methods
Study design and participants
This integrated post hoc analysis included data from two patient subpopulations from the randomized, double-blind, phase III studies of IXE in biologic-naïve and TNFi-experienced patients with active PsA: SPIRIT-P1 (NCT01695239) and SPIRIT-P2 (NCT02349295). Specific details of the study design and patient populations have been previously described.12,13 Patients were randomized to placebo (PBO), adalimumab 40 mg (active reference arm up to week 24 in SPIRIT-P1 only), and IXE 80 mg every 2 weeks (IXE Q2W) or every 4 weeks (IXE Q4W). The analysis populations described in this article were not prespecified but were developed as part of the study design for the presented analysis.
Only patients from the intent-to-treat populations of SPIRIT-P1 and SPIRIT-P2 who were initially randomized to either IXE Q4W (approved label dose) or PBO at week 0 were included in the present analysis. Axial manifestations in PsA patients were defined as Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) Question 2 score (Q2, back pain) ⩾4 and an average of BASDAI Q5 + Q6 (intensity and duration of morning stiffness in the spine) ⩾4 at baseline. Patients included in the first sensitivity analysis (subgroup 1) were <45 years of age in addition to overall analysis criteria. Patients included in the second sensitivity analysis (subgroup 2) were <45 years of age and had elevated C-reactive protein (CRP; >5 mg/L) at baseline in addition to overall analysis criteria.
Efficacy end points
The efficacy of IXE on axial manifestations was analyzed using BASDAI Q2 (back pain) and the average of BASDAI Q5 and Q6 (inflammation). We also assessed total BASDAI score, modified BASDAI score [mBASDAI, without Q3 (peripheral arthritis)], and Ankylosing Spondylitis Disease Activity Score (ASDAS) change from baseline. In addition, we assessed BASDAI individual questions Q1 (fatigue), Q3, Q4 (enthesitis), Q5 (intensity of morning stiffness), and Q6 (duration of morning stiffness). The proportion of patients achieving an improvement in BASDAI Q2 score of ⩾2 and 50% improvement in BASDAI (BASDAI50) response at weeks 16, 24, and 52 was also assessed.
All six individual questions of the BASDAI were collected as prespecified end points in patients with axial manifestations at baseline, in SPIRIT-P1 and SPIRIT-P2 trials. The calculation of the mBASDAI, BASDAI50, and the ASDAS was exploratory end points.
Statistical analysis
Treatment comparisons at weeks 16 and 24 were performed using logistic regression with treatment and study as covariates for categorical end points (BASDAI50 response, improvement in BASDAI Q2 score of ⩾2, and the proportion of patients with BASDAI Q2 score <4), and an analysis of covariance model with treatment, study, and baseline values as covariates for continuous end points (BASDAI individual and total scores, mBASDAI score, and ASDAS) using nominal p-value. Observed data after week 16 for inadequate responders of week 16 were excluded from the analysis. Missing data for categorical and continuous end points were imputed by non-responder imputation (NRI) and modified baseline observation carried forward (mBOCF), respectively. When mBOCF was used, missing data were imputed by baseline value for patients who discontinued treatment due to an adverse event and by last post-baseline observation for the remaining patients. Data are summarized with least square mean and standard error for continuous end points and the proportion of patients for categorical end points. Statistical analyses were performed using SAS® Version 9.2 or higher (SAS Institute, Cary, NC, USA).
Results
Participants
A total of 313 patients were included in the present analysis: 313 patients in the main analysis population (IXE, N = 162 and PBO, N = 151), 97 patients in sensitivity analysis subgroup 1 (IXE, N = 56 and PBO, N = 41), and 63 patients in sensitivity analysis subgroup 2 (IXE, N = 33 and PBO, N = 30; Figure 1).
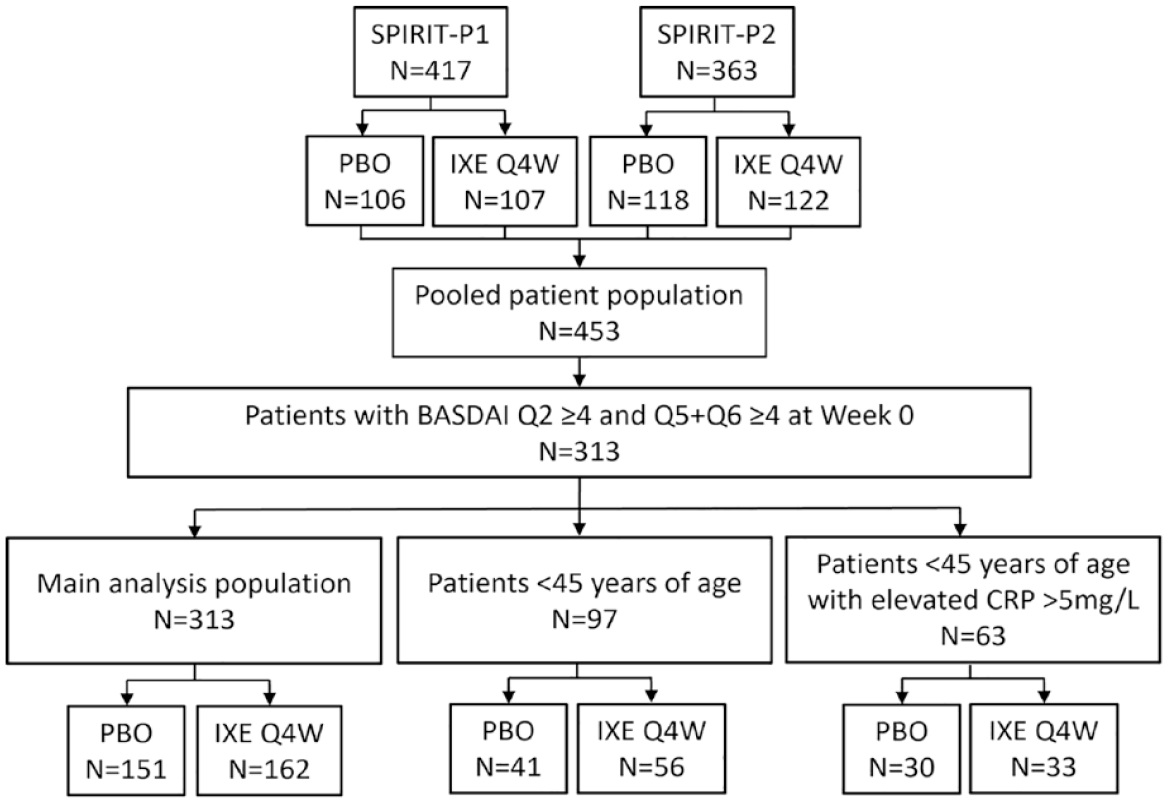
Flow chart of patients included in this post hoc analysis (patients with BASDAI Q2 ⩾ 4 and Q5 + Q6 ⩾ 4 at week 0) pooled from SPIRIT-P1 and SPIRIT-P2 trials.
Patient demographics and baseline disease characteristics for the three analysis populations studied are presented in Table 1. The mean age of patients was 51.0 years in the main analysis population and 36.9 and 36.8 years in subgroups 1 and 2, respectively. More female PsA patients presented with axial manifestations at baseline compared to those without axial manifestations. Dactylitis and enthesitis were numerically more common in patients with PsA and axial manifestations compared to those without axial manifestations. Mean tender joint count (TJC) 68 was also somewhat higher in patients with PsA and axial manifestations, while psoriasis, swollen joint count 66, and CRP were similar.
Baseline demographics and disease characteristics in the overall analysis and sensitivity analysis subgroups.

Data presented as mean (SD) unless otherwise specified. Measures of disease activity were not calculated for PsA patients without axial manifestation at baseline. Fatigue, BASDAI Q1; back pain, BASDAI Q2; peripheral arthritis, BASDAI Q3, enthesitis, BASDAI Q4; intensity of morning stiffness, BASDAI Q5; duration of morning stiffness, Q6; inflammation, average of BASDAI Q5 and Q6.
†p < 0.01, *p < 0.05 versus placebo for baseline demographics.
ASDAS, Ankylosing Spondylitis Disease Activity Score; BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; IXE, ixekizumab; mBASDAI, modified BASDAI; PBO, placebo; PsA, psoriatic arthritis; Q4W, every 4 weeks; SF-36 PCS, Short-Form-36 physical component summary; SJC, swollen joint count; TJC, tender joint count.
Main analysis population
In the main analysis population, patients treated with IXE exhibited a significantly greater improvement in axial manifestations such as back pain (BASDAI Q2) and morning stiffness (average of BASDAI Q5 and Q6) at weeks 16 and 24 (p < 0.001 for all; Figure 2). Patients treated with IXE also experienced a significantly greater improvement in total BASDAI score compared to PBO at weeks 16 (p < 0.001) and week 24 (p < 0.001; Figure 2); these improvements were sustained through week 52 for patients who received IXE.
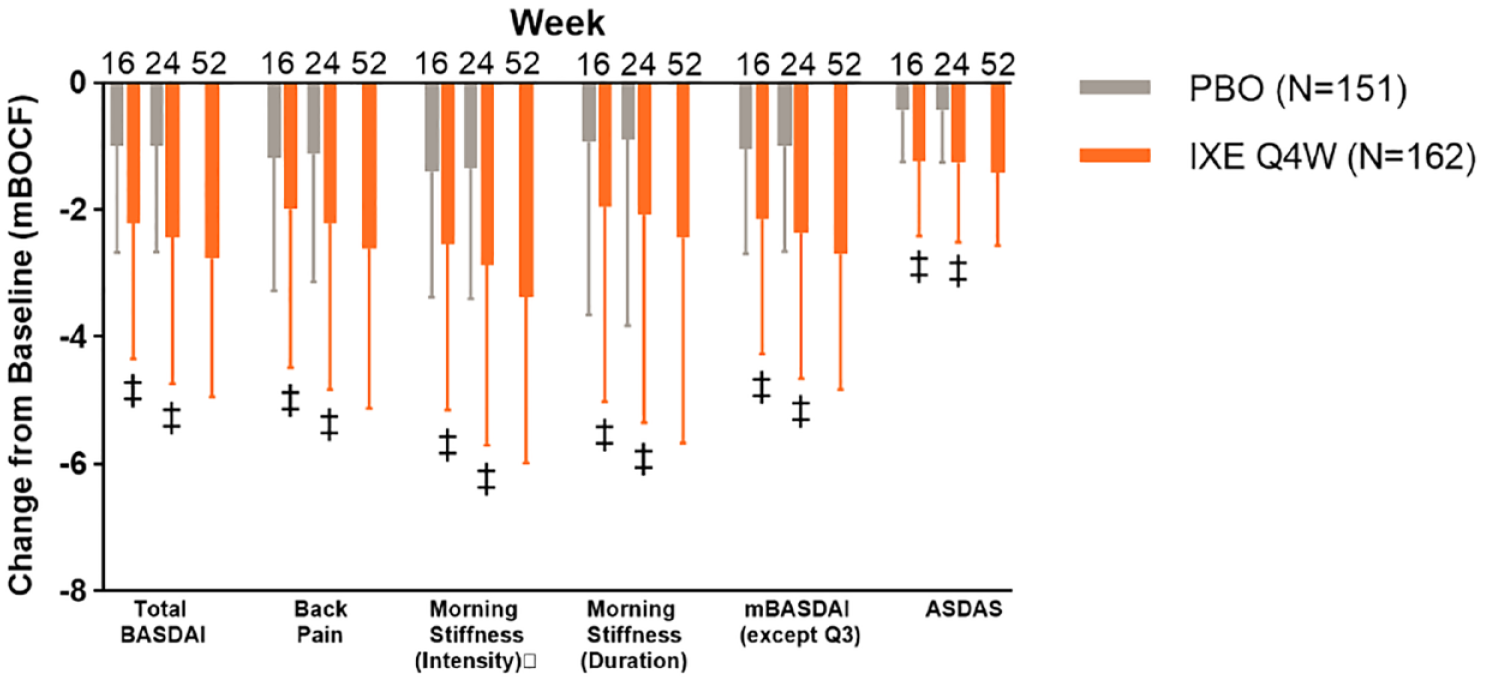
The change from baseline (mBOCF) in BASDAI- and ASDAS-related end points in patients treated with either ixekizumab or placebo in the overall analysis population.
At weeks 16 and 24, a significantly greater improvement from baseline in the other individual BASDAI questions was observed in patients who received IXE compared to those who received PBO (p < 0.001 for all; Table 2), an improvement which was sustained through week 52 for patients who received IXE.
The change from baseline (mBOCF) in BASDAI questions 1 (fatigue), 3 (peripheral arthritis), 4 (enthesitis) and the average of questions 5 and 6 (inflammation) in patients treated with either ixekizumab or placebo in patients in the overall analysis population, patients who are <45 years of age with BASDAI Q2 total score ⩾4 and BASDAI Q5/Q6 ⩾4 at baseline and patients who are <45 years of age with BASDAI Q2 total score ⩾4 and BASDAI Q5/Q6 ⩾4 and CRP >5 mg/L at baseline.
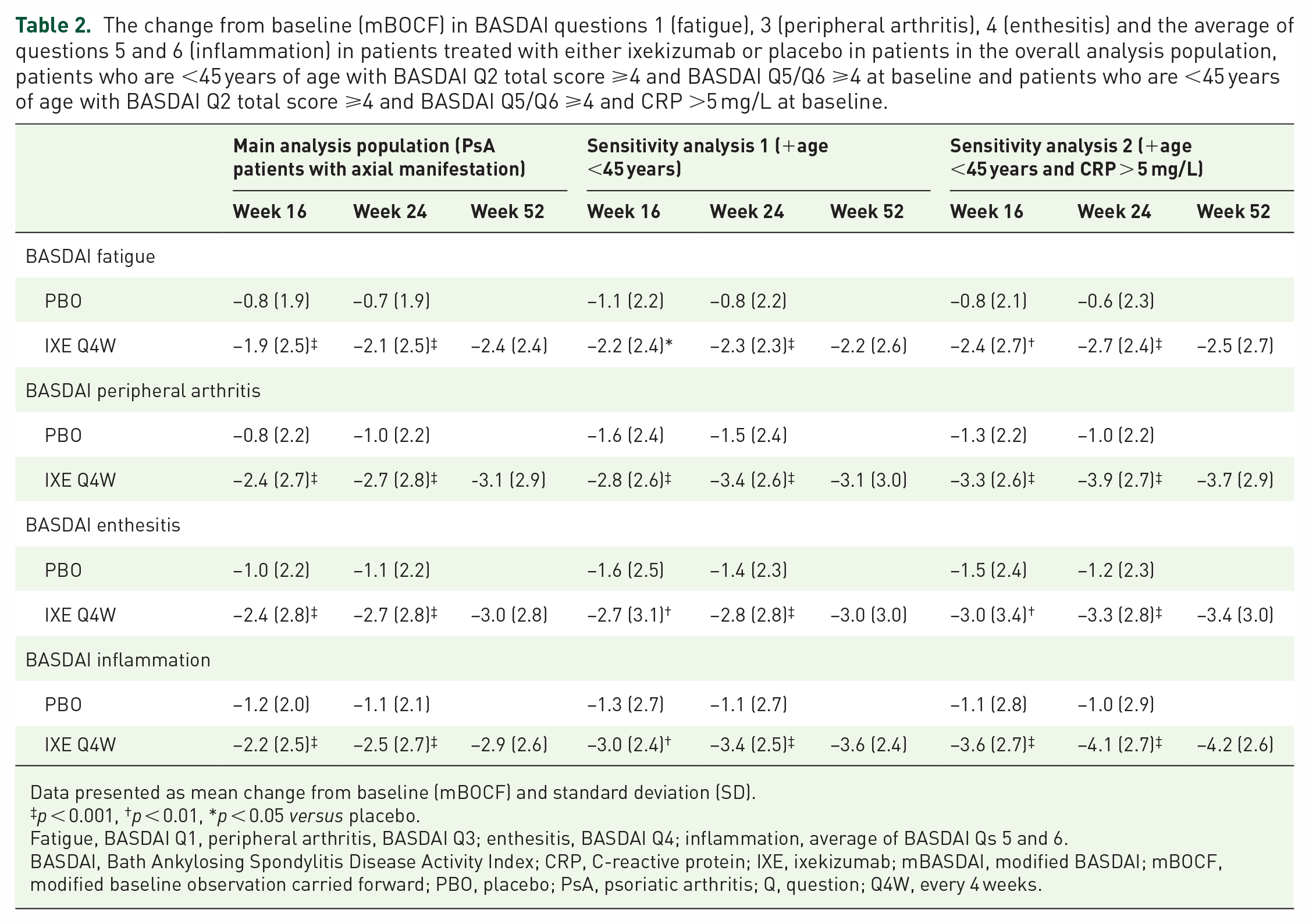
Data presented as mean change from baseline (mBOCF) and standard deviation (SD).
p < 0.001, †p < 0.01, *p < 0.05 versus placebo.
Fatigue, BASDAI Q1, peripheral arthritis, BASDAI Q3; enthesitis, BASDAI Q4; inflammation, average of BASDAI Qs 5 and 6.
BASDAI, Bath Ankylosing Spondylitis Disease Activity Index; CRP, C-reactive protein; IXE, ixekizumab; mBASDAI, modified BASDAI; mBOCF, modified baseline observation carried forward; PBO, placebo; PsA, psoriatic arthritis; Q, question; Q4W, every 4 weeks.
At weeks 16 and 24, a significantly greater improvement from baseline in ASDAS was observed in patients who received IXE compared to those who received PBO (p < 0.001 for all) (Figure 2).
At weeks 16 and 24, significantly more patients who received IXE than PBO achieved BASDAI50 [p < 0.001 for both; Figure 3(a)].

BASDAI 50 responses at weeks 16, 24, and 52 in patients treated with either ixekizumab or placebo in (a) patients in the overall analysis population, (b) patients who are <45 years of age with BASDAI Q2 total score ⩾4 and BASDAI Q5/Q6 ⩾4 at baseline, and (C) patients who are <45 years of age with BASDAI Q2 total score ⩾4 and BASDAI Q5/Q6 ⩾4 and CRP > 5 mg/L at baseline.
At weeks 16 and 24, significantly more patients treated with IXE compared to PBO achieved a BASDAI Q2 (back pain) score improvement of ⩾2 (p < 0.001 for both, Figure 4).
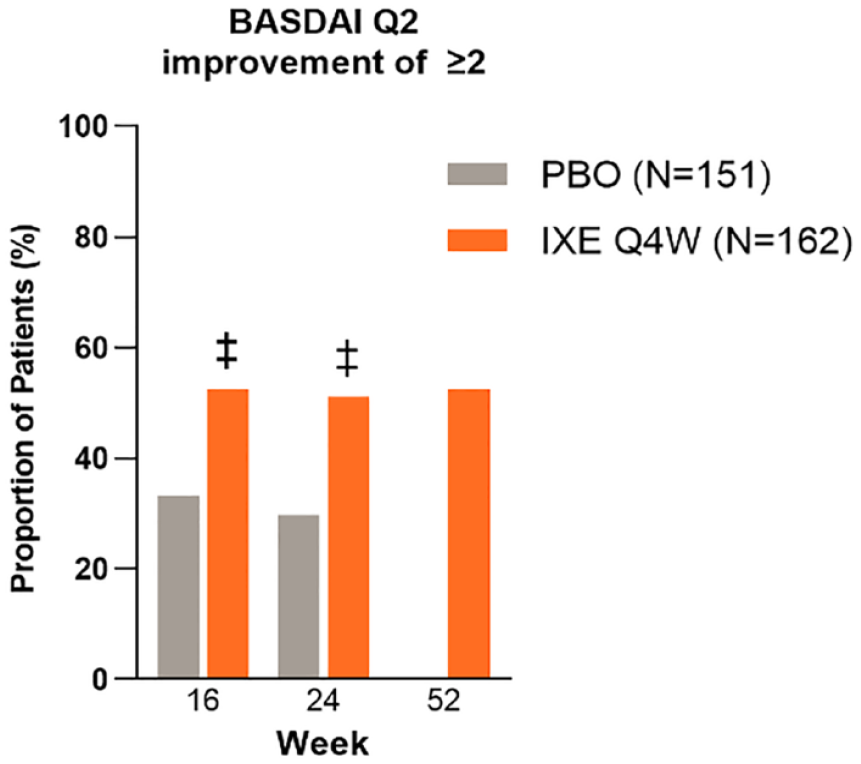
The proportion of patients who achieved an improvement of ⩾2 points in BASDAI Q2 at weeks 16, 24, and 52 in patients treated with either ixekizumab or placebo in the overall analysis population. Data are presented as non-responder imputation.
The effect of IXE on improvements in ASDAS and the proportion of patients who achieved BASDAI Q2 improvement of ⩾2 and BASDAI50 was sustained at week 52.
Sensitivity analysis
Similar to the effects observed in the main analysis population, patients <45 years with spinal pain and inflammation at baseline (the first sensitivity analysis) and those treated with IXE exhibited a significantly greater improvement in back pain at week 16 (p = 0.008) and week 24 (p < 0.001, Figure 5). Similar improvements in morning stiffness were observed in patients treated with IXE versus PBO at week 16 (p < 0.001) and week 24 (p < 0.001).
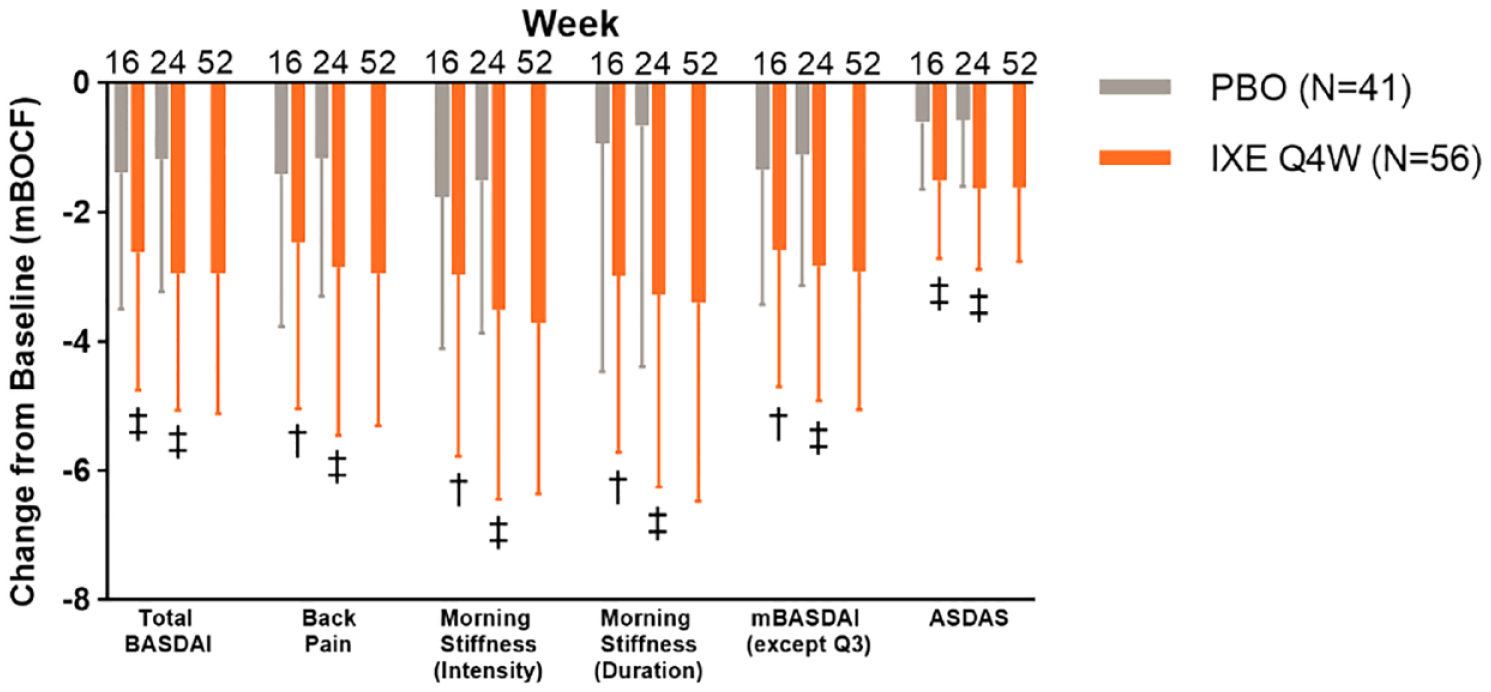
The change from baseline (mBOCF) in BASDAI- and ASDAS-related end points in patients treated with either ixekizumab or placebo who are <45 years of age with BASDAI Q2 total score ⩾4 and BASDAI Q5/Q6 ⩾ 4 at baseline.
Patients treated with IXE also experienced a significantly greater improvement in total BASDAI score compared to PBO at week 16 (p < 0.001) and week 24 (p < 0.001, Figure 5). This effect was sustained through week 52 for patients who received IXE. At weeks 16 and 24, significantly greater improvements from baseline in BASDAI Q1 (fatigue; week 16, p = 0.013; week 24, p < 0.001, Table 2), BASDAI Q3 (peripheral arthritis; weeks 16 and 24, p < 0.001, Table 2), BASDAI Q4 (enthesitis; week 16, p = 0.001; week 24, p < 0.001, Table 2), BASDAI Q5 (intensity of morning stiffness; week 16, p = 0.005; week 24, p < 0.001, Figure 5), and BASDAI Q6 (duration of morning stiffness; week 16, p = 0.004; week 24, p < 0.001, Figure 5) were observed in patients who received IXE compared to those who received PBO; these improvements were sustained through week 52 for patients who received IXE.
There was also a significantly greater improvement from baseline in mBASDAI score in patients who received IXE compared to those who received PBO at week 16 (p = 0.001) and week 24 (p < 0.001; Figure 5), an improvement which was sustained through week 52 for patients who received IXE. At weeks 16 and 24, a significantly greater improvement from baseline in ASDAS was observed in patients who received IXE compared to those who received PBO (p < 0.001 for all, Figure 5).
At weeks 16 and 24, significantly more patients who received IXE than PBO achieved BASDAI50 [p = 0.005, Figure 3(a)], with the exception of the subset of patients who are <45 years of age with BASDAI Q2 total score ⩾4 and BASDAI Q5/Q6 (morning stiffness) ⩾4 at baseline (the second sensitivity analysis) at week 16 [Figure 3(b)].
The effect of IXE on improvements in mBASDAI score, ASDAS, and the proportion of patients who achieved BASDAI50 was sustained at week 52.
Among patients in the second sensitivity analysis, patients <45 years who had back pain, inflammation, and CRP greater than 5 mg/L at baseline, and those treated with IXE exhibited a significantly greater improvement in back pain and morning stiffness at weeks 16 and 24 (p < 0.001 for all, Figure 6). Patients treated with IXE also experienced a significantly greater improvement in BASDAI score compared to PBO at week 16 and week 24 (p < 0.001 for both, Figure 6). This effect was sustained through week 52 for patients who received IXE. At weeks 16 and 24, significantly greater improvements from baseline in BASDAI Q1 (week 16, p = 0.002; week 24, p < 0.001, Table 2), BASDAI Q3 (week 16 p < 0.001; week 24, p < 0.001, Table 2), BASDAI Q4 (week 16, p = 0.002; week 24, p < 0.001, Table 2), BASDAI Q5 (week 16, p = 0.002; week 24, p < 0.001, Figure 6), and BASDAI Q6 (week 16, p = 0.002; week 24, p = 0.003, Figure 6) were observed in patients who received IXE compared to those who received PBO; these improvements were sustained through week 52 for patients who received IXE.
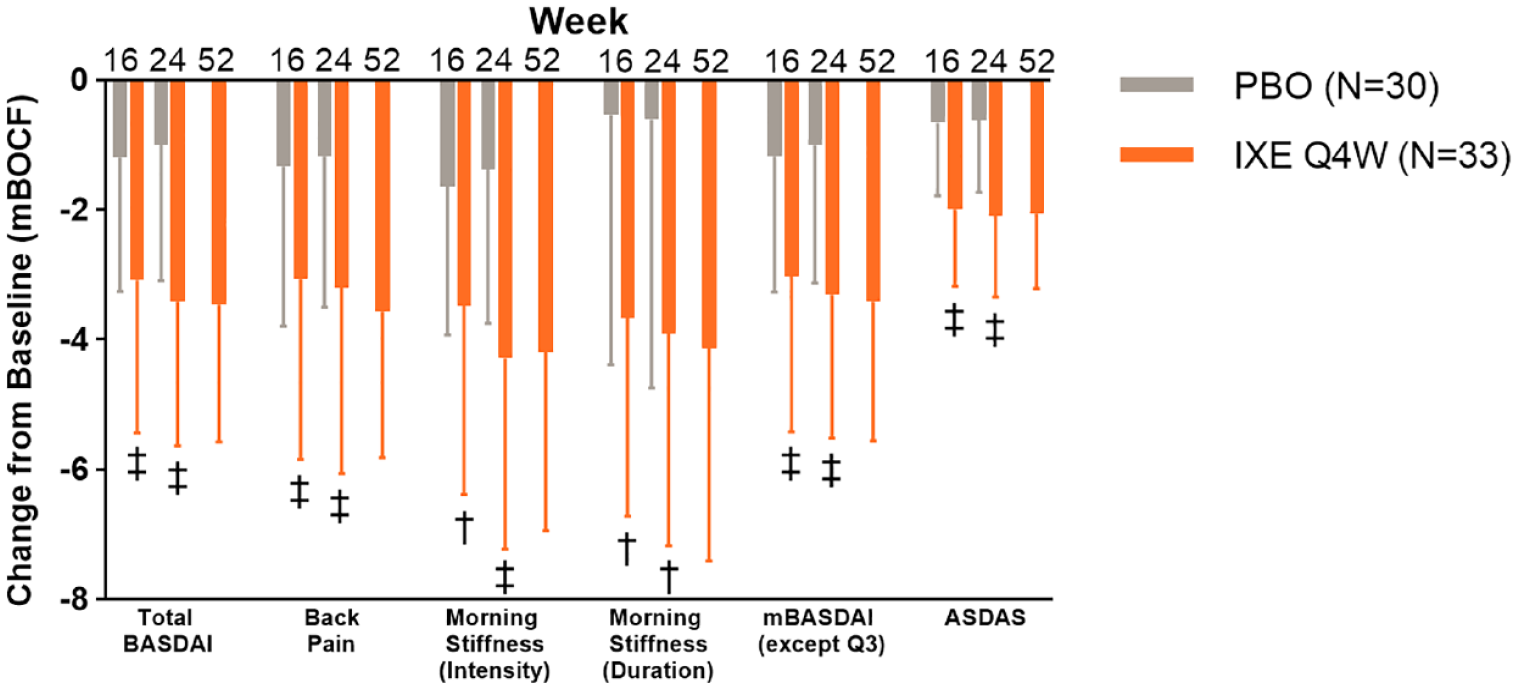
The change from baseline (mBOCF) in BASDAI- and ASDAS-related end points in patients treated with either ixekizumab or placebo who are <45 years of age with BASDAI Q2 total score ⩾4 and BASDAI Q5/Q6 ⩾4 and CRP >5 mg/L at baseline.
There was also a significantly greater improvement from baseline in mBASDAI score in patients who received IXE compared to those who received PBO at weeks 16 and 24 (p < 0.001 for both, Figure 6), an improvement which was sustained through week 52 for patients who received IXE. At weeks 16 and 24, there was a significantly greater improvement from baseline in ASDAS among patients who were treated with IXE when compared to those who received PBO (p < 0.001 for both, Figure 6).
Significantly more patients who received IXE compared to PBO achieved a BASDAI50 response at week 16 (p = 0.005) and week 24 (p = 0.005, Figure 3(c)).
The effect of IXE on improvements in mBASDAI score, ASDAS, and the proportion of patients who achieved BASDAI50 was sustained at week 52.
Discussion
The present post hoc analysis assessed the efficacy of IXE on axial manifestations suggestive of axial involvement in patients with PsA who were biologic disease-modifying antirheumatic drug (bDMARD) naïve (SPIRIT-P1) and TNFi-experienced (SPIRIT-P2) patients. In patients with axial manifestations, greater severity of PsA-related symptoms such as enthesitis, dactylitis, and TJC was observed compared to patients without axial manifestations. Significant improvements from baseline were reported across multiple clinical end points, including purely axial symptoms such as back pain and morning stiffness in the spine, in patients who were treated with IXE compared to PBO at weeks 16 and 24.
The present findings also indicate that patients from the pooled SPIRIT-P1 and SPIRIT-P2 studies with active PsA and axial manifestations experience more severe disease characteristics compared to all other remaining patients. These findings are in line with the 2018 analysis of patients with PsA who were enrolled in the US Corrona PsA/SpA Registry, which also described that those who exhibited axial manifestations at baseline had worse disease activity as assessed by a variety of different clinical variables. 14
To date, two observational studies have assessed the use of biologics for the treatment of patients with symptoms suggestive of axial PsA.15,16 Both adalimumab and etanercept were shown to be effective for the treatment of patients with PsA and axial symptoms.15,16 A post hoc analysis which included a pooled subset of patients from SELECT-PsA 1 and SELECT-PsA 2 with symptoms of axial involvement also revealed that the JAKi upadacitinib improved BASDAI- and ASDAS-related end points compared to PBO at weeks 16 and 24. 17 Unlike the present analysis, axial involvement was assessed by the investigator based on the totality of information available, not using defined criteria. MAXIMISE was the first phase III, randomized, double-blind, PBO-controlled trial assessing the efficacy of IL-17A inhibitor secukinumab in patients with PsA and axial involvement. 18 Patients included had a BASDAI score ⩾4, a spinal pain score ⩾40 by Visual Analog Score, and were bDMARD naïve. The authors report a significant improvement in ASAS20 response at week 16 with secukinumab versus PBO. Inflammation was assessed by magnetic resonance imaging but was not required at study entry. 18 In a post hoc analysis of the guselkumab trials DISCOVER-1 and DISCOVER-2, investigators confirmed the presence of sacroiliitis either by documented prior imaging or by pelvic radiography at screening. 19 The authors report that guselkumab improved the symptoms of axial disease in patients with PsA who had the evidence of sacroiliitis. 19
At present, there are no specified criteria to properly identify patients with PsA and axial disease, which has ranged from unilateral advanced sacroiliitis to a condition similar to r-axSpA. 20 A multinational, multi-center, cross-sectional study investigating axial involvement in a cohort of patients who fulfill the Classification Criteria for Psoriatic Arthritis (CASPAR) for PsA (AXIS) aims to develop a data-driven definition of axial involvement in PsA and determine its prevalence. There is still an important unmet need in patients with PsA and axial involvement. This analysis supports ixekizumab as a potential treatment for these patients.
A limitation of the present analysis is the lack of a validated definition of axial PsA, making the analysis populations difficult to define. To overcome this, three subsets of patients with PsA and axial manifestations suggestive of axial involvement were analyzed; in each of these subsets, similar effects of IXE on disease activity and quality of life were observed. Also, there are two important factors which were not considered in this cohort of patients, the HLA-B27 status or radiographs of the sacroiliac joint spine, as they were not part of the SPIRIT clinical trials. A strength of this analysis is the addition of two sensitivity analysis subgroups, allowing for additional interpretation of the data.
In conclusion, IXE demonstrated efficacy in improving axial manifestations (back pain and morning stiffness), disease activity, and fatigue through 52 weeks in three subsets of patients with active PsA and axial manifestations. These data provide clinically relevant information about some of the disease characteristics of patients with PsA and axial manifestations as well as support for using IXE as a potential therapeutic option for these patients.
Footnotes
Acknowledgements
The authors would like to thank Edel Hughes, PhD, of Eli Lilly and Company for writing and editorial support.
