Abstract
Background:
Abatacept (ABT) is known to lower infection risk than other biologics and is effective and safe in elderly patients with rheumatoid arthritis (RA). However, there were inconsistent reports on the impact of ABT on malignancies which are more common in the elderly and strongly related to prognosis.
Objectives:
This study aimed to evaluate the efficacy and safety of ABT in patients with RA with previous malignancy in clinical practice.
Design:
A multicenter, retrospective study.
Methods:
Patients who received ABT for RA in two hospitals in Yokohama until May 2022 were included in this study. The patients were divided into two groups according to the presence or absence of a history of malignancy (no previous malignancy: NP group, previous malignancy: PM group). The collected parameters were compared between the groups using propensity score matching.
Results:
In this study, 312 patients were included, of whom 73 had previous malignancies when starting ABT. The age at ABT initiation was significantly higher in the PM group, the rate of methotrexate use was significantly lower in the PM group, and the Steinbrocker stage was significantly higher in the PM group. After matching these 3 factors, 68 patients were selected from each group. No significant differences in the ABT continuation rate, and malignancy incidence were observed between the two groups after ABT initiation. In addition to these factors, when matched for smoking history, interstitial lung disease, disease duration, sex, and inflammatory status, which are known risk factors for malignancy in RA, 40 patients were selected from each group. No significant differences in the ABT continuation rate, and malignancy incidence were observed between the two groups after ABT initiation.
Conclusion:
In our clinical practice, ABT was as effective and safe in patients with a history of malignancy as in those without.
Introduction
Rheumatoid arthritis (RA) is a systemic autoimmune disease that causes bone destruction due to inflammation and causes various lesions and complications in other organs, in addition to joints. 1 Malignancy, among others, is a crucial complication that directly affects the quality of life and life prognosis of patients and is a significant reason for death in patients with RA.2,3 Several risk factors for malignancy in RA have been reported, including age, smoking, interstitial lung disease, disease duration, and disease activity.4,5 Aging is associated with the risk of malignancy in the general population, 6 and with the aging of the population worldwide recently, 7 addressing malignancy will continue to be a significant clinical challenge. Particularly in RA practice, the safety of disease-modifying antirheumatic drugs (DMARDs) against malignancy remains poorly understood, 5 which often makes treating RA difficult in elderly patients at a high risk of malignancy and in patients with a history of malignancy.
Abatacept (ABT) is a recombinant soluble fusion protein, composed of the extracellular domain of human CTLA-4 and the Fc domain of human IgG1, and selectively inhibits T-cell activation by binding to CD80/86 on antigen-presenting cells. 8 In 2019, a study based on post-marketing surveillance (PMS) data in Japan has reported that ABT was as effective and safe in elderly patients as non-elderly patients. 9 Results from several randomized controlled trials (RCT) and PMS data have not shown that ABT increases the risk of developing all malignancies in RA compared with placebo or the general population10–13; however, some cohort studies have suggested that ABT is associated with a slightly increased risk of skin cancer compared with other biological DMARDs.14–18
Notably, although numerous studies on the risk of developing malignancy with ABT have been conducted,9–11,13,15–18 data on the efficacy and safety of ABT in patients with RA with previous malignancy compared with those in patients without previous malignancy remain scarce. In this study, we examined the safety of ABT in patients with RA with previous malignancy and compared it with that in patients without previous malignancy in clinical practice in two hospitals in Japan. We compared the efficacy and safety of ABT between patients with RA with and without previous malignancy, matched for age, sex, and other known risk factors for malignancy in RA4,5 and found no significant differences between the two groups in the ABT continuation rate or the incidence of new or recurrent malignancy or progression after ABT initiation.
Design and methods
Patients
In this study, 276 and 36 patients with RA treated with ABT from August 2010 to May 2022 in the Rheumatology Departments of Yokohama Minami Kyosai Hospital and Saiseikai Yokohamashi Nanbu Hospital, respectively, were enrolled. RA was clinically diagnosed based on the 2010 American College of Rheumatology/European League Against Rheumatism classification criteria. 19 The observation period was from the initiation of ABT administration to ABT discontinuation or September 2022. Clinical data, including sex, age, and disease duration at ABT initiation, history of previous malignancy, concomitant drugs, physical examination findings, laboratory findings, and continuation period of ABT, were retrospectively collected from the clinical charts. The patients were divided into two groups according to the presence or absence of a history of malignancy at the intiation of ABT [no previous malignancy (NP group) and with previous malignancy (PM group)]. The collected parameters, ABT continuation rates, and occurrence rates of new or recurrent malignancy or progression after ABT initiation were compared between the groups using propensity score matching (PSM).
The study was conducted according to the Declaration of Helsinki, and informed consent was obtained from all patients in an opt-out approach on the website. The Clinical Ethics Committee of Yokohama Minami Kyosai Hospital approved the study design (1-21-1-42). Reporting of this study complied with the checklist of the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 20 (Supplemental Material).
Clinical efficacy and safety
Physical examination findings, laboratory findings from ABT initiation to the last observation or discontinuation, new diagnosis, and any recurrence and progression of malignancy during the ABT therapy were retrospectively collected. Disease Activity Score-28 for RA with C-reactive protein (DAS28-CRP) and Simplified Disease Activity Index (SDAI) were used to analyze disease activity, and missing values were complemented by the last observation carried forward method. For the analysis of the ABT continuation rate, ABT discontinuation was defined as when the clinician determined that ABT discontinuation was medically advisable due to ineffectiveness or complications and stopped ABT.
Statistical analysis
All statistical analyses were performed using GraphPad Prism (version 8.0; GraphPad Software, La Jolla, CA, USA), Statistical Package for the Social Sciences (version 29.0; IBM Corp., Armonk, NY, USA), and R (version 4.2.3; The R Project for Statistical Computing). Unpaired two-tailed Student’s t-test was used for the parametric analysis of continuous variables. The Mann–Whitney U test was used for the nonparametric analysis of continuous variables. The chi-square test or Fisher’s exact test was used to analyze categorical variables. The log-rank test was used to compare the ABT continuation rates, and the Cox proportional hazards model was used to compare the incidence rates of malignancy. The propensity score was calculated using binomial regression analyses using R. PSM was performed using the 1:1 pair with or without a history of malignancy using calipers with a width of 0.2 of the standard deviation. Differences with p-values <0.05 were considered statistically significant in all analyses.
Results
In this study, 312 patients were included, of whom 73 (23.4%) had previous malignancies during ABT initiation. The details on PM history are shown in Table 1. In that order, the most common previous malignancies were lymphoma, breast cancer, stomach cancer, and lung cancer. Sixty-two patients had one PM, 10 had two previous malignancies, and one had three previous malignancies. Disease activity was significantly improved 3 months after ABT initiation (Figure 1(a) and (b)). In patients with RA with PM, the median time from the most recent malignancy diagnosis to ABT initiation was 2.7 years [interquartile range (IQR): 0.81–7.1 years]. At the time of initiation of ABT, five patients were tumor-bearing; two were under treatment for lung cancer, two were under follow-up for methotrexate-related lymphoproliferative disease, and one was under hormonal treatment for prostate cancer. Eleven patients had newly diagnosed malignancy, and one experienced recurrence of pre-existing malignancy after 3.6 ± 3.1 years of ABT treatment. The incidence of malignancy after the initiation of ABT was 1080.3 per 100,000 person-years. The median time from the start of ABT to the onset of malignancy was 4.6 years (IQR: 2.6–6.3 years; Table 2). The only case of recurrent malignancy in the present study was malignant lymphoma of the thyroid gland. The patient had developed RA 5 years before the initial diagnosis of malignant lymphoma and was being treated with methotrexate (MTX); when the diagnosis of malignant thyroid lymphoma was made, MTX was discontinued, and a total thyroidectomy was performed. 1 year after discontinuing methotrexate and total thyroidectomy, RA flared up without any treatment, and a subcutaneous ABT injection was started. After that, the disease activity improved. However, 4.4 years after the start of ABT, a recurrence of a thyroid tumor and tracheal invasion were found during a close examination of blood sputum, and ABT was discontinued. Radiotherapy was given for the malignant tumor, but the patient’s progress was poor, leading to death.
Details of previous malignancy history.


Disease activity after ABT initiation. Disease activity after ABT initiation in all patients (a and b); the unadjusted groups (c and d); after adjusting for the age at ABT initiation, MTX use, and the Steinbrocker stage (e and f); and after adjusting for the age at ABT initiation, MTX use, the Steinbrocker stage, smoking history, interstitial lung disease, disease duration, sex, and serum CRP levels (g and h). The blue line represents the NP group, and the red line represents the PM group. The p-values versus previous visits were calculated using the paired t-test (*p < 0.05 and **p < 0.01). Values are shown as means ± SD.
Details of newly diagnosed or recurrence of malignancy during ABT therapy.

Comparing patient background at the start of ABT between patients with and without prior malignancy, significant differences in age, MTX combination, and the Steinbrocker stage were observed. However, no significant differences in other backgrounds were observed between the two groups (Table 3). Disease activity up to 60 months after ABT initiation was not significantly different between the two groups (Figure 1(c) and (d), and Supplemental Table 2). The 1- and 10-year continuation rates of ABT were 83.5% per year and 50.2% per 10 years, respectively, in the NP group and 85.6% per year and 56.2% per 10 years, respectively, in the PM group without significant differences (p = 0.78, Figure 2(a)). ABT was discontinued during the observation period in 74 patients in the NP group and 21 in the PM group. Dropouts or loss to follow-up during the observation period were 13 patients in the NP group and 7 in the PM group. There were no significant differences in reasons for discontinuation or dropout between the two groups (Supplemental Table 1).
Patient background at ABT initiation.

Tumor-bearing status included the following: two cases of lung cancer on chemotherapy, two cases of methotrexate-related lymphoproliferative disease on follow-up, and one case of prostate cancer on hormone therapy.
ABT, abatacept; Anti-CCP Ab, anti-cyclic citrullinated peptide antibody; b/tsDMARDs, biological and targeted synthetic disease-modifying antirheumatic drugs; CDAI, clinical disease activity index; CRP, C-reactive protein; DAS, disease activity score; ESR, erythrocyte sedimentation rate; IQR, interquartile range; MMP-3, matrix metalloproteinase 3; MTX, methotrexate; NP group, no previous malignancy group; PM group, with previous malignancy group; PSL, prednisolone; PSM, propensity score matching; RF, rheumatoid factor; SASP, salazosulfapyridine; SD, standard deviation; SDAI, simplified disease activity index; TAC, tacrolimus.
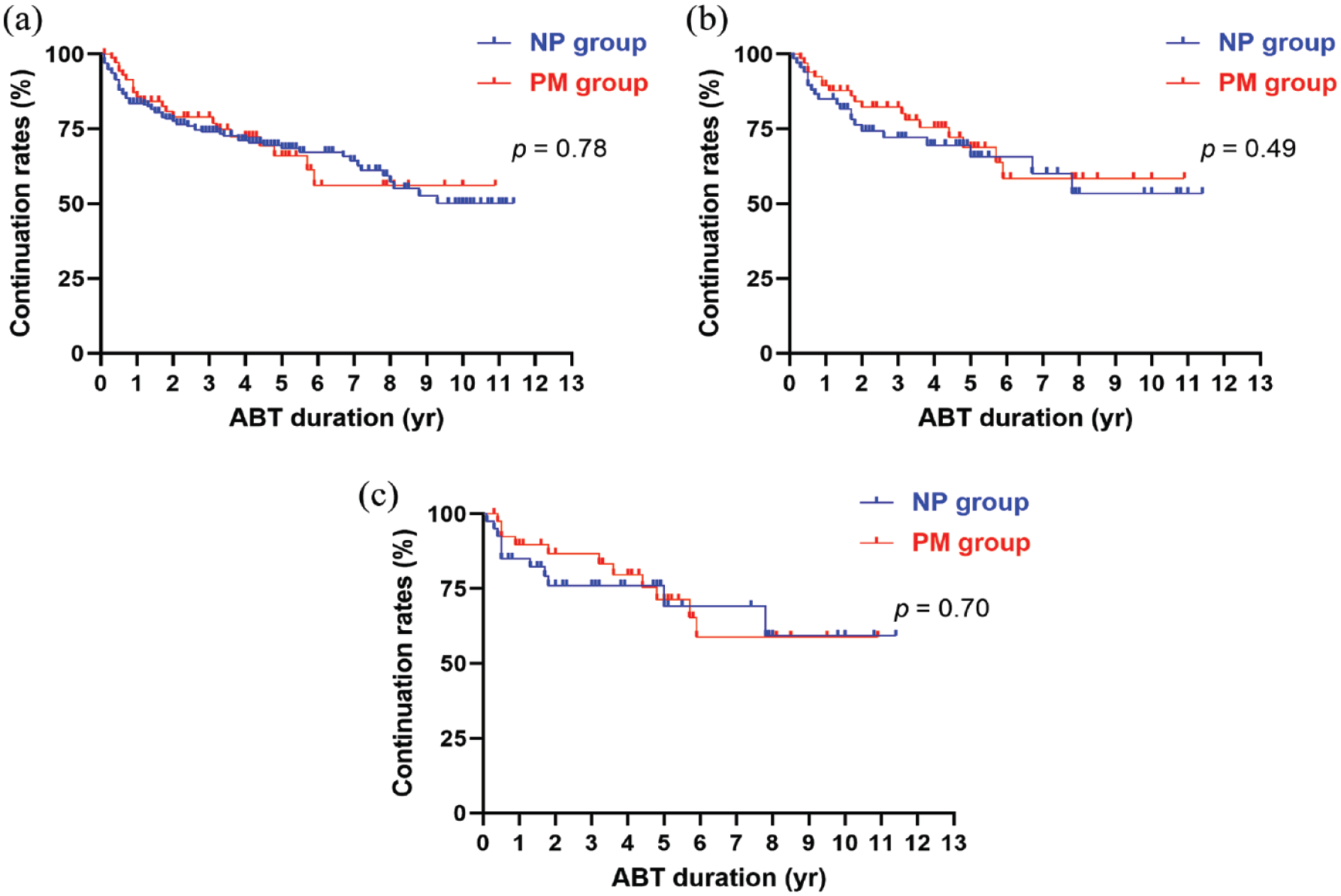
Continuation rates of ABT. The continuation rates of ABT in the unadjusted groups (a); after adjusting for the age at ABT initiation, MTX use, and the Steinbrocker stage (b); and after adjusting for the age at ABT initiation, MTX use, the Steinbrocker stage, smoking history, interstitial lung disease, disease duration, sex, and serum CRP levels (c). The blue line represents the NP group, and the red line represents the PM group. The p-values were calculated using the log-rank test.
Significant points were that the NP and PM groups have patient background differences, particularly in age and the Steinbrocker stage, which reflects the duration of disease and activity; both are essential risk factors for malignancy.4,5 Furthermore, a significant difference in the MTX concomitant use rate was observed, making it challenging to compare ABT efficacy in the two groups. Next, to adjust for these problems, PSM was used to match the NP and PM groups by adjusting background factors. First, we adjusted for three background factors that were significantly different between the NP and PM groups and then selected matched pairs – 68 cases in each group were selected, without significant differences in background factors (Table 4, PSM1). Disease activity up to 60 months after ABT initiation was not significantly different between the two groups (Figure 1(e) and (f), and Supplemental Table 3). The 1- and 10-year continuation rates of ABT were 85.0% per year and 53.5% per 10 years, respectively, in the NP group and 89.4% per year 58.5% per 10 years, respectively, in the PM group, without a significant difference (p = 0.49, Figure 2(b)). In addition to these factors, we adjusted for smoking history, interstitial lung disease, disease duration, sex, and serum CRP levels. Forty cases in each group were selected, without significant differences in background factors (Table 4, PSM2). Disease activity up to 60 months after ABT initiation was not significantly different between the two groups (Figure 1(g) and (h), and Supplemental Table 3). The 1- and 10-year continuation rates of ABT were 85.0% per year and 59.3% per 10 years, respectively, in the NP group and 89.7% per year and 58.8% per 10 years, respectively, in the PM group, without a significant difference (p = 0.70, Figure 2(c)).
Patient backgrounds at ABT initiation after PSM.

Adjusted for the age at ABT initiation, MTX use, and the Steinbrocker stage.
Adjusted for the age at ABT initiation, MTX use, the Steinbrocker stage, smoking history, interstitial lung disease, disease duration, sex, and serum CRP levels.
ABT, abatacept; Anti-CCP Ab, anti-cyclic citrullinated peptide antibody; b/tsDMARDs, biological and targeted synthetic disease-modifying antirheumatic drugs; CDAI, clinical disease activity index; CRP, C-reactive protein; DAS, disease activity score; ESR, erythrocyte sedimentation rate; MMP-3, matrix metalloproteinase 3; MTX, methotrexate; NP group, no previous malignancy group; PM group, with previous malignancy group; PSL, prednisolone; PSM, propensity score matching; RF, rheumatoid factor; SASP, salazosulfapyridine; SD, standard deviation; SDAI, simplified disease activity index; TAC, tacrolimus.
Finally, we compared the incidence of malignancy after ABT initiation between the NP and PM groups. In the unadjusted comparison, the incidence of malignancy after ABT initiation in the NP and PM groups was 1183.1 and 779.0 per 100,000 person-years, respectively, without a significant difference [hazard ratio (HR) = 0.72; p = 0.68] (Table 5). In the comparison after matching the three significantly different background factors, the incidence of malignancy after ABT initiation in the NP and PM groups was 1249.9 and 788.9 per 100,000 person-years, respectively, without a significant difference (HR = 0.71; p = 0.71; Table 5). In addition to these factors, in comparisons after matching for known malignancy risk factors in RA, the incidence of malignancy after ABT initiation in the NP and PM groups was 1370.0 and 1,160.6 per 100,000 person-years, respectively, without a significant difference (HR = 0.99; p = 1.00; Table 5).
Hazard ratio of malignancy incidence after ABT initiation.

Adjusted for the age at ABT initiation, MTX use, and the Steinbrocker stage.
Adjusted for the age at ABT initiation, MTX use, the Steinbrocker stage, smoking history, interstitial lung disease, disease duration, sex, and serum CRP levels.
ABT, abatacept; CI, confidence interval; NP group, no previous malignancy group; PM group, with previous malignancy group; PSM, propensity score matching.
Discussions
The increased risk of developing malignancies in the elderly 6 is an important clinical issue to be considered, as it directly affects the treatment of comorbidities, quality of life, and prognosis.2,3 Whether patients with RA are clearly at a higher risk of developing any malignancies than the general population remains controversial; however, several reports have shown that patients with RA have an increased risk of lung cancer and malignant lymphoma compared with the general population.21–23 Evidence of the safety of most DMARDs in patients with previous malignancies and tumor-bearing individuals remains poor, 5 and in clinical practice, antirheumatic treatment for these patients poses a significant challenge.
ABT is a biological DMARD that targets T-cell co-stimulatory signals and has a stronger ability to lower the risk of infection than other biologics. 16 Furthermore, it has been reported that the efficacy and safety of ABT in patients aged ⩾ 65 were comparable to those in patients aged <65 years. 9 Due in part to this evidence of safety, ABT is often used in clinical practice for treating elderly patients with RA and those presenting with more comorbidities. 24 Several RCTs and PMS data have reported no increase in the incidence of malignancy with ABT in patients with RA compared with placebo10–13 or other biologics. 16 In contrast, other studies have reported slightly increased malignancy rates compared with other biologics (primarily tumor necrosis factor (TNF) inhibitors), with particular attention to skin cancer.11,14,15,17,18 Based on the aforementioned statements, whether ABT increases the risk of developing malignancies remains unclear. Furthermore, the effect of ABT on patients with previous malignancies or those with cancer-bearing conditions is uncertain.
The incidence of malignancy in patients with RA treated with ABT was reported as 1026 and 1021 per 100,000 person-years in the Swedish cohort and Japanese 3-year surveillance, respectively.12,14 In this study, the risk of malignancy after ABT initiation was 1080.3 per 100,000 person-years, which did not differ significantly from those in previous reports, although the background factors were different; for example, our cohort was older than previous cohorts.12,14 To investigate the association between ABT and the risk of malignancy, this study sought to determine the continuation rate of ABT and the rate of new diagnosis, recurrence, and progression of malignancy in patients with RA treated with ABT who had a history of malignancy at the start of ABT administration and to compare these patients with those without a history of malignancy. We first matched the two groups for age, MTX use, and the Steinbrocker stage, which were significantly different in the backgrounds of the two groups being compared. We found no significant differences in the continuation rate of ABT or incidence of malignancy between patients with and without previous malignancy. However, a history of smoking and age is a significant risk for developing malignancy in RA patients. 25 Since many other factors were reported to be associated with the development of malignancy in RA, we next matched and compared all these supposed risk factors for developing malignancy among the items we could collect and found no significant difference in the continuation rate of ABT or the incidence of malignancy after ABT initiation between patients with and without previous malignancy.4,5 However, note that all patients in this study were East Asians. This racial group does not have an inherently high risk of skin cancer, 26 and in fact, only one patient had a history of skin cancer and no recurrence or new skin cancer was observed after ABT was started. Of the malignancies reported during ABT use, six were cancers of the digestive system (including five colorectal cancers), and about half of the malignancies observed during ABT use in this study were of the digestive system. Colorectal cancer is the most common malignancy among Japanese, which may also be influenced by race. 27 Therefore, the results of this study cannot directly reflect the risk of skin and colorectal cancer in patients treated with ABT.
The safety of biologics in RA patients with precancerous conditions, prior malignancies, or undergoing treatment for malignancies has not been proven. 28 Patients with these conditions are not being promoted to receive biologics, including ABT, which is an essential topic for future research.28,29 A recent meta-analysis suggests that TNF inhibitors may reduce the risk of developing malignancy in patients with inflammatory disease compared to Janus kinase inhibitors and placebo. 30 However, for example, we do not know how long a biologic can be safely used after a malignancy treatment or for what treatments for cancer types a biologic can be administered. In this study, 73 patients with a history of malignancy were initiated on ABT at a median time of 2.7 years. Of these, 41 had a history of malignancy within 5 years, and five had started ABT during treatment or follow-up of malignancy. However, there were no significant safety or efficacy alarms with the introduction of ABT during the observation period. Although the results of this study do not provide a clear answer to the question of how many years after a history of malignancy ABT can be used safely or whether it is safe to use ABT in RA patients with tumor-bearing conditions, it is essential to note that in actual clinical data, the 10-year continuation rate of ABT in RA patients with a history of malignancy was well maintained, exceeding 50%. They may help us consider the efficacy and safety of ABT in real-world practice when it is unavoidable to use it in these patients when disease activity becomes a problem.
This study has several limitations. First, this was a retrospective study; thus, some missing data and background differences should be included. Furthermore, the frequency of screening for malignancy in individual cases was not evaluated. However, we addressed these issues by adjusting for background factors using PSM. Second, the study encompassed a limited region and race. Generalization to different populations and races may be difficult, particularly for different cancer types and ages. Third, verifying whether ABT specifically affects malignancy is impossible since this study did not have a negative control. Finally, this study might be underpowered due to the small number of malignancy cases. Further evidence on the safety and efficacy of antirheumatic drugs, including ABT and other biologics, in patients with previous malignancy or tumor-bearing conditions is warranted.
In conclusion, the efficacy and safety of ABT were similar among patients with previous malignancy and those without in our clinical practice. ABT may also be effective in older patients with a history of malignancy and may not specifically increase the risk of developing malignancy or relapse.
Supplemental Material
sj-pdf-1-tab-10.1177_1759720X231186874 – Supplemental material for Efficacy and safety of abatacept in patients with rheumatoid arthritis with previous malignancy
Supplemental material, sj-pdf-1-tab-10.1177_1759720X231186874 for Efficacy and safety of abatacept in patients with rheumatoid arthritis with previous malignancy by Yosuke Kunishita, Kento Ichikawa, Yuji Uzawa, Masaki Mitsuhashi, Yuji Yoshioka, Tadanobu Okubo and Shouhei Nagaoka in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
