Abstract
Background:
Patients with idiopathic inflammatory myopathies (IIMs) are at risk of reduced bone mineral density (BMD).
Objectives:
To compare the prevalence of reduced BMD between patients with IIMs and controls and to determine its risk factors.
Design:
This was a single-center case-control study.
Methods:
BMD was assessed by dual-energy X-ray absorptiometry. The prevalence of reduced BMD in IIM patients and age-and sex-matched non-rheumatological controls was compared. The BMD results of female IIM were also compared to age-matched female rheumatoid arthritis (RA) and systemic lupus erythematosus (SLE) patients. Independent factors associated with reduced BMD in IIM patients were identified by multivariate analyses.
Results:
A total of 230 patients (IIM: 65, non-rheumatological controls: 65, RA: 50, SLE: 50) were recruited. The mean age of IIM patients was 58.6 ± 11.0 years and 76.9% were females. Significantly, more IIM patients had reduced BMD (73.8% versus 43.1%, p = 0.043) and osteoporosis (29.2% versus 13.8%, p = 0.033) than non-rheumatological controls. Multivariate analysis confirmed that IIM was independently associated with reduced BMD (OR: 2.12, p = 0.048, 95% CI: 1.01–4.46). The prevalence of reduced BMD was not significantly different between IIM, RA, and SLE patients but the mean hip BMD was the lowest in the IIM group (0.641 ± 0.152 g/cm2 versus 0.663 ± 0.102g/cm2 in the RA group versus 0.708 ± 0.132 g/cm2 in the SLE group, p = 0.035). Lower body mass index and more advanced age were independently associated with lower BMD in IIM patients.
Conclusion:
Reduced BMD was more prevalent in IIM patients than in non-rheumatological controls. Hip BMD was lower in patients with IIMs than RA or SLE. Close monitoring and early treatment are encouraged especially in patients with risk factors.
Summary
In this case-control study, bone mineral densities (BMD) among patients with inflammatory myopathies were compared with controls. Reduced BMD was more prevalent in patients with myositis. Close monitoring of BMD and early treatment should be considered in patients with risk factors.
Introduction
Osteoporosis and osteopenia, collectively known as reduced bone mineral density (BMD), are characterized by reduced bone mass, bone microarchitectural distortion, and fragility, leading to increased fracture risks and significant socioeconomic burden. 1 Up to one-fifth of osteopenic women older than 65 years suffered from fragility fracture, signifying the importance of early recognition patients with reduced BMD. 2
Idiopathic inflammatory myopathies (IIMs) are a group of multisystem autoimmune connective tissue diseases which could affect musculoskeletal, cutaneous, and respiratory systems. The mainstay of treatment includes glucocorticoids (GCs) and immunosuppressants. Systemic inflammation, reduced mobility, and the use of GCs all contribute to accelerated bone loss in IIM patients. We have previously found a high prevalence of reduced BMD in IIM patients with 23.7% and 47.4% of them having osteoporosis and osteopenia, respectively. 3 However, it was unsure whether the apparent high risk of poor bone health in IIM patients was independently associated with the disease or due to other known risk factors.
Data comparing the prevalence of reduced BMD in IIM patients and patients with other systemic conditions or rheumatological diseases are scarce. Therefore, this study was conducted to compare the prevalence of reduced BMD in IIM patients with non-rheumatological controls, rheumatoid arthritis (RA), and systemic lupus erythematosus (SLE). The factors associated with reduced BMD were also examined.
Methods
Study design and patients
This was a single-center, retrospective, case-control study. The objectives were (1) to compare the prevalence of reduced BMD in IIM patients versus age- and gender-matched controls with non-rheumatological conditions; (2) to compare the prevalence of reduced BMD in female IIM patients with age-matched female patients with RA and SLE; and (3) to evaluate the clinical determinants of BMD in IIM patients.
Consecutive Chinese IIM patients followed up in the medical clinics of a regional hospital in Hong Kong with dual-energy X-ray-absorptiometry (DXA) scan performed from 1 January 2010 to 31 December 2020 were identified by the Clinical Data Analysis and Reporting System (CDARS). The CDARS is an electronic system created by the Hong Kong Hospital Authority in 1991, mainly for audit and research purposes. The system has been extensively used in large-scale epidemiological studies.4,5 IIM patients had to fulfill the 2017 European League Against Rheumatism (EULAR)/American College of Rheumatology (ACR) classification criteria for adult and juvenile IIM patients. 6 Non-rheumatological, RA or SLE controls were identified by the same system. Eligible controls who were age (within 2 years) and sex matched with the IIM patients were recruited consecutively at a 1:1 ratio. RA and SLE patients fulfilled the 2010 ACR/EULAR classification criteria for RA or Systemic Lupus International Collaborating Clinics Classification Criteria for SLE, respectively.7,8 Patients with overlap syndrome and juvenile disease onset (<18 years old) were excluded. Patients and controls with documented metabolic bone disorders (e.g. hyper/hypoparathyroidism, untreated chronic hypothyroidism, renal osteodystrophy, and bone metastases) were also excluded.
Clinical factors assessment
The clinical data were retrieved from the electronic health record system. Demographic data including age, sex, body weight, height, smoking and drinking history, and comorbidities including chronic kidney disease, chronic liver disease, thyroid disease, and malignancy were recorded. Disease duration, history of fragility fractures, and medication history including dosage and duration of GCs, immunosuppressants, calcium/vitamin D supplementation, and anti-osteoporotic drugs were documented. High-dose GC was defined as a dose equivalent to oral prednisolone ⩾0.5 mg/kg/day for more than 2 weeks or the use of intravenous pulse GC. For non-rheumatological controls, diagnoses and indications for DXA scans were documented. For IIM patients, classification of subgroups by the EULAR/ACR classification criteria and diagnostic criteria of antisynthetase syndrome (ASS) proposed by Connors et al., 9 myositis-specific autoantibodies status and results of manual muscle testing 8 (MMT8) at the time of DXA scan were documented. 10 Serum levels of creatine kinase (CK), albumin, and C-reactive protein (CRP) at diagnosis and at the time of DXA scans were recorded. The 10-year probabilities of major osteoporotic (clinical spine, hip, shoulder, or forearm) and hip fracture in patients over 40 years of age were estimated by the World Health Organization (WHO) fracture risk assessment (FRAX) tool (including BMD measurement) using the web-based algorithm adapted for Hong Kong at http://www.shef.ac.uk/FRAX and were adjusted for the GCs dosage. 11 Accordingly, the risk of major osteoporotic fractures and hip fracture were increased by 15% and 20%, respectively for patients on equivalent prednisolone dosage of 7.5 mg or more; and for patients on equivalent prednisolone dosage of less than 2.5 mg, the major osteoporotic fractures and hip risk were decreased by 20% and 35%, respectively.
BMD assessment
BMD determined by DXA scans was recorded by absolute value in g/cm2, T-score (number of standard deviations above or below mean results of young adults), and Z-score (number of standard deviations above or below mean results of age-matched population) at lumbar spine L1-4 and neck of femur (NOF). The prevalence of reduced BMD at lumbar spine, or NOF, of the IIM patients was compared to that of non-rheumatological, RA, and SLE patients. Reduced BMD was defined as either osteopenia or osteoporosis at lumbar spine or NOF. In postmenopausal women and men, osteoporosis was defined as a T-score of ⩽−2.5 and osteopenia was defined by a T-score between ⩽−1.0 and −2.5 according to the WHO criteria. 12 For premenopausal women, Z-score was used for comparison and a Z-score of ⩽−2.0 was regarded as osteoporosis according to the definition from the International Society for Clinical Densitometry. 13
Statistical analysis
The mean BMD in Hong Kong females at the age 50 years was 0.94 g/cm2 with a standard deviation of 0.10 g/cm2 at lumbar spine. 14 In a local cross-sectional study of female IIM patients, the mean BMD was 0.87 g/cm2 at lumbar spine. 3 The required sample size was 63 to detect the difference and achieve an 80% power assuming a 5% type I error.
Descriptive statistics were expressed as frequencies with percentages for categorical variables, mean with standard deviation, or median with range for continuous variables. Categorical variables were compared by chi-square test. Continuous variables were compared by Independent Student’s t-test or Mann–Whitney U test. Chi-square test was used to compare the primary outcome. Binary logistic regression was used to adjust for potential confounders. The prevalence of reduced BMD between IIMs, RA, and SLE were compared by a 2 × 3 contingency table and the BMD results were compared by ANOVA test. Where appropriate, Pearson’s or Spearmen’s correlation tests were used for comparison of continuous variables with BMD. The demographics and clinical variables independently associated with BMD were determined by multivariate linear regression. A p-value of <0.05 was denoted as statistically significant. Statistical analyses were performed by IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 15
Results
A total of 230 patients were included in this study, of which 65 were IIM patients, 65 were non-rheumatological controls, 50 were RA patients, and 50 were SLE patients. The demographic features and medication use of IIM patients and non-rheumatological controls are shown in Table 1. The mean age of IIM patients was 58.6 ± 11.0 years and 50 (76.9%) were female. The most common IIM subtype was dermatomyositis (DM), accounting for 36.9%, followed by polymyositis (PM) or immune mediated necrotizing myopathies (IMNM) at 29.2%. The median disease duration of IIMs patients from diagnosis to the time of DXA was 51 months (2–432 months). Almost all IIMs patients (98.5%) had received GCs and 83.1% of IIMs patients were active GC user at the time of the DXA scan, compared to 52.3% and 35.4%, respectively in the non-rheumatological controls (both p < 0.001). Most IIM patients (87.7%) had received high-dose GCs which was significantly more common than the non-rheumatological controls (36.9%, p < 0.001). There were significantly more IIM patients on immunosuppressants (92.3% versus 38.5%, p < 0.001) and biologic or targeted synthetic disease modifying anti-rheumatic drugs (b/tsDMARDs) (13.8% versus 1.5%, p = 0.01). There was no difference in smoking, drinking, menopausal status, and comorbidity status between the two groups. The clinical characteristics of the IIM patients are presented in Table 2. Among them, 78.5% had positive myositis-specific autoantibodies, with anti-MDA5 (16.9%), anti-TIF1 (12.3%), and anti-SRP (12.3%) being the most common autoantibodies identified. The mean MMT8 at the time of DXA scan was 73.9 ± 6.7. The diagnoses of the non-rheumatological controls and the indications of DXA are summarized in Supplemental Tables 1 and 2.
Demographics and medications of idiopathic inflammatory myopathy patients and non-rheumatological controls.

b/tsDMARDs, biologic or targeted synthetic disease modifying anti-rheumatic drugs; BMI, body mass index; DXA, dual-energy; GC, glucocorticoids; IIM, inflammatory myopathies; X-ray-absorptiometry.
Clinical characteristics of the 65 recruited patients with idiopathic inflammatory myopathies.

CK, creatine kinase; CRP, C-reactive protein; DXA, dual-energy.
The prevalence of reduced BMD, osteopenia, and osteoporosis in the IIM group were 73.8%, 44.6%, and 29.2% compared to 56.9%, 43.1%, and 13.8%, respectively in the non-rheumatological controls (Table 3). The prevalence was significantly higher for reduced BMD (p = 0.043) and osteoporosis (p = 0.033). The mean BMD at lumbar spine was 0.886 ± 0.181 g/cm2 and the mean BMD at NOF was 0.651 ± 0.144 g/cm2, significantly lower than those of the control group (lumbar spine: 0.960 ± 0.143 g/cm2, p = 0.011; NOF: 0.751 ± 0.127 g/cm2, p < 0.001), as shown in Figure 1. Fragility fractures occurred in 13 IIM patients and 10 non-rheumatological controls (p = 0.491), with vertebral fracture being the most common in both groups. Multivariate analysis showed that IIM was independently associated with reduced BMD (odds ratio: 2.12, p = 0.048, 95% CI 1.01–4.46). Ever usage of GCs, immunosuppressants, or b/tsDMARDs were not associated with increased prevalence of reduced BMD. In the subgroup analysis, the mean BMD of lumbar spine and NOF were still significantly lower in IIM patients [BMD at lumbar spine: 0.891 ± 0.159 g/cm2 (IIMs) versus 0.966 ± 0.149 g/cm2 (controls), p = 0.037; BMD at NOF: 0.663 ± 0.130 g/cm2 (IIMs) versus 0.764 ± 0.137 g/cm2 (controls), p = 0.002] compared to matched non-rheumatological controls who were also on GCs. The median major osteoporotic fracture scores were 8.05% in the IIM group and 3.75% in the non-rheumatological controls (p = 0.005) and the median hip fracture scores were 1.77% in the IIMs group and 0.70% in the control group (p = 0.016). Eleven IIM patients had an estimated 10-year major osteoporotic fracture risk of more than 20% by FRAX, compared to one in the non-rheumatological controls (p = 0.002). A total of 23 IIM patients had an estimated 10-year femur fracture risk of more than 3% by FRAX versus 11 in the non-rheumatological controls (p = 0.015).
Bone health parameters in idiopathic inflammatory myopathy patients and non-rheumatological controls.
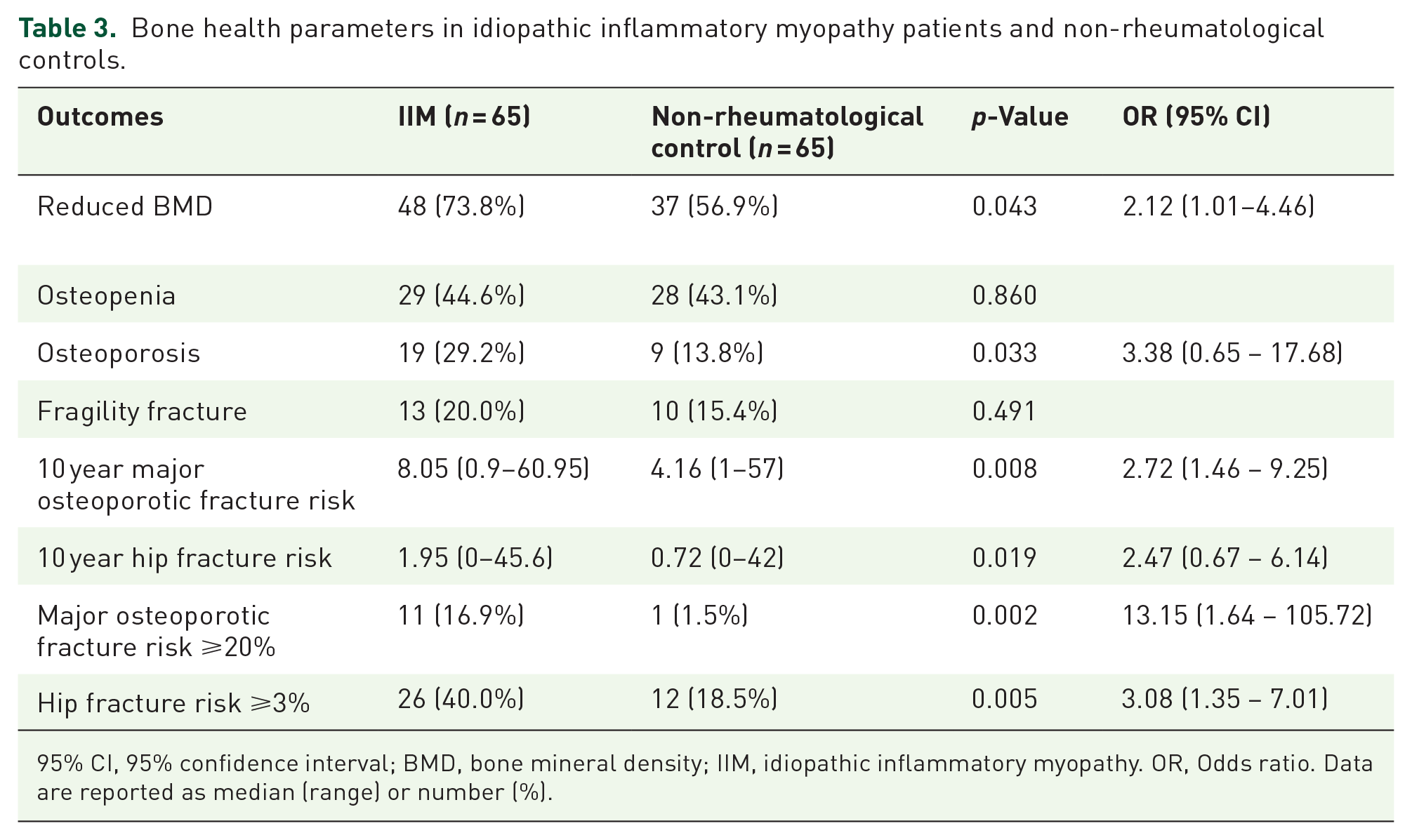
95% CI, 95% confidence interval; BMD, bone mineral density; IIM, idiopathic inflammatory myopathy. OR, Odds ratio. Data are reported as median (range) or number (%).

Bone mineral density in idiopathic inflammatory myopathy patients and non-rheumatological controls.
The female IIM patients were compared with age-matched RA and SLE controls. Only female IIM patients were included in this comparison due to an imbalanced number of male RA/SLE controls. The mean age of the female IIM patients was 59.3 ± 10.4 years. Among the RA controls, 43 (86%) had received GCs and 28 (56%) remained on GCs at the time of DXA scan, at an average dose of prednisolone 3.3 ± 3.4 mg per day. The mean DAS-28 by Erythrocyte Sedimentation Rate (ESR) was 3.93, corresponding to moderate disease activity. Among the SLE patients, almost all (98%) had received GCs and 45 (90%) were active GC users at the time of DXA scan with a mean prednisolone dose of 5.0 ± 5.5 mg per day. The median disease duration was the longest in the SLE group (198 months, p < 0.001), versus 96 months in the RA group and 63.5 months in the myositis group. More RA patients were on b/tsDMARDs than IIM and SLE patients (RA: 30%, IIM: 10%, SLE: 4%, p = 0.001). The demographic and clinical characteristics of the three groups were shown in Table 4.
The comparison of demographics, clinical variables, and bone health parameters between female idiopathic inflammatory myopathy patients versus RA and SLE controls.

b/tsDMARDS, biologic/targeted synthetic disease modifying anti-rheumatic drugs; BMI, body mass index; DAS28, Disease Activity Score 28; DXA, dual-energy X-ray absorptiometry; GCs, glucocorticoids; IIMS, inflammatory myopathies; MMT8, Manual muscle testing 8; SLE, systemic lupus erythematosus; SLEDAI, Systemic lupus erythematosus disease activity index.
Data are reported as mean ± standard deviation, median (range), or number (%).
Thirty-eight (76%) female IIM patients had reduced BMD, compared to 38 (76%) in the RA group and 31 (62%) in the SLE group (p = 0.205). Among those, 18 (36%) IIM patients had osteoporosis versus 14 (28%) in the RA group and 8 (16%) in the SLE group (p = 0.076). However, the mean BMD at the NOF was the lowest in the myositis group (0.641 ± 0.152g/cm2 versus 0.663 ± 0.102 g/cm2 in the RA group versus 0.710 ± 0.130 g/cm2 in the SLE group, p = 0.029), as shown in Figure 2. The mean BMD at the lumbar spine was not significantly different between the groups (IIM: 0.882 ± 0.195g/cm2 versus RA: 0.896 ± 0.175 g/cm2 versus SLE: 0.906 ± 0.171 g/cm2, p = 0.819). The prevalence of osteoporosis and the mean BMD at the NOF were compared between IIM and RA controls, as well as IIM and SLE controls separately. Osteoporosis was more prevalent in IIM patients than SLE controls (p = 0.023), while there was no difference between IIM and RA group (p = 0.391). Similarly, the mean BMD at the NOF was significantly lower in IIM patients compared to SLE group (p = 0.021), but such difference was not observed between IIM and RA group (p = 0.392). Multivariate analysis by linear regression confirmed that IIMs were independently associated with lower hip BMD (p = 0.013), while ever usage of prednisolone (p = 0.332), disease duration (p = 0.646) and the use of b/tsDMARDs (p = 0.959) were not.
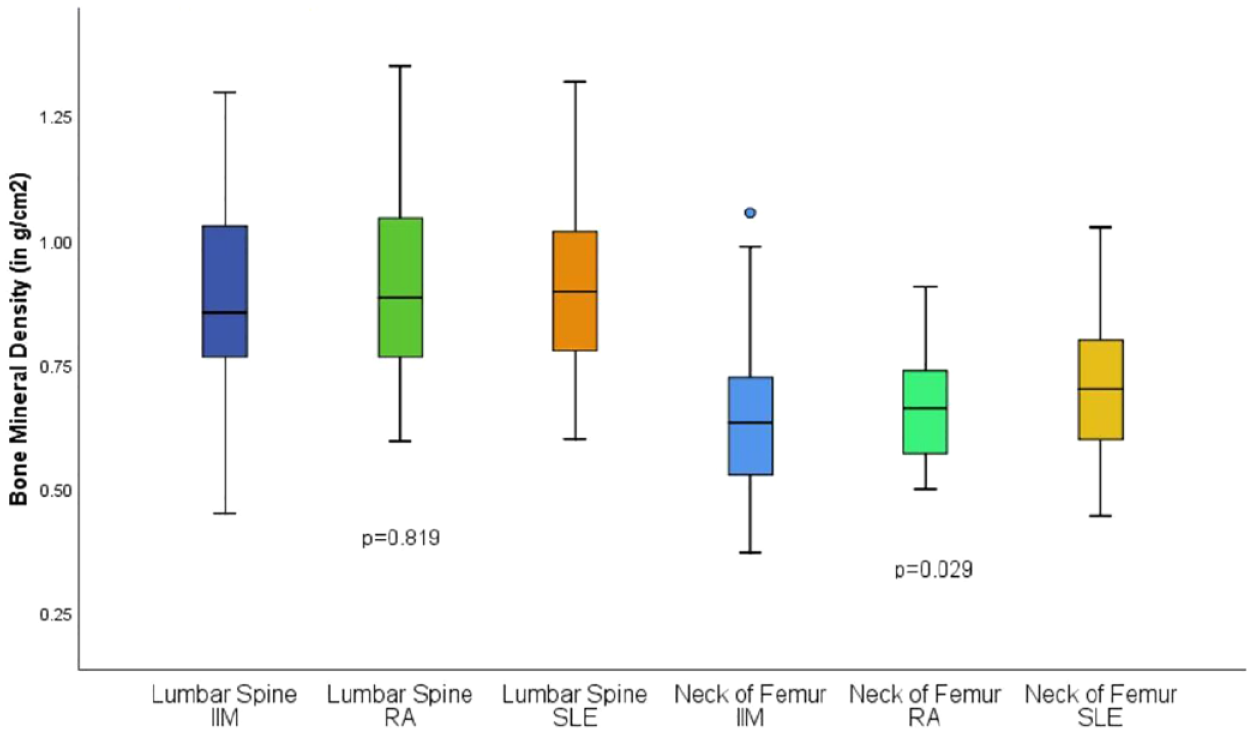
Bone mineral density in idiopathic inflammatory myopathy, rheumatoid arthritis, and systemic lupus erythematosus patients.
Among IIM patients, advanced age (p = 0.006) and lower BMI (p = 0.001) were found to be correlated with lower BMD at lumbar spine while ever usage of high-dose GCs tended to be associated with lower lumbar spine BMD (p = 0.053). Regarding the hip, in addition to advanced age (p < 0.001) and lower BMI (p = 0.003), postmenopausal status (p = 0.006) and personal history of fragility fracture (p = 0.015) were associated with lower BMD. Lower MMT8 (p = 0.051) and the presence of comorbidities (p = 0.052) demonstrated tendencies to be associated with lower hip BMD. Otherwise, no association between BMD with myositis subtype (DM/PM/IMNM/clinically amyopathic DM/ASS), disease duration, CK or CRP was found. In the multivariate analysis, lower BMI and more advanced age were found to be independently associated with lower BMD in both NOF (β = 0.369, p = 0.003 and β = −0.623, p < 0.001, respectively) and lumbar spine (β = 0.399, p = 0.001 and β = −0.336, p = 0.006, respectively).
Discussion
To our knowledge, this is the first study comparing BMD in IIM patients with non-rheumatological, RA, and SLE controls. It was found that reduced BMD and osteoporosis were common in IIMs, affecting 73.8% and 29.2% of IIM patients, respectively. The BMD at lumbar spine and NOF were significantly lower in IIM patients when compared to non-rheumatological controls. Having IIM was associated with a 2.1 times independent risk of reduced BMD compared to patients with non-rheumatic diseases. Though the prevalence of reduced BMD did not differ significantly between the IIMs, RA, and SLE patients in this study, the mean hip BMD was the lowest in patients with IIM. RA patients were selected as controls, as RA is a well-recognized inflammatory condition associated with accelerated bone loss. 16 SLE patients were selected due to their comparable usage of GCs as in IIMs patients. In a recent Taiwan study by Lee et al. comparing the incidence of osteoporosis or fractures in DM or PM patients with propensity score-matched controls, DM/PM patients were three times more likely to have osteoporosis. DM or PM patients were also more likely to sustain nontraumatic fractures, with an adjusted hazard ratio of 3.77. 17 A cross-sectional study in Hungary by Vincze et al. comparing the prevalence of osteoporosis in IIM and RA patients also found that IIM patients had elevated risks for osteoporosis. 18 It was shown that 60% and 13.5% of participating IIM patients had osteopenia and osteoporosis, respectively, in contrast to 39.5% and 7% in the RA controls.
There are several possible explanations to the increased risk of osteopenia or osteoporosis in IIM patients. Firstly, significantly more IIMs patients were on GCs than the non-rheumatological controls and the majority required high-dose GCs. GCs induced apoptosis of mature osteoblasts and osteocytes. 19 It also stimulated the production of Wnt pathway inhibitors such as Dickkopf-1 and sclerostin and increased the lifespan of osteoclasts by stimulating receptor activator for nuclear factor-kB ligand (RANKL) and suppressing osteoprotegerin.20,21 However, the use of GCs was not found to be independently associated with increased risk of reduced BMD in the multivariate analysis. This could be related to the high proportion of GC exposure in both IIM patients and controls. On the other hand, examining the cumulative GC dose might be more reflective of the total burden of the medication. Another explanation is the effect of disability on bone health. A cross-sectional BMD study identified reduced physical activity to be associated with axial bone loss in female RA patients. 22 Disability is a common problem in IIMs, 38.8% of IIM patients had mild degree of disability on health assessment questionnaire disability index (HAQ-DI) and 43.7% had moderate to severe disability in the study by Ponyi et al. 23 Unfortunately, HAQ-DI was not assessed in this study. Further study to evaluate the association of functional disability and bone loss in IIM patients might be insightful.
Among all the rheumatological patients included in this study, 71% had reduced BMD and 26.7% had osteoporosis. Systemic inflammation was associated with upregulated bone resorption and impaired bone formation. Proinflammatory cytokines such as TNF, IL1, and IL6 enhanced osteoclastogenesis mediated by RANKL and macrophage-colony stimulating factor 1. 24 These proinflammatory cytokines were upregulated in IIMs and played a major role in the co-stimulation, activation, and transmigration of inflammatory cells into muscle fibers. 25 We previously demonstrated that a higher Myositis Disease Activity Assessment Visual Analog Scales (MYOACT) score was associate with lower BMD. 3 In the current study, although MYOACT score was not documented, lower MMT8 tended to correlate with lower BMD, though a low MMT8 could be contributed by both disease activity or residual damage.
Thirteen (20%) IIM patients had a history of fragility fracture in this study, with five cases of vertebral fracture and eight cases of non-vertebral fracture. A cross-sectional study by Gupta et al. revealed 46% of Indian IIM patients had asymptomatic vertebral fractures, with 19% having more than one fracture. 26 The prevalence of osteoporosis in that study was 26.9%, which was comparable to our result. The apparently lower incidence of fractures in our study could be explained by the lack of universal spinal radiographs to detect asymptomatic morphometric vertebral fractures. The majority of fragility fractures that occurred in our study were symptomatic, and more than half involved non-vertebral sites. The significantly higher FRAX scores in IIM patients compared to non-rheumatological controls again highlighted the increased risk of poor bone health in these patients. In fact, a recently published guideline from the British Society for Rheumatology recommended that bone health assessment should be performed and appropriate treatment instigated regardless of GC treatment for IIM patients in view of the heightened fracture risk. 27
There are several limitations to this study. First, only BMD was compared, which only measured the quotient of the bone mineral content divided by the bone area but could not reflect the actual bone quality. The use of trabecular bone score in addition to BMD would be able to reflect the microarchitecture better and has been shown to improve fracture risk prediction. 28 Second, this study was not adequately powered to detect the difference in the prevalence of fragility fracture between the groups. The non-rheumatological controls recruited in this study were heterogeneous and some of them had underlying risk factors of osteoporosis, which might explain the similar rates of fragility fracture in this study. Third, the current study included IIM patients with various disease duration. Reduced BMD in rheumatic diseases is a complex process in which systemic inflammation, disability, and the use of GCs all contribute to the accelerated bone loss. GCs undoubtedly could adversely affect bone health but on the other hand, its use to suppress disease activity and improve mobility early in the disease course might reduce the detrimental effect of inflammation and immobility on bone. Prospective studies assessing BMD together with disease activity by MYOACT and disability by HAQ-DI regularly at different time points would be helpful to delineate the exact role of individual risk factors. Fourth, the optimal usage of anti-osteoporotic treatment was limited by the local reimbursement policy, which could affect the generalizability of the results. Lastly, due to the retrospective nature of the study, selection bias introduced by the various indications of DXA could not be ruled out.
Conclusions
IIM patients were at higher risks of reduced BMD than the age- and sex-matched non-rheumatological controls independent of GC use. Compared to patients with RA and SLE, the hip BMD was the lowest in IIM patients. Older age and lower BMI were associated with lower BMD at hip and lumbar spine in patients with IIMs. Vigilant monitoring of BMD and timely initiation of bone protective therapies should be considered in IIM patients, especially those who are at advanced age and underweight.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X231181968 – Supplemental material for Reduced bone mineral density in patients with idiopathic inflammatory myopathies: a case-control study
Supplemental material, sj-docx-1-tab-10.1177_1759720X231181968 for Reduced bone mineral density in patients with idiopathic inflammatory myopathies: a case-control study by Iris Yan Ki Tang, Lucas Luk, Victor Wong, Steve Pang, Virginia Lao and Ho So in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
All authors critically revised thearticle for important intellectual content. Specific roles included: study design (IT, HS), data collection (IT, LL, VW, SP, VL, HS), data analysis (IT, HS), and drafting of article (IT, VL, HS).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
