Abstract
Objective
To investigate the prevalence of low bone mineral density (BMD) along with its possible risk factors in male Han Chinese patients with alcohol dependence (AD).
Methods
This retrospective, cross-sectional study included male patients with AD, classified into normal and low BMD groups according to bone densitometry T scores. Demographic and alcohol-related data, and routine laboratory parameters were compared between the two groups. Binary logistic regression analysis was employed to evaluate risk factors associated with low BMD, and correlations between the T-score and demographic, alcohol-related, and routine laboratory data were evaluated.
Results
Among a total of 107 patients with AD included in the study, the prevalence of low BMD was 70.09% (75/107). Patients with low BMD were older, consumed more alcohol daily, and had higher lactate dehydrogenase (LDH) and lower Ca2+ levels than patients with normal BMD. Regression analysis revealed that increased daily alcohol intake, low serum Ca2+ levels, high serum LDH levels, and comorbid hypertension was related to low BMD in patients with AD. Further correlation analysis revealed a positive association between T-score and serum Ca2+ levels.
Conclusion
Increased daily alcohol intake, low serum Ca2+ levels, high serum LDH levels, and comorbid hypertension may be risk factors for low BMD.
Introduction
According to the 2018 Global Status Report on Alcohol and Health estimates by the World Health Organization (WHO), the harmful use of alcohol accounts for approximately three million deaths. 1 Harmful alcohol consumption causes significant health issues, along with high economic costs for society and the healthcare sector. 2 Notably, the prevalence of low bone mineral density (BMD) is significantly increased in alcoholic patients, due to associated risk factors including lack of exercise, poor nutrition, medication, and a high rate of smoking.3,4
Osteopenia and osteoporosis, characterized by low BMD, increase the risk of fragility fractures and bone injuries. 5 However, an association between alcohol consumption and fracture outcomes is not yet clearly established. A meta-analysis reported an association between higher alcohol intake (42 drinks per day) and an increased risk of non-spinal fractures, including osteoporotic and hip fractures. 3 Alcohol dependence (AD) leads to several medical problems, including liver dysfunction, vitamin D deficiency, and hyperparathyroidism, which may increase the risk of developing osteopenia and osteoporosis. 6 Moreover, chronic alcohol misuse commonly causes secondary osteoporosis in men. 7 More than four times higher incidence of fractures has been reported in patients with chronic alcoholism than in age-matched controls, 8 and an in vitro study showed that ethanol inhibits osteoblast proliferation and function. 9 Conversely, other studies did not find a significant association between moderate daily alcohol consumption and the risk of hip or vertebral fractures.10,11 These differences may be attributed to the diverse patterns and habits of alcohol consumption, as well as the varying classifications of drinking across different countries and regions. In China, AD is a significant public health concern, particularly among males, and high-quality studies have estimated the current and lifetime prevalence of AD in China as 2.2% and 3.7%, respectively. 12 Furthermore, a cross-sectional study revealed a high prevalence of osteoporosis (5.0% among males aged ≥ 40 years) and fractures (10.5% among males) in China. 13 However, studies on the prevalence of low BMD and its possible risk factors in male Han Chinese patients with AD remain limited.
Considering the high prevalence of AD and low BMD in China, and the association between AD and low BMD, the aim of the present study was to investigate the prevalence of low BMD and its potential risk factors in male Han Chinese patients with AD. To the best of our knowledge, this study is the first to investigate the potential risk factors for low BMD in male Han Chinese patients with AD. The present findings may have significant clinical implications for the prevention and treatment of osteopenia and osteoporosis associated with AD.
Patients and methods
Study population
Patients hospitalized at the Anhui Mental Health Centre between August 2021 and August 2022 were consecutively selected for enrolment into this observational, cross-sectional, retrospective study. The inclusion criteria were as follows: (1) diagnosis of AD by a psychiatrist according to the International Classification of Diseases 10th Revision criteria; (2) aged between 18 and 65 years; and (3) completion of benzodiazepine replacement therapy to reduce AD. Benzodiazepines are administered to hospitalized patients to lessen the symptoms of alcohol withdrawal, and consequently, patients usually attain a relatively stable state for cooperation with bone health measurements. The exclusion criteria were as follows: (1) severe long-term infections, chronic bedridden conditions, and/or severe disorders of the heart, lung, liver (including fibrosis, cirrhosis, hepatocellular carcinoma, and liver failure), and/or renal system; (2) long-term use of glucocorticoids or immunosuppressants; and/or (3) diseases of the adrenal glands and gonads, bone tumours, hyper- or hypothyroidism, and/or other diseases affecting bone metabolism. All participants provided written informed consent before study participation, and the study protocol adhered to the principles of the Helsinki Declaration of 1975, revised in 2013. All patient details were de-identified. The study was approved by the Ethics Committee of the Hefei Fourth People’s Hospital, Anhui Mental Health Centre (approval No. IRB-HFSY-YJ-LW-LY [2022003], dated 12 August 2022) and the reporting of the study conforms with STROBE guidelines. 14
Clinical data collection and bone health measurements
Routine laboratory parameters were retrieved from participant’s electronic medical records. Demographic and clinical data, including age, body mass index, alcohol use (years of drinking and daily alcohol intake), comorbid diseases, smoking history, were collected using a short questionnaire, with alcohol-related data and smoking history based on retrospective self-reporting.
Bone health measurements were performed on the right calcaneus bone using a SONOST 3000 portable ultrasound bone densitometer (OsteoSys; Seoul, South Korea). The SONOST 3000 measures broadband ultrasound attenuation and the speed of sound, both of which are temperature-dependent. Quantitative ultrasound effectively predicts fracture risk and BMD, potentially reducing the reliance on dual-energy X-ray absorptiometry. 15 Each participant was evaluated for 15 s. The procedure was performed by a single trained evaluator with substantial experience (LG). BMD was assessed by the T-score derived using the SONOST 3000 bone mineral densitometer in accordance with WHO guidelines. 16 In general, bone density is indicated by T-scores as follows: a score of ≥ –1 is normal; –2.5 to –1 indicates osteopenia, a condition where the bone density is below normal, which may lead to osteoporosis; and ≤ –2.5 indicates osteoporosis. Patients with a T-score of ≥ –1 SD and < –1 SD were assigned to normal and low BMD groups, respectively.
Statistical analyses
Data are presented as mean ± standard error of the mean (SEM) or n (%) prevalence. The normality of continuous data distribution was analysed using one-sample Kolmogorov–Smirnov test. Between-group differences in normally distributed continuous data (demographic data, alcohol-related data, and routine laboratory parameters in normal and low BMD groups) were analysed by Student’s t-test for independent samples, while χ2-test was used to analyse between-group differences in categorical data regarding comorbid diseases and smoking history. Variables with a P-value ≤0.1 by Student’s t-test or χ2-test were entered into a binary logistics regression model to evaluate risk factors related to low BMD. The area under the curve (AUC) of the influencing factors was examined for their ability to distinguish patients with low BMD from those with normal BMD using receiver operating characteristic (ROC) curve analysis. The correlation between T-score and demographic data, alcohol-related data, routine laboratory parameters, comorbid diseases, and smoking history was evaluated using Pearson’s correlation coefficient for continuous data or Spearman’s rank correlation coefficient for categorical data. All statical analyses were performed using IBM SPSS software, version 17.0 (IBM Corp., Armonk, NY, USA) and a P value <0.05 was considered statistically significant.
Results
Comparison of demographic and alcohol-related data, comorbid diseases, smoking history, and routine laboratory parameters between the groups
A total of 107 male Han Chinese patients with AD were included in the study, all of whom had alcoholic fatty liver. Participants were classified into normal and low BMD groups based on their T-scores, and comprised 32 patients (29.91%) with normal BMD and 75 patients (70.09%) with low BMD. Demographic and alcohol-related data, routine laboratory parameters, comorbid diseases, and smoking history of the two groups are summarised in Table 1. Notably, patients with low BMD were older, consumed more alcohol daily, and had higher lactate dehydrogenase (LDH) and lower Ca2+ levels than those with normal BMD (all P < 0.05). No statistically significant differences in other variables were observed between the groups (all P > 0.05; Table 1).
Comparison of demographic and clinical data between 107 male Han Chinese patients with alcohol dependence, categorised according to normal or low BMD.

Data presented as n prevalence or mean ± SEM.
Normal BMD, T score ≥ –1 SD; low BMD, T score < –1 SD.
BMD, bone mineral density; BMI, body mass index; TBIL, total bilirubin; DBIL, direct bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; γ-GGT, γ-glutamyl-transpeptidase; TBA, total bile acid; TC, total cholesterol; HDL, high density lipoprotein; TG, triglyceride; Apo, apolipoprotein; BUN, blood urea nitrogen; UA, uric acid; CK, creatine kinase; CK-MB, creatine kinase-myocardial band isoenzyme; LDH, lactate dehydrogenase; PHOS, phosphorus; TT3, total triiodothyronine; TT4, total thyroxine; fT3, free triiodothyronine; fT4, free thyroxine; TSH, thyroid stimulating hormone.
Student’s independent samples t-test for normally distributed continuous data (demographic and alcohol-related data, and routine laboratory parameters); or χ2-test for categorical data regarding comorbid diseases and smoking history.
Factors associated with BMD in patients with AD
Variables with a P value < 0.1 for between-group comparisons were entered into a binary logistic regression model (Table 2). Age, daily alcohol intake, Ca2+, LDH, hypertension, and globulin levels were used as variables to estimate the risk of low BMD. Notably, increased daily alcohol intake, low serum Ca2+ levels, high serum LDH levels, and comorbid hypertension were found to be significantly associated with low BMD in male Han Chinese patients with AD (all P < 0.05; Table 2).
Binary logistics regression analysis of risk factors associated with low BMD in 107 male Han Chinese patients with alcohol dependence.
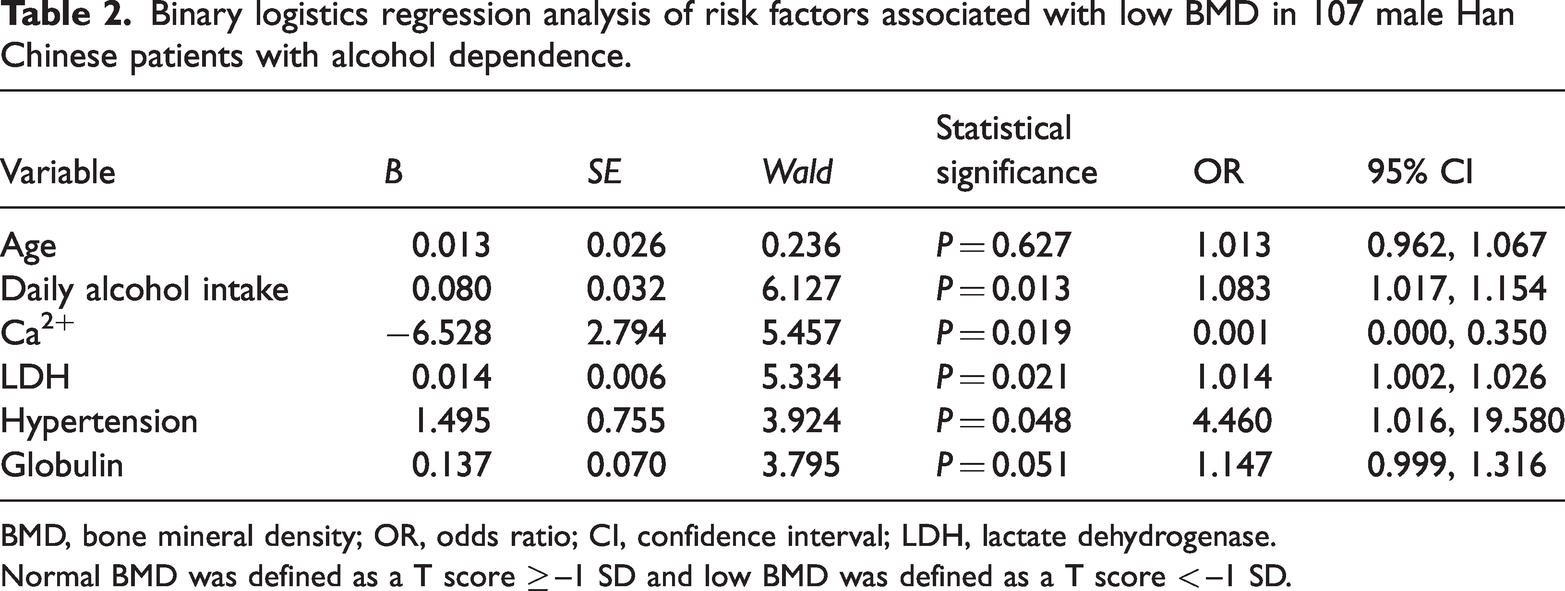
BMD, bone mineral density; OR, odds ratio; CI, confidence interval; LDH, lactate dehydrogenase.
Normal BMD was defined as a T score ≥ –1 SD and low BMD was defined as a T score < –1 SD.
Value of associated factors in distinguishing between patients with low versus normal BMD
The value of the associated factors in distinguishing patients with low BMD from those with normal BMD was analysed using ROC curves (Figure 1). Notably, a combined panel of daily alcohol intake, Ca2+, LDH, and hypertension could differentiate patients with low BMD from those with normal BMD with high accuracy (AUC = 0.783, P < 0.001; 95% CI 0.693, 0.873).

Receiver operating characteristic curve of the value of associated factors (a combined panel of daily alcohol intake, Ca2+, lactate dehydrogenase, and hypertension) in differentiating patients with low bone mass density (BMD) from patients with normal BMD in 107 male Han Chinese patients with alcohol dependence. AUC, area under the curve.
Relationship between T-score and demographic and alcohol-related data, comorbid diseases, smoking history, and routine laboratory parameters
The relationships between T-scores and demographic and alcohol-related data, comorbid diseases, smoking history, and routine laboratory parameters were further analysed using Pearson’s correlation coefficient or Spearman’s rank correlation coefficient (Figure 2). Notably, T-score was positively associated with serum Ca2+ levels (r = 0.271, P = 0.005).

Correlation between T score and the demographic and alcohol-related data, comorbid diseases, smoking history, and routine laboratory parameters in 107 male Han Chinese patients with alcohol dependence. BMI, body mass index; ALP, alkaline phosphatase; TSH, thyroid stimulating hormone; UA, uric acid; TG, triglyceride; CK-MB, creatine kinase-myocardial band isoenzyme; CK, creatine kinase; DBIL, direct bilirubin; TBIL, total bilirubin; HDL, high density lipoprotein; TBA, total bile acid; γ-GGT, γ-glutamyl-transpeptidase; AST, aspartate aminotransferase; LDH, lactate dehydrogenase; ALT, alanine aminotransferase; Apo, apolipoprotein; TC, total cholesterol; BUN, blood urea nitrogen; FT3, free triiodothyronine; TT3, total triiodothyronine; FT4, free thyroxine; TT4, total thyroxine; PHOS, phosphorus. ×, no statistically significant correlation (P > 0.05).
Discussion
The present study compared demographic and alcohol-related data, comorbid diseases, smoking history, and routine laboratory parameters between normal- and low-BMD groups to investigate the prevalence of low BMD and its possible risk factors in male Han Chinese patients with AD. The study yielded four major findings. First, the prevalence of low BMD in patients with AD was 70.09%; secondly, patients with low BMD were older, consumed more alcohol daily, and had higher LDH and lower Ca2+ levels than those with normal BMD; thirdly, increased daily alcohol intake, low serum Ca2+ levels, high serum LDH levels, and comorbid hypertension were significant predictors of low BMD in male Han Chinese patients with AD; and fourthly, T-score was positively associated with serum Ca2+ levels.
The present 70.09% prevalence of low BMD in male Han Chinese patients with AD is consistent with previous epidemiological data reporting bone density loss in 88.6% of male patients with AD, along with 34.1% incidence of osteoporosis. 17 The present results support the notion that patients with AD experience increased bone density loss compared with the healthy population that has a reported 30.9% prevalence of low BMD. 18
Advanced age is the most reliable risk factor for low BMD, 19 and fractures due to osteoporosis increase with age.20,21 Specifically, the combined effect of a decline in physical activity, intestinal calcium absorption, and the ability of the kidney to synthesize 1,25-(OH)2D3 contributes to age-related bone loss.22,23 Additionally, older adults may experience a slower rate of bone turnover because of relative and secondary increases in parathyroid hormone levels. 24 As expected, patients with low BMD were older than those with normal BMD in the present study.
Although light-to-moderate alcohol consumption may be beneficial to increasing bone density and reducing age-related bone loss,25,26 chronic alcohol consumption may increase the likelihood of osteoporosis and fractures. 27 Bone density has been shown to significantly increase in the lumbar spine and femur of male patients with AD after 3–4 years of abstinence. 17 In the present study, the daily alcohol intake was significantly higher in patients with low BMD than those with normal BMD. Moreover, increased daily alcohol intake was a significant predictor of low BMD in male Han Chinese patients with AD. Alcohol-induced low BMD may be due to the following: (1) AD may cause several medical issues, including liver insufficiency, vitamin D deficiency, and hyperparathyroidism, all of which increase the risk of osteoporosis; 28 (2) alcohol may interfere with bone remodelling, enhance bone fragility, and increase bone loss; 29 and (3) alcohol may interfere with bone metabolism through direct toxic effects on osteoblasts.30,31 Notably, no significant difference between the two BMD groups was observed in the present study regarding years of drinking, suggesting that daily alcohol intake, rather than the years of drinking, is related to low BMD in male Han Chinese patients with AD.
Lactate dehydrogenase is an intracellular enzyme widely distributed throughout the body, with high levels in tissues that use glucose as an energy source. 32 The present results indicated that LDH levels were significantly higher in patients with low BMD than those with normal BMD, and LDH was a significant predictor of low BMD in male Han Chinese patients with AD. Similarly, high serum LDH activity has been observed in male Japanese patients with AD. 33 A relationship between high serum LDH activity and low BMD has also been observed in sickle-cell disease. 34 Although the possible mechanisms underlying this relationship remain unclear, it may be related to liver function damage due to AD. As serum LDH activity increases in the early stages of liver disease, 35 AD may cause damage to liver function, leading to an increase in serum LDH levels.
Common complications in patients with AD include hypertension, 36 diabetes, 37 and liver cirrhosis, 38 all of which are associated with low BMD and fracture risk. 39 Two previous studies reported a significant association between low BMD and hypertension.33,40 Consistent with other studies, the present study revealed that comorbid hypertension was an influencing factor for low BMD in male Han Chinese patients with AD. Although the mechanism underlying the association between hypertension and low BMD remains largely unknown, it may partly involve shared risk factors, such as age, as well as common underlying mechanisms involved in regulating the skeletal and cardiovascular systems.41,42
Ca2+ is an important nutrient required for bone development and accretion. 43 Persistent low Ca2+ diet increases the risk of low BMD, osteoporosis, and fractures. 44 Moreover, Ca2+ and vitamin D are the most commonly discussed nutrients for preventing low BMD. 45 Alcohol has been reported to reduce serum Ca2+ concentrations in several animal and clinical studies.46,47 Furthermore, excessive alcohol consumption may cause vomiting and diarrhoea, resulting in reduced Ca2+ absorption. 48 In the present study, serum Ca2+ levels were significantly lower in patients with low BMD versus those with normal BMD, and binary logistic regression analysis revealed low serum Ca2+ levels to be associated with low BMD in male Han Chinese patients with AD.
Liver disease can potentially reduce BMD,49,50 and secondary osteoporosis and increased bone fragility are underrecognized complications in patients with chronic liver disease. 51 Notably, up to one-third of these individuals fulfil the diagnostic criteria for osteopenia or osteoporosis. 51 Similarly, individuals with chronic liver disease exhibit a significant risk of nontraumatic bone fractures, 52 with the prevalence ranging from 7% to 35%, 53 and the incidence of fractures is two to three times higher in patients with end-stage chronic liver disease than in healthy controls. 54 In the present study, all patients had alcoholic fatty liver. However, due to the retrospective study design, the duration and severity of the illness was not documented. Consequently, direct assessment of the impact of duration and severity of liver disease on bone density was not possible. However, liver function-related indicators, including TBIL, DBIL, ALT, and AST levels, were compared between the normal- and low-BMD groups. The results indicated no significant between-group differences, suggesting that liver function may not be a contributing factor for low BMD in male Han Chinese patients with AD. Nevertheless, further studies are warranted to investigate the effects of chronic liver disease severity on BMD.
The results of the present study may be limited by several factors. First, the sample size in this single-centre study was relatively small. Secondly, several potential influencing factors associated with low BMD, such as 25-hydroxyvitamin D and parathyroid hormone levels, could not be evaluated due to the retrospective study design. Thirdly, all patients with AD in the present study population were male, which limits the generalizability of the results. Fourthly, the study evaluated a large number of risk factors using a limited sample size, which increases the possibility of false-positive associations in the correlation analysis. Consequently, independent studies with large sample sizes and smaller P-values are warranted. Fifthly, as liver disease may contribute to reduced BMD, 49 consideration of the severity of alcoholic fatty liver disease is crucial in these patients. Sixthly, this study involved a nonstandardized survey questionnaire, which was constructed based on the available literature, and the study results may have been affected by potential recall bias, as information on alcohol intake was based on retrospective self-reporting. Lastly, due to the observational, cross-sectional study design, a definitive conclusion regarding cause and effect cannot be made. Further studies with larger sample sizes and longitudinal design may enable more definitive conclusions to be made.
In conclusion, the present study revealed that male Han Chinese patients with low BMD were older, with higher daily alcohol intake and LDH levels and lower Ca2+ levels, compared with those with normal BMD. To the best of our knowledge, the present study is the first to confirm that increased daily alcohol intake, low serum Ca2+ levels, high serum LDH levels, and comorbid hypertension may be factors influencing low BMD in male Han Chinese patients with AD. These findings highlight that more attention should be paid to low BMD in the clinical treatment of male Han Chinese patients with AD, particularly in the presence of high-risk factors.
Footnotes
Acknowledgements
The authors of this study would to thank all study participants for their valuable role in this study.
Author contributions
Yu Liu, Qianhui Xia, and Zenghui Ding conceived and designed the experiments, analysed and interpreted the data, and wrote the paper; Lina Gu and Yayun Xu analysed and interpreted the data; and Yongmei Wang and Xulai Zhang conceived and designed the experiments.
Availability of data and materials
The analysed data sets generated during the study are available from the corresponding author on reasonable request.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was supported by the Major projects of Anhui Science and Technology (No. 202103a07020004), the National Natural Science Foundation of China (No. 61701482), and National Key Specialty Construction of Clinical Projects of China.
