Abstract
Osteoarthritis (OA) is the most common form of arthritis resulting in joint discomfort and disability, culminating in decline in life quality. Attention has been drawn in recent years to disease-associated molecular biomarkers found in readily accessible biofluids due to low invasiveness of acquisition and their potential to detect early pathological molecular changes not observed with traditional imaging methodology. These biochemical markers of OA have been found in synovial fluid, blood, and urine. They include emerging molecular classes, such as metabolites and noncoding RNAs, as well as classical biomarkers, like inflammatory mediators and by-products of degradative processes involving articular cartilage. Although blood-based biomarkers tend to be most studied, the use of synovial fluid, a more isolated biofluid in the synovial joint, and urine as an excreted fluid containing OA biomarkers can offer valuable information on local and overall disease activity, respectively. Furthermore, larger clinical studies are required to determine relationships between biomarkers in different biofluids, and their impacts on patient measures of OA. This narrative review provides a concise overview of recent studies of OA using these four classes of biomarkers as potential biomarker for measuring disease incidence, staging, prognosis, and therapeutic intervention efficacy.
Keywords
Introduction
Osteoarthritis (OA), the most prevalent form of arthritis, results in joint pain and disability. OA can affect any articulating joint in the body, often multiple joints at a time. The most common clinical symptoms of OA present as joint pain and stiffness, restricting mobility and function. 1 While the underlying causes of OA remain unknown, it is thought to begin with molecular, anatomical, and/or physiological anomalies in joint tissues. These perturbances initiate, usually over many years, a cascade of progressive, dynamic, whole-joint changes involving local inflammation, articular cartilage degradation, and bone remodeling.
Pathological tissue changes can be detected by different imaging modalities. 2 Primary assessment methods to diagnose OA are based on clinical evaluations and patient-reported outcome measures3,4 combined with radiographic analysis of osteophytes, bone sclerosis, and joint space narrowing, using the Kellgren and Lawrence (KL) grading scale. 5 Even though X-ray and magnetic resonance imaging (MRI) are employed to diagnose joint disorders, these have a limited ability to monitor very early molecular changes occurring in joint tissues that precede the morphological changes they detect.2,6,7 Moreover, findings from imaging modalities do not always correlate with patient symptoms. 8 Joint alterations detected by radiographs generally identify structural modifications detected relatively late in disease pathogenesis. However, these late-stage alterations are the result of ongoing molecular changes that are only detectable using assays capable of detecting these specific molecular entities. 9 With populations rapidly aging 10 and the close association of OA prevalence with increasing age,1,11 there is an urgent need for cost-efficient and sensitive methods to identify molecular changes occurring in joints that precede imaging-detected pathologies. Use of these molecules as biomarkers would enable earlier detection and more effective treatment of OA. As OA appears to diagnose and prognose joint disease progression over weeks or months, as opposed to years. In this regard, recent studies suggest the association of biomarkers, such as noncoding RNAs, 12 metabolites, 13 inflammatory markers,14–16 and cartilage degradation products17,18 with OA pathology (Figure 1), as well as other biomarker entities (e.g. epigenetic and microbial) reviewed in detail elsewhere.19–22 This narrative review includes discussion of some key emerging and classical molecular indicators found in human fluids that associate with OA diagnosis, progression, and prognosis, published in the past 5 years.
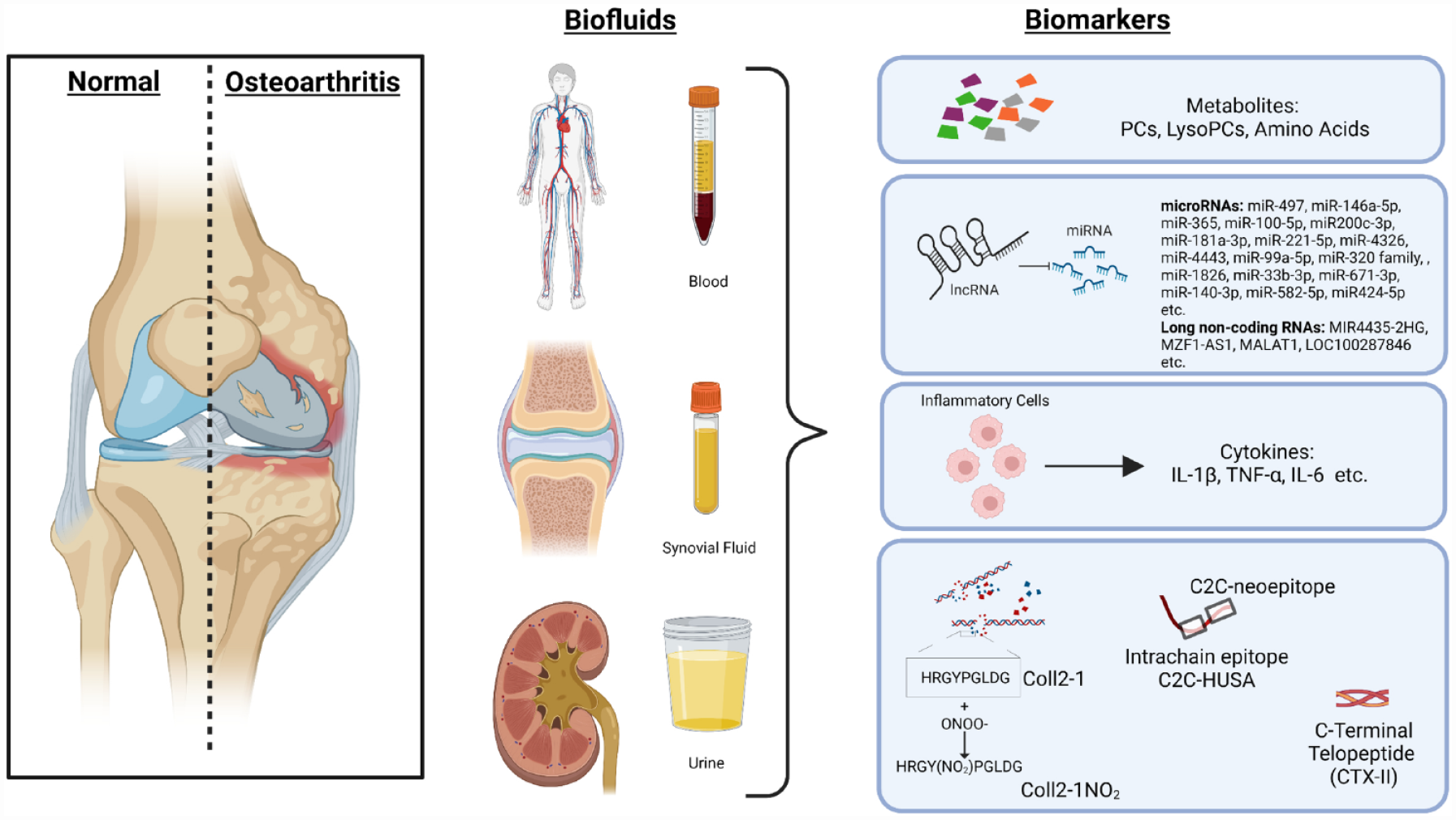
Diagrammatic representation of some key biomarkers in biofluids from patients with osteoarthritis. Metabolites, noncoding RNAs, inflammatory markers and type II collagen cleavage products have been investigated as potential biomarkers of OA diagnosis, progression and prognosis in multiple biofluids including blood isolates (plasma and serum), synovial fluid and urine.
Emerging molecular biomarkers
Metabolites
Metabolites are small molecules produced as by-products of metabolic processes and include lipids, amino acids, sugars, and their derivatives. A metabolome includes all metabolites found in a biological unit (i.e. any given cell, tissue, or biological fluid of interest). Methods to detect metabolite levels can be targeted or untargeted. Targeted approaches have the advantage of measuring concentrations of specific metabolites. Conversely, untargeted approaches more comprehensively measure metabolites within a metabolome and have the potential to uncover novel entities, but with limitations, as metabolite identification is generally based on comparison of measurement spectra to database records. In the past 5 years, multiple studies have investigated metabolomes of urine, synovial fluid, and blood (serum and plasma), as potential sources of biomarkers for OA.
Blood is a whole-body metabolome to which several tissues, including those in the joints, contribute. Blood-derived metabolites have been investigated as a source for diagnostic and prognostic biomarkers of OA. Untargeted metabolomics using gas chromatography, coupled with time-of-flight mass spectrometry (GC/TOF-MS), found 58 significantly changed metabolites in the plasma of OA patients compared to healthy volunteers, of which succinic acid, xanthurenic acid, and
Up to 34% of patients with end-stage KOA do not show reduced pain or improved function after total knee arthroplasty (TKA). 27 Circulating biomarkers could contribute to the decision-making process involved in determining the likelihood of beneficial surgical outcomes. For instance, differential correlation network analysis of plasma metabolites from TKA patients (n = 461) at baseline identified 12 and 23 metabolites that were positively associated with pain and function nonresponders, respectively. 28 In the same cohort, pain nonresponders were positively associated with ratios of acetyl-carnitine (C2) to PC acyl-alkyl (ae) C40:1, and negatively associated with PC diacyl (aa) C36:4 to isoleucine, while function nonresponders were negatively associated with the ratio of glutamine to isoleucine. 29 Thus, in addition to individual metabolites, their ratios might also be good candidates as biomarkers of OA prognosis.
Specific patient characteristics may also contribute to differences in the circulating metabolome. For instance, network analysis of plasma and synovial fluid metabolites from advanced KOA patients (males and females, both n = 50) found sex-specific differences in synovial fluid versus plasma, with individual metabolites serving as key ‘nodes’ for the differential networks. 30 Furthermore, plasma cystine, uric acid, and tyrosine were associated with OA severity and effusion synovitis in female volunteers (n = 596). 31 However, a comparison to males is needed to see if these metabolite-OA disease associations are female-selective. Weight is another potential contributing factor to the circulating metabolome. LC/Q-TOF/MS/MS analyses of metabolites in the serum of control, normal weight KOA, and obese KOA subjects found differences in levels of 21 metabolites in KOA patients versus controls and 15 differences in metabolite levels between healthy weight and obese KOA patients. 32 However, targeted nuclear magnetic resonance (NMR)-spectroscopy of serum from 31 KOA patients showed no detectable differences in metabolites between underweight and obese individuals. 33 This suggests that the contribution of weight to OA is complex. Age is another likely contributor to the metabolome of OA patients, as analysis of plasma from a cohort of 346 subjects (152 KOA, 194 healthy volunteers) by targeted metabolomics found that individual levels of select lysoPC and PC analogs were associated with individuals with KOA in older populations, particularly males. 34 Thus, multiple patient characteristics should be considered when investigating the role of metabolites in OA prognosis. The challenge lies in their identification, which is essential as their inclusion in computational modeling would further help improve metabolite discovery as well as biomarker accuracy for diagnosis and prognosis, optimizing precision medicine approaches.
While research has mainly focused on metabolite levels in the blood, examining more local fluids, like synovial fluid, may be a more direct, and potentially earlier, indicator of OA and its progression. OA-affected tissues (cartilage, synovium, osteophytes, fat pad, subchondral bone, etc.) secrete their metabolic by-products, directly contributing to the fluid’s composition. Untargeted metabolite profiling identified 10 metabolites that were found at different levels in synovial fluid from primary KOA patients (n = 10) versus control individuals (n = 5). 35 Untargeted metabolomics of synovial fluid from OA and healthy individuals (n = 5 each) identified 35 metabolites as potential biomarkers, including phosphatidylcholines, lysophosphatidylcholines, ceramides, myristate derivatives, and carnitine derivatives. 36 Synovial fluid metabolite levels of glutamine, 1,5-anhydroglucitol, gluconic lactone, tyramine, threonine, and 8-aminocaprylic acid, detected by untargeted GC/TOF-MS, were significantly different in a sample of KOA patients (n = 25) compared to controls (n = 10). A validation set of 11 controls and 24 OA subjects with targeted metabolomics of the synovial fluid found glutamine, 1,5-anhydroglucitol, gluconic lactone and 8-aminocaprylic acid, or 1,5-anhydroglucitol and gluconic lactone in combination, could differentiate control and OA patients. 37 Metabolites might also be helpful in identifying the stage of OA, as levels of 28 metabolites were differentially detected by GC/TOF-MS in synovial fluid from KOA patients divided into KL1/2 (n = 8) and KL3/4 (n = 7), which related to fatty acid metabolism, glycerophospholipid metabolism, and glycerolipid metabolism. 38 Metabolites in synovial fluid offer the potential to expand our understanding of the local changes in joint metabolic processes during OA. However, this approach is limited by the increased chance of joint insult during acquisition, which could increase the risk of OA or exacerbate its progression. Thus, the need remains to identify OA-specific metabolites in more readily accessible biofluids, like blood.
Urine is an even easier and less invasive biofluid to acquire than blood, but metabolites in urine have been less frequently studied over the past 5 years as compared to plasma and synovial fluid. To our knowledge, only a single study was identified that used untargeted liquid chromatography-high resolution mass spectrometry of urine from KOA patients classified into inflammatory OA (n = 22) and noninflammatory (n = 52), compared to non-OA controls (n = 68) to identify 26 metabolites that could distinguish inflammatory OA versus non-OA controls, but no metabolites that could distinguish noninflammatory OA versus non-OA controls. 39
Multiple studies indicate that metabolite levels in biofluids of OA patients may be helpful in providing diagnostic and prognostic information. However, future studies should focus on establishing congruency between joint metabolite changes associated with disease progression and their detection in systemic fluids while controlling for changes resulting from additional clinical, anthropometric, and demographic factors. This would help to better define the links between local pathogenesis and systemic biomarkers. Additional investigations probing putative urinary metabolite biomarkers and studying their possible interrelationships with other body fluids may be helpful in understanding the relationships of metabolite biomarkers to OA.
Noncoding RNAs
In the past 5 years, several studies have provided intriguing data to support noncoding RNAs as biomarkers for OA, particularly microRNAs (miRNAs). These noncoding RNAs are encoded in DNA, transcribed into RNA but not translated into protein. MicroRNAs bind mRNA and either promote mRNA degradation or suppress protein translation. 40 Some studies have identified miRNAs as diagnostic of OA. For instance, higher levels of miR-497 were found in serum of women with hip OA (KL IV; n = 23) compared to controls (n = 52) and its levels were also an effective discriminator of individuals with OA (AUC = 0.89). 41 Plasma miR-146a and miR-365 were also increased in KOA patients (KL I-III; n = 42) compared to healthy controls (n = 28) and could distinguish individuals with OA (AUC=0.8843 and 0.8182, respectively). 42 However, not all miRNAs are increased with OA. Compared to trauma patients without KOA (n = 10), serum levels of let-7e were lower in KOA patient patients (n = 10). 43 Similarly, serum samples from primary KOA patients (KL III/IV) and healthy individuals (both groups, n = 12) were screened by microarray and three miRNAs (miR-33b-3p, miR-671-3p, miR-140-3p) were found to be downregulated in KOA patients, with subsequent validation by qRT-PCR. 44 In a study using whole blood, miR-582-5p, and miR-424-5p were reduced in KOA patients compared to control individuals and were found to be reduced in expression in KOA patients with sclerotic (n = 6) compared nonsclerotic (n = 4) subchondral bone. 45 In a larger cohort study using synovial fluid, levels of miR-100-5p, miR-200c-3p, and miR-1826 were lower in KOA patients (KL II-IV, n = 150) compared to controls (n = 150), with each found to be fair to good at distinguishing individuals with KOA based on AUC analysis. 46 Overall, these studies suggest that miRNAs in different biofluids from OA patients may help in diagnosing OA, potentially even individual pathologies associated with the disease. However, a better understanding of the differences between healthy control subjects and OA patients at different stages of disease progression is needed.
Identification of biomarkers capable of distinguishing OA at early stages would help provide interventions to slow disease progression in a timelier manner. Some studies have investigated miRNA levels in biofluids from early compared to late-stage OA patients. For instance, miR-210 in synovial fluid was lower in healthy volunteers (n = 10) compared to both KL I/II and KL III/IV KOA patients (both KOA groups, n = 20), suggesting that miR-210 may be a putative early diagnostic marker of OA. 47 In a separate study of early (KL 0/I; n = 41) and late (KL III/IV; n = 50) KOA subjects, a distinct panel of 11 plasma miRNAs was able to distinguish the 2 groups. 48 Finally, in women with KOA, serum miR-186-5p was found to be positively associated with incident KOA, while miR-146a-5p was also found to be positively associated with prevalent KOA in the same sample. 49 Further studies involving longitudinal and early OA patients would help identify novel miRNA biomarkers or validate already identified miRNA biomarkers of early disease, enabling earlier intervention to slow disease progression and prolong the time to potential surgical intervention.
Additional recent studies have focused on biofluid miRNAs as prognostic biomarkers of OA. Using hip and KOA subjects from Research Arthritis and Articular Cartilage (RAAK) (n = 22) and Genetics osteoARthritis and Progression (GARP) (n = 71) study cohorts, a panel of 7 miRNAs in plasma (miR140-3p, miR-1307-5p, miR-181a-3p, miR-221-5p, miR-4326, miR-443, and miR-99a-5p) was capable of predicting OA progression over 2 and 5 years. 50 Furthermore, in a KOA patient sample from the Osteoarthritis Initiative (OAI) cohort (n = 106), an association was found between plasma levels of the miR-320 family (miR-320b/c/d/e) and fast-progressing radiographic KOA subjects over a 4-year period. 51 Finally, in a sample of KOA patients (KL II-IV, n = 136), baseline levels of 3 miRNAs (miR-146a-5p, miR-145-5p, and miR-130b-3p) were higher in individuals with lower pain relief, with mir-146a-5p trending as an independent predictor of postoperative pain relief. 52 Taken together, these findings suggest that miRNAs may act as important predictors of OA progression.
In addition to predicting OA progression, biomarker tracking of therapeutic intervention efficacy could also be useful, alerting clinicians to when therapy is no longer effective and to try alternative interventions. For instance, in a study of OA patients (KL II/III, n = 28) who underwent high tibial osteotomy for medial compartmental KOA, miR-30c-5p was reduced in 6-month postoperative synovial fluid compared to preoperative, which also associated with postoperative pain relief. 53 Additionally, miR-98 levels were higher in the serum of KOA patients meeting the American College of Rheumatology Classification Criteria for OA compared to controls (both groups, n = 20), which were reduced with treatment with glucosamine sulfate. 54 Future studies are required to determine if the decrease in miR-98 levels with treatment corresponded with improvements in patient outcome measures.
In addition to miRNAs, some of their regulators have also begun to be investigated as potential biomarkers of OA. Long noncoding (lnc) RNAs act as miRNA “sponges,” effectively inhibiting miRNA-RNA interactions, reducing the overall epigenetic contributions of miRNAs. 40 The lncRNA MIR4435-2HG was downregulated in both plasma and synovial fluid of OA patients (stage 3/4, n = 78) compared to healthy volunteers (n = 58), with synovial fluid levels being an excellent OA diagnostic indicator (AUC = 0.96). In this study, OA treatments, including exercise, reduction of joint burden, and nonsteroidal anti-inflammatory drugs, increased MIR4435-2HG plasma levels up to 3 months after treatment initiation. 55 The sustained elevation of this lncRNA suggests it might be a good biomarker of therapeutic efficacy; however, further study is needed to correlate its plasma levels to pain and function outcome measures. Additionally, a study of 20 preoperative KOA patients revealed that serum levels of MZF1-AS1, MALAT1, and LOC100287846 were lower preoperatively in the chronic postoperative pain group (n = 10) compared to the normal postoperative pain recovery group (n = 10). 56 Thus, in addition to miRNAs, biofluid lncRNAs may also be good candidate biomarkers for OA diagnosis, prognosis, and therapeutic efficacy tracking. More extensive studies investigating lncRNAs, their links to individual miRNAs, and changes with OA stage, symptoms and outcomes are necessary.
Overall, additional longitudinal validation studies with larger sample numbers are needed to confirm the applicability of these noncoding RNAs as biomarkers of OA. In addition to blood isolates, studies should also focus on other biofluids, like synovial fluid and urine, which may reveal connections between joint-specific production, presence in circulation, and eventual clearance, helping to understand molecular dynamics of noncoding RNAs for biomarker purposes.
Classical molecular biomarkers: Recent progress
Inflammatory markers
Compared to rheumatoid arthritis (RA), inflammation in OA is considered a low-grade process. Despite this, inflammation is still an important contributor to the development and progression of OA, even in the early stages. 18 Structural alterations in the OA-inflicted joint are hypothesized to be linked, at least in part, to multiple proinflammatory cytokines. 57 The role of inflammatory mediators in human OA has been extensively reviewed.15,58 Many of the identified cytokines are abundantly expressed by various cell types involved in OA, including immune cells, chondrocytes, osteoblasts, and synoviocytes, and have become attractive biomarkers for OA diagnostic and therapeutic intervention efficacy tracking purposes. 59 Of all the inflammatory mediators, interleukin (IL)-1β, tumor necrosis factor (TNF)-α, and IL-6 have received notable attention due to their modulation of a myriad of OA-relevant signaling pathways. 60 Some recent studies provide support for further investigations into the potential of these inflammatory factors as biomarkers of OA.
A study of synovial fluid from 83 patients with primary KOA found negative correlations between IL-1β, TNF-α, or IL-6 with patient-reported visual analog scale (VAS) pain scores or KL grade; however, a positive relationship between KL grade and VAS pain scores was identified. 61 These findings suggest that these inflammatory mediators are larger drivers of pain in KOA patients with less severe radiographic damage compared to those at later stages, despite their chronic presence. In a study of 46 subjects, proinflammatory cytokines (IL-1β, TNF-α, or IL-6) were detected at higher serum levels of both American College of Rheumatology Stage 3 primary and secondary KOA subtypes compared to healthy controls. 62 Furthermore, exploratory analysis of patients with early KOA (n = 139; >80% KL ⩽ II) showed that serum IL-6 was associated with increases in Western Ontario and McMaster Universities Arthritis Index (WOMAC) function and MRI features (osteophytes, synovitis, and meniscus extrusion), synovial fluid IL-6 was solely associated with increases in MRI effusion, and serum TNF-α was associated with increases in osteophytes, cartilage loss, synovitis, and effusion MRI features. 63 In contrast, in later stage KOA subjects (n = 20; >70% KL ⩾ II), only serum IL-6 was associated with increased WOMAC pain and decreased function. 63 Thus, proinflammatory mediators, like IL-1β, TNF-α, and IL-6, have the potential to be used to help subclassify OA patients (early versus late or primary versus secondary), facilitating the administration of precision medicine procedures. Of note, in the later stages of KOA, the grading of the severity of OA has a considerable ceiling effect, reducing the capacity to uncover associations with increasing levels of proinflammatory biomarkers. Therefore, future studies should strive to evaluate KOA at earlier disease stages.
Evidence suggests that IL-6 may have interesting roles in regulating OA symptoms. IL-6 levels were reportedly higher in the serum of KOA patients undergoing TKA (n = 127) compared to healthy participants (n = 39). 64 Linear regression modeling of this patient sample demonstrated that circulating IL-6 levels were negatively correlated with pain intensity. However, in synovial fluid of KOA patients undergoing TKA (n = 104), which is more closely situated to OA-affected tissues, IL-6 levels were positively associated with synovitis of the parapatellar subregion and pain (WOMAC or neuropathic). 65 Similarly, a study of KOA subjects (n = 70; KL⩾II) also found IL-6 synovial fluid levels were positively associated with pain measured on the Likert scale during movement. Synovial TNF-α was also positively associated with WOMAC total pain and WOMAC or Likert scale scores for pain during movement or at rest, while IL-1β was negatively associated with Likert scale scores for pain during movement. 66 Another study (n = 200) reported a signature of 11 serum cytokines, which included IL-1β and IL-6, were associated with increased WOMAC dysfunction scores and MRI-detected bone marrow lesions over 2 years. 67 Undoubtedly, IL-6 is likely a key mediator in OA; however, its detection in specific biofluids, association with distinct stages of OA pathology, and links to pain at different stages of disease require further investigation. Similarly, measures of other fluid-based cytokines, such as TNF-α and IL-1β, and their relationships with one another must be better understood to uncover mechanisms and relationships with OA pain and pathology.
Another inflammatory mediator that has gained considerable attention is C-reactive protein (CRP), an acute phase protein primarily expressed downstream in response to injury and the release of inflammatory cytokines. TNF-α and IL1-β trigger the synthesis of IL-6, which subsequently induces the production of CRP. 68 A comprehensive systematic review of 32 studies recently concluded that, compared to healthy controls, OA patient levels of serum high-sensitivity (hs-)CRP corresponded with increased pain and decreased physical function. 69 Similarly, CRP metabolite (CRPM), but not CRP itself, was predictive of KOA radiographic progression as KOA patients (n = 152) with ⩾9 ng/mL CRPM serum levels were more likely to develop contralateral KOA after 2 years (KL change ⩾2). 70 A positive association between the levels of hs-CRP and KOA patients with MRI-defined synovitis (n = 58), compared to healthy controls (n = 33), has been reported, with no association to pain intensity as measured by VAS. 71 In a separate study of KOA subjects with varying degrees of pain intensity measured by VAS (n = 281), no relationship between either hs-CRP or CRPM, and clinical pain intensity was found, although hs-CRP did associate with KL grade. 72 Furthermore, CRP was positively associated with VAS-measured pain severity in an independent cross-sectional study of a larger cohort of advanced OA subjects (knee or hip; n = 770), but not with any items of the WOMAC questionnaire. 73 Finally, CRPM serum levels correlated with knee joint inflammation, particularly synovitis, as measured by etarfolatide radionucleotide imaging in KOA patients (n = 25). 68 CRP and its metabolites appear to be involved in OA-related pain, but because measuring pain is typically a subjective measure, its analysis and outcome interpretations are complicated. 61 Additional investigations are needed to clarify the associations between hs-CRP, CRPM, pain severity, and OA pathologic severity, to further support its use as a diagnostic or prognostic biomarker for OA.
Cartilage degradation markers
Collagen type II, the principle component of the extracellular matrix (ECM) of healthy hyaline cartilage, forms an extensive collagen fibrillar network. 74 Over time, it is susceptible to degradation, notably by collagenases, resulting in peptide fragments that can be readily assayed in biofluids. If the balance is shifted such that degradation occurs faster than anabolic processes, loss of cartilage ensues, exacerbating OA pathology. Various degradation products of type II collagen have been identified, including the C-telopeptide of cross-linked type II collagen (CTX-II), 45-mer peptide (C2C), a specific 45-mer peptide containing the downstream C2C neoepitope and intrachain epitope (IB-C2C-HUSA), and a 9-mer peptide (108HRGYPGLDG116) released during degradation from the triple helical region of type II collagen (Coll2-1). Immunoassays detecting these degradative type II collagen fragments have emerged as promising tools in the search for molecular biomarkers of OA.17,75,76
In 2016, a longitudinal nested case–control study (194 cases and 406 OA comparators) of the OAI cohort was published that evaluated 18 biochemical molecules for their ability to predict OA progression between 24 and 48 months, defined as both progression of radiographic joint space loss and persistent progressive pain worsening. 75 This study identified that, baseline, 12-, and 24-month time-integrated levels of urinary CTX-II, and 24-month time-integrated level of urinary IB-C2C-HUSA™, were promising predictors of OA progression status. Furthermore, both time-integrated urinary CTX-II and urinary IB-C2C-HUSA™ levels at baseline, 12 months, or 24 months were able to predict OA progression, as defined by radiographic joint space loss, persistent progressive pain worsening, or both.
CTX-II levels in urine, serum, or synovial fluid are increased in KOA patients with higher radiographic KL grades.66,76-82 Of note, no significant differences in urine CTX-II levels were detected between patients with early radiographic stages of OA (KL1) and healthy individuals, 80 suggesting that the rise of CTX-II levels might result from processes occurring at later stages of OA. Other patient characteristics, like age, sex, and body mass index, can also influence urine CTX-II levels. For instance, premenopausal women with early KOA (n = 146; KL ⩽ 1) had significantly higher urine CTX-II levels compared to controls (n = 108), an effect not observed in men. 83 Urine CTX-II has also been found to be associated with pain duration and body weight of Mexican females with KOA (n = 155). 82 Moreover, a longitudinal study showed baseline urine CTX-II levels as a risk factor for the incidence of total joint replacement in postmenopausal women (n = 478). 84 Thus, patient characteristics likely influence urine CTX-II levels, which may need to be considered when examining the role of this cartilage degradation product in OA studies.
Elevated IB-C2C-HUSA™ in urine was first reported in detail to discriminate subjects with early from advanced OA 85 and has been shown to predict the risk of emerging KOA over a 12-year span. 86 Higher baseline IB-C2C-HUSA™ levels were also shown to associate with, and increase the risk of, progressive radiographic cartilage loss over 3 years in the human population-based cohort study originally using IB-C2C-HUSA™. 87 Moreover, it was noted that IB-C2C-HUSA™ may be an effective preoperative marker of symptom outcome of total knee replacement in females. 88 Similarly, an analysis of 600 OA patient samples from the Foundation for the National Institutes of Health study suggested a positive association with MRI-measured infrapatellar fat pad signal intensities with IB-C2C-HUSA™ and uCTX-II. 89
Serum levels of another collagen degradation product, Coll2-1, and its nitrated form, Coll2-1NO2, were reported to be associated with KOA features identified by whole-organ MRI scoring (n = 121) 90 or radiographic analysis (n = 598). 76 Overall, these recent findings further indicate that CTX-II, IB-C2C-HUSA™, and Coll2-1 (and its nitrated form) may be promising cartilage degradation biomarkers found in patient biofluids, which will further enhance diagnostic and prognostic accuracy. To support their usefulness in precision medicine, it will be imperative to consider how patient demographic, anthropometric, or clinical characteristics influence levels of these putative biomarkers in large-scale future studies.
Concluding remarks
It is evident that newly emerging and classical OA-related molecules, including metabolites, miRNAs, inflammatory cytokines, and type II collagen degradation fragments, could be helpful biomarkers for assessing disease incidence, staging, prognosis, and therapeutic intervention efficacy. However, to increase the chances of their success at adding value to patient care and advance prognostic and diagnostic precision medicine approaches, additional large, longitudinal, multicenter studies are required to uncover likely interrelationships between the various identified biomarkers and their levels in different biofluids. For example, although blood-based biomarkers tend to be frequently evaluated, the use of synovial fluid, a more isolated biofluid in the synovial joint, can provide insightful information on local disease activities. Similarly, urine is another worthwhile biofluid for the exploration of biomarkers. As a result, studies investigating the convergence of biomarkers from all three fluids might also be of interest to intersect local, systemic, and excreted OA biomarkers. Finally, investigating OA populations to generate models using multiple molecular entities that link to OA metrics, while considering the influence of various patient characteristics, would be helpful to further define and refine OA biomarkers signatures best associated with specific OA pathologies.
