Abstract
Despite decades of research efforts and multiple clinical trials aimed at discovering efficacious disease-modifying osteoarthritis (OA) drugs (DMOAD), we still do not have a drug that shows convincing scientific evidence to be approved as an effective DMOAD. It has been suggested these DMOAD clinical trials were in part unsuccessful since eligibility criteria and imaging-based outcome evaluation were solely based on conventional radiography. The OA research community has been aware of the limitations of conventional radiography being used as a primary imaging modality for eligibility and efficacy assessment in DMOAD trials. An imaging modality for DMOAD trials should be able to depict soft tissue and osseous pathologies that are relevant to OA disease progression and clinical manifestations of OA. Magnetic resonance imaging (MRI) fulfills these criteria and advances in technology and increasing knowledge regarding imaging outcomes likely should play a more prominent role in DMOAD clinical trials. In this perspective article, we will describe MRI-based tools and analytic methods that can be applied to DMOAD clinical trials with a particular emphasis on knee OA. MRI should be the modality of choice for eligibility screening and outcome assessment. Optimal MRI pulse sequences must be chosen to visualize specific features of OA.
Introduction
Imaging plays an important role not only in osteoarthritis (OA) research in general 1 but specifically also in disease-modifying OA drugs (DMOAD) clinical trials. 2 Although the OA research community has been aware of the limitations of conventional radiography as an imaging tool, 3 magnetic resonance imaging (MRI) and other more advanced modalities have not resulted in regulatory approval of a DMOAD to date. MRI enables detailed structural assessment of OA-affected joints that is not possible using radiography.4,5 Despite decades of research efforts and multiple clinical trials to try to develop efficacious DMOADs, we still do not have a drug that has been approved by regulatory agencies.
It has been discussed that the radiography-based definition of structural eligibility is one of the reasons for failure of DMOAD trials.6–10 Several plausible explanations exist to elaborate on this statement. First, the definition of OA disease severity based on radiography is limited due to lack of reproducibility of radiographic joint space measurements. 3 Second, there are only weak associations between radiography-depicted structural changes and pain. 11 Third, radiography cannot depict potentially detrimental findings, which indicate an increased risk of articular collapse or rapid disease progression. 2 Last, radiography cannot depict most of the articular and periarticular tissues [such as menisci, cartilage, bone marrow lesions (BMLs), ligaments, and synovitis]. 3
In this perspective article, we will describe how we can wisely utilize available imaging modalities and techniques for DMOAD trials, with an emphasis on MRI and knee OA. We will explain available MRI-based semiquantitative (SQ) scoring systems that can be applied to DMOAD trials, how to select appropriate MRI pulse sequences depending on the specific target tissue of the trial, how radiography can still be utilized in DMOAD trials in combination with MRI, and the need to consider different phenotypes of OA when designing DMOAD trials.
X-ray-based patient selection/screening for DMOAD trials
Radiographic SQ assessment of knee radiographs is typically performed to select/screen participants for DMOAD trials. Structural disease severity of OA is defined by the Kellgren and Lawrence (KL) 12 grading system, which assigns a score based on the presence or absence of osteophytes and joint space narrowing. Investigators can stratify patients into those who are eligible (the presence of definite OA but not end-stage OA) and those who are ineligible (the absence of definite OA or the presence of end-stage OA). Subjects who have mild OA (KL grade 2) and moderate OA (KL grade 3) are usually enrolled in DMOAD trials. Because the radiographic appearance of joint space width (JSW) can vary significantly depending on the knee positioning or angulation of the X-ray beam, it is important to acquire standardized weight-bearing anteroposterior bilateral knee X-rays at the time of eligibility screening. 13 For this purpose, positioning devices such as Synaflexer™ should be used, or fluoroscopy-guided X-ray acquisition should be performed.3,14 Despite using positioning devices, false-positive or false-negative longitudinal change in JSW may be observed as shown in Figure 1 in an exemplary fashion.

Reproducibility limitations of radiography and superiority of magnetic resonance imaging (MRI) in depicting osteoarthritis as a whole-joint disease. (a) Baseline anterior–posterior (a.p.) radiograph shows a normal medial tibiofemoral joint space width (arrows). (b) At 2 years follow-up, there is apparent definitive joint space narrowing (arrowheads). Soft tissues are not assessable on the radiograph. (c) Baseline MRI of the same knee shows discrete superficial cartilage thinning of the medial tibia (arrowhead) while the cartilage of the medial femur is apparently normal. There is minimal medial meniscal extrusion of 2 mm still considered physiologic. (d) Two years later, no definite cartilage loss is observed (arrowheads) and meniscal extrusion has not progressed (arrow). Apparent progression on the a.p. radiograph is due to positioning errors with minimal change in beam angulation leading to false-positive joint space narrowing.
At baseline, JSW should be measured as a surrogate for the integrity of cartilage and menisci. There are pros and cons for selecting low or high JSW thresholds for enrolling patients, and there is mixed literature evidence to support either choice. On one hand, investigators may wish to include knees with sufficiently preserved cartilage, especially for studying potential anti-catabolic drug effects on articular cartilage. For example, prior DMOAD trials have used a threshold value of the remaining medial JSW to be ⩾2 mm or 2.5 mm.15–17 Using ⩾2 mm minimal JSW (mJSW) will lead to inclusion of a higher proportion of knees because a larger number of KL grade 2/3 patients (including those with diffuse full-thickness chondral loss) would fulfill that criterion.16,17 On the other hand, a recent clinical trial of Sprifermin showed that selection of patients with low minimum JSW and moderate to high knee pain at baseline resulted in more rapid progression of OA and knees with advanced OA showed symptom modification by the drug. 18 Furthermore, KL grade 3 knee OA was shown to progress more rapidly than KL grade 2 knee OA. 19
Trained and experienced musculoskeletal radiologists play a key role in X-ray-based screening for DMOAD trials. First, they should perform the X-ray-based eligibility reading in a centralized fashion 14 based on KL grading to exclude subjects without radiographic OA (KL grade 0 or 1) or end-stage OA (KL grade 4). Second, they should perform the assessment of the minimum JSW, although the same task may also be performed in a semi-automated fashion using validated tools being quality checked by expert readers after the initial assessment. Third, they should exclude additional subjects at eligibility who meet predefined radiographic exclusionary findings described below. These exclusionary findings include advanced osteonecrosis, subchondral insufficiency fractures, severe varus or valgus malalignment, large subchondral cysts which may have a high risk of collapse during a trial, femoral or tibial fracture, and radiographically appreciable rheumatic/neoplastic/metabolic disease. 2
MRI-based eligibility screening and phenotypic stratification of subjects
Following successful radiography-based screening and consideration of relevant exclusionary/inclusionary criteria, MRI should be utilized as an additional eligibility screening tool. ROAMES 20 is a relatively new scoring system (published in January 2020) and data from clinical trials using ROAMES are yet to be published. In ROAMES, SQ assessment of cartilage, menisci, BMLs, osteophytes, synovitis (‘Hoffa-synovitis’), and joint effusion (‘effusion-synovitis’) is performed. Moreover, diagnoses of exclusion including subchondral insufficiency fractures and meniscal root tears are recorded as ‘present’ or ‘absent’ (Figure 2). An important aim of ROAMES is to perform phenotypic stratification (Table 1) of potentially eligible participants and to detect exclusionary findings which cannot be depicted by radiography.20,21 A recent study showed that it is uncommon to find high-risk exclusionary MRI findings that potentially precludes safe participation in a DMOAD trial. 22 However, such exclusionary findings are found in about 3% of KL grade 2 knees and about 12% of KL grade 3 knees. This study highlights the value of using MRI screening. 22

Diagnoses of exclusion using MRI as an instrument to define patient eligibility. (a) Axial T2-weighted MRI shows a complete posterior root tear of the medial meniscus (arrows). (b) Corresponding coronal intermediate-weighted fat-suppressed image shows corresponding medial meniscal extrusion due to mechanical instability of the medial meniscus (arrow). Root tears are considered high-risk findings for rapid progression of cartilage loss and subsequent articular collapse. For this reason, patients exhibiting root tears should not be included in clinical DMOAD trials as joints exhibiting root tears are likely not amenable to any pharmacologic DMOAD effects. (c) Coronal intermediate-weighted fat-suppressed image shows articular collapse due to subchondral insufficiency fracture of the medial femoral condyle. There is an osteochondral depression at the fracture site (arrow) and corresponding large bone marrow edema (asterisk). In addition, there is a large nonspecific subchondral cyst (arrowhead). Bone cysts that potentially increase the risk for fracture are considered exclusionary at screening.
Phenotypes of knee OA based on ROAMES.

MOAKS, Magnetic resonance Osteoarthritis Knee Score; OA, osteoarthritis; PFJ, patellofemoral joint; ROAMES, Rapid OsteoArthritis MRI Eligibility Score; TFJ, tibiofemoral joint.
Based on the aim of DMOAD trials and types of agents being tested, several factors should be considered when deciding patients belonging to which phenotype will be most suitable for inclusion in a trial. Of note, in this article, we focus our discussions to structural phenotypes that are relevant to imaging-based outcome criteria. However, there are clinical phenotypes (intra-articular/extra-articular/secondary/age-related and systemic) and molecular endotypes (bone and cartilage/inflammatory/low repair/metabolic) of OA that are of interest to the broader OA research community, and those are described in other dedicated publications.23,24 If one is testing a compound that is aimed to regenerate cartilage, there should be enough cartilage remaining in the knee joint so the drug’s efficacy in cartilage regeneration at the site of cartilage damage can be demonstrated. Inclusion of knees with the ‘cartilage-meniscus’ phenotype is likely to be most relevant for such a study. However, in a recent FNIH study, only 5% of 485 subjects (after excluding KL grade 1 knees and those having meniscal root tears) fulfilled the cartilage/meniscus phenotype based on the original ROAMES definition. 21 These knees had diffuse full-thickness chondral damage and meniscal tears/maceration in medial and lateral tibiofemoral compartments. Application of a less stringent definition with a single compartment needing to demonstrate meniscal tear, the number of participants classified as cartilage-meniscus phenotype rose to 21%. A recent analysis based on the FNIH cohort showed phenotypic stratification of the cartilage-meniscus phenotype in various subtypes can be done, and may help to define trial cohorts at the time of screening. 25 In that analysis, KL grade 2 knees and all definitions demonstrated raised odds of progression, while KL grade 3 knees demonstrated an apparent protective effect. This latter finding was likely because KL grade 3 knees stratified by the suggested definitions had relatively mild chondral defects at the time of screening. 25
ROAMES is a tool that can be used to perform for phenotyping of knees that exhibit severe BMLs (=predominant subchondral bone changes), (severe BMLs), severe effusion/synovitis (=predominant inflammatory changes), or mixture of structural phenotypes in different combinations (e.g. mixed cartilage-meniscus/subchondral bone and mixed subchondral bone/inflammatory). Depending on the exact type of DMOAD under investigation, one must determine whether severe synovitis or BMLs are contraindicated for therapy, or they may interfere with the desired effects of the compound. Figure 3 shows examples of subchondral BMLs in the context of phenotypic stratification. Using ROAMES one can stratify phenotypes to determine whether the efficacy of the drug differs in various phenotypes.

Subchondral bone phenotype of knee osteoarthritis. Phenotypic stratification may help in selecting patients most likely to benefit from a specific candidate DMOAD molecule. Compounds targeting the subchondral bone may have an impact on bone marrow lesions. For this reason, knees with large bone marrow lesions or those with multiple lesions are included in such trials. (a) Sagittal intermediate-weighted fat-suppressed image shows a large bone marrow lesion in the medial femoral condyle fulfilling the definition of the subchondral bone phenotype (arrows). In addition, there is a minor subchondral cyst and widespread full-thickness cartilage damage. Note that knees with extensive widespread full-thickness cartilage loss are likely not responsive to any anti-catabolic mode of action as there is not sufficient cartilage to preserve and measure structural DMOAD effects. Phenotypes may overlap and one knee may exhibit more than one specific phenotype. This knee also exhibits large effusion-synovitis and thus fulfills the inflammatory phenotype, in addition. (b) Sagittal intermediate-weighted fat-suppressed MRI of another patient shows several tibial and femoral bone marrow lesions (arrows). In comparison with the bone marrow lesion in (a), these are smaller in size or volume but numerous and thus defining this knee as exhibiting the subchondral bone phenotype. Note that bone marrow lesions are nonspecific findings and multiple differential diagnoses apply. In this case, there is an identical-appearing signal change at the femoral metaphysis consistent with red marrow conversion in the typical location. In contrast, subchondral OA-related bone marrow lesions are localized directly adjacent to the subchondral plate.
Choice of MRI sequences suitable for screening and evaluation of different outcome measures
There are two ways of performing MRI screening at the time of eligibility assessment of trial participants. One may choose to obtain a complete series of MRI sequences for a comprehensive whole joint assessment at the time of eligibility screening. In this case, the investigators must accept the risk of incurring extra cost of imaging for those who are excluded after screening process. A benefit of this approach is simplified logistics and patient convenience. Alternatively, an abbreviated protocol with two quick sequences [i.e. a sagittal and coronal PDW FS or IW FS, 22 or a 3 min three-dimensional (3D) FSE sequence, e.g. SPACE and VISTA] can be used at the time of eligibility screening, and if a patient is indeed eligible for inclusion, the patient will then return to complete a full set of sequences of comprehensive whole joint MRI assessment. This option is logistically more challenging because there is a need for two visits for included subjects in a relatively short period. Some patients might decline to return for the second full exam. So long as there is sufficient budget, the first option would be a preferable option for both the participants and the researchers.
For the full MRI protocol, appropriate technical considerations should be given. A dedicated knee coil should be used to ascertain the best image quality. Optimization of all MRI acquisition parameters should be performed, including, but not limited to, patient positioning, signal homogeneity, image orientation, and spatial resolution and signal-to-noise ratio. This is an important step to optimize quality of imaging and minimize image degradation secondary to artifacts. 26 Additional factors to consider are minimization of patient discomfort during the MRI scan without sacrificing image quality, and imaging cost within the budgetary constraint. From the radiological point of view, the most important issue is the choice of most appropriate pulse sequences for each specific pathological feature to be evaluated. The use of an incorrect pulse sequence will preclude the meaningful interpretation of acquired images. Table 2 presents the summary of suggested MR pulse sequences for optimum SQ analysis of each knee OA feature, based on the available literature evidence and authors’ own expertise.26–28 Suggested protocols are optimally performed on a 3T scanner, using multichannel phased-array extremity coils for an optimal signal-to-noise ratio. 29 However, 1.5T scanners will also provide images with sufficient quality to perform reproducible SQ analyses. 26
List of MRI pulse sequences that are suitable for evaluating various OA features using semiquantitative scoring.
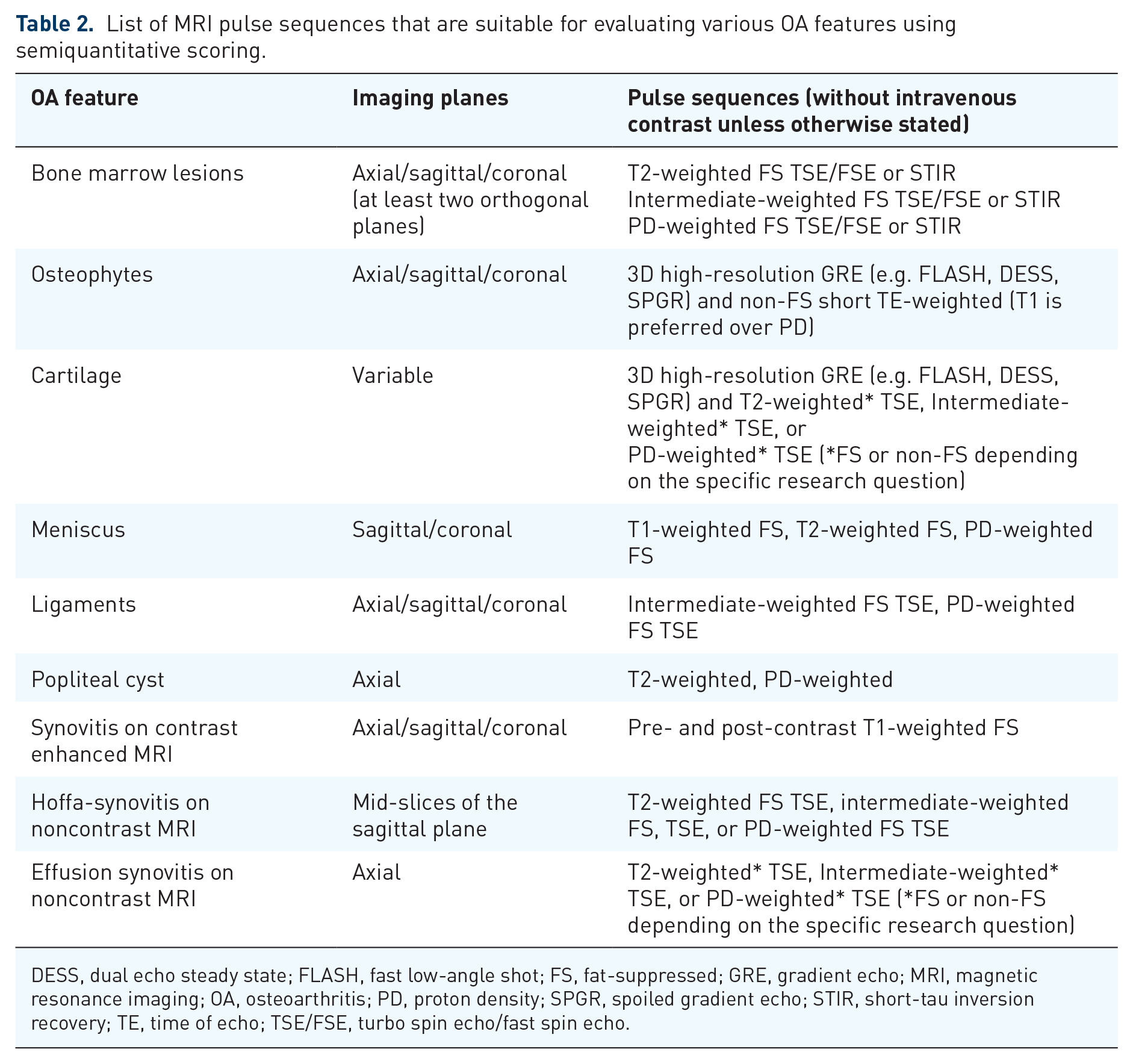
DESS, dual echo steady state; FLASH, fast low-angle shot; FS, fat-suppressed; GRE, gradient echo; MRI, magnetic resonance imaging; OA, osteoarthritis; PD, proton density; SPGR, spoiled gradient echo; STIR, short-tau inversion recovery; TE, time of echo; TSE/FSE, turbo spin echo/fast spin echo.
Fluid-sensitive fast spin echo (FSE) or turbo spin echo (TSE) sequences [which include, for example, T2-weighted, intermediate-weighted, or proton density (PD)-weighted fat-suppressed sequences] obtained in three orthogonal planes are time-efficient to deploy and particularly important for evaluation of BMLs. 30 Use of GRE sequences for BML assessment 31 is less suitable for SQ assessment than fluid-sensitive fat-suppressed FSE/TSE sequences because GRE sequences are relatively insensitive to BMLs and can lead to underestimation of the lesion size.32,33 A direct comparison of BML visualization using GRE and FSE/TSE sequences is shown in Figure 4.
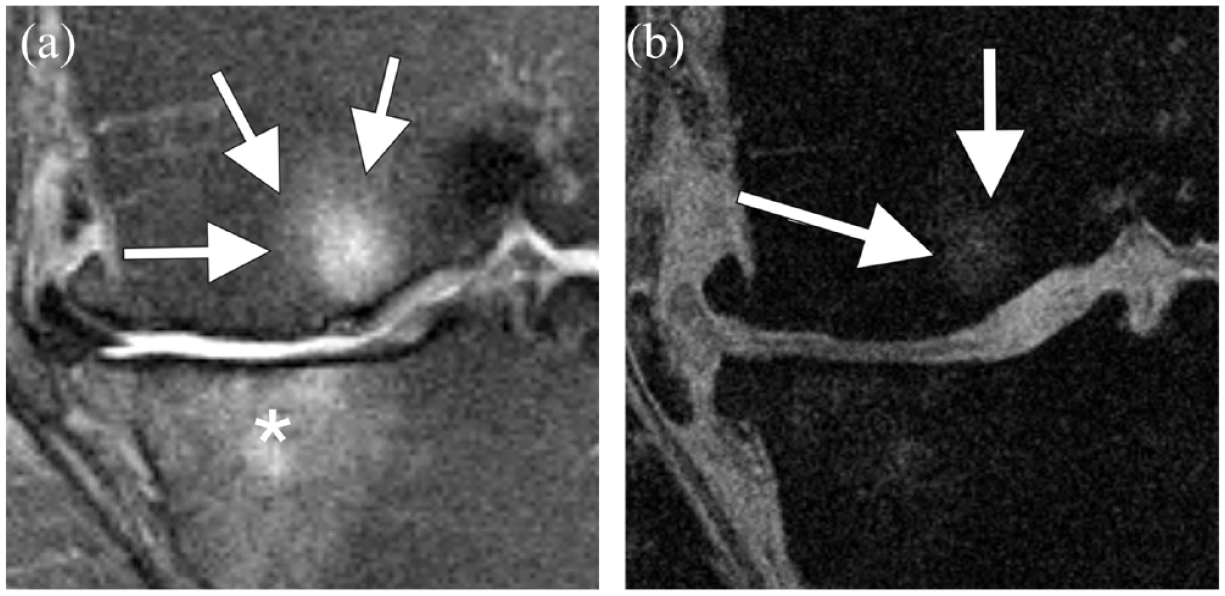
Relevance of sequence selection of feature-specific assessment. (a) Coronal intermediate-weighted fat-suppressed sequence shows the medial tibiofemoral compartment. There are large bone marrow lesions at the medial femur (arrows) and tibia (asterisk) reflected as areas of high signal intensity contrasting the normal fatty marrow that is depicted with low signal. In addition, there are other signs of advanced structural knee OA including widespread cartilage damage, marginal osteophytes, and meniscal extrusion. (b) Coronal fast low-angle shot (FLASH) with water excitation (WE) MRI, a 3D high-resolution sequence, is commonly used for cartilage quantification. This type of sequence, a gradient echo sequence, is prone to magnetic susceptibility and thus relatively insensitive to BMLs and will lead to underestimation of the lesion size as shown by the arrows. The tibial lesion is hardly depicted at all.
GRE sequences are ideally deployed for 3D quantitative cartilage analysis (e.g. thickness and volumetric measurements), but not for focal cartilage defects which should be evaluated using fluid-sensitive FSE/TSE or short-tau inversion recovery sequences.34,35 Another thing to consider is that GRE sequences are prone to magnetic susceptibility artifacts. One should be aware that intra-articular vacuum phenomenon is depicted as linear or punctate hypointensity within the joint space, and misinterpreting such artifact as meniscal damage or a chondral defect must be avoided. 36
Addition of a T1-weighted FSE/TSE or Dixon sequence allows evaluation of subchondral sclerosis or intra-articular loose bodies with high sensitivity. Angulating the imaging plane specifically for certain structures to be assessed may be helpful, for example, paracoronal T2-weighted sequence is helpful for differentiating anterior cruciate ligament partial tears versus complete tears. Fat-suppressed 3D FSE/TSE sequences can be used as an alternative triplanar two-dimensional (2D) TSE sequences. Both these techniques can provide comparable results for SQ assessment of knee OA, although 3D TSE sequences exhibit different image characteristics (e.g. increased blurriness). 37
Thanks to modern advanced MRI techniques, highly accelerated acquisition of imaging became possible and scan time can be decreased to a fraction of conventional scanning method. Examples of such techniques include parallel imaging and improvements in 3D FSE imaging, which enables the acquisition of triplanar MRI of the knee in less than 5 min.38–40 Artificial intelligence shows additional promise regarding image acceleration41,42 (Figure 5).

Artificial intelligence applied to accelerate image acquisition. Trained convolutional neural networks (CNNs) are used for post hoc image reconstruction. The original MRI data set is undersampled and the missing structural information is re-created by the CNN resulting in almost equivalent image quality. (a) Example shows coronal intermediate weighed fat-suppressed images acquired with a 7T ultrahigh-field system. A super high-resolution matrix of 720 × 720 pixels is used with an in-plane resolution of 0.15 mm × 0.15 mm, 3 mm thickness, acquired in 9 min 30 s. (b) Fourfold undersampling with post-acquisition AI reconstruction results in a decrease in imaging acquisition time down to 2 min 22 s. The image overall exhibits a smoother image impression but the overall quality seems comparable. As CNNs always need extensive training data, the future will need to show if rare findings are depicted with confidence and determination of the ideal acceleration factor without losing relevant structural information needs to be shown in the future.
Imaging-guided intra-articular injection of investigational drugs
Some investigational drugs and emerging biologic treatment need to be administered through an intra-articular injection.43–46 Such drugs include, but not limited to, nerve growth factor agents, fibroblast growth factors, platelet-rich plasma, mesenchymal stem cells, etc. 47 Intra-articular injections of the knee should ideally be performed under imaging guidance.48,49 A systematic review revealed the superolateral approach was investigated most and had the highest pooled accuracy rate of correct injection of 91% [95% confidence interval (CI) of 84–99%]. 50 An investigational compound is unlikely to work if the injection is extra-articular. Therefore, if an extra-articular injection is documented, affected subjects should be excluded from any outcome analysis to prevent artificial reduction of demonstrated clinical efficacy of the DMOAD being evaluated. Also, the drug may cause an adverse event if it is extra-articularly injected (e.g. development of heterotopic ossification or other structural side effects) at follow-up. 51 Examples of extra-articular administration with X-ray documentation are shown in Figure 6.

Example of documentation of extra-articular injection. The documentation of an intra-articular route of administration is paramount and most easily achieved using air administered at the time of injection. (a) Lateral radiograph shows air within Hoffa’s fat pad but not intra-articularly (arrows). (b) Another lateral X-ray shows air in the prefemoral fat pad (arrow) but not within the joint cavity. (c) Another example shows an air collection in the subcutaneous tissue but not in the joint (arrows).
To confirm correct intra-articular needle placement, a lateral projection X-ray should be obtained after injection of a small amount of intra-articular air prior to the injection of the investigational compound or placebo itself. 52 Audible squishing sounds after intra-articular injection of air can be used as an additional proof of successful injection. 53 Alternatively, ultrasound-guided intra-articular drug delivery can be performed. 54 This technique is advantageous over X-ray guidance in that real-time visualization of the needle tip position is possible during the procedure. The presence of joint fluid in the medial or lateral gutters can be helpful, providing an additional target for needle placement. However, one may note that a recent study showed that neither ultrasound-guided nor palpation-guided intra-articular knee injections provide a 100% success rate, using intra-articular air visualization on lateral projection X-ray as a reference. 55 Although ultrasound-guided injection demonstrated somewhat higher success rates than palpation-guided injection, documenting the presence of intra-articular air seems important to avoid subsequent extra-articular injection of DMOAD and to help minimize artificial reduction of efficacy of such drugs.
SQ MRI scoring systems applicable to DMOAD trials
There are several published SQ scoring systems for the assessment of the articular and periarticular tissues in knee OA. To overcome the limitation of radiography-based evaluation, MRI enables scoring of the whole joint including cartilage, menisci, BMLs, osteophytes, joint effusion, synovitis, subchondral cysts, ligaments, and intra-articular bodies. Some scoring systems such as MRI Osteoarthritis Knee Score (MOAKS) 56 can score the OA features of the whole joint, whereas others may target only select features, e.g. BMLs, synovitis, menisci, and osteophytes.20,27,57 Of these, Rapid OsteoArthritis MRI Eligibility Score (ROAMES) 20 was created so that it can be used to evaluate the eligibility of subjects in DMOAD trials. Investigators can use MRI SQ scoring tools to assess multi-tissue changes between the baseline and follow-up time points for the determination of DMOAD efficacy and safety.2,26,58 For instance, for the evaluation of anabolic compounds, SQ assessment enables investigators to capture safety concerns such as increased ossification and osteophyte growth. Other concerning imaging findings that can be detected include the occurrence of subchondral insufficiency fracture, osteonecrosis, and others. 20 When compared with quantitative volumetric cartilage assessment (which evaluates quantitative changes invisible to the human eye over a period by addressing an entire knee joint compartment or plate), 59 SQ assessment is suited to evaluate superficial, partial-thickness, and full-thickness focal chondral defects. For a more global assessment of articular cartilage across the entire compartment or a plate, SQ evaluation may also depict chondral loss over time (in periods as short as 6 months) 60 but has limited ability to capture anabolic effects like cartilage growth.
Importance of within-grade scoring
To increase the sensitivity to detect small changes between time points, ‘within-grade’ SQ MRI scoring is typically performed. 61 Using this methodology, even a small morphologic change that does not fulfill the criteria for a full-grade change (i.e. score change of 1) is still recorded as a longitudinal change. 62 Recently, it could also be shown that within-grade assessment is associated with longitudinal quantitative cartilage thickness loss supporting the assumption that within-grade change reflects real cartilage damage progression. 63 Within-grade changes are also applied for BML assessment and have been shown to be clinically valid, which is illustrated in Figure 7. 61

Within-grade assessment. Semiquantitative MRI assessment is based on expert evaluation of MRIs applying validated scoring systems. While definitive visual change may be apparent, often lesions (particularly bone marrow lesions and cartilage alterations) do not fulfill the definition of a so-called full-grade change. For this reason, and particularly to increase sensitivity to change, so-called within-grade changes have been introduced that are able to document definite change despite not fulfilling a full-grade change. Within-grade changes have been shown to be clinically valid and to correspond to quantitative cartilage loss. (a) Coronal short tau inversion recovery (STIR) image shows a small bone marrow lesion at the central medial femur (arrow). (b) Follow-up MRI 1 year later shows a definite increase in size that does not fulfill the criteria for a full-grade change (arrowhead). This is a typical example of a within-grade increase of a subchondral bone marrow lesion.
MRI interpretations without blinding to time points
MRI evaluation is performed at multiple time points in a DMOAD trial to observe the structural change between the baseline and the follow-up time points. It is a routine practice within the OA research community to perform MRI SQ scoring at follow-up time points without readers being blinded to the time points. 64 Scoring of MRIs in chronological order is known to increase sensitivity in the detection of clinically relevant longitudinal changes. If SQ scoring is done in a random order and a blinded manner, readers may not be able to capture a meaningful longitudinal score changes for each imaging feature. 64
Utility of the delta-sum and delta-subregion approaches
SQ scoring of MRI data requires careful consideration so that investigators can best deploy it to record score changes at different timepoints in DMOAD clinical trials. To begin with, adding together all subregional scores from the knee joint is not an ideal way of assessing longitudinal score changes for the joint. As an example, BMLs can show changes in size in a short time period, and worsening and improvement of BMLs in different subregions can occur at the same time in the whole joint. In this case, there may be no apparent change in an overall score calculated by addition of all subregional scores. Thus, the use of a ‘summation score’ can mask what is truly happening in the joint.65,66 Another important consideration is the use of ‘delta-sum and delta-subregion’ method. 67 In this method, all subregional scores are summed up to calculate the overall deterioration (>0), unchanged (0), or improvement (<0). For example, the knee joint is divided into 14 subregions in MOAKS. For cartilage evaluation, no change in two subregions, worsening in seven subregions, and improvement in four subregions yield a delta-subregion change of +3 for the whole knee. One can use alternative methodology for assessment of longitudinal changes, such as the use of maximum grades and a latent class analysis. 68
Conclusions and future prospects
The use of MRI from screening to outcome assessment, likely in combination with X-ray-based KL grading, particularly to define eligibility, is encouraged in DMOAD trials, rather than relying solely on radiography-based imaging criteria. It is important to recognize various structural phenotypes of OA to perform more targeted clinical trials. SQ analysis tools such as ROAMES are available to facilitate longitudinal evaluation of OA features and to assess the efficacy of DMOADs. The choice of appropriate MR pulse sequences and protocols are the key to a meaningful evaluation of imaging features of OA. A tailored abbreviated protocol of two sequences usually takes less than 7 min depending on the MRI parameters, which is notably shorter than a standard MRI protocol that takes around 15 min. Artificial intelligence approaches will help speed up image acquisition by a factor of around 2 in the near future. 69 Technically successful imaging-guided intra-articular injection of investigational drugs is important to prevent unwanted reduction in efficacy of such drugs. We anticipate that DMOAD research and development will focus more on early knee OA (i.e. knees with KL grade 0 and 1), painful and symptomatic knee OA, and knees with imaging features fulfilling the MRI diagnosis of OA. At present, however, an MRI definition of early OA is yet to be fully determined, and regulatory agencies are unlikely to approve a drug that is targeting a disease status without a validated definition (i.e. ‘early’ MRI-defined OA). Therefore, it is likely that candidate DMOADs will have to show efficacy in symptomatic patients with established structural OA first. If we can identify patients in the earliest possible stage of OA and treat them with available efficacious DMOADs, we may be able to prevent patients with early knee OA from progressing into more advanced OA with irreversible damage for which total knee arthroplasty will be the only current treatment option. Of note, the US Food and Drug Administration recently proposed that DMOADs should help patients feel and function better. 70 Our research efforts and journey toward the discovery and clinical development of an efficacious DMOAD must continue to accomplish these aims.
