Abstract
While criteria for early-stage knee osteoarthritis (OA) in a primary care setting have been proposed, the role of imaging has been limited to radiography using the standard Kellgren–Lawrence classification. Standardized imaging and interpretation are critical with radiographs, yet studies have also shown that even early stages of radiographic OA already demonstrate advanced damage to knee joint tissues such as cartilage, menisci, and bone marrow. Morphological magnetic resonance imaging (MRI) shows degenerative damage earlier than radiographs and definitions for OA using MRI have been published though no accepted definition of early OA based on MRI is currently available. The clinical significance of structural abnormalities has also not been well defined, and the differentiation between normal aging and structural OA development remains a challenge. Compositional MRI of cartilage provides information on biochemical, degenerative changes within the cartilage matrix before cartilage defects occur and when cartilage damage is potentially reversible. Studies have shown that cartilage composition can predict cartilage loss and radiographic OA. However, while this technology is most promising for characterizing early OA it has currently limited clinical application. Better standardization of compositional MRI is required, which is currently work in progress. Finally, there has been renewed interest in computed tomography (CT) for assessing early knee OA as new techniques such as weight bearing and spectral CT are available, which may provide information on joint loading, cartilage, and bone and potentially have a role in better characterizing early OA. In conclusion, while imaging may have a limited role in diagnosing early OA in a primary care setting, there are advanced imaging technologies available, which detect early degeneration and may thus significantly alter management as new therapeutic modalities evolve.
Introduction
Osteoarthritis (OA) is a highly prevalent joint disease affecting 22.9% of individuals over the age of 40 knee 1 and has demonstrated a significant increase in prevalence of 9.3% from 1990 to 2017. 2 Given our aging population and the increasing obesity epidemic, we are expecting these numbers to grow gradually and lead to even higher rates of disability and health care costs. 3 Therapy is limited and mostly consists of lifestyle modification at earlier disease stages and joint replacement at more advanced disease stages. Though there is the perception that there are no efficient treatments available, 4 it is generally accepted that if detected at early stages, management strategies are available that help effectively to reduce disease burden. 5 This creates a window of opportunity that should engage physicians to diagnose OA at the earliest stages possible. However, there is significant uncertainty and a lack of consensus of what represents early OA, which also further challenges the execution of rigorous clinical trials to treat OA. 6
In 2018, classification criteria were proposed for early-stage OA intended for a primary care setting. 6 This was based on expert opinions and resulted in the recommendation of the following diagnostic criteria: (i) pain, function, and quality of life (using questionnaires such as the Knee injury and Osteoarthritis Outcome Score (KOOS)); (ii) clinical examination including symptoms such as joint line tenderness; and (iii) knee radiographs demonstrating a Kellgren & Lawrence (KL) grade 0 or 1. At this time, no other imaging criteria were recommended. MRI, while considered as highly effective in diagnosing early structural abnormalities, was considered as not sufficiently standardized, and it was felt that there was a too high population prevalence in asymptomatic individuals to use MRI to diagnose early knee OA. In 2021, these draft criteria were tested using longitudinal outcome data from the Osteoarthritis Initiative (OAI) cohort with encouraging performance in terms of enrichment for structural and clinical progression. 7
Given the ongoing controversy of the role of imaging, we are revisiting the early definition of OA in this review article from an imaging perspective with a focus on the knee as the most clinically significant manifestation. We will evaluate the role of standard radiographs, MRI morphological and compositional techniques, and computed tomography (CT)-based methods based on published data and ongoing initiatives to achieve better standardization of imaging methods.
Standard radiographic techniques
The KL classification to define knee OA was first published in 1957 8 and yet is still considered as a standard in diagnosing knee OA. According to the previously published classification by Luyten et al., 6 criteria for early OA include KL grades 0 and 1, which is inherently problematic as it includes not only early stages of OA but also normal joints without any manifestations of OA. Thus, these criteria should be best used if patients also meet clinical criteria. Earlier definitions included also KL2 (osteophytes only) in addition to KL0 and 1 along with pain.9,10 Figure 1 demonstrates representative knee radiographs with KL grades 0, 1, and 2. While considered a standard, KL scores have limited reproducibility and include terminology ‘doubtfully’ and possibly as pertains to joint space narrowing, which is inherently imprecise. Also, joint space narrowing is dependent on the imaging technique (weight-bearing extension anteroposterior or flexion posteroanterior (bend) radiographs), which further complicates a reproducible and standardized diagnosis. 11 Moreover, it should be noted that even in individuals with KL grade 1 and 0, cartilage damage is not rarely found. Taguchi et al. 12 found cartilage defects, mostly at the medial femoral condyle, in 26% of women aged 50 years and older in their study investigating MRI studies in patients with KL1 and 2, normal leg alignment and no obesity. This study clearly documents advanced and irreversible cartilage damage already in patients with nearly normal radiographs based on KL scores.

Radiographs of the knee (posterior–anterior, bent projection) demonstrating a normal knee radiograph without osteophytes (KL0) in 35-year-old man (a), a knee radiograph with tiny osteophytes (arrows) of doubtful significance (KL1) in 43-year-old woman (b), and a knee radiograph with definite osteophytes (arrows) in the medial joint compartment (KL2) in a 42-year-old woman (c).
Given the limitations of KL scoring other systems have been suggested such as the atlas-based Osteoarthritis Research Society International (OARSI) classification. Culvenor et al. 13 compared KL and OARSI atlas criteria in their definition of the presence of knee OA. The investigators used a standard KL grading of 2 and higher to define tibiofemoral OA, while for the OARSI atlas-based grading, OA was diagnosed if there was grade 2 or higher joint space narrowing and/or osteophytes sum score grade 2 and higher and/or grade 1 joint space narrowing with grade 1 osteophyte. Based on the gradings of 1178 knee radiographs, a diagnosis of knee OA was made in 17.3% (n = 203) of knees using the KL score and in 26.2% (n = 309) of knees using the OARSI atlas-based score. Intra- and inter-rater reliability were in a similar range for both scoring systems with kappa values between 0.86 and 0.97 for intra-rater reliability and between 0.63 and 0.73 for inter-rater reliability. These results show that the OARSI atlas criteria identify a larger number of individuals with knee OA, but to date, there is limited information how valid these data are in identifying early stages of disease. Clearly a better standardization is required to define (early) radiographic OA.
Given the inherent limitations of semiquantitative scores with respect to reproducibility, truly quantitative measurements may be more attractive to better define disease burden in knee OA. Researchers have therefore worked on developing methods that would allow an accurate and reproducible assessment of joint space width.14–16 The additional advantage of measurements of joint space narrowing is their sensitivity to change, which improves assessment of disease progression. However, to date, there are no established cut-off values that would allow the definition of abnormal joint space narrowing. Establishing a threshold would require a reproducible radiographic technique, an adjustment for joint size, and a measurement technique that is accurate and likewise reproducible. But most of all, this cut-off value would need to be validated through longitudinal studies demonstrating that it can in fact predict development of more advanced stages of knee osteoarthritis.
Given the limitations of standard semiquantitative and quantitative assessment of knee radiographs in diagnosing knee OA, researchers have developed computer-aided/machine learning tools to predict knee OA based on radiographs and found encouraging results.17,18 One of these studies analyzed trabecular bone texture in knee radiographs using convolutional neural networks in knee radiographs from the OAI and Multicenter Osteoarthritis Study (MOST) cohorts. 17 Using progression of joint space narrowing as an outcome, they found better prediction with trabecular bone texture than with KL-based models in the MOST cohort and similar prediction in the OAI cohort.
In summary, while radiographs are an inexpensive imaging technique, a rigorous diagnosis of early OA requires standardized imaging, image readings, and measurement techniques. Still, even with optimized image acquisition and analysis, radiographs are neither sensitive nor specific for early OA disease stages. Using a combination of radiographic (KL0 and 1) and clinical findings as suggested by Luyten et al. 6 may be a reasonable compromise and was recently longitudinally validated. 7
Magnetic resonance imaging: morphological imaging
Prevalence of structural pathology in knees without radiographic OA
As already outlined above, it has been acknowledged that a radiographic status of KL grade 0 or 1 may not be equivalent to absence of disease.19,20 A large population-based observational study reported presence of any MRI-detected OA-related abnormalities in 89% of knees with apparently completely normal radiographs (i.e. KL grade 0). 19 Particularly, MRI-detected cartilage damage and osteophytes seem to be highly prevalent in the medial patellofemoral and medial posterior tibiofemoral joints. 21 An increasing prevalence of features, including cartilage damage, bone marrow lesions, and meniscal damage, is found in older patients. 19 While the prevalence of MRI pathology did not show significant differences between the patients with and without knee pain for most definitions of pain, the prevalence of ‘any MRI abnormality’ was markedly increased in those with Western Ontario and McMaster Universities OA index (WOMAC) pain compared with those without. However, it needs to be noted that MRI abnormalities were very frequent in participants without pain (up to 86%). 19 Another study based on the Osteoarthritis Initiative (OAI) focused on KL0 knees with and without risk factors (i.e. obesity, family history of total knee joint replacement, repetitive knee bending, previous knee trauma and/or surgery, and presence of Heberden’s nodes) observed that 96% of persons with knee OA risk factors but without radiographic knee OA had at least one MRI finding present and 87% of persons without such risk factors. 22 The prevalence of any MRI pathology was higher (prevalence ratio adjusted for age, sex, and BMI 1.3 [CI 1.1–1.6]) in individuals with risk factors for knee OA than in those without risk factors. The prevalence of MRI-defined OA in the tibiofemoral joint using the definition by Hunter et al. 23 was 23% (CI 19–28%) in subjects with OA risk factors and 9% (CI 3–21%) in those without. In addition, MRI-defined structural tissue pathology was far more prevalent in an early OA model of KL0 knees with established contralateral radiographic OA, compared with a healthy reference sample with KL0 in both knees and no risk factors. 24 This has been shown for all MRI features, apart from effusion-synovitis. Regarding longitudinal change, incident cartilage damage (defined semiquantitatively) and quantitative cartilage loss (based on 3D segmentation) was more commonly seen in the contralateral OA model group,24,25 supporting the assumption that patients with a radiographically normal knee but contralateral radiographic OA represent a suitable model for testing disease-modifying osteoarthritis drugs (DMOADs) in clinical trials of early, nonradiographic OA.
Risk of structural OA development
As multiple MRI features of OA are frequently found in a knee with structural disease and increase the risk of progression, 26 it would be relevant to understand the time course of events to be able to address specific pathology early in order to avoid or delay progression to more advanced disease. Although cartilage loss is one of the hallmark features of structural disease, the evidence that OA commences with cartilage lesions is sparse. 27 Several authors have suggested that incidental meniscal pathology might be one of the main triggers of disease incidence. 28 However, as meniscal damage is frequently found in individuals without symptoms, the role of the meniscus in disease onset is still under debate.29,30 Synovitis, which is reflected on MRI as joint effusion and synovial thickening, increases the risk of cartilage damage and might play an important yet not fully understood role in early disease.31,32 Furthermore, subchondral bone marrow abnormalities appear to have an impact on disease progression, and animal models have suggested that bone marrow alterations may be the earliest structural reflections of disease onset. 33 Finally, cartilage abnormalities markedly impact risk for progressive disease and may be important triggers for progression on a subregional level and subsequently of the whole joint leading to radiographic OA.34,35 Eventually, a more or less concomitant presence of these lesions may be possible. 36 It has been reported before that knees exhibiting specific structural joint damage on MRI, especially within the 2 years prior to disease incidence, concomitant presence of several features, and occurrence of such features at one or more time points expose the joint at increased risk of developing incident radiographic OA compared with matched knees not showing these features of structural damage. 37 Figure 2 gives an example of isolated and concomitant structural MRI features in knees without radiographic OA (KL0).
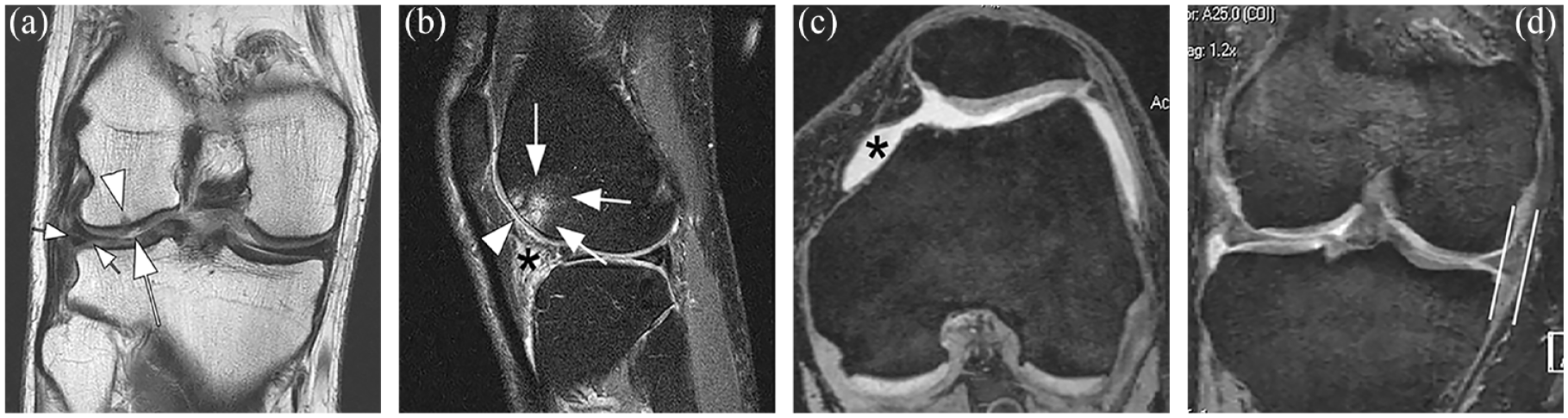
Structural tissue damage is highly prevalent and often concomitantly present in knees without radiographic osteoarthritis. (a)–(d) Structural abnormalities in MRI studies that are not visualized in radiographs but represent disease features that have been shown to lead to progressive OA. (a) Coronal intermediate-weighted (IW) MRI shows a superficial focal cartilage lesion at the lateral femur (large arrow) and a minor adjacent subchondral bone marrow lesion (arrowhead) in this KL0 knee. In addition, there is a discrete but definitive horizontal-oblique tear of the body of the medial meniscus reaching the meniscal base and the undersurface (small arrows). (b) Sagittal IW fat-suppressed MRI of another KL0 knee shows a large subchondral bone marrow lesion (arrows) at the lateral femoral trochlea with adjacent superficial cartilage damage (arrowhead). In addition, there is marked Hoffa-synovitis (asterisk). (c) Other findings commonly observed in KL0 knees include effusion-synovitis (asterisk) as shown on this axial reformation of a dual echo at steady state (DESS) MRI, which has been shown to predict longitudinal cartilage loss in knees without radiographic OA. (d) Meniscal extrusion is a risk factor for cartilage damage and is also commonly observed in knees without radiographic OA. Coronal DESS image shows marked extrusion of the medial meniscal body (parallel lines). Note absence of osteophytes at the joint margins in this KL0 knee.
Clinical relevance of pre-radiographic findings
Another study focused on a subset of the OAI and evaluated subjects without radiographic OA at two defined time points, that is, at 12 and 48 months, using cartilage loss and incident knee symptoms as the outcome parameters. This analysis found that structural tissue damage was associated with incident persistent knee symptoms and that more concomitant features were associated with an increased risk of symptom outcomes and incident tibiofemoral cartilage damage. 36 From this, the authors concluded that the observed findings should not be considered incidental in individuals with higher risk for OA and may rather reflect early degenerative disease. The relevance of knee pain in early structural knee OA has been emphasized in other studies. Peat et al. 38 found that incident radiographic OA is preceded by prodromal symptoms lasting at least 2–3 years. It should also be noted that this early phase of disease constitutes a window of opportunity and management strategies are available that help effectively to reduce disease burden. 5 These include lifestyle changes including addressing risk factors for disease such as obesity and lack of physical activity.
MRI definitions of OA and early OA
Compared to radiography, MRI is able to visualize the different knee tissues much more comprehensively. 37 Figure 2 highlights structural abnormalities in MRI studies that are not visualized in radiographs but represent disease features that have been shown to lead to progressive OA and may in part be considered as signs of early OA. In 2011, an MRI definition of structural knee OA was introduced by an expert group using Delphi methodology. 23 Consequently, this MRI definition of OA includes more joint tissues and pathologies than the radiographic KL definition, suggesting that it may be more sensitive to detect knee OA at an earlier stage, or that it may result in a more valid definition compared with radiography. In this exercise, tibiofemoral OA was defined on MRI as the presence of a definite osteophyte and full-thickness cartilage loss, or a definite osteophyte or full-thickness cartilage loss plus two of the additional features: (1) subchondral bone marrow lesion or cyst not associated with meniscal or ligamentous attachments, (2) meniscal subluxation, maceration or degenerative tearing (such as a horizontal tear), (3) partial-thickness cartilage loss, or (4) bone attrition. In a subsequent validation exercise, Schiphof and colleagues compared KL grading with the MRI definition in the population-based Rotterdam study and showed that the MRI definition detected more cases of structural OA (9%) compared with radiography (4%). 20 In addition, the association between the definitions and knee pain at baseline was higher when the MRI definition was included. This was similar for the association between the definitions and persistent knee pain, and between the definitions and BMI. 20 This study, however, did not focus particularly on early OA or an early OA definition by MRI.
Magnusson and colleagues investigated participants from the OAI study to study the differences in prevalence of MRI abnormalities over time in those with and without risk factors for knee OA. 39 An operational MRI definition of OA was included but not specified further, a radiographic definition was defined as KL ⩾ 1 and a knee pain definition defined as pain on most days of a month in past 12 months. The study reported that the development of structural OA was comparable irrespective of the presence of risk factors for OA. In addition, the overlap with symptoms was quite limited, in particular in the subgroup without risk factors for OA. These findings suggest that structural tissue damage without symptoms should probably not be defined as OA, but as risk factors for development of clinically symptomatic disease and that structural changes may be part of aging, but not necessarily OA.
Previous studies have suggested classification criteria for early knee OA.9,10 In two of these, early knee OA was defined as a combination of (1) knee pain, (2) KL grade 0 or 1, and (3) arthroscopic findings of cartilage lesions and/or MRI findings demonstrating articular cartilage degeneration and/or meniscal degeneration and/or subchondral BMLs. These criteria were updated after additional consensus meetings, suggesting that MRI should not be part of the criteria for early OA in routine clinical practice or primary care, in light of lack of validated consensus criteria and the high prevalence of structural joint changes detected by this method in the general population. 6 Others have emphasized the role of MRI in the research setting due to its superior sensitivity to change and validity in the context of early OA but currently not thought appropriate for the routine clinical care setting because of its high cost and potential risk of over-diagnosis. 40
In summary, currently no accepted definition of early OA based on morphologic MRI is available. Structural features of OA are highly prevalent in knees without radiographic OA, but their clinical importance is still unclear and differentiation between normal aging and structural OA development remains a challenge. As OA is most likely best treated in early and potentially still reversible stages, development of an MRI definition of early OA seems warranted particularly in the context of clinical DMOAD development.
Magnetic resonance imaging: compositional imaging
Cartilage compositional MRI techniques are sensitive to changes in the extracellular matrix of articular cartilage. 41 Compositional MRI in early OA has the potential to detect the initial stages of cartilage degeneration, at a stage where these changes may still be more amenable to lifestyle or pharmaceutical intervention. 42 This would provide a considerable advance on conventional (morphological/structural) MRI, even with ongoing improvements in MRI acquisition and analysis. Structural cartilage damage implies damage to the collagen matrix; such changes are likely to be irreversible given the limited innate regenerative potential of collagen in adults. 43
A number of different compositional MRI techniques have been described over the past two decades (Table 1). The most widely studied of these is T2 (transversal relaxation time) mapping, followed by T1ρ (longitudinal relaxation time in the transverse plane in the presence of a radiofrequency [spin-lock] field) mapping. While other techniques offer improved specificity for a particular cartilage component (e.g. delayed gadolinium-enhanced MRI of cartilage [dGEMRIC] is more specific for proteoglycan/glycosaminoglycan [GAG] content than T1ρ, 44 although both techniques are correlated to proteoglycan/GAG based on a recent systematic review) 41 , the greater accessibility, lack of requirement for gadolinium-based contrast agent, and feasibility at more commonly available field strengths are all factors which have driven the wider uptake of T1ρ and, in particular, T2 mapping. Meta-analyses have shown that T2 and T1ρ mapping are reliable and have discriminative validity both in the setting of established OA and, importantly, in individuals at risk of OA.45,46
Compositional MRI techniques.

CEST, chemical exchange saturation transfer; dGEMRIC, delayed gadolinium-enhanced MRI of cartilage; DTI, diffusion tensor imaging; DWI, diffusion-weighted imaging; GAG, glycosaminoglycan; gagCEST, GAG chemical exchange saturation transfer; MRI, magnetic resonance imaging; MSK, musculoskeletal; SAR, specific absorption rate; SNR, signal-to-noise ratio; T, Tesla; UTE, ultrashort echo time.
Ability of compositional MRI to predict OA incidence & progression
The OAI MRI protocol included T2 mapping. Given the size, extensive follow-up, and well-characterized nature of this dataset, it is unsurprising that the majority of studies evaluating the ability of compositional MRI to predict OA incidence and worsening have focused on T2 mapping. A number of approaches to assessing ability of compositional MRI to predict outcome are available; analysis can be done at the level of an individual focal lesion/abnormality, at the level of a cartilage plate (e.g. medial femur) or a defined subregion thereof (e.g. central medial femur), or at the level of a global value for the entire knee. It is encouraging that T2 mapping has demonstrated predictive validity across these different scales. For example, Kretzschmar et al. 47 examined cartilage plates from knees, which developed new-onset cartilage lesions over a 4-year period, comparing against cartilage plates from control knees at both the focal lesion and cartilage plate level. The authors showed that, at the local level, cartilage T2 values were significantly higher in case knees at 1 year prior to lesion onset, and at 2 years prior to onset at plate level (Figure 3). Razmjoo et al. 48 leveraged advances in automated segmentation and registration techniques to analyze the T2 values of the entire OAI at a cartilage plate level (4796 knees). The authors demonstrated strong associations between compartmental T2 values and incident radiographic OA within 2 years for all cartilage plates except the patella.

Magnified views of the lateral femoral condyle in an individual with an incident cartilage defect. While the cartilage appears normal on the morphological sequence at 12 months prior to lesion onset (a), local T2 elevation of the cartilage T2 map (b, white arrow) is demonstrated in the lesion equivalent area 12 months prior to lesion onset (c, white arrow). Reproduced from Kretzschmar et al. 47 with permission.
Standardization of compositional MRI
Like any quantitative imaging technique, compositional MRI requires careful attention to detail across acquisition and analysis pipeline to ensure that values obtained are accurate, reproducible, and interpretable. Prior work has demonstrated variability across vendors, RF coils and pulse sequences for cartilage compositional MRI.49–53 Additional sources of variability lie in image analysis approaches, including quantification methods (e.g. exponential decay function fitting for parameter mapping), in particular in situations where the image signal-to-noise ratio (SNR) is low, and manual image processing methods such as cartilage segmentation are used.54,55
To address these issues, a musculoskeletal subcommittee was formed under Radiology Society of North America (RSNA) – Quantitative Imaging Biomarker Alliance (QIBA) taskforce in April 2017. The first profile of the committee has been focused on standardizing data acquisition techniques for cartilage T2 and T1ρ imaging as potential imaging biomarkers for OA. 56 3D T1ρ and T2 imaging sequences with the same structure (termed as MAPSS) have been developed on three major MR platforms. 57 With harmonized protocols and centralized data processing, the inter-vendor inter-site variations have been reduced substantially as compared with earlier studies. 57 These results highlight the importance of standardizing data acquisition methods, including sequence structures and acquisition protocols, and demonstrate the great promise of minimizing inter-site inter-vendor differences by improving sequence performance (e.g. more robust to B0 and B1 inhomogeneity) and by standardizing data acquisition.
In addition to data acquisition, standardization and automation of post-processing pipelines are critical for clinical translation of compositional MRI. Significant advances in the field have been made for automatic cartilage segmentation based on deep-learning methods. Further work to standardize post-processing methods, such as constructing T2/ T1ρ maps, is needed. 58
Clinical utility of compositional MRI
Despite the availability of compositional MRI techniques for more than 20 years, they have yet to make a real impact on clinical care for OA. This may partially relate to some of the technical challenges described above (such as standardization) as well as the long acquisition time required. However, the major reason is the lack of disease modifying drug treatments for OA (DMOAD). This means that the identification of early disease has limited impact on patient management. Even in the absence of DMOAD therapy, improved standardization of compositional MRI could see clinical utility being realized for indications such as monitoring of cartilage repair tissue and assessment of suitability for joint preserving surgery (e.g. high tibial osteotomy and unicompartmental knee arthroplasty). Ongoing RSNA-QIBA efforts will continue to improve standardization in readiness for clinical utility.
Computed tomography
The strength of CT modalities in the assessment of OA lies in their ability to acquire more detailed imaging of mineralized joint tissues compared with radiography and MRI. 59 CT techniques overcome radiographic superposition of joint structures by acquiring a 3D data volume and involve faster and more easily standardizable acquisitions, lower cost, and the ability to acquire reconstructable 3D data sets with greater ease than MRI. Here, we focus on clinical scale techniques in vivo for early diagnosis of degenerative disease, although it should be noted that much relevant research has been undertaken ex vivo with microCT, not just in bone but also the assessment of articular soft tissues such as the meniscus.60,61
With focus on the knee, a good place to start is the role of CT in the assessment of osteophytes as a cardinal feature of early OA. 23 One cohort study of individuals without radiographic OA showed that osteophytes were the most common finding on MRI in 74% (527/710), 19 raising doubts over the value of radiography in their detection. Segal et al. 62 confirmed these by showing that cone beam CT (CBCT) was superior to radiography in knee osteophyte detection with a sensitivity of 93% compared with 60% and an accuracy of 95% compared with 79%. Omoumi et al. 63 have subsequently demonstrated quantification of osteophytes by volume and spatial distribution around the proximal tibia using CT. Although it seems reasonable to consider CT as the new gold standard for osteophyte detection, no studies have yet confirmed this in place of MRI, likely due to the difficulty in setting a true reference standard.
The precise role of subchondral bone in early osteoarthritis remains unclear; however, animal studies have continued to demonstrate the importance of bone remodeling in OA development, including how early inhibition of bone remodeling can slow post-traumatic OA development.64,65 In humans, significant differences in subchondral bone mineral density (BMD) have been demonstrated with multidetector CT between severe OA and non-OA knees with higher density in the OA-affected medial compartment and lower density in the lateral compartment in a pattern supported by a biomechanical hypothesis. 66 In 2020, Shiraishi et al. 67 used high-resolution peripheral quantitative CT (HRpQCT) to show strong relationships between subchondral bone microstructure at the anterior medial tibia and KL grade, suggesting it may be a useful index in monitoring treatment. Optimum quantitative assessment of BMD (and other relevant morphometric parameters) with CT means that further work needs to be done with clinical CT techniques to define bone behavior more clearly in early knee OA.
CBCT has recently found application at the knee in the context of OA due to the ability to perform weight bearing acquisitions, 68 comparisons naturally being made with radiography. In 2016, Segal et al. 69 showed that greater surface area with a low joint space width (JSW) measured on weight bearing CT (WBCT, Figure 4) correlated more strongly with tibial cartilage lesion severity than radiographic minimum JSW. In 2021, Turmezei et al. 70 subsequently demonstrated that 3D joint space mapping using WBCT yielded a smallest detectable difference in tibiofemoral JSW measurement of +/−0.1 mm for KL 0 and 1 knees, a sensitivity twice that of radiography. 71 WBCT therefore looks to be a logical successor to radiography in the measurement of early joint space narrowing in OA if protocol standardization can be achieved. 72

Bilateral knee weight-bearing cone beam CT in coronal (a) and sagittal (b, right knee) planes. The dotted lines demarcate the limits of the cone beam, like a discus in 3D, outside of which the imaging data reconstruction is unreliable. A substantial advantage to weight bearing is the ability to look at the physiologically loaded joint space in 3D, here controlled by a positioning device (P) to image in 20 degrees of knee joint flexion. Joint space narrowing is seen at both medial tibiofemoral compartments, worse on the left as marked between the white arrowheads. CT is also excellent for the visualization of osteophytes (white arrow), subchondral sclerosis (black arrowhead, also between the white arrowheads), and subchondral cysts (black arrow). In addition to 3D quantification of joint space width, it is also possible to detect meniscal extrusion (asterisk), evaluate patellofemoral disease, and study 3D joint morphology from the same acquisition that is only marginally more dose than traditional radiography. Imaging data courtesy of Professor Neil Segal, Kansas University Medical Center, Kansas City, USA.
One of the most important recent developments across all clinical imaging has been spectral CT, which has been achieved due to the realization of photon counting detector technology in clinical scanner systems. 73 In their study on a large animal model of OA with intra-articular contrast, Rajendran et al. 74 demonstrated simultaneous high-resolution and multi-energy imaging that allowed morphological assessment of cartilage loss as a marker of joint disease. Noncontrast spectral imaging has also been performed on ex vivo human knee specimens suggesting optimal virtual mono-energetic imaging of cartilage and bone simultaneously at 60 to 70 keV, 75 but further research is required to see how this may enhance our understanding of early human OA in vivo.
In a similar principle to the diffusion of contrast medium into articular cartilage with dGEMRIC, CT arthrography has shown excellent correlation of attenuation values with sulfated glycosaminoglycan content (R 2 = 0.90, p < 0.0001). 76 CT arthrography has also been able to depict articular cartilage lesions with superior performance and greater reliability compared with MR arthrography at the ankle, 77 but wider clinical application still remains limited by the need for an invasive intra-articular injection of contrast medium.
In keeping with its versatility, there are a number of other interesting CT applications with relevance to early OA such as semiquantitative scoring of intra-articular mineralization as a predisposing factor, 78 whole body scoring of OA load, 79 and the use of CT as the anatomical framework for metabolic imaging. An example of metabolic imaging with SPECT/CT has shown significantly higher bone tracer uptake in the medial tibiofemoral compartment in the presence of degenerate or torn menisci compared with normal menisci, which may be of value in identifying individuals with early OA. 80
The notion of identifying early OA with CT has been in researchers’ minds for decades, 81 but it has been recent developments such as WBCT and spectral CT that have pushed it concertedly to the forefront. This has been allied with the advent of lower scan doses that make CT techniques more palatable for investigation in large numbers, in some cases with the dose equivalence of one day of background radiation per scan, just twice the reported dose of equivalent radiography.82,83
Discussion
Although OA is a clinical diagnosis, symptoms occur relatively late in the overall disease course. Imaging has an important role to play in identifying preclinical pathological processes that have the potential to identify targets for prevention and treatment. In this review, we have evaluated the role of standard radiographs, morphological and compositional MRI techniques, and CT-based methods in the definition of early OA. Figure 5, taken from reference, 42 well illustrates and summarizes the role of the different imaging techniques in diagnosing OA. We found that these methods show great promise and are likely to have complementary roles in unraveling the complexity of OA and thus providing new therapeutic approaches tailored to a personalized approach. Using novel imaging modalities to better understand the pathophysiology at early OA stages will provide targets for interventions, that can be performed before there is irreversible tissue damage. This includes, for example, joint inflammation/synovitis which can be well visualized with MRI and potentially treated when structural damage is not yet present. This concept has been studied in the evolution of post-traumatic OA84,85 and may lead to novel treatment approaches.

Disease stages in OA as defined by imaging.
Radiographs have been in widespread use in OA since the 1950s. 8 They have provided the cornerstone of clinical and research work in OA. However significant, well described limitations exist in the use of radiographs for defining early OA as, by the time the first radiographic changes occur, significant pathological changes are already present. 19 Furthermore, it is now well accepted that OA is a disease of the whole joint, yet radiography can only directly visualize bone, with other structures such as articular cartilage inferred from measurement of JSW. However, as radiographs are a very good way to assess bone, there has been a rekindling of work refining assessment of trabecular bone texture. 17 This is promising as it has been shown to be a better predictor of progression of joint space narrowing than with KL-based models in a number of studies. 17
MRI has a major advantage over radiographic techniques in that the whole joint and different tissue components can be visualized. However, to date, there is still no accepted definition of early OA based on morphologic MRI and there remains a lack of clarity as to what structural changes constitute early OA. Structural features of OA are common and predate radiographic OA. 19 However, their clinical importance remains unclear and differentiation between normal aging and structural OA development remains a challenge. Understanding this will be very important as these structural changes are likely to identify different phenotypes of knee OA and different joint responses to risk factors. Pathological responses to important risk factors such as obesity are likely to occur in early life and understanding which can be attributed to aging and which are a pathological response will be important if we are to prevent the development of knee OA and treat it at the very early stages.
Compositional imaging adds important information on the health of the knee joint by providing rich data on the composition of cartilage and other tissues (such as menisci) before the development of morphological changes. The promise of cartilage compositional MRI in early OA lies in its ability to demonstrate cartilage abnormality at an earlier stage than conventional, structural MRI. 42 However, as with any quantitative imaging technique, compositional MRI requires careful attention to detail across acquisition and analysis to ensure that values obtained are accurate, reproducible, and interpretable. There will also be the need for better standardization of post-processing of images and automated segmentation. It is also likely that the full benefit of compositional imaging will be seen when these data are used in concert with morphological and other imaging information so that a picture of the health of the knee joint, and any response to pathological processes, can be captured.
Recent developments such as WBCT and spectral CT have rekindled interest in CT as a method for defining early OA. The strength of CT modalities lies in the ability to acquire more detailed imaging of mineralized joint tissues compared with radiography and MRI.59,62 The advent of lower scan doses makes it more feasible to use CT in large studies as it is now possible to do CT at doses only twice that of radiography. 82 CT techniques can also be used to acquire a 3D volume data, but the relative advantage compared with MRI remains unclear.
Conclusion
In a primary care setting, the role of imaging in defining early knee OA is limited to standard radiographs and KL grades 0 and 1. However, radiographs have significant limitations in characterizing early degenerative change. Both morphological and compositional MRI show abnormalities at earlier stages. While these technologies are substantially more sensitive, there is no established definition for early OA using MRI. Compositional MRI has the potential to diagnose cartilage matrix degenerative changes at the earliest stages, yet clinical application of this technology is still limited. New CT techniques may also provide new insights into early knee degeneration. The role of advanced imaging technologies in OA will have increased clinical significance as new therapeutic modalities evolve, which will require an earlier definition of damage of joint tissues and better identification of therapeutic targets.
