Abstract
Ocular disease, such as scleritis and peripheral ulcerative keratitis (PUK), may be a serious ocular complication. We present a patient with severe and refractory PUK treated with baricitinib. A review of the literature on Janus kinase inhibitors (JAKINIB) in refractory ocular surface pathology was also performed. For the literature review, the search in PubMed, Embase, and the Cochrane library was carried out from inception until 31 May 2021, including conference proceedings from four major rheumatology congresses. All original research articles studying JAKINIB treatment in patients with inflammatory eye disease were included. We present an 85-year-old woman with rheumatoid arthritis (RA) and secondary Sjögren’s syndrome refractory to methotrexate, leflunomide, certolizumab pegol, adalimumab, and tocilizumab (TCZ). However, 10 months after starting TCZ, the patient suffered a perforation secondary to PUK, requiring urgent surgical intervention. In the absence of infection, she was treated with boluses of intravenous methylprednisolone followed by oral prednisone at high doses in a decreasing pattern together with baricitinib at a dose of 2 mg/day with a very rapid and persistent favorable response to eye and joint symptoms. After 18 months of treatment, the patient had not presented serious side effects or signs of reactivation of her disease. In addition to this report, three other studies including one PUK associated with RA and two non-infectious scleritis treated with tofacitinib were included in this literature review. All three patients had experienced an insufficient response to conventional treatment, including biologic agents, before being switched to JAKINIB, leading to a complete or partial recovery in all of them without significant adverse effects so far. JAKINIBs (baricitinib and tofacitinib) may be an effective and safe therapy in patients with severe autoimmune and refractory ocular surface pathology, such as scleritis and PUK.
Keywords
Scleritis and peripheral ulcerative keratitis (PUK) can be a serious ocular complication isolated or related to immune-mediated inflammatory diseases (IMID). PUK is a destructive inflammatory disease that affects outer corneal stroma.1,2 It is estimated that the incidence of PUK is around three cases per million inhabitants/year and that it affects both sexes equally. 3
PUK may occur isolated or associated to IMIDs. The best known association is with rheumatoid arthritis (RA), but it can also be associated with antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis, systemic lupus erythematosus (SLE), relapsing polychondritis, or polyarteritis nodosa.4–6 If present, PUK is one of the most destructive and serious ophthalmologic manifestations of IMIDs. In fact, it is a serious complication with a high risk of eye perforation and sudden permanent visual loss. 7
In RA, PUK occurs more frequently in patients with very aggressive and advanced disease, with positive rheumatoid factor (RF) or positive anti-citrullinated protein (aCCP) antibodies. The appearance of PUK may indicate the transformation of RA into systemic vasculitis and, as with other IMIDs, the association of RA and PUK indicates a poor prognosis.4,5 In fact, it has been observed that patients with untreated RA and PUK are associated with a higher mortality rate, with a 5-year mortality 50%. 8
PUK can progress from marginal corneal thinning to perforation, which represents a more severe ocular condition that can lead to vision loss.9–11 Therefore, early diagnosis and treatment are mandatory. Although there is no universal consensus on the management of PUK, treatment generally depends on the severity of the corneal damage and the activity of the underlying disease. In addition to topical treatment and systemic glucocorticoids, conventional immunosuppressive therapy is generally required. However, despite this strategy, the proportion of patients experiencing visual loss remains inappropriately high.5,12,13 In many of these patients, biologic therapy has been shown to be effective.14–16 However, despite this treatment with both TNF inhibitors (TNFi) and non-TNFi biologic drugs, there are patients in whom PUK remains refractory.
Baricitinib is an oral, targeted synthetic disease-modifying antirheumatic drug (DMARD) that inhibits JAK1 and JAK2, that has been approved for use in RA patients refractory or intolerant to ⩾ 1 DMARDs. 17 However, there are few data in the literature on the use of Janus kinase inhibitors (JAKINIBs) in patients with inflammatory ocular disease.18–20
Taking all these considerations into account, we reported for the first time, to the best of our knowledge, the clinical response to baricitinib in a patient with severe PUK refractory to several conventional synthetic and biological systemic DMARDs and immunosuppressive therapy. In addition, a literature review was conducted on the effect of JAKINIB on ocular surface pathology.
Case report and literature review
Case report
We followed the CARE (Case Report) guidelines for case report publication. 21 We have de-identified all patient details.
We present the case of an 85-year-old woman undergoing follow-up at the rheumatology outpatient clinic since July 2014 for seropositive RA (RF 92 UI/ml; reference range 0–22) and aCCP antibodies higher than 2777 (reference range 0–20) and secondary Sjögren’s syndrome (SS).
The patient had received treatment with conventional DMARDs, methotrexate (MTX) and leflunomide, which were discontinued due to poor tolerance (severe asthenia and alopecia). She also was treated with TNFi certolizumab pegol (CZP) and adalimumab (ADA) that had to be suspended due to persistence of active joint disease. Because of that, treatment with subcutaneous tocilizumab (TCZ) at a weekly dose of 162 mg was started at the beginning of February 2019 (DAS28-CRP at TCZ onset was 5.25, which indicated severe activity). However, in March 2019, she presented an episode of dry eye associated with bilateral hyperemia. Because of that she was referred to the Ophthalmology outpatient clinic. A slit-lamp examination disclosed a peripheral crescentic ulceration with an epithelial defect, thinning, and subepithelial infiltration near the limbus. Topical treatment with preservative-free lubricants, low-dose prophylactic antibiotic drops (tobramycin) along with topical glucocorticoids (fluorometholone ophthalmic) was prescribed with good response, but only partial control of the joint symptoms (DAS28-CRP: 4.3; moderate activity) had been achieved at that time. Several months later, in December 2019, she presented to the Ophthalmology emergency room due to a 10-day history of hyperemia, pain, photophobia, and decreased visual acuity in her right eye. The ophthalmological examination showed an 6 mm epithelial defect with stromal thinning and an extensive perforation near the limbus.
Because the corneal glue did not close the corneal perforation, urgent surgical intervention was performed using corneal suture to repair the corneal perforation and hospital admission for ophthalmic monitoring (Figure 1). Penetrating keratoplasty was subsequently ruled out due to the general characteristics of the patient.

Peripheral.
Regarding joint activity, the patient persisted with a DAS28-CRP of 4.1 and CRP 3.2 mg/dl. During admission, she was evaluated by Rheumatology, deciding, in the absence of infection data, to administer iv boluses of methylprednisolone (iv MP) at a dose of 250 mg/day for three consecutive days and followed by oral prednisone at an initial dose of 30 mg/day that was gradually tapered to a current dose of 2.5 mg/day.
In January 2020 and after performing chest radiographs, Mantoux, booster, quantiferon, and serologies to exclude infection, it was decided to start baricitinib at a dose of 2 mg/day, after obtaining the informed consent of the patient to start this treatment. To take care of ocular surface, treatment was maintained with free lubricants, autologous serum, and oral doxycycline.
After 2 weeks of treatment, an excellent evolution was observed both at the ocular (Figure 2) and joint (DAS28-CRP: 1.94) levels. Since then, she has not experienced any new episodes of corneal ulcers and her disease activity remains under control. After 18 months from the start of baricitinib, DAS28-CRP score was 2.1 and CRP 1.5 mg/dl. Currently, the patient remains in ocular and joint remission and the only adverse effect she has had has been mild alopecia.

Optical coherence tomography (OCT) of the same patient shown in Figure 1 with PUK after treatment with baricitinib.
Literature Review
For this literature review, a search in PubMed, Embase, and the Cochrane library was conducted from their inception to 31 May 2021, including conference proceedings from four major rheumatology congresses. Original research articles studying JAKINIB treatment in patients with inflammatory eye disease were included.
Table 1 shows the main literature studies on ocular surface pathology under treatment with JAKINIBs.
Literature review of patients with refractory ocular surface pathology (PUK, scleritis) including the present case.
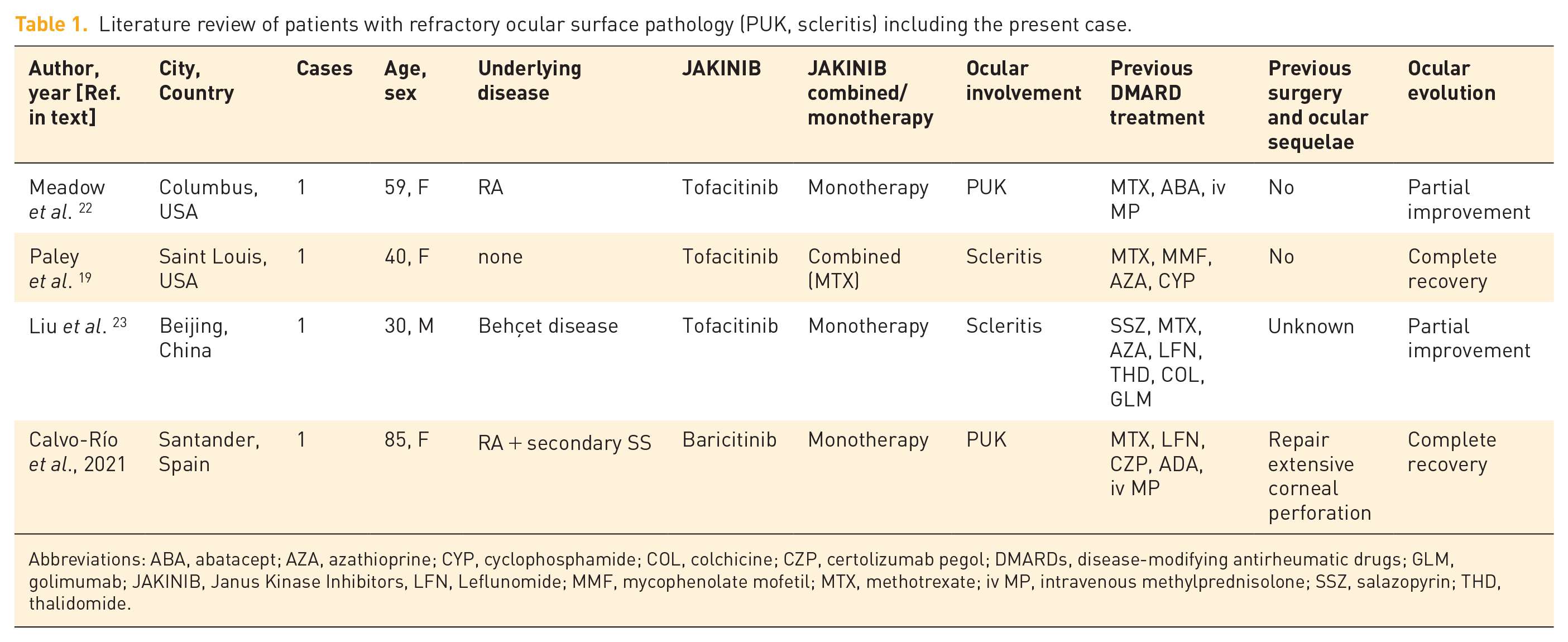
Abbreviations: ABA, abatacept; AZA, azathioprine; CYP, cyclophosphamide; COL, colchicine; CZP, certolizumab pegol; DMARDs, disease-modifying antirheumatic drugs; GLM, golimumab; JAKINIB, Janus Kinase Inhibitors, LFN, Leflunomide; MMF, mycophenolate mofetil; MTX, methotrexate; iv MP, intravenous methylprednisolone; SSZ, salazopyrin; THD, thalidomide.
Discussion
We present a patient with RA refractory to conventional synthetic DMARDs, TNFi and TCZ, and secondary PUK, in whom the efficacy of baricitinib was verified. To our knowledge, only a few cases of use of JAKINIB in PUK and scleritis have been reported.
The pathogenesis of PUK is not fully understood; both humoral and cellular immunity may be involved. Different immune cells, primarily macrophages, and mast cells are recruited to the peripheral cornea. These cells release metalloproteases and collagenases that are mainly responsible for the destruction of the external corneal stroma. Both TH1 and TH17 lymphocytes are involved in the pathogenesis of ocular inflammation. These lymphocytes produce cytokines, such as IL-2, IL-6, IL-17, IL-23, interferon (IFN)-γ, and TNF, that recruit leukocytes from the circulation and contribute to tissue damage. A high level of cytokines, such as TNF and interleukin (IL)-6, is present in the sclera-corneal limbus, promoting the local invasion of immune cells and then the progression of the ulceration. 24 JAKINIBs inhibit both the Th1 and Th17 response, thus they may be useful in the treatment of PUK. 25
The cornea is an avascular structure that receives oxygenation from tears, aqueous humor, and limbal vessels that are in contact with the cornea but without invading the tissue. The peripheral cornea, the typical location of PUK, is distinct from the central cornea in both anatomical and physiological characteristics. The peripheral cornea is close to the capillary bed and partly obtains its nutrients from the capillary arches. The proximity to this vascular and lymphatic arch may explain the peripheral location of PUK as inflammatory cells and inflammation mediators can more easily access this part of the cornea.26,27
PUK can be associated with various infectious and non-infectious eye and systemic diseases. Most cases are due to local or systemic immunological processes, of which RA is the most common cause, accounting for 34% of non-infectious PUK cases. It occurs mainly in patients with destructive articular and nodular RA with high titers of RF and aCCP antibodies.5,11 In RA, PUK usually occurs after a long course of the disease. Studies report a 17- to 20-year delay between the diagnosis of RA and the onset of the corneal ulcer.1,3,11 It is bilateral in more than one-third of these patients. Its appearance late in the course of the disease can be a warning sign that indicates a worsening of the underlying disease. In fact, some authors believe that the appearance of PUK may indicate the evolution of RA toward a phase of systemic vasculitis.
PUK is often a manifestation of a systemic disease, indicating a high degree of inflammatory activity. In the last years, the incidence of PUK has decreased, probably due to the advances in the treatment of autoimmune diseases that allow a better control of the activity of the disease.26,28 In addition, although it is likely that the vast majority of PUK cases are detected by a medical specialist, in some patients, PUK may remain inactive and these cases may not be reported, underestimating the percentage of patients affected by this entity.
There is no universal consensus on the management of patients with PUK. In general, treatment is based on the severity of the clinical manifestations. Combined glucocorticoids with conventional synthetic DMARDs are the cornerstone therapy of PUK, but it is not always effective.8,29 In these cases, biologic therapy can be useful. Most of the scientific evidence comes from case series. Several studies support the efficacy of TNFi. In this regard, infliximab (IFX)7,11,14,26,30 and ADA26,29,31 have been the most widely used biologic agents in PUK refractory to conventional synthetic DMARDs. There are also studies that have demonstrated the efficacy of other TNFi, such as etanercept,26,32 golimumab (GLM), and CZP. 26
Among biologics other than TNFi, rituximab (RTX) is the most widely used. There are several studies that show its effectiveness, also leading to a reduction in the need for eye surgery. In the review of the literature that we present in Table 1, none of the patients who received JAKINIB had been treated with RTX. However, we include RTX in Figure 3 based on previous reports.9,26,33–36 Ocular manifestations in RA not only may be related to the underlying disease but also it may be due to the adverse ocular reactions secondary to treatment with nonsteroidal anti-inflammatory drugs (NSAIDs) and DMARDs, both synthetic and biological, in patients with RA. 37

Proposed therapeutic strategy for PUK.
TCZ is a humanized monoclonal antibody against IL-6 receptor, which has been approved for the treatment of inflammatory diseases, such as RA, systemic and polyarticular juvenile arthritis, and giant cell arteritis. 38 TCZ has also shown efficacy in other refractory inflammatory diseases, including ocular diseases.39–45 TCZ may be useful in PUK associated to RA. 26 Paradoxically, PUK has been described in RA patients receiving TCZ, which can be interpreted as a lack of efficacy or as a paradoxical reaction to the drug. 46
Abatacept is a recombinant fusion protein that selectively modulates the co-stimulatory signal necessary for T-cell activation. Abatacept is indicated for the treatment of moderate-to-severe active RA in adults. 47 Efficacy of abatacept has been demonstrated in one patient with PUK and RA refractory to other biologic drugs. 26
During the last years, JAKINIBs have been used in different IMIDs, including RA, psoriatic arthritis, spondyloarthritis, ulcerative colitis, and other diseases. JAKINIBs are likely to be effective agents in refractory non-infectious inflammatory eye disease, including uveitis,18–20,48 scleritis,19,23 sicca syndrome,49–52 and conjunctivitis. 53 Several abovementioned inflammatory cytokines that have been implicated in the pathogenesis of ocular inflammation are known to use the JAK/signal transducer and activator of transcription (STAT) signaling pathway, including IL-2, IL-23, and IL-6.19,54,55 As JAKINIBs are small molecules that can pass through blood-aqueous or blood-retinal barrier more efficiently than biologics, topical administration of small molecules may control ocular inflammation with better tolerability. In support of this hypothesis, tofacitinib administered topically was effective in a mouse model of corneal inflammation. Table 1 shows a literature review on ocular surface pathology under treatment with JAKINIB drugs. To our knowledge, there is only one study that demonstrates the efficacy of tofacitinib in a patient with PUK and corneal melting secondary to RA, who had been refractory to boluses of iv MP, MTX, and abatacept. 22 There are two other studies that demonstrated the efficacy of tofacitinib in two patients with scleritis, in one case secondary to Behçet’s disease and in the other to RA with secondary SS. These patients had been refractory to systemic glucocorticoids, conventional synthetic, and biological DMARDs.19,23
Overall, 70% of patients with Behçet’s disease will have ocular involvement. Although the most frequent is panuveitis, anterior uveitis, and posterior uveitis, there are some published cases with ocular involvement in the form of PUK, although we have not found cases of Behçet’s in the literature associating PUK and scleritis. 54
There is also a three-arm clinical trial that has demonstrated the efficacy of different doses of topical tofacitinib in 233 patients with dry eye syndrome compared with cyclosporine ophthalmic emulsion and placebo (vehicle). This study demonstrated a tendency for tofacitinib to improve dry eye signs and symptoms with a reasonable safety and tolerance profile. 51 Another study has shown that the topical administration of tofacitinib in mice with experimental allergic conjunctivitis reduces the symptoms and decreases the inflammatory response and histamine release. 53
Based on the above mentioned considerations and a previous report 23 of our group, we have proposed a therapeutic scheme for the treatment of PUK isolated or secondary to IMID, which is detailed in Figure 3. Conventional systemic DMARDs are at the base of the pyramid. When these drugs are not effective, must be discontinued due to intolerance or side effects, or are contraindicated, the first-line drugs should be TNFi, especially IFX and ADA due to the previous experience with these biologic agents. In case of secondary failure of these drugs, we can consider switching to other TNFi agents, such as GLM or CZP, while in those patients with primary failure, we can change the therapeutic target to TCZ or RTX. At the top of the pyramid, we can use JAKINIBs such as baricitinib or tofacitinib.
Conclusion
In conclusion, to our knowledge, we report the first case of effective treatment with baricitinib in a patient with PUK secondary to RA refractory to conventional synthetic DMARDs, TNFi, and TCZ. Although our results are certainly promising, more studies with larger series of patients are needed to confirm our findings.
Supplemental Material
sj-png-1-tab-10.1177_1759720X221137126 – Supplemental material for Baricitinib in severe and refractory peripheral ulcerative keratitis: a case report and literature review
Supplemental material, sj-png-1-tab-10.1177_1759720X221137126 for Baricitinib in severe and refractory peripheral ulcerative keratitis: a case report and literature review by Vanesa Calvo-Río, Lara Sánchez-Bilbao, Carmen Álvarez-Reguera, Santos Castañeda, Iñigo González-Mazón, Rosalía Demetrio-Pablo, Miguel A. González-Gay and Ricardo Blanco in Therapeutic Advances in Musculoskeletal Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
