Abstract
Blau syndrome (BS) is an autoinflammatory disorder characterized by non-caseating granulomatous dermatitis, arthritis, and uveitis. We present a case of refractory and severe BS that was treated with the Janus kinase inhibitors (JAKINIBS), Tofacitinib (TOFA) and then Baricitinib (BARI). Our aim was to describe the clinical and immunological outcomes after treatment with JAKINIBS. Blood tests and serum samples were obtained during follow-up with TOFA and BARI. We assessed their effects on clinical outcomes, acute phase reactants, absolute lymphocyte counts (ALCs), lymphocyte subset counts, immunoglobulins, and cytokine levels. A review of the literature on the use of JAKINIBS for the treatment of uveitis and sarcoidosis was also conducted. TOFA led to a rapid and maintained disease control and a steroid-sparing effect. A decrease from baseline was observed in ALC, CD3+, CD4+, CD8+, and natural killer (NK) cell counts. B-cells were stable. Serum levels of interleukin (IL)-4 and tumor necrosis factor alpha (TNF-α) increased, whereas IL-2, IL-6, IL-10, and IL-17 maintained stable. TOFA was discontinued after 19 months due to significant lymphopenia. The initiation of BARI allowed maintaining adequate control of disease activity with an adequate safety profile. The literature review showed seven patients with uveitis and five with sarcoidosis treated with JAKINIBS. No cases of BS treated with JAKINIBS were found. We report the successful use of JAKINIBS in a patient with refractory and severe BS.
Introduction
Blau syndrome (BS) is an autoinflammatory disorder characterized by non-caseating granulomatous dermatitis, symmetric arthritis, and recurrent uveitis with childhood onset. It is associated with mutations in the NOD2/CARD15 gene.1–3 BS is inherited in an autosomal dominant pattern. When the same phenotype is presented without family history, it is also called Early Onset Sarcoidosis (EOS), but the term BS can be used for both inherited and sporadic forms.4–6
Eye involvement is usually the most severe and refractory manifestation of BS. Chronic granulomatous uveitis can evolve into cataract, high intraocular pressure (IOP), and band keratopathy, frequently requiring surgery.3,7
Non-steroidal anti-inflammatory drugs (NSAIDs), corticosteroids, and conventional immunosuppressive agents, such as methotrexate (MTX) and azathioprine (AZA), are initially used. More recently, biologic agents such as tumor necrosis factor alpha (TNF-α) blockers, interleukin (IL)-1, and IL-6 receptor inhibitors have demonstrated effectiveness in refractory cases.8–10
Tofacitinib (TOFA) and Baricitinib (BARI) are two Janus kinase inhibitors (JAKINIBS) that are approved for their use in several immune-mediated inflammatory diseases (IMIDs) but not in granulomatous diseases. Janus kinases (JAK) may be involved in the pathogenesis of uveitis and sarcoidosis; however, there are only a few case reports published on the successful use of JAKINIBS.11–14
We present a case of severe BS refractory to multiple lines of immunosuppressive therapy, treated with TOFA and then BARI. In addition, a literature review was performed on the use of JAKINIBS in uveitis and sarcoidosis.
Patients and methods
We present the case of a 25-year-old woman with BS followed by the Rheumatology and Ophthalmology unit of our hospital. At the age of 2 years old, she was initially diagnosed with Juvenile Idiopathic Arthritis (JIA) after presenting several episodes of polyarthritis. She was initially treated with acetylsalicylic acid that was discontinued due to drug-induced hepatitis. MTX (3.75 mg/week) and oral methylprednisolone (0.5 mg/kg/day) were then started. From the age of 4, she developed several episodes of bilateral anterior uveitis and required topical corticosteroids and cycloplegics. The ophthalmologic examination showed peripapillary granulomas and mild vitritis.
In addition to relapsing bilateral anterior uveitis, when she was 10 years old, she had a micropapular erythematous rash in groins and both feet. The skin biopsy revealed non-caseating granulomas. BS was then suspected, and genetic laboratory testing reported a mutation in NOD2/CARD15 (R334Q) gene. This mutation was not found in her parents; therefore, she was diagnosed with EOS/sporadic BS. Biological therapy was started due to refractory manifestations. She received etanercept, anakinra, and abatacept, but none of them were effective to induce remission of the disease.
At the age of 14 years old, she experienced an episode of bilateral panuveitis. Adalimumab (ADA; 40 mg every other week (EOW)) was prescribed. In the subsequent years, she developed other severe episodes of uveitis, IOP, and cataract in her right eye. IOP required surgery.
Five years later, she required intensified therapy with ADA (40 mg weekly) and oral MTX up to 20 mg weekly to control ocular, cutaneous, and articular manifestations. Despite good adherence to treatment, systemic and topical corticosteroids were frequently required for recurrent episodes of anterior, posterior, and panuveitis (up to 3–4 per year).
At the age of 25 years old, in addition to erythematous rash (Figure 1(a)), the ophthalmologic examination showed cystoid macular edema in her left eye. The central foveal thickness measured by Optical Coherence Tomography (OCT) was 372 µm, and the best-corrected visual acuity in her left eye was 0.5/1. (Figure 1(c)).

Skin rash before (a) after treatment with Tofacitinib (b). Optical coherence tomography of left eye showing cystoid macular edema before (c) and after treatment with Tofacitinib (d).
Due to the persistence of inflammatory activity, and based on the rationale of JAK involvement in the pathogenesis of BS, 11 written informed consent to treatment was obtained, because TOFA is an off-label indication of BS. We followed the CARE (Case Report) guidelines for case report publication. 15 All the patient details were de-identified, and the signed consent to publish was also provide from the patient. It was approved by the Local Clinical Research Ethics Committee (Name: Comité Ético de Investigación médica de Cantabria), approval number: 2020.437.
In addition to methylprednsiolone 6 mg/day, TOFA at a standard dose of 5 mg twice daily was started. ADA and MTX were stopped. TOFA led to a rapid and sustained control of previously refractory clinical manifestations. Arthritis signs in both knees and wrists and erythematous plaques on chest and lower extremities disappeared 1 month after TOFA initiation. Macular edema and anterior uveitis signs improved from the first weeks of treatment and were not present at ophthalmological evaluation after 2 months. A progressive reduction in corticosteroids dose up to withdrawal was observed.
The patient was asymptomatic at cutaneous, ocular, and articular levels during TOFA therapy. She only experienced one relapse consisting of a micropapular rash in the infraumbilical area and flogotic signs in both wrists when TOFA was discontinued during a mild upper respiratory infection.
Nineteen months after TOFA onset, the dose was reduced to 5 mg/daily because of significant lymphopenia (700 cells/µl). Subsequently, the patient experienced clinical worsening consisting of arthritis of both wrists. TOFA was finally discontinued due to persistence of marked lymphopenia (500 cells/µl). Four weeks after TOFA discontinuation, an improvement in lymphocyte count to normal range levels was observed.
Due to BS worsening, ADA was restarted (40 mg EOW) while waiting for other therapeutic options. After 2 weeks of ADA, a new off-label treatment with BARI at a standard dose of 4 mg daily was started. Since then, cutaneous, ocular, and joint manifestations have remained stable.
Laboratory Immunological study
Blood tests and serum samples were obtained at baseline and at months 1 and 12 after TOFA onset and at baseline and month 4 after BARI onset. We assessed their effects on acute phase reactants, absolute lymphocyte counts (ALCs), lymphocyte subset counts, red blood cell count, platelet count, immunoglobulins, and cytokine levels.
Ethylenediaminetetraacetic acid (EDTA) peripheral blood samples were collected to analyze B- and T-lymphocytes subsets using flow cytometry in a Navios EX Flow Cytometer (Beckman Coulter, Barcelona, Spain). Monoclonal antibodies from Beckman Coulter were used to characterize T-, B-, and natural killer (NK) cells (available upon request).
IgG, IgM, IgA, and IgG subclasses were measured in serum samples by turbidimetry using the Optilite® analyzer (Binding Site, Barcelona, Spain).
Peripheral blood mononuclear cells (PBMCs) were isolated by using Ficoll-Paque gradient from sodium heparin blood samples for the in vitro polyclonal activation. Peripheral Blood Mononuclear Cells (PBMCs) were seeded on 96-well plates at a final concentration of 0.5 × 106 cells/µl, cultured at 37°C in 5% CO2 with supplemented Roswell Park Memorial Institute 1640 medium (R10), and stimulated with magnetic beads coated with anti-CD3 and anti-CD28 antibodies (Dynabeads®; Thermo Fisher Scientific, Madrid, Spain) in 1:1 cell:bead ratio. After 72 h, T-cell proliferation was analyzed measuring carboxyfluorescein succimidyl ester (CFSE) by flow cytometry, and supernatants were collected and stored at –80°C until analysis.
In the serum and culture supernatant, IL-2, IL-4, IL-6, IL-10, IL-17A, and TNF-α levels were analyzed by using the Human High Sensitivity T-cell panel from Merck Millipore (Merck Life Science S.L.U., Madrid, Spain) in a Luminex 200 platform following manufacturer’s instructions. The normal or abnormal levels of cytokines were established following two different criteria: the pretreatment levels of the patient and the levels of five healthy age and sex-matched controls.
Results
Laboratory Immunological study
Table 1 shows the laboratory test results throughout the follow-up of treatment with TOFA and BARI. A decrease from baseline in the ALC, CD3+, CD4+, and CD8+ cell counts was progressively observed during follow-up with TOFA and during the first 4 months with BARI. The values of anti-CD3/28 (1 µl/500,000 cells) were normal [>0.7 (4 × 107 Dynabeads/ml)] before TOFA therapy and at 1 and 12 months after TOFA onset. They were assessed to evaluate the possible influence of JAKINIBS on the activation of T-lymphocytes through stimulation via the second signal (CD28). Both CD4 and CD8 Central Memory T-cells (TCM), Effector Memory T-cells (TEM), and Effector Memory Recently Activated T-cells (TEMRA) also suffered a progressive decrease after 12 months of treatment. The most remarkable change was observed in CD4+ and CD8+. Naïve cells showed a deep delayed reduction, falling under the normal range with TOFA.
Complete laboratory and immunological test results throughout follow-up with TOFA and BARI therapy.

TOFA was stopped at month 19 due to severe lymphopenia. BARI was started 6 weeks after TOFA withdrawal.
BARI, Baricitinib; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; NK, natural killer; TCM, Central Memory T-cells; TEM, Effector Memory T-cells; TEMRA, Effector Memory Recently Activated T-cells; TOFA, Tofacitinib; Tregs, regulatory T-cells.
Regarding the ALC, a drop to 500 cells/µl together with a clinical worsening forced to stop TOFA. Lymphocyte counts transiently increased during the 2 weeks of treatment with ADA and remained stable during BARI therapy. However, after 4 months of treatment with BARI, a new episode of lymphopenia (800 cells/µl) was observed (Figure 2(a)).
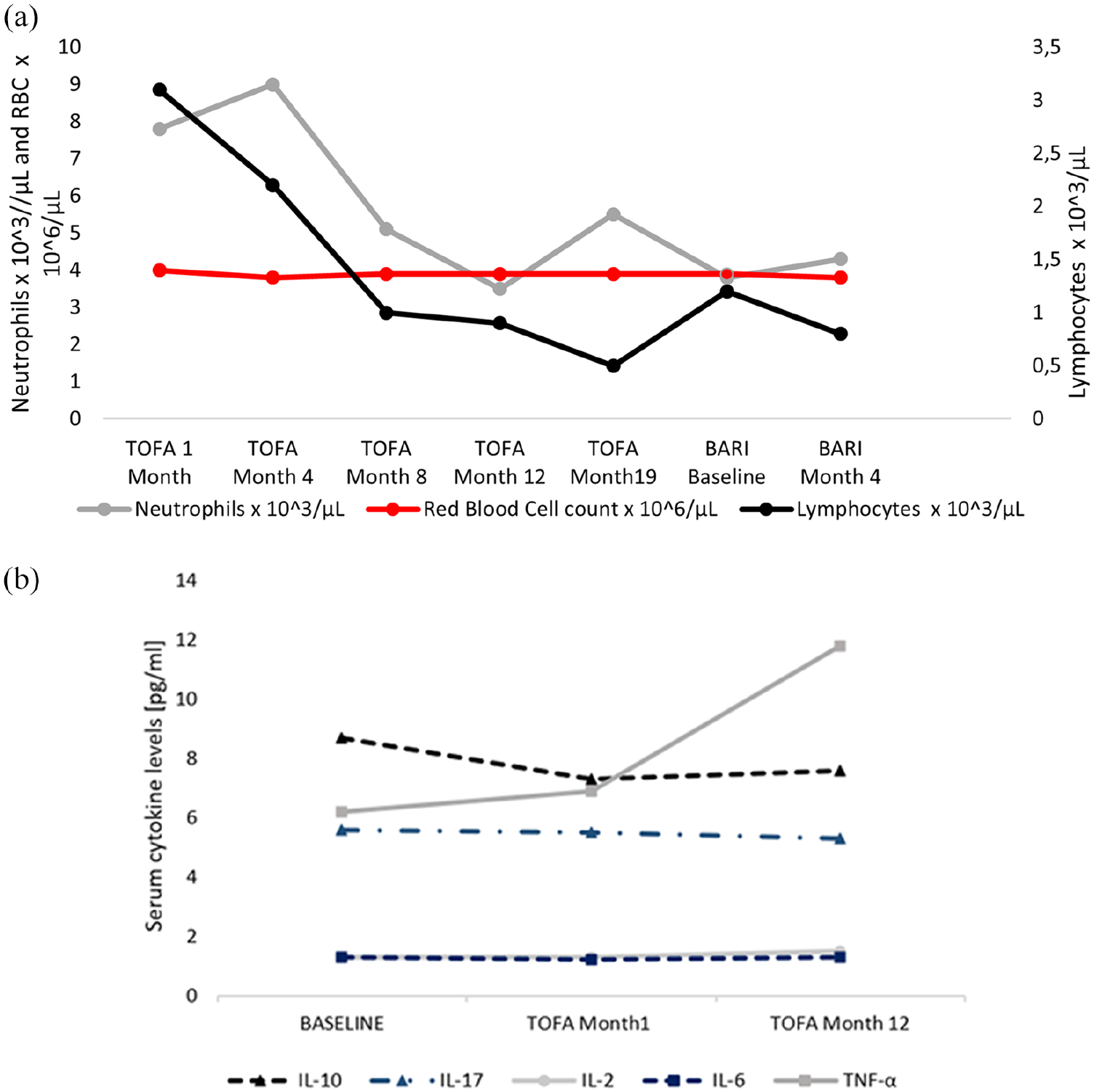
(a) Leukocytes, lymphocytes, and hemoglobin evolution through follow-up with Tofacitinib and Baricitinib therapy. (b) Cytokines evolution through follow-up with Tofacitinib.
B-cell (CD19+) and NK cell counts remained within the reference range during follow-up. However, a mild decrease in cell count was observed. IgG, IgM, and IgA levels stayed within the reference range after 19 months of treatment with TOFA and 4 months of treatment with BARI. IgG levels increased (mainly IgG1 and IgG3), and IgM showed a small reduction.
After 12 months of treatment with TOFA, IL-4 and TNF-α serum levels increased, whereas IL-2, IL-6, IL-10, and IL-17A did not modify their serum levels. Regarding cytokine levels after in vitro culture with anti-CD3/CD28 stimulation, IL-2 and IL-4 showed a marked reduction in their production, whereas IL-6, IL-10, IL-17A, and TNF-α levels increased in a significant way (Figure 2(b)).
Literature review
For literature review, a search was conducted in PubMed, Embase, and the Cochrane Library, from their inception to 31 July 2021, and conference proceedings from the four major rheumatology conferences. Original research articles on JAKINIBS in uveitis and sarcoidosis were included.
Table 2 summarizes the studies published on the use of JAKINIBS for the treatment of uveitis. Bauermann et al. were the first authors to describe the successful use of TOFA in a case of severe refractory recurrent anterior uveitis and associated macular edema in an adult patient with JIA in 2018. 16 In 2019, Paley et al. 17 published the positive effect of TOFA in a case of idiopathic uveitis refractory to topical and local steroid injections and different disease-modifying antirheumatic drugs (DMARDs). The same group also reported a case of idiopathic scleritis remission using MTX and TOFA, achieving a steroid-sparing effect in both cases. A case series of four consecutive patients with long-term history of JIA and severe associated uveitis was published by Miserocchi et al. 18 They observed a good response of ocular inflammation in all four cases. However, the articular disease did not respond adequately.
Review of the literature on uveitis treated with Janus kinase inhibitors.
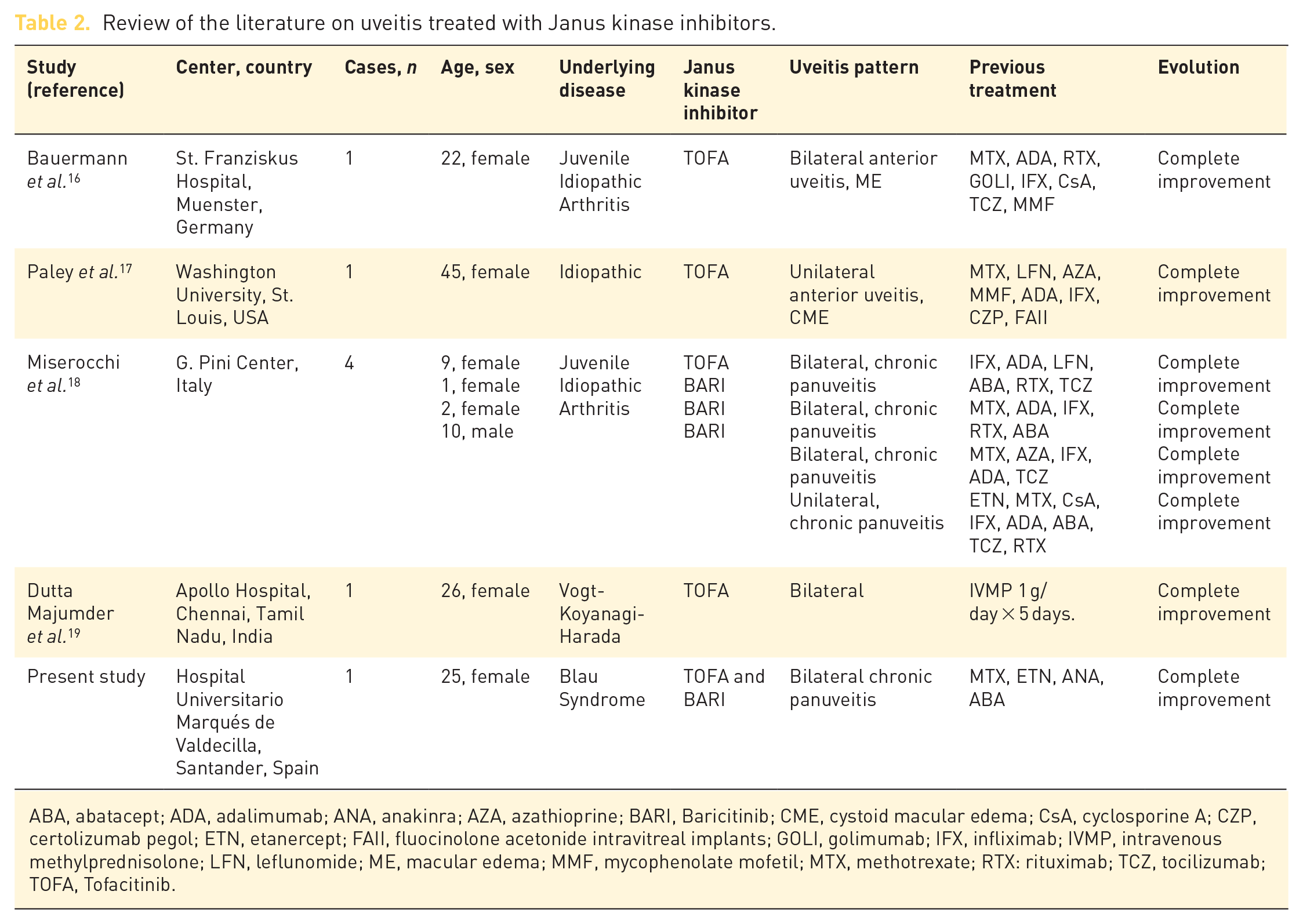
ABA, abatacept; ADA, adalimumab; ANA, anakinra; AZA, azathioprine; BARI, Baricitinib; CME, cystoid macular edema; CsA, cyclosporine A; CZP, certolizumab pegol; ETN, etanercept; FAII, fluocinolone acetonide intravitreal implants; GOLI, golimumab; IFX, infliximab; IVMP, intravenous methylprednisolone; LFN, leflunomide; ME, macular edema; MMF, mycophenolate mofetil; MTX, methotrexate; RTX: rituximab; TCZ, tocilizumab; TOFA, Tofacitinib.
Dutta Majumder et al. 19 reported a case of a young girl with exudative retinal detachment due to Vogt-Koyanagi-Harada disease that was steroid-resistant. One month after the initiation of TOFA, her visual acuity improved and fundus examination revealed resolution of exudative retinal detachment.
Regarding the usefulness of JAKINIBS for sarcoidosis, Damsky et al. were the first to describe the efficacy of TOFA in a patient with cutaneous sarcoidosis and afterward in multiorgan sarcoidosis.12,13,20 Other groups have published multiorgan and cutaneous sarcoidosis improvement after the initiation of ruxolitinib (RUXO), a JAK1 and JAK2 inhibitor 12 (Table 3). Other phase III trials are currently being conducted on cutaneous (NCT03910543) and systemic sarcoidosis (NCT03793439 and NCT02920710).
Review of the literature on sarcoidosis treated with Janus kinase inhibitors.

ADA, adalimumab; APR, apremilast; AZA, azathioprine; CP, cyclophosphamide; CsA, cyclosporine A; HCQ, hydroxicloroquine; INF, infliximab; IVIG, intravenous immunoglobulin; LEF, leflunomide; MMF, mycophenolate mofetil; MTX, methotrexate; PnbUVB: phototherapy (nbUVB); RTX, rituximab; RUXO, ruxolitinib; 13-CRA, isotretinoin; TNF, tumor necrosis factor; TOFA, Tofacitinib.
Discussion
To the best of our knowledge, we report for the first time the successful use of TOFA and then BARI in a patient with severe and refractory EOS/sporadic BS. Remission was achieved with TOFA and was subsequently maintained with BARI when TOFA was discontinued due to severe lymphopenia.
As previously described, scarce evidence is available on the use of JAKINIBS in the treatment of uveitis and sarcoidosis. The reason for the use of TOFA and BARI for treating our patient was based on the granulomatous nature of the disease. The activation of macrophages in the granuloma is considered to be dependent on CD4+ cells that secrete several cytokines such as IL-2 and TNF-α.3,24 JAK1 and JAK3 pathways are known to be constitutively active in sarcoidosis and other granulomatous diseases. TOFA inhibits JAK1, JAK3, and to a lesser extent JAK2, and BARI inhibits JAK1 and JAK2, thereby blocking the proinflammatory cytokine expression at sites of inflammation. 25
A dose-dependent mean decrease in ALCs was reported in TOFA-treated rheumatoid arthritis (RA) patients. This was also the case with our patient. Reductions in hemoglobin levels, hematocrit, and red blood cell counts could be explained by the inhibition of JAK2-dependent erythropoietin signaling. 26 Since lymphopenia has been associated with an increased risk of serious infections during TOFA treatment, evaluation of the ALC at baseline and monitoring every 3 months is recommended. Treatment with JAKINIBS should be interrupted if the ALC is 500 cells/µl.
TOFA partially and reversibly inhibits the signaling of IL-2, IL-4, IL-7, IL-9, and IL-15. These cytokines are important for the development and proliferation of CD3+, CD19+, and NK cells. In RA patients treated with TOFA, a gradual decline to steady state of CD3+, CD4+, CD8+, and NK cell counts has been described. In contrast, CD19+ cell counts showed dose-dependent increases. 26 Cell-mediated immunity data suggest that TOFA does not significantly impair T-cell function. Noteworthy, CD4+ cell count did not fall below 200 cells/µl, which is known to be a risk factor for opportunistic infections.27,28
Lymphopenia is a well-known analytical finding of TOFA. However, in both RA and atopic dermatitis, BARI has not been usually associated with a decrease in ALC. The decrease after 4 months of treatment with BARI could be due to lymphopenia associated with sarcoidosis or some other mechanism that is still unknown.
In patients with RA treated with TOFA, only slight decreases in serum IgG, IgM, and IgA levels are observed. This may be due to changes in disease activity rather than a direct effect on B-cell function.29,30 In fact, in the case of our patient, IgG levels increased (mainly IgG1, 3 and 4 subclasses), which could indicate that plasma cells resident in the bone marrow were not completely affected by the action of the treatment.
In conclusion, TOFA and then BARI were useful to achieve rapid and maintained clinical remission in a patient refractory to multiple lines of immunosuppressive therapy. Although these preliminary results are promising, further studies with larger series of patients and randomized clinical trials are needed to confirm our findings.
Footnotes
Author contribution(s)
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Professor Miguel A. González-Gay has received grants/research supports from Abbvie, MSD, Jansen, and Roche, and had consultation fees/participation in company sponsored speaker’s bureau from Abbvie, Pfizer, Roche, Sanofi, and MSD. Dr Ricardo Blanco received grants/research supports from Abbvie, MSD, and Roche, and had consultation fees/participation in company sponsored speaker’s bureau from Abbvie, Pfizer, Roche, Bristol-Myers, Janssen, Lilly, and MSD. Dr Carmen Álvarez-Reguera, Diana Prieto-Peña, Alba Herrero-Morant, Lara Sánchez-Bilbao, José Luis Martín-Varillas, Elena González-López, María Gutiérrez-Larrañaga, David San Segundo, Rosalía Demetrio-Pablo, and Gonzalo Ocejo-Vinyals have not declared financial disclosures.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by Redes Temáticas de Investigación Cooperativa en Salud (RETICS) Program, RD16/0012 Red de Investigación en Inflamación y Enfermedades Reumáticas (RIER) from ISCIII from ‘Instituto de Salud Carlos III’ (ISCIII) (Spain).
