Abstract
Interleukin (IL)-6 and IL-1 blockade showed beneficial results in patients with severe COVID-19 pneumonia and evidence of cytokine release at the early disease stage. Here, we report outcomes of open-label therapy with a combination of blocking IL-6 with tocilizumab 8 mg/kg up to 800 mg and IL-1 receptor antagonist anakinra 100–300 mg over 3–5 days. Thirty-one adult patients with severe COVID-19 pneumonia and signs of cytokine release, mean age 54 (30–79) years, 5 female, 26 male, and mean symptom duration 6 (3–10) days were treated. Patients with more than 10 days of symptoms, evidence of bacterial infection/elevated procalcitonin and other severe lung diseases were excluded. Computed tomography (CT) scans of the lung were performed initially and after 1 month; inflammatory activity was assessed on a scale 0–25. Twenty-five patients survived without intubation and mechanical lung ventilation, two patients died. C-reactive protein decreased in 19/31 patients to normal ranges. The mean activity CT score decreased from 14 (8–20) to 6 (0–16, n = 16). In conclusion, most of our patients recovered fast and sustained, indicating that early interruption of cytokine release might be very effective in preventing patients from mechanical ventilation, death, and long-term damage.
Introduction
The novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), was first identified in December 2019 as the cause of a respiratory illness designated coronavirus disease 2019, or COVID-19. 1 Most patients have mild to moderate disease, but some patients become severely ill. This severe disease is driven by immunoreaction and no more by virus-replication. This concept is further supported, as anti-viral treatment with remdesivir, and treatment with anti-COVID-19-spike antibodies (monoclonal or re-convalescence sera) did not improve the disease.2 –5 Severe and life-threatening COVID-19 pneumonia is often characterized by local and systemic immune-mediated hyperinflammation, so-called cytokine storm.6 –9 SARS-CoV-2 is thought to bind to toll-like receptors, activates the inflammasome and the cleavage of pro-interleukin (IL)-1β by caspase-1, followed by the production of active mature IL-1β, a powerful pro-inflammatory cytokine that starts an inflammatory cascade, overproduction of other pro-inflammatory cytokines including IL-6 and active leukocyte migration in the areas affected by inflammation. 10 Higher levels of IL-6 have been positively correlated with severe course of COVID-19, whereas lower levels of IL-6 have been correlated with mild disease.11,12 Elevated levels of IL-6 have been found to be correlated with the likelihood of mechanical ventilation. 13 Activated monocytes migrating to lungs cause the typical opac infiltrates, which lead to a reduction of oxygen uptake resulting in severe cases in respiratory insufficiency.14,15 Serum levels of biomarkers indicating the cytokine storm such as IL-6 and ferritin correlate with severe courses of the COVID-19 disease. 16 The hyperinflammatory nature of the lung affection in COVID-19 builds the rationale for the use of immune-modulating drugs in this disease.
Among immune-modulating drugs, which were studied in COVID-19, are corticosteroids, IL-6, and IL-1 as well as Janus kinase (JAK) inhibitors. A meta-analysis that included 1703 patients showed benefit for patients with severe acute respiratory distress syndrome when different regimes of corticosteroids were used. 17 In this prospective meta-analysis of clinical trials of critically ill patients with COVID-19, administration of systemic corticosteroids, compared with usual care or placebo, was associated with lower 28-day all-cause mortality. For anti-IL-6 therapies, several trials were published with heterogeneous study populations and heterogeneous results.18 –27 In the largest trial so far, with more than 2000 patients allocated to tocilizumab and more than 2000 patients allocated to standard of care (RECOVERY), tocilizumab improved survival and other clinical outcomes: 596 (29%) in the tocilizumab group and 694 (33%) in the usual care group [most of the patients (82%) in both groups received systemic steroids] died within 28 days.25,27 Although the difference mentioned above has reached statistical significance, the absolute mortality risk reduction was rather low. Because of these data, the National Institute of Health is now recommending tocilizumab as a single intravenous dose in combination with dexamethasone in patients who are exhibiting rapid respiratory decompensation because of COVID-19 if not longer than 3 days hospitalized, not longer than 24 h at the intensive care unit (ICU) and who require ventilation assistance; or who were recently hospitalized but not admitted to the ICU who have rapidly increasing oxygen needs and require non-invasive ventilation or high-flow nasal cannula (HFNC) oxygen and who have significantly increased markers of inflammation [C-reactive protein (CRP) ⩾75 mg/l].
The blockade of IL-1 has been shown to be effective in several immune-mediated disorders characterized by the development of a cytokine storm 28 and has been reported to be successful in improving survival based in COVID-19 on retrospective cohort studies.29–33 Furthermore, one placebo-controlled trial with 594 patients with a median disease duration of 9 days demonstrated efficacy of anakinra superior to placebo. 34 Therefore, Anakinra is approved for COIVD-pneumonia since December 2021.
Thus, based on the pathophysiological observations in COVID-19 and evidence for the efficacy of IL-6 and IL-1 in the hyperinflammatory COVID-19 disease and based on the fact that with the single blockage of one of these cytokines results in a rather small reduction of mortality, our hypothesis was that not all immune activation pathways may be blocked in these patients with severe hyperinflammation comparable with macrophage activation syndromes. Thus, we proposed a hypothesis that a dual blockade of both key cytokines (IL-6 and IL-1) might me associated with a favorable outcome in severe COVID-19 pneumonia as compared with the single blockage of one of these cytokines.
Methods
From May 2020 until April 2021 adult patients, aged ⩾18 years, hospitalized with moderate to severe COVID-19 pneumonia (confirmed by computed tomography – CT), duration of typical symptoms of ⩽10 days, decreased oxygen saturation (⩽95% with at least 4 l oxygen supplementation) and elevated serum markers indicating systemic hyperinflammatory reaction [ferritin, CRP, lactate dehydrogenase (LDH), and IL-6] were identified as potential candidates for a dual cytokine blockade. The presence of COVID-19 was confirmed by quantitative real-time-polymerase chain reaction (RT-PCR). We excluded patients with evidence of bacterial infection, patients with positive procalcitonin (PCT) values, patients with concurrent cardio-pulmonal disorders such as severe chronic obstructive pulmonary disease (COPD), grade III–IV and heart failure of grade II and higher according to the New York Heart Association (NYHA) classification, and patients who were more than 80 years old. All patients were treated in clinical routine and were informed about the off-label character of the therapy in reference to the statement of the Robert Koch Institute regarding therapies in ‘off-label use’ in COVID-19 patients after careful consideration of the individual risk–benefit ratio, and written individual consents about the off-label use were obtained. Written informed consents were also obtained according to an institutional protocol of the Charité (Ethical Committee of Charité Universitätsmedizin Berlin approval no. EA2/072/29). Patients’ information was de-identified for demographic and other patient information. In addition, the equator guideline for case series used (CARE) was followed, and the checklist is added as a Supplementary Material. 35
Laboratory examinations
Routine laboratory investigations were performed on regular base including PCT levels, CRP, ferritin, LDH, full blood count, and IL-6.
In addition, monocytes from four patients were examined for intracellular production of IL-1β, IL-6, and tumor necrosis factor (TNF). For this, thawed peripheral blood mononuclear cells were rested overnight in complete medium at 37°C in a humidified 5% CO2/70% O2 air atmosphere and incubated for additional 5 h in the presence of brefeldin A and monensin. Immunofluorescent staining of intracellular cytokines and cell markers was then performed, followed by flow cytometric analysis of gated CD68+ HLA-DR+ monocytes. Data were acquired on the FACSCanto (Becton Dickinson, BD) and analyzed with FlowJo software version 10.7.1.
Chest CT examinations
All 31 patients underwent chest CT examinations on admission and 16 patients had follow-up CT examinations 1 month after therapy onset. All CT images were reviewed by a single radiologist (JLV) with over 10 years of experience in chest imaging. Findings of COVID-19 pneumonia were assessed according to Radiological Society of North America expert consensus document. 36 A semi-quantitative scoring system was used to evaluate the pulmonary involvement of all these abnormalities on the basis of the area involved and each of the five lung lobes was visually scored on a scale of 0–5, with 0 indicating no involvement; 1, less than 5% involvement; 2, 5–25% involvement; 3, 26–49% involvement; 4, 50–75% involvement; and 5, more than 75% involvement. The total CT score was the sum of the individual lobar scores and ranged from 0 (no involvement) to 25 (maximum involvement). 37 We used the following group cutoffs to aggregate the total CT score: ⩽7 mild, 8-17 moderate, and ⩾18 severe changes. 38 Chronic lung changes were scored subjectively into four groups (absent, mild, moderate, and severe).
Included patients received a monoclonal antibody against IL-6-receptor tocilizumab 8 mg/kg body weight with a maximum dose of 800 mg in patients with more than 100 kg body weight as a single intravenous infusion followed by an IL-1 receptor antagonist anakinra given in a dose of 100–300 mg per day for 3–5 days.
Furthermore, all patients were treated according to the actual valid guidelines, which were modified over time regarding use of remdesivir, corticosteroids, time point of admission to the ICU, and type of ventilation and position of the patients. Therefore, only one patient received additional remdesivir and 19 patients received corticosteroids.
Results
Clinical course
The mean age of our patients was 54 (range = 30–79) years, five female, 26 male, with a mean symptom duration of 7 (range = 3–10) days agreed to be treated so far. Mean oxygen saturation without oxygen supplementation was 83% (range = 40–95%) before initiating therapy. Mean serum ferritin level was 1417 µg/l (range = 52.4–2639), mean IL-6 was 265 ng/l (range = 2.2–2504), and CRP was 107 mg/l (range = 12.0–290.9) (see Table 1).
Baseline characteristics of 31 patients treated with simultaneous interleukin-1 and interleukin-6 blockade.

CRP, C-reactive protein; CT, computed tomography; ECMO, extracorporeal membrane oxygenation; IL, interleukin; LDH, lactate dehydrogenase; WHO, World Health Organization.
All patients received tocilizumab as a single intravenous infusion as described above followed by anakinra 100 mg subcutaneously (sc.) over 3 days in two patients and 300 mg sc. over 5 days in all other patients. In 25 patients, we were able to prevent intubation and mechanic ventilation, 6 patients had to be mechanically ventilated, and of whom 3 patients were on extracorporeal membrane oxygenation (ECMO) ventilation management. Two of our patients died, while on ECMO ventilation. Five patients did not need to be referred to the ICU at all. The mean duration of hospital stay was 18 days (range = 5–73 days), one patient needed oxygen supplementation at the time of discharge, and one patient was referred to an external weaning center.
Laboratory results
CRP levels decreased in 19/31 patients after treatment to normal ranges (<5 mg/l) and in 23/31 patients below 10 mg/l with a mean CRP of 12.9 mg/l (range = 1.8–83.1) at the end of therapy. Also, IL-6 levels, ferritin, and LDH dropped in all patients after treatment (Table 2).
Course of laboratory parameters and chest CT changes in 31 patients with severe COVID-19 pneumonia and therapy with simultaneous interleukin-1 and interleukin-6 blockade.

CRP, C-reactive protein; CT, computed tomography; IL, interleukin; LDH, lactate dehydrogenase; NA, not available.
p < 0.05; **p < 0.005.
After 1 month, CRP levels were measured in 19/31 patients and normalized in 14 of these patients with a mean CRP of 9.6 mg/l (0.3–74.0). Mean IL-6 levels were 16.9 ng/l (1.5–74), mean LDH was 297.7 U/l (155–501), and mean ferritin was 408.4 µg/l (54–1872) (Table 2).
Intracellular cytokine production over time was determined in monocytes from four patients. Levels of IL-6 and IL-1β but not that of TNF decreased during treatment (Figure 1).

Monocyte production of interleukin (IL)-6, IL-1β, and tumor necrosis factor-alpha (TNF-α) as well as serum levels of IL-6 and ferritin over time in four patients before and during treatment with simultaneous tocilizumab and anakinra. Serum levels are given relative to the upper limit of normal ranges.
Chest CT examinations
The mean severity score for active COVID-19 lung changes in the acquired chest CT scans was 15 (range = 7–23, n = 31) at baseline and decreased from 14 (range = 8–20, n = 16) to 6 (range = 0–16, n = 16) 1 month after treatment. Chronic fibrotic changes were absent in any patient at baseline and mild in six patients 1 month after intervention (Figure 2 and Table 2).
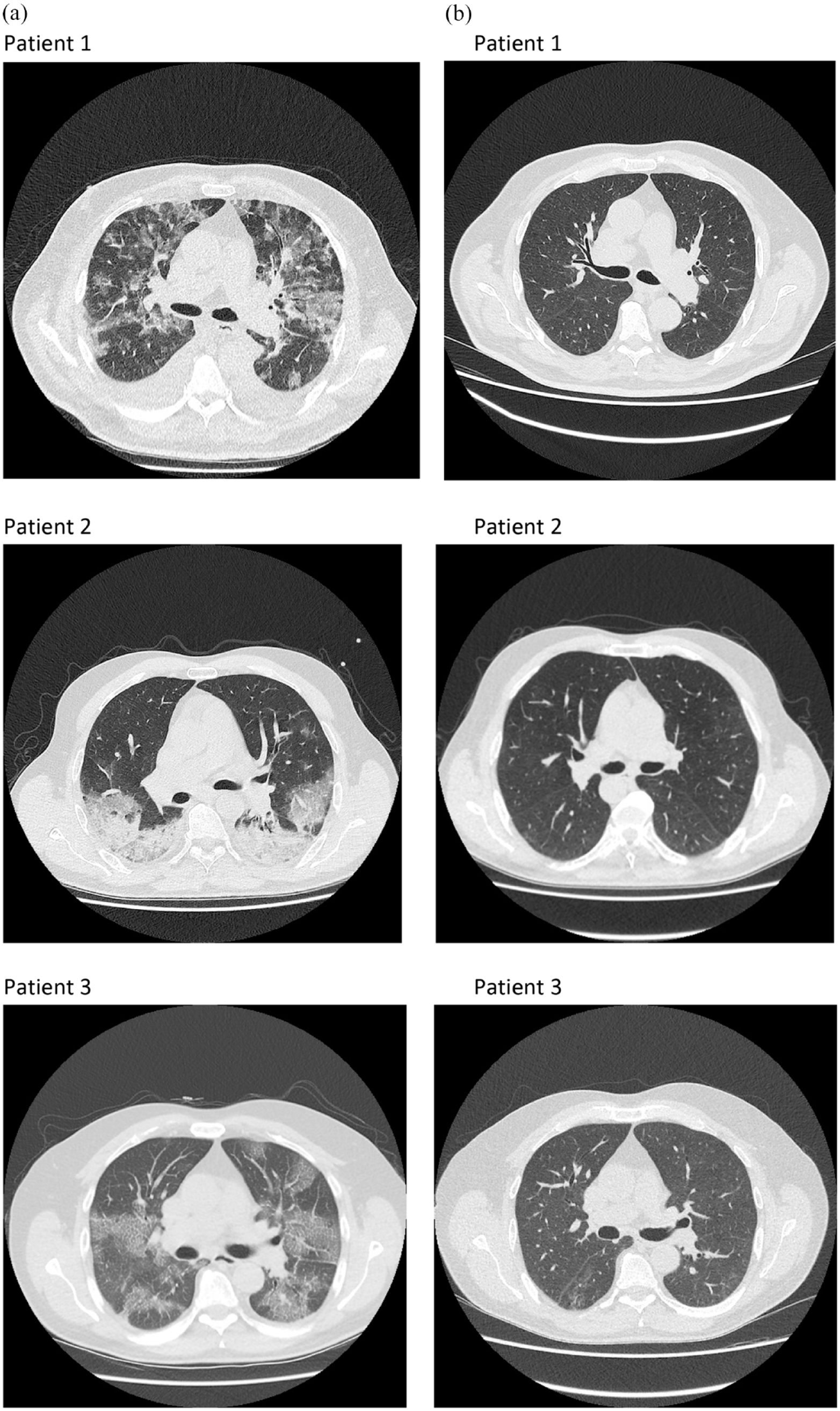
Chest CT of three patients with active COVID-19 infection before (a) and 1 month after treatment (b) with a combination of tocilizumab and anakinra.
Safety
In 21 of 31 patients, no adverse events were noticed, and 2 patients died on ECMO because of bacterial infection and bleeding. The following adverse events were seen in the remaining eight patients: lower abdominal pain, urinary tract infection, and gastrointestinal bleeding because of antrum ulcers Forrest III in one patient, atrial fibrillation, urinary tract infection, and apoplexy in two patients, and in the other six patients, bacterial infections occurred: increased PCT and antibiotic treatment, bacterial pneumonia, and pleural empyema, one with additional fungal lung infection with candida.
Discussion
In this case series, most of our patients treated with both tocilizumab and anakinra recovered mostly fast and sustained. Unfortunately, two patients died while they were treated on ECMO device, one because of bacterial infection and another one because of lung bleeding. Further four cases temporarily needed mechanical ventilation after treatment initiation. This is clearly superior when compared with the large trials with tocilizumab (RemapCap and Recovery).25 –27 In the RemapCap trial, with a quite similar selection of patients, the in-hospital mortality for the tocilizumab treated group was 27% and for the control group 36%, and in the Recovery trial, it was 29% in the tocilizumab group and 33% in the control group. Of note, in both trials more than 80% were additionally treated with corticosteroids. In our case series, only 61% patients were treated with dexamethasone. This was because the guideline for the use of dexamethasone was introduced later in 2020; some of our patients were already treated before.
We selected only patients with short disease duration of ⩽10 days and with evidence of respiratory insufficiency and signs of cytokine storm. Patients with signs of bacterial super-infection (PCT >0.5) were excluded. We hypothesize that early initiation of treatment in patients presenting with signs of hyperinflammation is crucial to disrupt the inflammatory cascade and to prevent severe lung damage. This is supported by histological data from COVID-19 patients showing perivascular CD 4 -T-cell infiltration of the lung tissue in the first days of the disease, while at later stages, alveolar macrophages dominate in the affected area. 39
The studies with tocilizumab and anakinra in COVID-19 as single substance were conducted without restriction of the disease duration that most likely was the reason for the heterogeneous study outcomes.24,40 In the COVACTA trial, the mean days form symptom onset to baseline were 12.1–11.4 days. 23 In one recent trial, patients were included who were treated with tocilizumab, sarilumab, or placebo in addition to steroids within hours after admission to the ICU, which occurred only 1.2 (0.9–2.8) days after being hospitalized. In this study, the hospital mortality was clearly reduced to 28.0% (98/350) for tocilizumab and to 22.2% (10/45) for sarilumab when compared with 35.8% (142/397) in the control group. In the SAVE MORE trial, 34 the 28 days mortality rate was low in both the placebo (6.9%) and the anakinra arm (3.2%) maybe because patients were treated early and were clearly identified to have active disease with soluble urokinase plasminogen activator receptor (suPAR) stratification.
Remarkably, even with IL-6 blockade given on the background of usual care (nowadays, normally including steroids), the mortality in severe COVID-19 disease remains with almost one-third of the patients very high. In our uncontrolled observation, two of the patients (6.4%) died. With all the limitations related to the small numbers and lack of control, the dual cytokine blockade represents a promising approach deserving a test in a randomized controlled trial.
Therapy of simultaneous use of tocilizumab and anakinra was safe. In 21 patients, no side effects were observed. In the 10 patients with adverse events, apoplexy is a typical manifestation of the COVID-19 disease and all but one of infections recovered, indicating that the combination of these two drugs was safe and well tolerated even in combination with corticosteroids in our patients. Therapy on ECMO is often fatal, which was also the case in our two patients who unfortunately progressed to ECMO. These two patients were male, suffered from co-morbidities, especially from severe adipositas.
The applied therapy was associated with reduction of systemic markers of inflammation as well as of inflammation detected by CT. This could be also measured by immediate decrease intracellular of IL-6 and IL-1β cytokine production in monocytes in four patients. The chest CT severity score was reduced by more than 60% 1 month after treatment. Also a subjective score for chronic pulmonary changes indicated that nearly no chronic changes occurred in the treated patients after 1 month, only in 6 of 16 patients mild fibrotic changes were found.
The median 12 days of stay in the hospital in our group was clearly less than what was published for a similar group of patients with severe course of the disease but without treatment in Germany, which was 18.1 days for patients with non-invasive ventilation 41 and less than in the COVACTA Study in which the median time to hospital discharge was 20–28 days in the survivors. 23 In our group, patients experienced a longer hospitalization because of stroke, bacterial infection, and because of rehabilitation.
Our case series was rather small, and we treated off label. The strength of the case series was that we treated consecutively a homogeneous, well-characterized group of patients with moderate to severe COVID-19 pneumonia. Ín 15 patients, we also had CT follow-up investigations as another objective parameter of therapeutic success of our treatment approach.
In conclusion, therapy with the combination of tocilizumab and anakinra in patients with severe course of COVID-19 presenting with a hyperinflammatory syndrome resulted in fast and sustained improvement of signs, symptoms, and markers of inflammations and prevented most of these patients from fibrotic changes in the lung as seen by CT with a favorable safety profile. Larger controlled trials should be conducted to confirm the role of dual cytokine blockade in severe COVID-19 disease.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221116405 – Supplemental material for Successful treatment of severe COVID-19 pneumonia, a case series with simultaneous interleukin-1 and interleukin-6 blockade with 1-month follow-up
Supplemental material, sj-docx-1-tab-10.1177_1759720X221116405 for Successful treatment of severe COVID-19 pneumonia, a case series with simultaneous interleukin-1 and interleukin-6 blockade with 1-month follow-up by Hildrun Haibel, Denis Poddubnyy, Stefan Angermair, Kristina Allers, Janis L. Vahldiek, Michael Schumann and Thomas Schneider in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
The authors thank the patients who participated, the staff on the COVID-19 wards and the colleagues on the ICU (Michaela Danassis and Anna Maria Nothnagel).
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
