Abstract
Background:
We aimed to evaluate the efficacy and safety of biologic agents targeting three main cytokines, that is, nerve growth factor (NGF), interleukin-1 (IL-1), and tumor necrosis factor-α (TNF-α), for osteoarthritis (OA) treatment.
Methods:
Databases (PubMed, Embase, and Cochrane Library) and ClinicalTrials.gov were systematically searched for randomized placebo-controlled trials (RCTs) of biologic agents from inception to November 15, 2020. The outcomes were the mean change in pain, function scores, and the risk of adverse effects (AEs).
Results:
Out of the 28 studies with 29 RCTs (8555 individuals) included, biologic agents were superior to placebo in pain relief (standardized mean difference [SMD] = 0.28, 95% confidence interval [CI] = 0.17–0.38, p < 0.001) and function improvement (SMD = 0.30, 95% CI = 0.18–0.43, p < 0.001). The incidence of any AEs (risk ratio [RR] = 1.09, 95% CI = 1.05–1.14, p < 0.001) and discontinuations due to AEs (RR = 1.39, 95% CI = 1.05–1.83, p = 0.021) were higher following treatment with biologic agents while no significant difference was found in serious AEs. Subgroup analyses showed that NGF inhibitors provided superior pain relief (SMD = 0.36, 95% CI = 0.26–0.47, p < 0.001) and function improvement (SMD = 0.41, 95% CI = 0.30–0.51, p < 0.001), whereas IL-1 inhibitors and TNF-α inhibitors did not. Meanwhile, NGF inhibitors increased the incidence of any AEs (RR = 1.12, 95% CI = 1.07–1.17, p < 0.001) and discontinuations due to AEs (RR = 1.48, 95% CI = 1.07–2.06, p = 0.018). IL-1 inhibitors and TNF-α inhibitors showed no difference in safety compared with placebo.
Conclusions:
The efficacy and safety of biologic agents vary by mechanism of action. NGF inhibitors can relieve OA-related pain and improve function but involve safety concerns. IL-1 inhibitors and TNF-α inhibitors are relatively safe options but with limited efficacy.
Introduction
Osteoarthritis (OA), the leading cause of pain and disability globally, is characterized as a whole joint disease involving cartilage, subchondral bone, and synovium.1,2 OA is a complex chronic disorder that most commonly affects the knee, followed by the hand and hip.1,2 With the combined impact of population aging and rise of obesity,2,3 the number of patients with OA worldwide has risen by 48% from 1990 to 2019 3 and is expected to increase continuously over the coming years, which seriously reduces patients’ quality of life and intensifies socioeconomic costs.4–6 The current pharmaceutical treatments for patients with OA, such as nonsteroidal anti-inflammatory drugs (NSAIDs), acetaminophen, duloxetine, and intra-articular glucocorticoid injection, can be effective but also have substantial limitations.7,8 For example, NSAIDs exhibit a small effect size in some patients and involve great safety concerns in prolonged use including NSAID-induced gastrointestinal toxic effects, especially in elderly individuals.9–11 Therefore, it is urgent to identify a novel therapeutic medication to treat this debilitating condition.
Biologic agents have achieved distinct effects in the treatment of rheumatic disorders such as rheumatoid arthritis (RA).12–14 This successful treatment approach has greatly encouraged the conduct of randomized controlled trials (RCTs) on biologic agents in OA. Biotherapeutic strategy as such has been used to treat OA by modulating or inhibiting the effects of major cytokines, which is of a similar mechanism to the successful strategy for the treatment of RA. 15 There were three main types of cytokine blockers used in OA, targeting the nerve growth factor (NGF), interleukin-1 (IL-1), and the tumor necrosis factor-α (TNF-α), respectively. These cytokines are all involved in pain pathways of OA. Specifically, TNF-α, IL-1, and NGF can modulate pain via nociceptor sensitization.16,17 In particular, the expression of NGF can be induced by the upregulation of IL-1 and TNF-α in the case of OA.18,19 The understanding of the cytokine network associated with the pathogenesis of OA has enhanced the rationale of studies exploring whether this biotherapeutic approach has an effect on symptom improvement.
Controversy concerning the efficacy and safety of biologic agents in OA remains in the existing literature, which presents mixed outcomes of success20–22 and failure.23–25 Several meta-analyses indicated that NGF inhibitors had shown effects of pain relief and function improvement relative to placebo in OA but with inconsistent safety performance.26–29 Contrary to the results of NGF inhibitors, two meta-analyses evaluated the efficacy of IL-1 inhibitors and TNF-α inhibitors, and ended up with conclusions of ineffectiveness for OA.30,31 Nevertheless, despite the increase of relevant clinical trials, no comprehensive meta-analysis has been undertaken to date to evaluate the efficacy and safety of these three main cytokine blockers in the treatment of OA. Therefore, the present meta-analysis was intended to examine the efficacy and safety of biologic agents based on the current RCTs investigating NGF inhibitors, IL-1 inhibitors, and TNF-α inhibitors.
Methods
Search strategy
We systematically searched the databases PubMed, Embase, and Cochrane Library for RCTs involving biologic agents in the treatment of OA from inception to November 15, 2020. As three main types of cytokine blockers, these biologic agents target NGF, IL-1, or TNF-α. Additional relevant trials were retrieved through ClinicalTrials.gov. There were no restrictions on language and publication date. The details of the search strategy are available in Supplementary Table 1. The references of the identified articles and previous review articles were manually searched to avoid omitting other related studies.
Selection criteria
The inclusion criteria were as follows: (1) population: adult patients diagnosed with OA of knee, hip, or hand; (2) intervention: intravenous, subcutaneous, or intra-articular administration of biologic agents; (3) comparator: only placebo acting as a control group; (4) outcomes: mean change from baseline in pain and function scores, incidence of any adverse events (AEs) and serious AEs, and incidence of discontinuations due to AEs; (5) study design: either full texts or abstracts of RCTs containing available data.
Exclusion criteria included case reports, letters, editorials, reviews, conference abstracts with unavailable indicators and other unrelated studies. Trials without placebo controls were excluded. In addition, if the intervention or control group of a trial was in combination with NSAIDs or other analgesics, the trial would be excluded. When there were several articles from the same study, only the most recent, complete, and relevant study was included to avoid duplication.
Data extraction
F.M. and H.L. independently screened each record in strict accordance with the inclusion and exclusion criteria using NoteExpress 3.3.0 software. F.M. and H.L. extracted data from eligible studies independently, including study name, author, year, publication type, study design, participant characteristics, the studied pain condition, intervention details, duration of follow-up, and outcome measures. In case of disagreement, a third reviewer would be consulted until reaching consensus. The data of mean change, standard error, standard deviation, and 95% confidence interval (CI) in tables and texts were obtained directly from the literature. For articles showing graphic results, the GetData Graph Digitizer software version 2.22 was used to extract the data. For crossover trials, data were only extracted from the first period. If multiple intervention doses were present in a trial, subgroups would be combined into one group for analysis. The scale with the highest sensitivity to change would be used in case of multiple pain scales reported in a study. 32 The function subscale of Western Ontario and McMaster Universities Arthritis Index (WOMAC) was used for the assessment of functional improvement. If a study did not measure or report WOMAC function, the function subscale of Australian/Canadian Osteoarthritis Hand Index score (AUSCAN) or one of the other functional measurement scales was used instead.
Quality assessment
F.M. and H.F. independently assessed the risk of bias of the included trials using Cochrane’s risk of bias tool, covering sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective outcome reporting and other sources of bias. 33 Each source of bias was rated as low risk of bias, high risk of bias or unclear risk of bias.
Statistical analysis
This study was registered on PROSPERO (CRD42021246922). Stata 12.0 software (Stata Corporation, College Station, TX, USA) was used for statistical analysis. In meta-analysis, continuous variables were represented by standardized mean difference (SMD), and binary data was expressed by risk ratio (RR). Both SMD and RR were reported along with 95% CI. The heterogeneity was reported using the Cochrane Q test 34 and the inconsistency index value (I²). 35 According to the size of heterogeneity, the pooled effects and their respective 95% CIs were calculated using fixed-effects or random-effects models. When I² value was less than 50%, we used a fixed-effects model. On the contrary, a random-effects model would be used. Funnel plots and Egger’s regression tests were used to detect publication bias. If funnel plot indicated asymmetry by Egger’s regression test, a trim and fill analysis would be conducted. A p value less than 0.05 was considered statistically significant.
Subgroup analyses were conducted in terms of the target of action (NGF, IL-1, or TNF-α). A sensitivity analysis was performed to evaluate the impacts of any single study on the pooled outcomes.
Results
Study selection
After an exhaustive literature search, a total of 1499 articles from databases and 65 studies from registers were preliminarily identified (Figure 1), from which 664 studies were removed due to duplication and 758 articles were excluded by reviewing titles and abstracts. After reading the full texts of the remaining articles, another 114 articles were excluded for the reasons of (1) conference abstract data duplicated or not extractable (n = 103); (2) no pain or clinical outcomes (n = 2); (3) no control group (n = 3); 4) control group that is not a placebo (n = 4); or (5) the intervention or control group in combination with NSAIDs or other analgesics (n = 2). Ultimately, 28 eligible studies comparing biologic agents with placebo in patients with OA were included in this meta-analysis.

Flow chart of the literature search and study selection.
Study characteristics
The baseline characteristics of the included studies are summarized in Table 1. Twenty-eight eligible studies (26 full texts and 2 abstracts) included 29 trials (one study 36 contained two trials), which were all randomized, double-blinded, and placebo-controlled. Except for one crossover trial, 37 the others were all parallel trials. For the two abstracts, data were obtained through ClinicalTrials.gov. Eventually, a total of 8555 individuals clinically or radiographically diagnosed with OA were included in this meta-analysis. All the included articles were published in English, between 2009 and 2020.
Baseline characteristics of studies included in the meta-analysis.

IA, intra-articular; IL-1, interleukin-1; IV, intravenous; MP, methylprednisolone; NA, data not available; NGF, nerve growth factor; RCT, randomized controlled trial; SC, subcutaneous; TNF-α, tumor necrosis factor-α.
Risk of bias assessment of included trials
According to the Cochrane Handbook for Systematic Reviews of Interventions (version 5.1.0), we assessed the risk of bias of each included study, and the details are shown in Figure 2. As for random sequence generation, 16 studies were rated as low risk of bias. The allocation concealment in 11 studies was not illustrated in detail. As for blinding of participants and personnel, all of the studies were rated as low risk of bias. The details of blinding of outcome assessment could not be adequately obtained from eight studies, which were therefore rated as unclear risk of bias. As for attrition bias and reporting bias, two and five studies were considered unclear risk, respectively. Other sources of bias were unclear in most of included studies (24/28).

Risk of bias assessment of included studies.
Efficacy of biologic agents in OA
Pain
A total of 24 RCTs including 7383 participants diagnosed with knee, hip, or hand OA reported the mean change from baseline in pain scores. Overall, biologic agents appeared to be statistically superior to placebo with regard to pain relief (SMD = 0.28, 95% CI = 0.17–0.38, p < 0.001, I2 = 71.2%) (Figure 3). Subgroup analyses against the mechanism of action demonstrated that NGF inhibitors (SMD = 0.36, 95% CI = 0.26–0.47, p < 0.001, I2 = 68.5%) were significantly superior to placebo in pain relief. On the contrary, both IL-1 inhibitors (SMD = −0.12, 95% CI = −0.45 to 0.21, p = 0.481, I2 = 75.0%) and TNF-α inhibitors (SMD = 0.24, 95% CI = −0.00 to 0.49, p = 0.050, I2 = 3.9%) were found no statistical significance in pain relief. Details of subgroup analyses are presented in Table 2 and Figure 4.

Forest plot for pain improvement of biologic agents compared with placebo in OA.

Forest plot for subgroup analyses in the improvement of pain conducted in accordance with mechanism of action. NGF = nerve growth factor; IL-1 = interleukin-1; TNF-α = tumor necrosis factor-α.
Results of overall and subgroup meta-analysis for the efficacy and safety of biologic agents in OA.
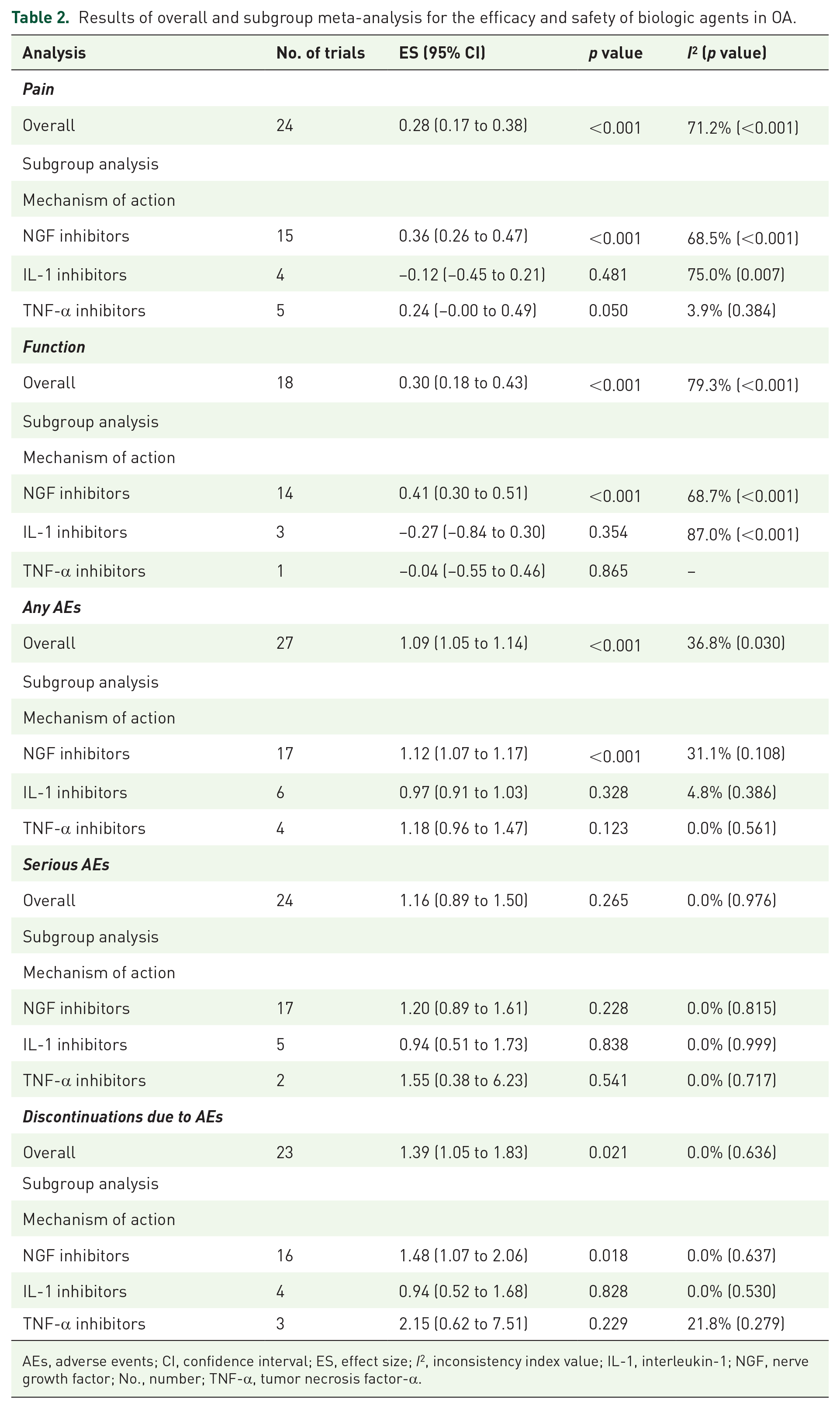
AEs, adverse events; CI, confidence interval; ES, effect size; I2, inconsistency index value; IL-1, interleukin-1; NGF, nerve growth factor; No., number; TNF-α, tumor necrosis factor-α.
Function. Data from 18 RCTs were pooled to evaluate the efficacy of biologic agents in function improvement. The results indicated that when compared with placebo, biologic agents achieved a significant improvement in terms of function scores (SMD = 0.30, 95% CI = 0.18–0.43, p < 0.001, I2 = 79.3%) (Figure 5). Specifically, NGF inhibitors were statistically superior to placebo (SMD = 0.41, 95% CI = 0.30–0.51, p < 0.001, I2 = 68.7%), but TNF-α inhibitors (SMD = −0.04, 95% CI =−0.55 to 0.46, p = 0.865) and IL-1 inhibitors (SMD = −0.27, 95% CI = −0.84 to 0.30, p = 0.354, I2 = 87.0%) were ineffective (Figure 6). The results of overall and subgroup meta-analyses for the function improvement of biologic agents in OA are shown in Table 2.

Forest plot for function improvement of biologic agents compared with placebo in OA.

Forest plot for subgroup analyses in the improvement of function conducted in accordance with mechanism of action. NGF = nerve growth factor; IL-1 = interleukin-1; TNF-α = tumor necrosis factor-α.
Safety of biologic agents in OA
Any AEs
All the included trials provided data on the incidence of any AEs. Nevertheless, one of them was a crossover randomized trial and was excluded on account of the data combining both treatment periods. Among any AEs, abnormal peripheral sensation, musculoskeletal and connective tissue disorders, gastrointestinal disorders, and infections were commonly observed in patients treated with anti-NGFs. Injection site reactions, neutropenia, and infections were more frequent with OA patients treated with IL-1 inhibitors. More subjects in the treatment of TNF-α inhibitors had injection site reactions and infections (Supplementary Table 2). Intravenous and subcutaneous administrations of biologic agents were generally similar in the incidence of AEs. The most common AE was musculoskeletal and connective tissue disorders such as arthralgia in patients treated with intra-articular IL-1 and TNF-α inhibitors injection. Overall, the incidence of any AEs was significantly different between biologic agents and placebo (RR = 1.09, 95% CI = 1.05–1.14, p < 0.001, I2 = 36.8%) (Figure 7). Subgroup analyses revealed that the incidence of any AEs of NGF inhibitors was significantly higher than that of placebo (RR = 1.12, 95% CI = 1.07–1.17, p < 0.001, I2 = 31.1%). On the contrary, there was no significant difference in any AEs compared TNF-α inhibitors (RR = 1.18, 95% CI = 0.96–1.47, p = 0.123, I2 = 0.0%) and IL-1 inhibitors (RR = 0.97, 95% CI = 0.91–1.03, p = 0.328, I2 = 4.8%) with placebo (Supplementary Figure 1).
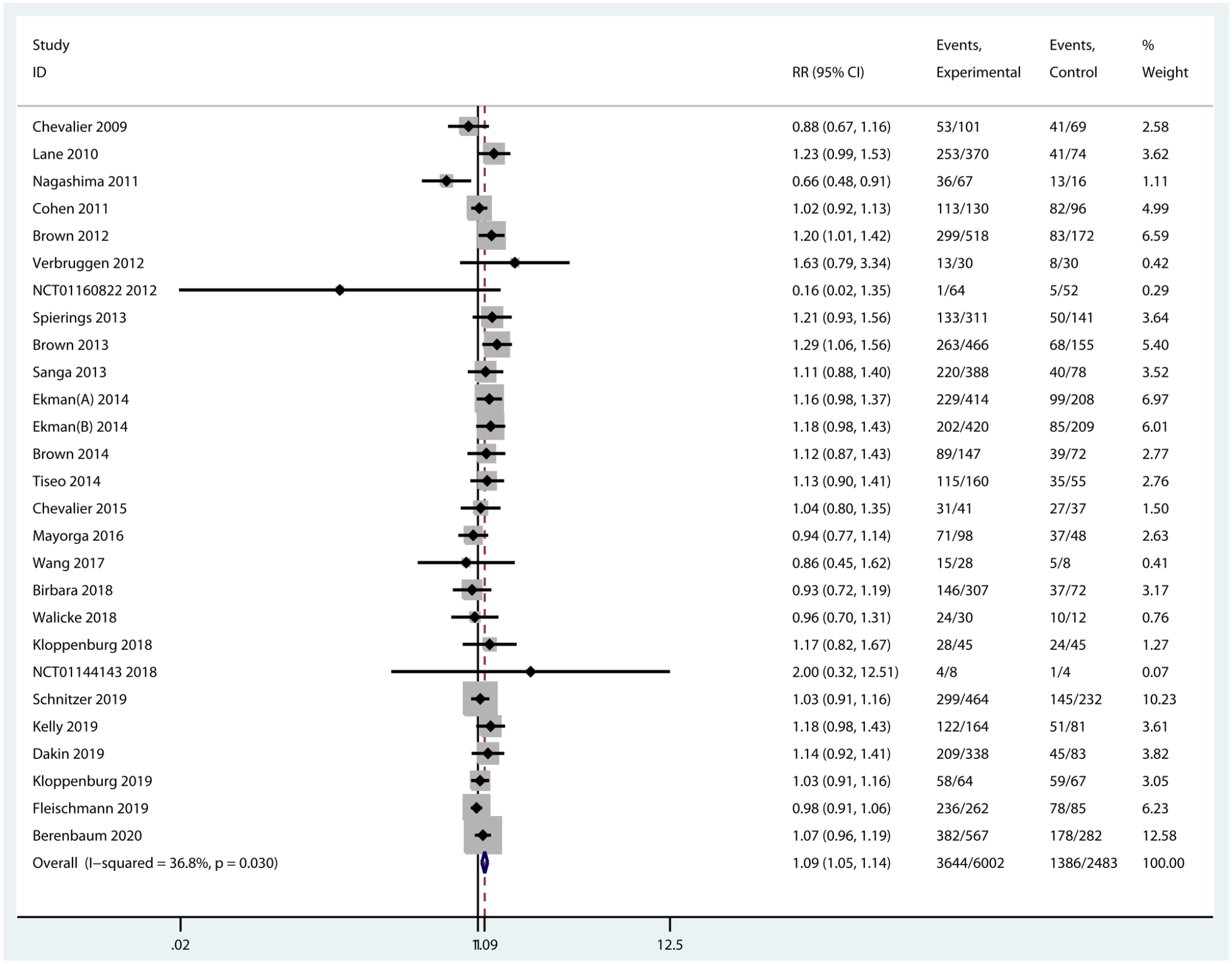
Forest plot for any AEs of biologic agents compared with placebo in OA.
Serious AEs
An AE, which was life-threatening, disabling, leading to hospitalization or death, or leading to a birth defect or congenital anomaly, was classified as a serious AE. Musculoskeletal and connective tissue disorders, infections, and gastrointestinal disorders were the most common serious AEs in subjects treated with NGF blockers. Serious infections were observed in RCTs of agents targeting IL-1 and few serious complications occurred in TNF antagonist therapy. Notably, no significant difference was found between biologic agents and placebo in terms of the incidence of serious AEs (RR = 1.16, 95% CI = 0.89–1.50, p = 0.265, I2 = 0.0%) (Figure 8). Compared with placebo, all the three types of cytokine blockers were not associated with any significantly increased incidence of serious AEs (Supplementary Figure 1). Moreover, no obvious difference was observed in serious AEs of biologic agents in different routes of administration.

Forest plot for serious AEs of biologic agents compared with placebo in OA.
Discontinuations due to AEs
The number of discontinued patients due to AEs was extracted from 23 RCTs with the data available. The incidence of discontinuations due to AEs was statistically higher in experimental groups than that in the control group (RR = 1.39, 95% CI = 1.05–1.83, p = 0.021, I2 = 0.0%) (Figure 9). Specifically, the incidence of discontinuations of NGF inhibitors was significantly increased compared with placebo (RR = 1.48, 95% CI = 1.07–2.06, p = 0.018, I2 = 0.0%). Nevertheless, differences in discontinuations due to AEs were not significant between the other two inhibitors with placebo (Supplementary Figure 1).
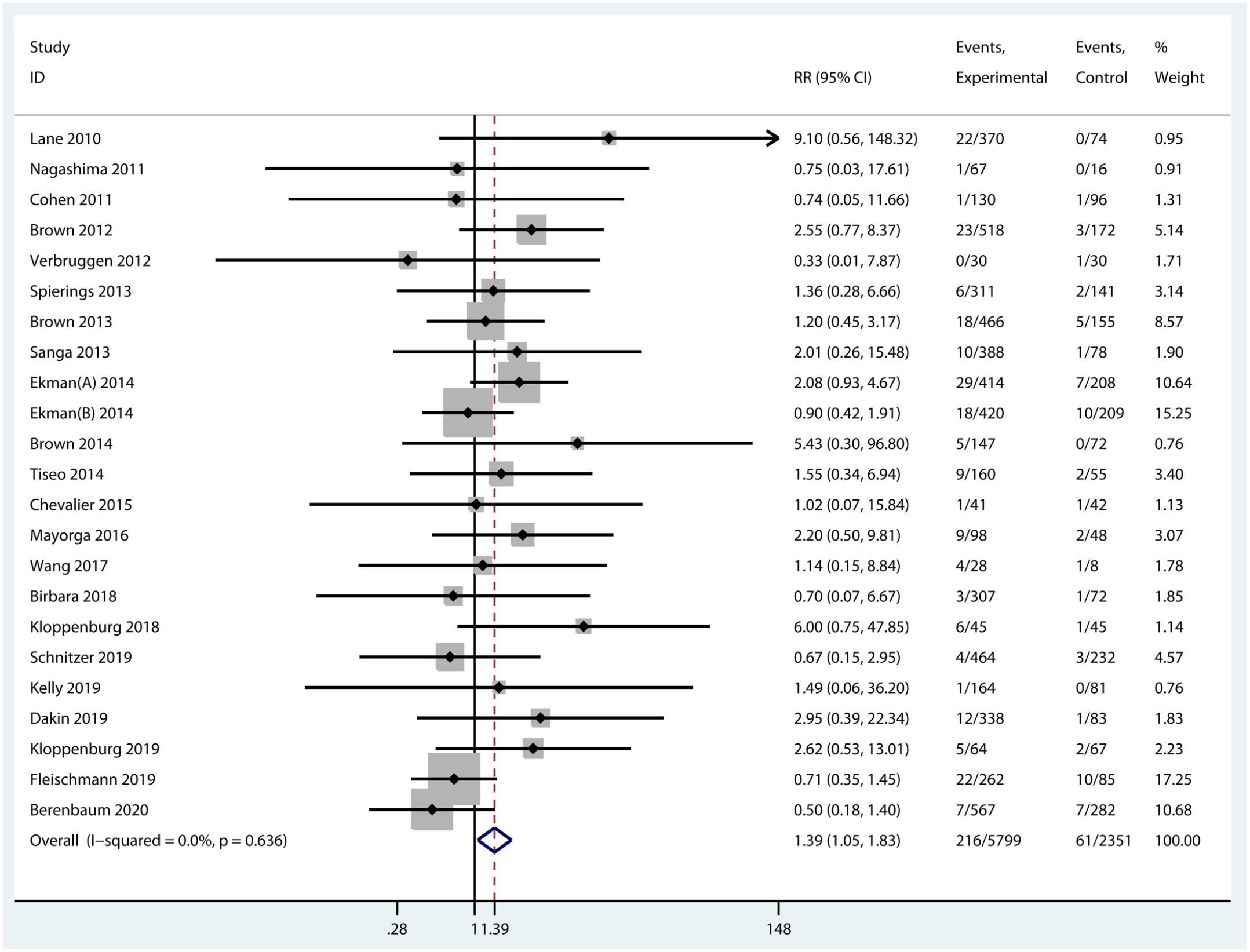
Forest plot for discontinuations of biologic agents due to AEs compared with placebo in OA.
Sensitivity analyses and publication bias
Sensitivity analyses were conducted to examine the influence of a single study on the pooled effects. After removing each individual study, the overall effect of each main outcome did not change statistically. All funnel plots showed no asymmetry by Egger’s regression tests (Supplementary Figure 2).
Discussion
This meta-analysis comprehensively investigated the efficacy and safety of biologic agents including NGF inhibitors, IL-1 inhibitors, and TNF-α inhibitors in patients with OA. The pooled results indicated that biologic agents were significantly superior to placebo in pain relief and function improvement with a higher incidence of any AEs and discontinuations due to AEs. Besides, NGF inhibitors had significant effects in pain relief and function improvement and were associated with higher risk of any AEs and withdrawals due to AEs. On the contrary, both IL-1 inhibitors and TNF-α inhibitors showed no difference compared with placebo in terms of efficacy and safety.
Comparisons with other studies
Several meta-analyses have been reported to evaluate the efficacy and safety of NGF inhibitors in OA.27–29,59–63 However, half of them only focused on tanezumab.28,29,60,61 Tanezumab demonstrated superiority in pain relief and function improvement compared with placebo, which was consistent with the results of this meta-analysis. The discovery of safety varied according to tanezumab dose explored. Yu et al. 28 evaluated the safety of low-dose tanezumab and no significant difference was found in terms of withdrawal due to AEs. Fan et al. 29 explored the safety of tanezumab administered as a fixed dosing regimen and there was significant difference in serious AEs compared with placebo. The results of safety for tanezumab in the other two studies were the same as this meta-analysis.60,61 In addition, there were four meta-analyses including all three NGF inhibitors, tanezumab, fulranumab, and fasinumab.27,59,62,63 Like the studies of tanezumab above, the efficacy results of anti-NGFs were consistent with this meta-analysis, but the safety findings were not absolutely the same. Among them, the article conducted by Yang et al. 27 indicated that pooled differences of AEs rates between experimental and control groups were not significant. However, it only included six OA trials and omitted several trials eligible for their inclusion criteria. 64 Schnitzer et al. published a meta-analysis in 2015 and found that safety, determined by odds ratios of withdrawals due to AEs, at the lower doses was better than higher doses and appeared similar to placebo. This study included 13 RCTs to evaluate the efficacy and safety of NGF inhibitors in the treatment of hip and knee OA. 59 Ever since Schnitzer et al.’s work, a certain number of high-quality RCTs investigating monoclonal NGF antibodies in the treatment of OA have been published.26,44,47,50,53-55,58 The network meta-analysis conducted by Cao et al. 62 compared the efficacy and safety of the anti-NGF antibody with NSAIDs and opioids in the treatment of OA and found that anti-NGFs were not associated with higher withdrawal rates related to AEs. But only nine RCTs of NGF inhibitors were included due to particular exclusion criteria which was dose-escalation studies of a single drug. Similarly, another meta-analysis conducted by Seah et al. 63 evaluated the effectiveness of NGF inhibitors in the treatment of hip and knee OA, and only included 13 eligible studies. This study found that anti-NGFs were not associated with higher incidence of serious AEs but were associated with significant increase of discontinuation due to AEs. As for IL-1 inhibitors and TNF-α inhibitors, there were two published studies evaluating the efficacy and safety of these two in OA.30,31 Persson et al. 30 reported that the efficacy of biologic disease-modifying anti-rheumatic drugs, IL-1 inhibitors and TNF-α inhibitors, was not superior to placebo in the treatment of OA. It is noteworthy that this meta-analysis included six related RCTs and contained four kinds of IL-1 inhibitors and TNF-α inhibitors (adalimumab, etanercept, anakinra, and infliximab) without inclusion of the other three inhibitors (i.e. lutikizumab, AMG 108, and canakinumab). Another meta-analysis conducted by Cao et al. 31 only evaluated the efficacy and safety of lutikizumab, an anti–IL-1α/β dual-variable domain immunoglobulin, and only included two RCTs of lutikizumab. Lutikizumab showed no improvement either in pain or function, but was of fine tolerance for patients with OA. Both of the two meta-analyses above did not include clinical trials investigating any kinds of IL-1 inhibitors and TNF-α inhibitors and only involved several blockers.
To update the evidence of biologic agents in the treatment of OA, we included 29 RCTs covering tanezumab (12 trials), fulranumab (4 trials), fasinumab (2 trials), adalimumab (3 trials), etanercept (1 trial), infliximab (1 trial), anakinra (1 trial), lutikizumab (3 trials), AMG 108 (1 trial), and canakinumab (1 trial) in our meta-analysis. Compared with previous studies which were either narrative review or meta-analysis only involving a few biological agents, the present work comprehensively evaluated the efficacy and safety of three main biologic agents targeting IL-1, TNF-α, and NGF. Meanwhile, considering that all the included trials are double-blinded randomized placebo-controlled trials, our subgroup analyses on the mechanism of action could provide indirect comparisons for these three main biologic agents.
Possible explanations
Although the pooled results indicated that biologic agents provided statistically significant effects in pain relief and function improvement, subgroup analyses on the mechanism of action showed that, except for NGF inhibitors, IL-1 inhibitors, and TNF-α inhibitors were found no statistical significance. These promising results of NGF inhibitors suggested that NGF played a significant role in pain pathways which had been confirmed by several experimental studies.16,65–67 Different from NGF inhibitors addressing the mechanisms of pain in a nonspecific way, IL-1 and TNF-α blockers target pro-inflammatory cytokines directly involved in the catabolic and anabolic of cartilage.68,69 However, taking into account the rapid clearance and short half-life of cytokine blockers, such disappointing results may be attributed to the insufficient drug exposure in the affected joint. 38 In consideration of the location of disease, hand OA often affects multiple joints compared with OA of the weight-bearing joints such as the knees and hips. We specially conducted a subgroup analysis for the treatment of hand OA with IL-1 and TNF-α blockers, and the results indicated that there was no significant difference in the efficacy and safety of the two blockers compared with placebo (Supplementary Figures 3 and 4). However, in several studies of patients with erosive hand OA, daily subcutaneous injections of 100 mg of anakinra improved pain after 3 months of treatment, 70 and subcutaneous treatment with 40 mg of adalimumab every 2 weeks for 1 year significantly decreased the number of new erosions in the patient subset with clinical interphalangeal joint swelling at baseline. 24 These promising studies implied that the efficacy of IL-1 and TNF inhibitors may depend on the inflammatory phenotype of OA. 15 In a trial of IL-1 inhibitors conducted by Schieker et al., 71 canakinumab can reduce the consequences of large joint OA such as total hip or knee replacement and were with less OA-related AEs. Such promising findings from the exploratory analysis of this RCT encouraged investigation of IL-1 blockers in OA treatment. Furthermore, the ratio of endogenous IL-1 receptor antagonist to IL-1β was high in the synovial fluid so that the effect of exogenous IL-1 inhibitors was limited. 72 The interaction between IL-1 and TNF-α in OA suggested that inhibitors targeting both of them simultaneously might be needed to effectively reduce the expression of matrix metalloproteinases and aggrecanases. 73 Nevertheless, serious adverse reactions including infection forced researchers to discreetly consider this strategy. 74
With respect to the safety of biologic agents, further subgroup analyses showed that NGF inhibitors had a higher incidence of any AEs and discontinuations due to AEs compared with the placebo group, whereas the other two blockers did not. Although NGF inhibitors have distinct effects in OA-related pain relief and function improvement, the US Food and Drug Administration (FDA) suspended all related trials until 2012 due to rapid progressive OA which happened in patients using NGF inhibitors and was in relation to dose and the combination of NSAIDs.75,76 The safety of subcutaneous tanezumab injection in patients with OA pain was generally similar to intravenous administration in terms of the incidence of rapid progressive OA.21,26,42,53 The tanezumab Adjudication Committee reviewed joint-related AEs in 249 of 386 patients and 68 of which were classified as rapid progressive OA in total 9810 patients treated with tanezumab monotherapy or tanezumab combined with NSAIDs. 77 Furthermore, a total of 18 of 88 patients reported as joint-related AEs were classified as rapid progressive OA among 1353 participants treated in the nine phase I and II studies of fulranumab. 78 Recently, the marketing application of tanezumab, an NGF antibody, for OA had been rejected by FDA in consideration of safety. 79 Therefore, the safety of NGF inhibitors reveals a demand for further investigations and great cautiousness in the upcoming trials. Contrary to NGF inhibitors, IL-1 inhibitors and TNF-α inhibitors were of favorable tolerance in the treatment of OA, but attention should still be paid to the risk of infection, even if there are no safety concerns.
Limitations
There were several limitations in this study. First, this study could not provide direct comparisons among the three targeted therapies, and a network meta-analysis was failed to be conducted due to lack of direct comparisons. However, as mentioned above, we retrieved all available data from double-blinded randomized placebo-controlled trials and most of the included studies were of high quality, which could provide indirect comparisons for their efficacy and safety. Second, in consideration of complete evidence capture, conference abstracts that were lack of stricter peer review were included as full texts in this study. Unfortunately, thorough examination of the method and critical assessment of the risk of bias were not allowed in conference abstracts. Third, as one of the factors affecting the efficacy and safety of biological agents, the potential effect of dose of injection was not examined, due to different divisions of dose subgroups in RCTs. A meta-analysis of tanezumab indicated that low-dose tanezumab (10 or 25 μg/kg and 2.5 mg) had similar effects in pain relief and function improvement and caused a lower incidence of AEs in OA. 61 Fourth, maintaining a steady drug concentration is important for the effectiveness of the medications and hence the lack of analysis for the effect of treatment duration on the efficacy of biologic agents was another limitation. Finally, all the 14 trials of tanezumab were sponsored by a pharmaceutical company (Pfizer Inc., New York, NY, USA), which might lead to overestimation of the effect of biologic agents.
Implication for research
Although previous studies and this meta-analysis supported the distinct efficacy of NGF blockers, controversy still exists based on the current evidence in the treatment of OA using anti-NGFs. The high risk of rapid progressive OA has led to marketing application failure. Considering that such AEs have not been observed in trials of other chronic diseases, such as low back pain, 80 whether characteristic and selection of patients in the trials of NGF inhibitors play a role in the incidence of rapid progressive OA deserves further clinical and preclinical studies, and identifying the phenotype of patients who showed significant efficacy without exhibiting a safety concern would be helpful for OA treatments. 81 Moreover, on account of rapid clearance and short half-life of IL-1 inhibitors and TNF-α inhibitors,15,38 it is necessary to build a relatively stable system to allow these two cytokine blockers to stay in the affected joints for a longer time. Nevertheless, due to the lack of efficacy of blockers inhibiting IL-1 and TNF-α, it should also be considered that IL-1 and TNF may not be the right targets for OA treatment.82,83 Deeper understanding of the molecular mechanism of pain in OA is urgently needed in the upcoming studies to develop more effective therapeutic medications. It is worth noting that more and more other cytokines such as IL-6, a cytokine in the synovial fluid, become attractive and promising targets,84–86 and maybe inhibitors targeting them can be novel treatment of OA in the future.
Conclusion
The results of combining all biologic agents showed statistically significant pain relief and function improvement. Specifically, NGF inhibitors provide significant pain relief and function improvement but with nonignorable safety concerns. Although with favorable tolerance, neither IL-1 inhibitors nor TNF-α inhibitors reduce OA-related pain and improve function. The results imply that the efficacy and safety of biologic agents vary by mechanism of action.
Supplemental Material
sj-docx-1-tab-10.1177_1759720X221080377 – Supplemental material for Efficacy and safety of biologic agents for the treatment of osteoarthritis: a meta-analysis of randomized placebo-controlled trials
Supplemental material, sj-docx-1-tab-10.1177_1759720X221080377 for Efficacy and safety of biologic agents for the treatment of osteoarthritis: a meta-analysis of randomized placebo-controlled trials by Fanqiang Meng, Hui Li, Haoran Feng, Huizhong Long, Zidan Yang, Jiatian Li, Yuqing Wang and Dongxing Xie in Therapeutic Advances in Musculoskeletal Disease
Footnotes
Acknowledgements
Everyone who contributed significantly to this study has been listed.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81930071, 81772413), the Key Research and Development Program of Hunan Province (2018SK2072, 2018SK2074), the Hunan Provincial Innovation Key Foundation for Postgraduate (CX20200389), the Central South University’s Innovation Key Foundation for Postgraduate (1053320192237), and the Postgraduate Independent Exploration and Innovation Project of Central South University (2018zzts256).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
