Abstract
Osteoarthritis (OA) is a highly prevalent condition worldwide associated with pain, progressive disability, reduced participation in social activities, and impaired quality of life. Despite its growing burden, the therapeutic options are still limited and almost exclusively addressed to symptoms’ management, while no disease-modifying OA drugs able to prevent or retard disease progression are actually available. For these reasons, in the last decades, relevant efforts to find new potential therapeutic targets in OA have been made and a number of existing conventional and biological disease-modifying anti-rheumatic drugs (DMARDs), including hydroxychloroquine (HCQ), methotrexate (MTX), tumor necrosis factor (TNF)-α, interleukin (IL)-1, and IL-6 inhibitors, commonly used to treat inflammatory rheumatic diseases, have been repurposed for the treatment of OA and explored also in hand osteoarthritis (HOA). The current narrative review was aimed to provide a comprehensive and updated understanding of the possibilities and the criticisms related to the treatment of HOA with conventional and biological DMARDs. Unfortunately, therapy with conventional and biologic drugs in HOA has not achieved the expected success, despite a rationale for their use exists. Thus, our findings outline the urgent need to enhance the exploration of HOA basic molecular mechanisms to find new potential therapeutic targets, personalized for each patient, and appropriate for the different subsets of HOA and for the different phases of disease.
Keywords
Introduction
Osteoarthritis (OA) is a highly common condition worldwide with an estimated prevalence in 2019 of 527.81 million, which has dramatically increased by 113.25% from 1990. 1 Hand osteoarthritis (HOA) was the second more prevalent OA joint localization, after knee, accounting for approximately 23.7% of the total prevalent cases of OA in 2019 and its prevalence increased by 91.84% from 1990 to 2019. 1 HOA consists of several subsets, including nodal HOA, erosive HOA (EHOA), and thumb-base OA; in addition, HOA can affect simultaneously multiple hand joints.2,3 This heterogeneity in the presentation and the variety of the resulted symptoms and signs makes HOA very complex to study.4,5 Indeed, its pathogenetic mechanisms still remain unclear.
The therapeutic options are limited and almost exclusively addressed to symptom management, mainly consisting of topical and oral non-steroidal anti-inflammatory drugs (NSAIDs) that have only small to modest analgesic benefits and several side effects for the oral formulations.5–7 Some positive, but preliminary results in improving pain and functionality were derived from the use in non-EHOA and EHOA of symptomatic slow-acting drugs for osteoarthritis (SYSADOAs), such as prescription-grade glucosamine sulfate (pGS) or prescription-grade chondroitin sulfate (pCS).8–11 Conversely, no disease-modifying OA drugs (DMOADs) that prevent or retard disease progression are actually available, although several clinical trials are currently ongoing.12–15
Traditionally, OA is thought to be a joint disease with damage and loss of cartilage, although in recent years, it has become evident that OA is a more complex disease affecting all tissues within the joint.16,17 Furthermore, especially in EHOA, the importance of local inflammation has been recognized and an important role in the pathogenesis of OA has been attributed to a cytokine imbalance in favor of pro-inflammatory mediators, such as tumor necrosis factor (TNF)-α, interleukin (IL)-1, and IL-6, that initiate a vicious cycle which leads to the activation of catabolic enzymes and a disintegrin-like and metalloproteinase with thrombospondin motif (ADAMTS) and, as final result, to cartilage damage.16,18,19 For these reasons, in the last decades, relevant efforts to find new potential therapeutic targets in OA have been made and novel drugs are currently in the pipeline. Furthermore, a number of existing conventional and biological disease-modifying anti-rheumatic drugs (DMARDs), including hydroxychloroquine (HCQ), methotrexate (MTX), TNF-α, IL-1 and IL-6 inhibitors, commonly used to treat inflammatory arthropathies, have been repurposed for the treatment of OA and explored also in HOA.7,14,20
The aim of the current narrative review is to provide a comprehensive and updated understanding of the possibilities and the criticisms related to the treatment of HOA with conventional and biological DMARDs.
Methods
Data sources and searches
The present narrative review was organized through the ‘Narrative Review Checklist’ proposed by the Academy of Nutrition and Dietetics. 21 The literature search was performed from 1 to 30 June 2022 across PubMed, Cochrane Library, MEDLINE, EMBASE, Web of Science, and Scopus databases using the following search terms: ‘conventional disease modifying anti-rheumatic drugs’, ‘conventional DMARDs’, ‘biological disease modifying anti-rheumatic drugs’, ‘biological DMARDs’, ‘hydroxycloroquine’, ‘methotrexate’, ‘TNFα inhibitors’, ‘etanercept’, ‘adalimumab’, ‘infliximab’, ‘interleukin 1 inhibitors’, ‘anakinra’, ‘canakinumab’, ‘lutikizumab’, ‘interleukin 6 inhibitors’, ‘tocilizumab’ in combination with ‘hand osteoarthritis’, and ‘erosive hand osteoarthritis’. A hand search of the reference lists of studies meeting the inclusion criteria was also performed to identify additional relevant reports. Furthermore, we searched www.clinicaltrials.gov for active or recently completed clinical trials testing conventional or biological DMARDs for HOA.
Inclusion/exclusion criteria
In this narrative review, we included studies evaluating the use of conventional or biological DMARDs for the management of HOA. In particular, articles were considered eligible if they met the following criteria: (1) diagnosis of HOA, according to the American College of Rheumatology (ACR) criteria; 22 (2) studies evaluating symptomatic or disease-modifying effect of conventional or biological DMARDs that are recommended or licensed for rheumatoid arthritis (RA); 23 (3) only randomized controlled trials (RCTs).
Open-label prospective studies with a not randomized controlled design and retrospective studies, as well as review articles, papers not published as a full article (conference abstracts), and articles not totally written in English language were excluded. No limits were set for publication year.
Selection of studies, data extraction, and outcomes
Following the removal of duplicates, all articles were first screened based on their title, keywords, and abstracts by two independent authors (O.B. and J-Y.R.). Then, a full-text evaluation of the selected studies was performed by the same authors (O.B. and J-Y.R.) to determine whether the trials met the inclusion criteria regarding design, study population, and interventions. Disagreement between the two reviewers was solved by involving a third author (N.V.). The relevant data, such as study design, participants’ characteristics, details on the interventions undertaken, types of outcome measures evaluated, duration of follow-up, loss to follow-up, and results, were independently extracted and aggregated into a Microsoft Excel® spreadsheet database by three authors (S.T., A.F., and S.C.). Patient-reported pain and function were considered the main outcomes of interest; possible side effects related to the investigated treatment were also recorded.
Results
Literature search results and trial characteristics
In total, 114 potential eligible studies were found; one additional paper was obtained by hand searching of references. Of these, 6 studies were excluded because they were written in a language other than English and 25 studies were excluded because they were review articles. Based on the title and the abstract content, 55 of these articles were not included in our review. The full texts of the remaining 28 studies were read, and a further 14 studies were excluded because 9 were not RCTs and 5 didn’t meet other inclusion criteria (Figure 1). We identified 14 assessable studies, 5 analyzing the effect of conventional DMARDs treatment in HOA patients, of whom 4 on HCQ and 1 on MTX treatment, and 9 evaluating the efficacy of biologic DMARDs; these included 7 studies dealing with TNF-α inhibitors, 1 with IL-1 antagonists, and 1 with IL-6 inhibitors. The main characteristics of the studies analyzing conventional DMARDs in HOA are summarized in Table 1, while Table 2 shows the relevant features of the trials about biologic DMARDs in HOA. In addition, we identified on ClinicalTrials.gov nine studies, of whom two ongoing and seven completed, but not yet published (main characteristics presented in Table 3).
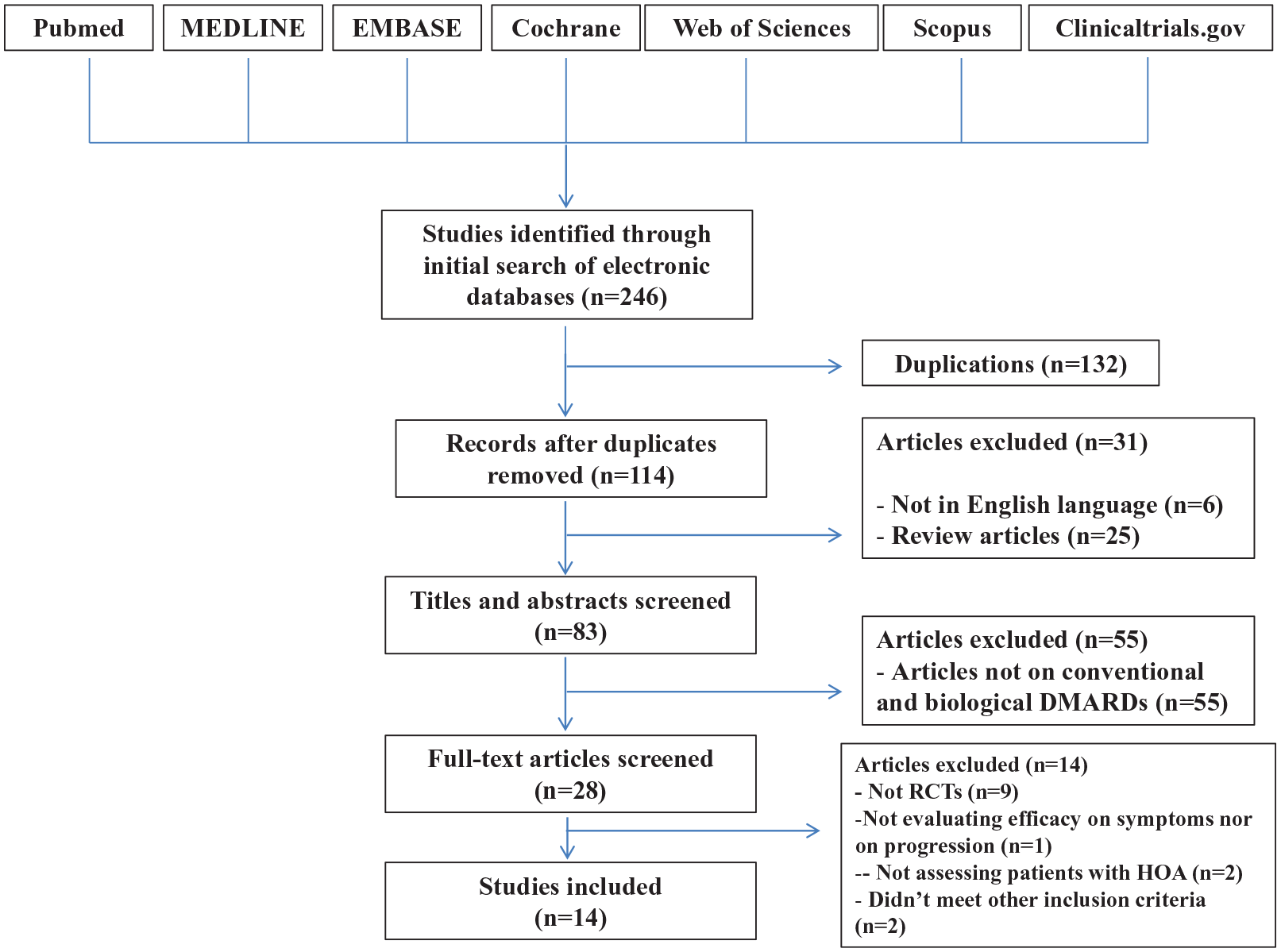
Study flow diagram.
Summary of randomized controlled trials (RCTs) investigating conventional disease-modifying anti-rheumatic drugs (DMARDs) for the treatment of hand osteoarthritis (HOA).

ACR, American College of Rheumatology; ADL scores, activities of daily living scores; AIMS2-SF, Arthritis Impact Measurement Scale 2 short form; AUSCAN, Australian Canadian Hand Osteoarthritis Index; BML, bone marrow lesions; CRP, C-reactive protein; DIPJs, distal interphalangeal joints; EHOA, erosive hand osteoarthritis; EQ-5D, EuroQol 5 dimensions; ESR, erythrocyte sedimentation rate; FIHOA, functional index for hand osteoarthritis score; GUSS, Ghent University Score System; HADS, Hospital Anxiety and Depression Scales; HAQ, health assessment questionnaire; HCQ, hydroxychloroquine; (HOOS)-12, Hip disability and Osteoarthritis Outcome Score; K-L, Kellgren and Lawrence; (KOOS)-12, Knee injury and Osteoarthritis Outcome Score; MCS SF-12, mental component score short-form 12; MCS SF-36, mental component score short-form 36; MHQ, Michigan Hand Outcomes Questionnaire; MRI, magnetic resonance imaging; MTX, methotrexate; N.A., not applicable; NRS; numerical rating scale; NSAIDs, non-steroidal anti-inflammatory drugs; OA, osteoarthritis; OAQoL, Osteoarthritis Quality of Life; OMERACT, Outcome measures in Rheumatology Clinical Trials; PCS, Pain Catastrophizing Scale; PCS SF-12, physical component score short-form 12; PCS SF-36, physical component score short-form 36; PD, power Doppler; PIPJs, proximal interphalangeal joints; PPT, pressure pain thresholds; PSQ, Pain Sensitivity Questionnaire; pts, patients; SAE, serious adverse event; SF-SACRAH, short form score for the assessment and quantification of chronic rheumatoid affections of the hands; SYSADOAs; symptomatic slow-acting drugs for osteoarthritis; US, ultrasonography; VAS, visual analogue scale; V-V score, Verbruggen–Veys anatomical score; WOMAC, Western Ontario and McMaster University Osteoarthritis Index.
Summary of randomized controlled trials (RCTs) investigating biologic disease-modifying anti-rheumatic drugs (DMARDs) for the treatment of hand osteoarthritis (HOA).

ACR, American College of Rheumatology; AUSCAN, Australian Canadian Hand Osteoarthritis Index; BML, bone marrow lesions; C2M/C3M, metallo proteinase-derived type II/III collagen fragments; col10neo, type X collagen neo-epitope; CRPM, matrix metalloproteinase-degraded C-reactive protein; CTX-I, carboxy-terminal collagen crosslinks I; CV, cardiovascular; DAS, disease activity score; DIPJs, distal interphalangeal joints; DMARDs, disease-modifying anti-rheumatic drugs; EHOA, erosive hand osteoarthritis; ESR, erythrocyte sedimentation rate; FIHOA, functional index for hand osteoarthritis score; GI, gastrointestinal; GUSS, Ghent University Score System; HA, hyaluronic acid; HCQ, hydroxychloroquine; HOA, hand osteoarthritis; HOAMRIS, Hand Osteoarthritis Magnetic Resonance Imaging Scoring System; hsCRP, high-sensitivity C-reactive protein; IPJs, interphalangeal joints; i.v., intravenous; K-L, Kellgren and Lawrence; MMP-3. matrix metalloproteinase-3; MPO, myeloperoxidase; MRI, magnetic resonance imaging; MTX, methotrexate; N.A., not applicable; NRS, numerical rating scale; OARSI, Osteoarthritis research society international; OMERACT HOAMRIS, Outcome measures in Rheumatology Hand Osteoarthritis Magnetic Resonance Imaging Scoring System; PCS, physical component score; PD, power Doppler; pts, patients; RA, rheumatoid arthritis; s.c., subcutaneous; SF, short-form; SHS, Sharp–van der Heijde score; OMERACT TOMS, Outcome measures in Rheumatology thumb base Osteoarthritis Magnetic Resonance Imaging Scoring System; US, ultrasonography; VAS, visual analogue scale; V-V score, Verbruggen–Veys anatomical score.
Summary of the registered on ClinicalTrials.gov phase II/III clinical trials investigating new potential biologic disease-modifying anti-rheumatic drugs.

ACR, American College of Rheumatology; ALT, alanine transaminase; anti-CCP antibodies, anticyclic citrullinated peptide antibodies; AST, aspartate transaminase; AUSCAN, Australian Canadian Hand Osteoarthritis Index; CMCJ, carpo-metacarpal joint; DIPJs, distal interphalangeal joints; DLCO, diffusing capacity of the lung for carbon monoxide; EHOA, erosive hand osteoarthritis; FEV1, forced expiratory volume in 1 second; FIHOA, functional index for hand osteoarthritis score; GUSS, Ghent University Score System; Hb, hemoglobin; IPJs, interphalangeal joints; HAQ-DI, health assessment questionnaire; HOA, hand osteoarthritis; MCS SF-36, mental component score short-form 36; MHOQ, Michigan Hand Outcomes Questionnaire; MRI, magnetic resonance imaging; N.A., not applicable; N.R., not reported; NRS; numerical rating scale; NSAIDs, non-steroidal anti-inflammatory drugs; OA, Osteoarthritis; OMERACT, Outcome measures in Rheumatology; PCS SF-36, physical component score short-form 36; PD, power Doppler; PIPJs, proximal interphalangeal joints; pts, patients; RAMRIS, Rheumatoid Arthritis Magnetic Resonance Imaging Score; RCT, randomized controlled trial; RF, rheumatoid factor; SACRAH, score for the assessment and quantification of chronic rheumatoid affections of the hands; s.c., subcutaneous; TB, Mycobacterium tuberculosis; US, ultrasonography; VAS, visual analogue scale; WBC, white blood cell.
HCQ
HCQ is an anti-malarian agent, currently used for the management of various dermatological and rheumatic disorders.40,41 HCQ exerts a variety of immunomodulatory effects, for which intracellular Toll-like receptors (TLR) were identified as key mediators. Since TLRs are upregulated in OA cartilage and thought to stimulate cartilage breakdown, HCQ was hypothesized as a potential treatment for OA. 42 For these reasons, HCQ has been used since many years as an unlicensed drug to treat, when other options have failed, patients with EHOA, considering its inflammatory features. 3 The first reports analyzing the effects of HCQ in EHOA dated back to the 1990s, when Robertson et al. 43 and Bryant et al. 44 reported, in two different retrospective studies, a clinical improvement in a limited number of patients treated with HCQ. Few years later, similar findings were observed in a small RCT on 15 patients with EHOA, of whom 7 received HCQ and 8 traditional analgesics and NSAIDs (Table 1). 24
More recently, negative findings were reported by the Hydroxychloroquine Effectiveness in Reducing Symptoms of Hand Osteoarthritis (HERO) trial, a randomized, double-blind trial with 12 months of follow-up, evaluating the efficacy of HCQ compared with placebo, in 248 symptomatic patients (VAS pain ⩾4/10) with HOA, with inadequate response or side effects to paracetamol, oral NSAIDs, or opioids (Table 1). 25 Furthermore, the economic evaluation of the HERO trial found that HCQ was not a cost-effectiveness treatment option when compared with placebo. 45
Similarly, the multicenter, randomized, double-blind, placebo-controlled trial by Lee et al. 26 failed to show the superiority of 24 weeks of HCQ treatment (400 mg daily) to placebo in a population of 196 symptomatic HOA patients. The number of adverse events was similar in the two groups, although three patients treated with HCQ experienced a mild to severe allergic or dermatologic reaction (Table 1).
More recently, the OA-TREAT trial, a randomized, double-blind, placebo-controlled, multicenter study evaluated the effectiveness of HCQ in patients with radiological evidence of EHOA and persisting symptoms and signs of inflammation despite the use of analgesics or NSAIDs with a pain score ⩾4 on a numerical rating scale (NRS) (0–10) and an AUSCAN function ⩾26. Seventy-five patients were randomized to HCQ and 78 to matching placebo. No significant difference between groups was found at 52 weeks in AUSCAN pain and function, as well as in radiological progression, NSAIDs consumption, and in the majority of secondary outcomes (Table 1). 27
The discouraging results from the above-reported clinical trials were recently confirmed by a systematic review and meta-analysis assessing the efficacy and safety of HCQ in OA. The authors found high-quality evidence supporting that this drug is no more effective than placebo or active comparator in controlling pain and improving functionality in patients with HOA, while the overall safety profile resulted acceptable. 46
MTX
MTX is an anti-metabolite agent approved since many years as a conventional DMARD for RA for which it still represents an anchor drug, being recommended as a first line of therapy by all the most prominent scientific societies.40,47–49 MTX suppresses the proliferation of immune and inflammatory cells in RA synovium and consequently inhibits the release of pro-inflammatory cytokines. For its mechanism of action, it was hypothesized that MTX can have a beneficial role in HOA patients with synovitis phenotype. 40
Our literature search found only one published study, a randomized, double-blind, placebo-controlled trial, analyzing the efficacy of MTX in symptomatic EHOA patients. The drug was administered orally to 32 patients, at the dosage of 10 mg weekly, while other 32 patients received an oral placebo for 12 months. At 3 and 12 months, no significant differences between the two groups in pain and functionality outcomes were observed. Furthermore, at 12 months, according to the Verbruggen–Veys (VV) anatomical score, erosive joints progressed significantly more to a remodeling phase in the MTX group than in the placebo group, while patients without erosions at baseline appeared to develop fewer erosions when treated with MTX at 1 year of follow-up, but these results were not statistically significant (Table 1). 28
Recently, a team of Australian researchers published the protocol of the study ‘METHODS’, a multicentre, randomized, double-blind, placebo-controlled trial, designed to investigate the efficacy of 20 mg MTX, administered orally, once a week, over 6 months in decreasing clinical symptoms and structural progression in patients with HOA and synovitis, determined from magnetic resonance imaging (MRI). The trial was temporarily stopped in March 2020 due to COVID-19 pandemic, after the enrolment of the first 80 patients, and resumed in November 2020. The trial is still ongoing (Table 1). 29
The search in ClinicalTrials.gov showed two other studies evaluating MTX treatment in patients with HOA, one early terminated due to COVID-19 pandemic and another actually still recruiting. The first one (study identifier: NCT01927484), a double-blind, randomized, placebo-controlled trial, was aimed to assess the efficacy of MTX in decreasing pain and inflammation in 120 knee OA patients with concomitant symptomatic HOA. MTX was administered orally, at the dosage of 25 mg weekly, for 28 weeks to 60 patients, while the other 60 received matching placebo (Table 1). Unfortunately, the results have not been posted. The other trial (study identifier: NCT04579848), a randomized, double-blinded, placebo-controlled, single-center, phase IV, superiority study started in August 2021 to assess the effect of MTX on pain, function, and structural outcomes in EHOA. The study was aimed to include 170 patients with symptomatic EHOA, with insufficient pain relief or with contraindications to paracetamol/NSAIDs to be allocated to MTX treatment, administered orally, once a week, at the starting dose of 15 mg for 2 weeks, followed by 20 mg for the remaining 50 weeks or to placebo. The primary outcome will be the difference in hand VAS pain at 6 months of treatment. A set of secondary outcomes will be also evaluated, including clinical data, pain, and functionality indexes and questionnaires assessing quality of life and mood disorders, as well as radiologic scores and a set of soluble biomarkers of extracellular matrix (ECM) turnover, inflammatory cytokines, and adipokines.
TNF-α inhibitors
TNF-α is a well-known pro-inflammatory cytokine which plays an important role in the development and progression of OA.16,50,51 In particular, TNF-α inhibits the synthesis of proteoglycan components and collagen and enhances the degradation of the ECM components by inducing the production of the main metalloproteinases (MMPs) and aggrecanases and increases the release of IL-6 and IL-8. Furthermore, it stimulates the synthesis of regulated upon activation normal T cell expressed and secreted (RANTES) and vascular endothelial growth factor (VEFG) and induces the production of iNOS, COX-2, and PGE-2 synthase.16,50,51 In addition, TNF-α, through the release of PGE-2, leads to the expression of the neurotrophin nerve growth factor (NGF), which is a key mediator of pain in OA. 52 Thus, considering the biological activity of TNF-α, together with the presence of signs of inflammation, such as synovitis, demonstrated by US and MRI in the interphalangeal (IP) joints of patients with HOA, it has been hypothesized that this cytokine could be a target for therapy to reduce pain and slow disease progression. 32 Therefore, several efforts have been made to treat HOA with TNF-α inhibitors with often limited and unsatisfactory results. 15
Our literature search found three RCTs evaluating treatment with adalimumab in HOA and one about etanercept, while there are no RCTs on other anti-TNF-α inhibitors; however, a double-blind placebo-controlled pilot study investigating the efficacy of intra-articular (i.a.) infliximab in 10 patients with symptomatic bilateral EHOA, who have had an inadequate response to NSAIDs or other conventional agents, deserves a mention.30–33,53 Indeed, even considering all the limitations inherent to a not randomized design and to a small sample size, the authors demonstrated a reduction of pain and a tendency toward stability or slight bone remodeling radiological progression, evaluated by the anatomical lesion progression system from Verbruggen and Veys, 54 only in the hand treated with infliximab after 12 months. 53
Adalimumab
Adalimumab is a currently approved treatment for RA, ankylosing spondylitis, psoriasis, Crohn’s disease, ulcerative colitis, psoriatic arthritis, juvenile idiopathic arthritis (JIA), uveitis, and hidradenitis suppurativa. 38 In HOA, adalimumab was first evaluated in an open-label trial dating back to 2007 which showed positive clinical effects in 12 EHOA patients, after a brief course of treatment. 55 Some years later, Verbruggen et al. 30 conducted a 12-month, double-blind, randomized, placebo-controlled trial to evaluate the safety and efficacy of adalimumab, 40 mg subcutaneous every 2 weeks, in controlling the structural damage in 60 patients with active EHOA in one or more of their IP joints. Fewer patients in the adalimumab group (26.7%) than in the placebo group (40%) developed at least one new erosive joint over 12 months, although these differences were not significant and the overall results did not demonstrate a therapeutic effect of adalimumab. However, in a subpopulation at higher risk of progression (patients with palpable soft tissue swelling at baseline), adalimumab seemed to prevent the evolution of structural damage to the erosive phase; these findings were also confirmed by the minor change toward progression of the scoring system Ghent University Score System (GUSS) after 6 months of treatment with adalimumab (Table 2). 30 The data on the symptomatic efficacy of adalimumab are not more encouraging than the above reported on the structural effect. Indeed, a well-conducted randomized, double-blind, placebo-controlled trial on 85 patients with painful HOA refractory to both analgesics and NSAIDs failed to demonstrate the superiority of adalimumab 40 mg, administered subcutaneously at a 15-day interval, for 6 months, over placebo in reducing pain at any time point. Similarly, no significant differences between the two groups were detected for all the secondary outcomes and in the dosage of some serum biomarkers of cartilage turnover (Table 2). 31 Similarly, another randomized double-blind placebo-controlled trial aimed to evaluate the efficacy of adalimumab in a population of 43 patients with painful EHOA and presence of synovitis detected on MRI failed to show an effect both for pain and for structure (Table 2). 32
Etanercept
Etanercept was the first TNF-α inhibitor approved for RA treatment in the United States and Europe. 56 The data about etanercept treatment in HOA are very scarce and limited to a recent multicentre, randomized, double-blind, placebo-controlled trial including 90 patients with EHOA and proven clinical and radiological signs of inflammation, other than hand pain >30 on a 0–100 mm VAS despite the use of NSAIDs. Patients were treated with subcutaneous etanercept 50 mg weekly for the first 24 weeks, followed by 25 mg weekly until 1 year, or placebo. No significant differences between the two groups in VAS pain were observed at 24 weeks, although a subgroup analysis of patients with signs of active inflammation at baseline showed a positive, albeit limited, symptomatic, and structural effect of etanercept. Indeed, in this study subpopulation, etanercept led to a relevant improvement in pain, after 1 year, based on a minimally clinically important difference in pain and to a significant reduction of GUSS score and of bone marrow lesions (BML) detected by MRI (Table 2). 33 Furthermore, the evaluation of a pattern of soluble biomarkers of inflammation and joint damage demonstrated a more significant reduction of MMP-3 levels in etanercept group versus placebo after 24 weeks, while no differences were found for the other analyzed biomarkers. 57
TNF-α inhibitors treatment for HOA in patients with concomitant RA
Primary HOA and RA are two distinct entities, which often occur simultaneously in the same patients, and even more frequently secondary HOA can develop in joints injured by RA. For these reasons, in the last decades, some researchers investigated the effects of TNF-α inhibitors, on incident and progressive secondary HOA.34–36 The main findings of these studies are summarized in Table 2. An exploratory observational longitudinal study analyzing data from the BeSt study, an RCT aimed to compare the efficacy of four treatment strategies in recent-onset active RA patients, investigated the effect of intravenous (i.v.) infliximab (dosage from 3 to 10 mg/kg/8 weeks) on the development and progression of radiographic secondary HOA over a period of 3 years. The radiographs of 416 patients at baseline and after 3 years were assessed and the osteophyte and the Sharp–van der Heijde scores were calculated. The authors found a trend toward an inhibitory effect of infliximab therapy on incident secondary HOA in proximal interphalangeal joints (PIPJs), but not in distal interphalangeal joints (DIPJs), which was independent from the effect of this biologic agent on inflammatory activity. Thus, it was speculated that TNF-α antagonists could be useful in preventing the development of secondary HOA through bone-linked pathways. 34 Some years later, similar results were reported in another observational longitudinal study analyzing 10 years of follow-up data from the above-mentioned BeSt study. Indeed, Loef et al. 35 observed a significant reduced risk of OA progression in the DIP joints for each month of treatment with TNF-α inhibitors, while no effect was showed in the development of incidental HOA. Conversely, Lechtenboehmer et al. 36 in a cohort study on data derived from RA patients in the Swiss Clinical Quality Management in Rheumatic Diseases registry found that biologic DMARD monotherapy was associated with an increased risk of radiographic DIP OA progression compared with conventional DMARD monotherapy in patients with pre-existing DIP OA. Furthermore, the risk was not significant in patients receiving a combination therapy of conventional and biologic DMARDs; this result seemed to be due mainly to an enhanced osteophytes growth. Instead, the risk of incident radiographic OA did not differ between patients treated with conventional DMARDs or biologics, either in monotherapy or combination therapy.
IL-1 inhibitors
IL-1β is one of the main important pro-inflammatory cytokines which plays a prominent role in the pathogenesis of OA.58,59 In particular, through the nuclear factor (NF)-ĸB pathway, it exerts catabolic effects, inhibits the synthesis of ECM and enhances the production of pro-inflammatory cytokines and chemokines. 16
Considering that several in vitro and animal studies showed that IL-1 receptor antagonists could have beneficial effects on OA progression and that EHOA resulted associated with an IL-1β gene polymorphism, this cytokine was investigated as new potential therapeutic target in OA in general and also in HOA.60–68 However, our literature found only one RCT exploring the effects of lutikizumab in HOA patients. 37
Lutikizumab
Lutikizumab is a novel human dual-variable domain immunoglobulin (DVD-Ig), which simultaneously binds and inhibits IL-1α and IL-1β without interfering with other human IL-1 family members. 37 It was developed for the treatment of hidradenitis suppurativa and OA. In animal models of OA, this monoclonal antibody resulted to reduce the cartilage degeneration and the threshold for evoked pain.37,69 A phase I trial in subjects with gonarthrosis demonstrated a good tolerability profile of lutikizumab and different pharmacological effects, such as decrease of neutrophils, hs-CRP, and of markers of synovitis. 69 For these reasons, in 2019, a phase II, placebo-controlled, double-blind, randomized study was conducted in patients with EHOA. One hundred ten patients diagnosed with EHOA in a phase of active inflammation completed the study, of whom 61 received placebo and 49 were treated with lutikizumab 200 mg subcutaneously every 2 weeks for 24 weeks. Injection site reactions, neutropenia, and discontinuations due to adverse events were more frequent in lutikizumab group than placebo (Table 2). However, lutikizumab demonstrated appropriate pharmacodynamic effects, including significant decrease of hs-CRP protein levels, IL-1α and IL-1β levels, and blood neutrophils; thus, the authors concluded that targeting IL-1 may not be effective for the treatment of EHOA. 37
IL-6 inhibitors
IL-6 is a pleiotropic cytokine, classified as an adipokine and myokine. 70 The exact role of IL-6 in OA is difficult to define, since both beneficial and detrimental effects of this cytokine were described. In vitro studies on chondrocytes demonstrated that IL-6 can induce the tissue inhibitor of metalloproteinases (TIMP)-1 and increase the expression of collagen type 2, while others showed an inhibition of collagen type 2 production and an enhanced expression of MMP-1, 3, and 13 and ADAMTS-4, 5/11.71,72 Furthermore, low innate capacity to produce IL-6 is associated with the absence of OA in old age, while targeting IL-6 or the signal transducer and activator of transcription 3 signaling pathway resulted to be associated with slow progression in an experimental mouse model of OA.73–75 Taken together, this evidence suggests a prominent role of IL-6 in the structural damage of OA.
Tocilizumab
Tocilizumab is an approved treatment for RA, systemic JIA (sJIA), JIA, COVID-19 disease, and cytokine release syndrome.76–78
The symptomatic efficacy of tocilizumab in HOA was evaluated in a recent randomized, double-blind, placebo-controlled, multicenter trial. Ninety-one patients with painful HOA were randomized to receive two infusions 4 weeks apart of tocilizumab (8 mg/kg) or placebo. The study failed to demonstrate the efficacy of this IL-6 inhibitor over placebo in improving pain and function. Overall, side effects were slightly more frequent in the tocilizumab group (n = 29, 69% versus n = 22, 53.7%) and consisted mainly in infections and neutropenia (Table 2). 39
Investigational conventional and biological DMARDs for the treatment of HOA, either active or completed
Our search in ClinicalTrials.gov allowed us to identify seven trials of investigational biological DMARDs in HOA, of whom three on gevokizumab, one on GSK3196165, one on denosumab, one on apremilast, one on iguratimod, other than two trials about MTX which we reported above in the related paragraph. The main characteristics of the first seven above-mentioned trials are summarized in Table 3.
Gevokizumab (also known as XOMA 052) is an experimental monoclonal IgG2 antibody that inhibits IL-1β binding to its receptor via an allosteric mechanism which potently neutralizes IL-1β signal activation without affecting IL-1Ra. Gevokizumab has entered clinical investigation for treating inflammatory or autoimmune diseases, such as Behçet’s uveitis and some dermatosis and diabetes and cardiovascular diseases.79–81
In 2012, a group of researchers from the United States decided to start a phase II proof-of-concept study of gevokizumab in patients with EHOA (ClinicalTrials.gov identifier: NCT01683396). Eighty-seven participants were enrolled and treated with subcutaneous injections of gevokizumab or placebo (dosage and schedule of treatment not specified). The authors aimed to evaluate the percentage of change from baseline in AUSCAN pain score at day 84. The study resulted completed in February 2014, but no results were posted. A similar study with the same characteristics and aims was registered in ClinicalTrials.gov in 2013 with the number NCT01882491. Furthermore, in 2014 the open-label safety extension study of gevokizumab in EHOA was registered (ClinicalTrials.gov identifier: NCT02293564) and enrolled 312 patients with the objective to analyze the incidence, severity, and type of treatment-emergent adverse events. Unfortunately, no results are yet available.
In 2016, a multicenter phase IIa double-blind, placebo-controlled study aimed to investigate the efficacy and safety of GSK3196165 in subjects with inflammatory HOA started and included different centers in the United States, Germany, Netherlands, Poland, and United Kingdom (ClinicalTrials.gov identifier: NCT02683785). GSK3196165 is a fully human monoclonal antibody that inhibits granulocyte-macrophage colony-stimulating factor (GM-CSF), a protein that plays a central role in a broad range of immune-mediated diseases, including RA. The trial enrolled a total of 44 patients diagnosed with HOA, according to the ACR criteria, and who have failed analgesics or NSAIDs (taken for at least 10 days in the past 3 months) and with active disease at screening, defined as the presence of at least two swollen and tender PIPJs or DIPJs in the affected hand and signs of inflammation in the MRI scan. Twenty-two patients received five weekly subcutaneous injections of GSK3196165, followed by three injections every other week, while the other 22 patients were treated with matching placebo. The follow-up was extended until week 22. The change from baseline in 24-hour average hand pain intensity, representing the primary outcome, was more pronounced, although not in a significant manner, in the GSK3196165 group versus placebo. Similarly, no significant differences between the two groups were found for the secondary outcomes.
Another recently completed study, of which we do not know yet the results, regards the evaluation of denosumab in EHOA (ClinicalTrials.gov identifier: NCT02771860). Denosumab, a monoclonal antibody to the receptor activator of NF-κB ligand (RANK-L), suppresses osteoclastic function and differentiation from immature to mature osteoclasts, resulting in strong anti-resorption efficacy for osteoporosis. 82 This is a randomized, double-blind placebo-controlled one-site proof-of-concept study in subjects with EHOA which foresaw the enrollment of a total of 100 subjects to be treated with subcutaneous injections of 60 mg of denosumab every 12 weeks, or matching placebo followed by a 48-week open-label phase in which all subjects will receive denosumab. The primary outcome will be the change in the negative evolution in GUSS score in the target IP joints from baseline to week 24.
We found another recent phase II, multicenter, randomized, double-blind, placebo-controlled trial investigating the efficacy and safety of apremilast in 30 patients with EHOA (ClinicalTrials.gov identifier: NCT01200472). Apremilast is a small-molecule phosphodiesterase 4 (PDE4) inhibitor; it is approved by Food and Drug Administration (FDA) and European Medicines Agency (EMA) for plaque psoriasis in adult patients who are candidates for phototherapy or systemic therapy, psoriatic arthritis in adult patients with moderately to severely active disease, and for oral ulcers associated with Behçet’s disease. 83 Patients received apremilast, as 10 mg capsules daily for 7 days, followed by 20 mg capsules two times a day for 91 days, or placebo; subsequently, all patients were treated with apremilast for other 77 days and a final 28-day phase of observation follow-up concluded the study. Unfortunately, no results were posted, but the researchers aimed to evaluate the proportion of subjects in each treatment group who achieve a significant 50% improvement in AUSCAN Index at day 84 after treatment.
Finally, our search in ClinicalTrials.gov allowed us to identify an RCT, recently started (March 2022), analyzing the effectiveness and safety of iguratimod in patients with HOA (ClinicalTrials.gov identifier: NCT05216757). Iguratimod is a novel synthetic small-molecule DMARD, approved only in Japan and China, as a new option for RA treatment. Pharmacological studies have demonstrated that this drug can reduce the production of immunoglobulins by acting on B cells and can also enhance bone formation by inhibiting the activation of osteoclasts and stimulating osteoblast differentiation. 84 A previous animal model study demonstrated that iguratimod can maintain the cartilage matrix homeostasis environment through an effect mediated by Wnt/β-catenin signaling pathway, through the inhibition of the production of inflammatory cytokines such as IL-6 and TNF-α and the reduction of MMP-13 levels in articular cartilage. 85 The researchers foresee to enroll about 150 participants diagnosed with symptomatic HOA with active signs of inflammation in at least one IP joint. Patients will be then randomized into two groups: one treated with oral iguratimod at the dosage of 25 mg two times a day and the other one with matching placebo for 12 weeks. Change from baseline to week 12 in patient’s assessment of VAS pain was chosen as a primary outcome; a set of secondary outcomes, including AUSCAN, functional index for hand osteoarthritis score (FIHOA), health assessment questionnaire (HAQ), SF-36, Michigan Hand Outcomes Questionnaire, US findings, and OMERACT HOA MRI score will be also evaluated.
Discussion
The present narrative review summarized the available evidence about the treatment of HOA with the conventional or biological DMARDs. Despite the great success that such kind of drugs have demonstrated in inflammatory joint diseases, so that they changed the history of their management, the results in HOA are mainly unsatisfactory and inconsistent.
In particular, the initial enthusiasm on HCQ derived from case series or studies with very small sample size24,43,44 was held back by the results of three high-quality rigorous trials with long follow-up duration25–27 and confirmed by a recent systematic review and meta-analysis. 46 Indeed, these RCTs showed that HCQ was no more effective than placebo in reducing pain, improving physical function and quality of life in patients with erosive and non-EHOA, despite a favorable safety profile.25–27,46 In addition, HCQ did not result a cost-effective option management of HOA. 45 Among the conventional DMARDs, the results are no more encouraging about MTX use in patients with HOA, although in this case, the evidence is limited to a single double-blind RCT which failed to demonstrate significant differences in pain progression after 3 and 12 months between MTX and placebo group. 28 However, the authors stated that a possible explanation of these negative results could be attributed to the low dose of the MTX employed (10 mg/week) or to the high placebo effect reported in OA or can be linked to the multifactorial origin of pain in HOA. Interestingly, although this trial was not designed to investigate a structure-modifying effect of MTX, fewer erosions detected by radiographs or MRI were observed after 1 year of treatment in patients without erosions at baseline, suggesting a potential role of MTX in HOA progression, when administered in early phases of the disease. 28 More evidence will be obtained in the future, as soon as the ongoing trials on MTX will be available.
Over the last decades, the advances in the understanding of OA pathophysiology showing that inflammatory cytokine network plays a prominent role in the development and progression of the disease have driven the investigation on the potential effectiveness and usefulness of drugs targeting specific cytokines, the so-called biologics. 86 Unfortunately, clinical trials did not meet the expectations, since at least six well-conducted RCTs failed to demonstrate either significant symptomatic or structural effects of TNF-α inhibitors, IL-1, and IL-6 antagonists in patients with HOA.30–33,37,39 However, the findings of some studies suggested that specific subsets of HOA may benefit more from biological therapy. Indeed, in the study by Kloppenburg et al., 33 etanercept resulted superior compared with placebo in controlling pain and reducing structural progression in a subgroup of EHOA patients with more pronounced inflammatory symptoms. Similarly, Verbruggen et al. 30 found less erosive progression in EHOA patients receiving adalimumab compared with placebo only in a subpopulation with soft tissue swelling at baseline, considered a prognostic factor for disease progression. In this respect, in recent years, great emphasis was given to the identification of risk factors for radiologic progression in EHOA, and other than swollen joints, also the presence of synovitis in the small joints of the hand, documented by US or MRI, and BML at MRI were found as independent predictors for the development of joint erosions.87–90
Furthermore, we have to consider that the failure of biologic treatment in HOA could be influenced by several factors. First of all, the number of high-quality studies exploring biologics in HOA is still limited; the study population differs from a study to another, as well as the investigated drugs or the comparators, and the follow-up duration is often too short. An important issue that strongly limits the uniformity of clinical trials in HOA is represented by the heterogeneity of OA phenotypes, the fluctuating phases of the disease which turns period of low and high activity, and the definition itself of HOA. Indeed, the majority of the analyzed trials referred to the ACR criteria and K-L radiological score and this selection may miss the inclusion of patients in the early phases of OA who could more benefit from the treatment. Similarly, a consensus definition of ‘early OA’ is still lacking. Another reason may be related to the way of drug administration. Indeed, in almost all RCTs on biologics, the pharmacological agents were administered by subcutaneous way, while only one study assessed the intra-articular delivery which is known as the optimal strategy to maximize the local therapeutic effect, reducing adverse events.53,91 Indeed, although limited by a small sample size and by a not randomized design, the study by Fioravanti et al. 53 showed that i.a. infliximab resulted effective in reducing pain and it demonstrated some effects also in slowing radiological progression. In this respect, a very recent network meta-analysis found that infliximab was the most effective treatment in OA compared with all other biologics regarding pain relief. 92
Another possible explanation behind the failure of biologic DMARDs therapy in HOA can be attributed to the involvement of more complex interactions among various inflammatory cytokines. 14 Indeed, it is possible that inflammation might not be the right target, considering that the degree of synovitis can fluctuate in the same patient over time and that some evidence exists demonstrating low serum levels of TNF, IL-1, and IL-6 in EHOA.18,20,93 Furthermore, it is likely that blocking one cytokine is not be sufficient to effectively treat OA; in fact, in vitro studies on synovial cells showed that IL-1 and TNF act in a simultaneous parallel manner in OA. 93 Furthermore, we have to take into consideration that in the development and progression of HOA, an important role was played by the mechanical load, consisting mainly in the repetitive stress of some occupational or re-creational activities. 94 In addition, actually, we know that another pivotal risk factor for HOA is represented by obesity with its network of inflammatory mediators released by adipose tissue.2,95,96
Pain in HOA is the result of a complex mechanism, in which the excess of nociception due to structural damage, as well as central and peripheral sensitization, plays an important role.97,98 Growing evidence supports the involvement of the innate immune pathways in OA pain, as those mediated by C-C motif chemokine ligand 2 and NGF or aggrecan fragment which activates TLR 2 on joint nociceptor.39,99 For these reasons, in recent years, NGF blockade was investigated in patients with knee and hip OA with positive results on effectiveness, but serious concerns in safety profile, although no studies are conducted in patients with HOA. 14
Even more complex is the search for effective DMOADs, able to delay or reverse the progression of the structural damage of the joint, simultaneously leading to clinical improvement in symptoms. Indeed, in this regard, many issues are still under debate, as if it is better to continue to use the radiographic measure of joint space width, which is considered the gold standard for imaging trials in OA, or rather to refer to MRI which can evaluate cartilage morphology or the presence of BML. 15
The main limitation of this review can be attributable to its narrative nature with all the limitations inherent to a nonrigorous systematic review. In particular, our literature search did not identify the quality and the strength of the discussed trials, and has not been built on a robust methodology structure. Further limitations are those intrinsic to the included papers which are very heterogeneous from each other.
Conclusion
In conclusion, our narrative review highlights how, despite a major public health burden with increasing prevalence and consequent disability, the management of OA, in general, and of HOA, in particular, remains largely palliative with an unmet need for effective treatments. The therapy with conventional and biologic drugs in HOA has not achieved the expected success, despite a rationale for their use exists. Taken together, the results of our review outline that the heterogeneity and the complexity of HOA make it urgent to enhance the exploration of its basic molecular mechanisms which can account for the diversity of HOA symptoms and phenotypes. It should be equally important to reach universally accepted classification of the different HOA subsets and to contribute to the development of more advanced imaging and sophisticated biomarkers tools which can help to evaluate the effects of the investigational drugs and to define the different phases of the disease. Furthermore, we believe that is not more the era of ‘one-fits-all’ treatment guidelines, but an approach toward personalized OA treatment for each patient should be strongly encouraged.
